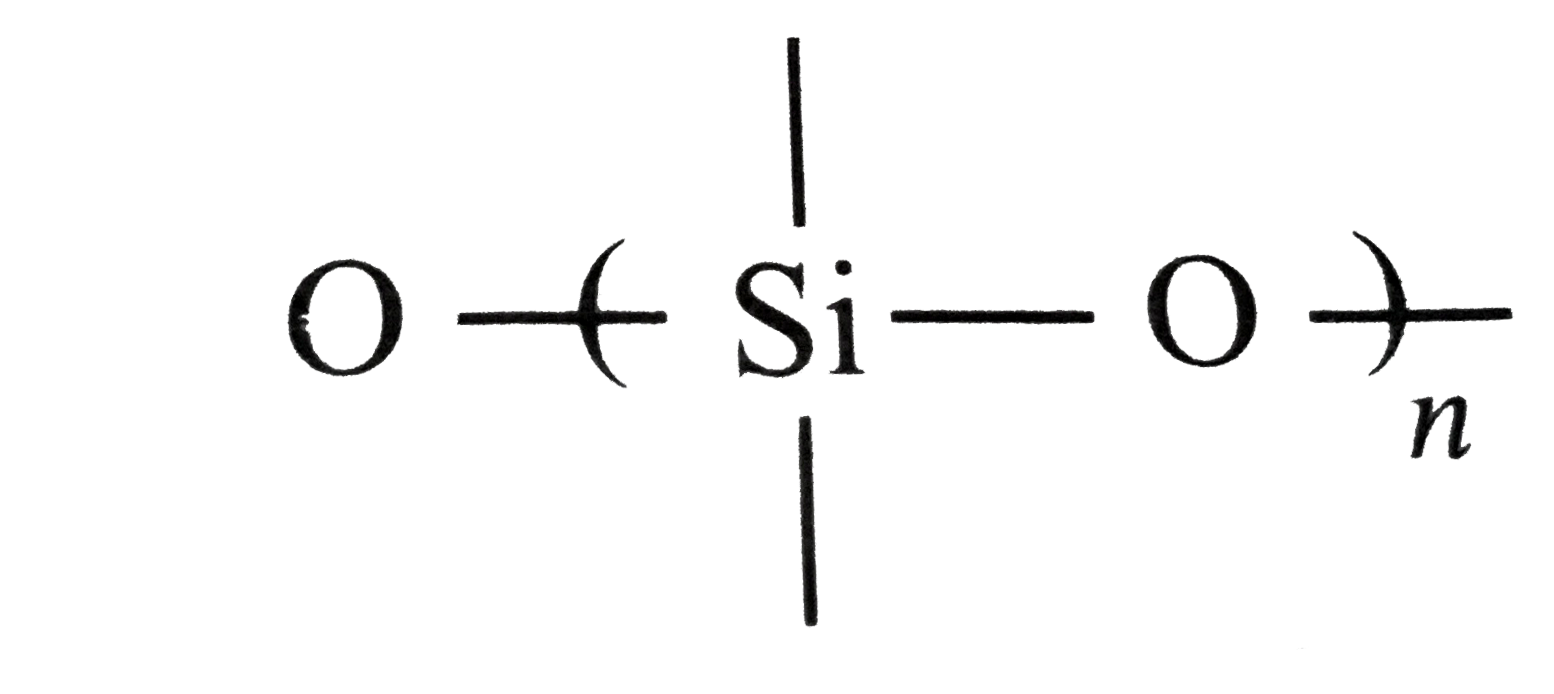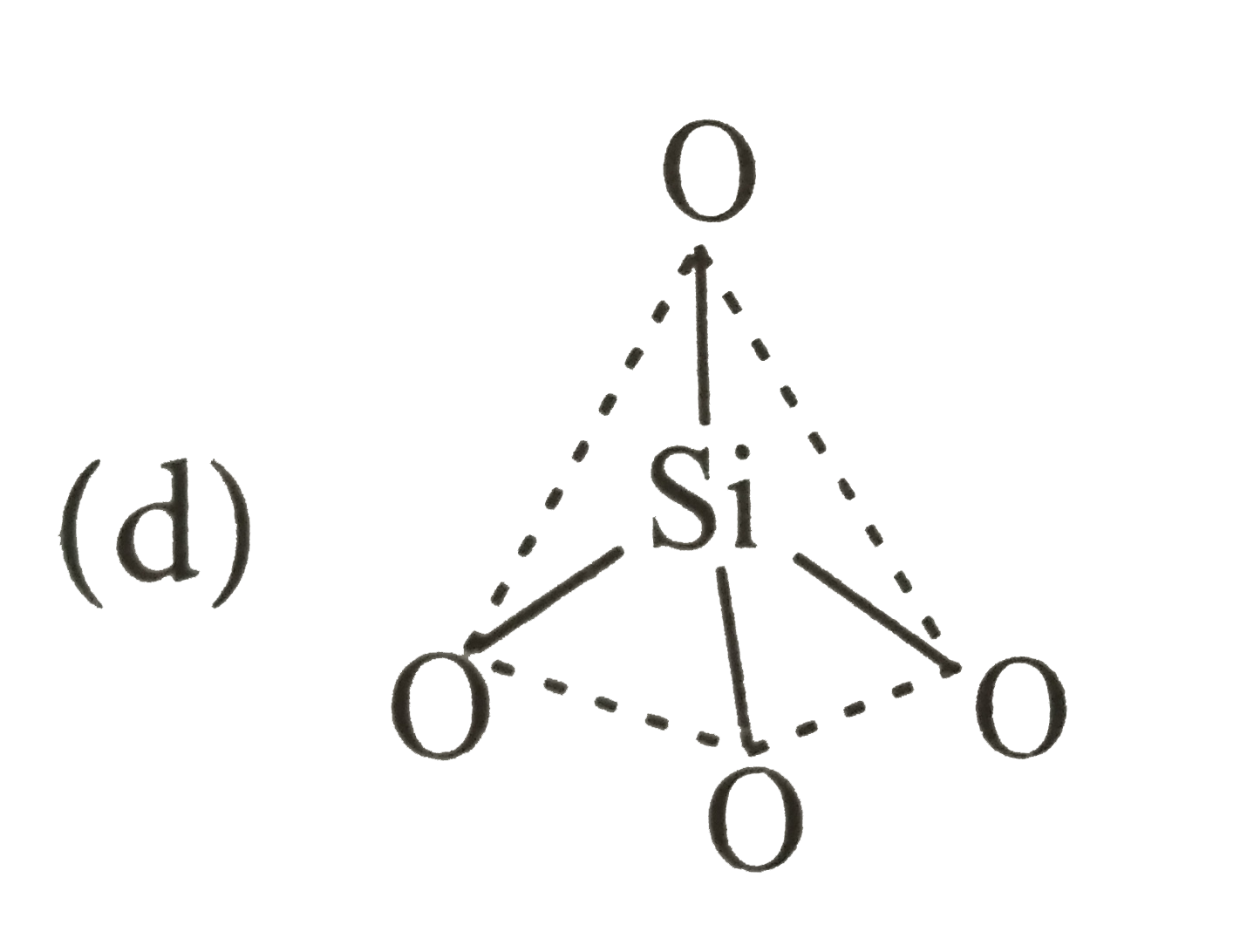Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following is an example of non-benzenoid aromatic compound? |
|
Answer» Toluene |
|
| 2. |
Whichof the followinghas thelargestsize ? |
|
Answer» `S^(2-)` THUS`Te^(2-)` has thelargestsize . |
|
| 3. |
The solubility of Sr(OH)_2 at 298 K is 19.23 g/L of solution. Calculate the concentrations of strontium and hydroxyl lons and the pH of the solution. |
|
Answer» Solution :Molecular MASS of `Sr(OH)_2`= Sr+2[OH] =87.6+34= 121.6 g `mol^(-1)` Concentration of `Sr(OH)_2 (M)` `="WEIGHT (g)"/"Volume (L) X Molecular mass"` `=(19.23 gL^(-1))/(121.6 "g mol"^(-1))=0.1581 "mol L"^(-1)` or M `Sr(OH)_2` which is totally soluble is COMPLETELY ionized. `Sr(OH)_2=Sr_((aq))^(2+) + 2OH_((aq))^(-)` `THEREFORE [Sr^(2+)]=[Sr(OH)_2] `=0.1581 M `[ObarH]=2[Sr(OH)_2] = 2xx0.1581`=0.3162 M pOH=-log `[OH^-]` =-log (0.3162) =-(-0.5) =+0.5 pH=14.0-pOH=(14.0-0.5) =13.5 |
|
| 4. |
Which one of the following metal cannot be extracted by using Al as a reducing agent ? |
|
Answer» Na from `Na_(2)O` |
|
| 5. |
Two grams of a mixture of propane and actylene is burnt in excess oxygen. The unreacted oxygen is removed by adsorption into basic pyrogallol. The ratio of volumes of gaseous CO_(2) and H_(2)O is 3:2. Determine the mass of unsaturated hydrocarbon in the given mixture. |
|
Answer» Solution :The stoichimetric equations for the conbustion of hydrocarbons are `C_(8)H_(8)overset(O_(2))to3CO_(2)+4H_(2)O,C_(2)H_(2) overset(O_(2))to2CO_(2)+H_(2)O` LET the number of moles of `C_(2)H_(2)` is x Let the number of moles of `C_(3)H_(8)` is y Ratio of VOLUMES is equal to ratio of moles `(" number of moles of "CO_(2))/("number of moles of "H_(2)O )=(3)/(2)=(2x+3y)/(x+4y)` SOLVING, x:y =6:1 The ratio of weights of `C_(2)H_(2)` and `C_(3)H_(8)` = `6xx26:1xx44=156:44` MASS of `C_(2)H_(2)` in the given mixture `=(156)/(156+44)xx2=1.56` grams |
|
| 6. |
Two atoms are said to be isobars if.... |
|
Answer» they have same atomic number but different MASS number. 
|
|
| 7. |
The wrong statement about fullerene is |
|
Answer» it has 5 membered carbon ring. |
|
| 8. |
Which of the following compounds will give racemic mixture on nucleophilic substitution by OH-ion? (i) CH_(3)-underset(C_(2)H_(5))underset(|)(C)H-CH_(2)Br (ii) H_(3)C-underset(Br)underset(|)overset(CH_(3))overset(|)(C)-C_(2)H_(5) (iii) CH_(3)-underset(Cl)underset(|)overset(H)overset(|)(C)-C_(2)H_(5) |
|
Answer» (i) |
|
| 9. |
What is the origin of petroleum in nature ? |
| Answer» Solution :Like COAL and natural gas, petroleum was formed from the remains of ancient marine ORGANISMS, such as plants, algae, and bacteria. . Millions of years ago, algae and plants lived in shallow SEAS. After dying and sinking to the seafloor, the ORGANIC material mixed with other sediments and was buried | |
| 10. |
Which hybird orbitals are used by carbon atoms in the following molecules?CH_3-CH=CH_2 |
Answer» SOLUTION :
|
|
| 11. |
What is the experimental evidence in support of the idea that electronic energies in an atom are quantized ? |
| Answer» Solution :The line spectrum of any element has lines corresponding to definite wavelengths. Lines are obtained as a result of electronic transitions The w avelength is DISTANCE betw een TWO SUCCESSIVE peaks or two successive troughs of a wave. So, X = 4 x 2.16 pm = 8.64 pm 37 between the ENERGY levels. So, the electrons in these levels have fixed energy, i.e., quantized values. | |
| 12. |
What is the reason that sodium reacts with water more vigorously than lithium? |
| Answer» SOLUTION :Because SODIUM is more electro-postive than LI. | |
| 13. |
Which cation forms an insoluble chloride and an insoluble sulphide? |
| Answer» Answer :D | |
| 14. |
Which xenon compound is not possible in following chemical reaction ? |
|
Answer» `XeO_3+ 6HF to XeF_6 + 3H_2O` |
|
| 15. |
Which law represents the pairing of electron in a sub-shell after each orbital is filled with one electron |
|
Answer» Pauli's exclusion principle |
|
| 16. |
The rms velocity of CO_(2) at a temperature, T (in kelvin) is x eras^(1). At what temperature (in kelvin), the rms velocity of nitrous oxide would be 4 x ems^(-1)? (Atomic weights of C, N and O are 12, 14 and 16 respectively), |
|
Answer» 16 T |
|
| 17. |
Which temperature shows liquification of gas ? |
|
Answer» `T_(C )` |
|
| 18. |
Which one of the following is wrong Statement |
|
Answer» MILK of magnesia is used as antacid in medicine |
|
| 19. |
What is Oxidation Number? What is the oxidation Number of Cl is KClO_(3)? |
|
Answer» Solution :Oxidation NUMBER is the oxidation state of an ELEMENT in a compound ascertained according to a set of rules fomulated on the basis that the electrons in a co-valent BOND BELONG entirely to the more electronegative ELEMENTS. Oxidation number of Cl in `KClO_(3)` is +5. |
|
| 20. |
The temperatures at which real gases obey the ideal gas laws over a wide range of pressure is called .. |
|
Answer» CRITICAL temperature The TEMPERATUREAT whichrealgasesobeythe ideal gaslawsovera widerangeofpressureiscalledboyle temperature |
|
| 21. |
Which CI_(2) is passed through hot NaOH solution, oxidation number of CI changes from E238 |
|
Answer» `-1 to 0` |
|
| 22. |
Why group 2 elements are harder than alkali metals? |
|
Answer» Solution :The strength of metallic bond in alkaline EARTH metals is HIGHER than alkali metals DUE to the presence of 2 electrons in its outermost shell as COMPARED to alkali metals, which have only 1 electron in valence shell. Therefore, alkaline earth metals are harder than alkali metals. (ii) The alkaline earth metals have greater nuclear charge and more valence electrons, thusmetallic BONDING is more effective. Due to this they are harder than alkali metals |
|
| 23. |
What is mean by inactivators of groups ? Explain with suitable examples. |
|
Answer» Solution :DEFINITION : The GROUP which enter first in the benzene ring by the first substitution method, which makes the second ELECTROPHILICSUBSTITUTION reaction difficult such groups are known as inactivators groups. Examples : All meta directing groups are inactivator groups, `-COOH, -NO_(2), -SO_(3)H, -COCl, -COR, -COOR, -CN` ETC. Explaination : (a) RESONANCE effect : In meta directing group the `pi` electron pair moves towards groups from the rings. and ring possess less electron density and becomes positive.  (b) Inductive effect : Majority of groups attached by (-I) effect attract the electrons towards themselves and reduce the electron density in the rings. Due to both resonance and electron attracting effect of meta directing groups, there is dereases in the electron density in the ring which makes meta directing group in activator in reactivity. 
|
|
| 24. |
What is following reaction is indicated ? (i) X_((g)) to X_((g))^(+) + e^(-) (ii) X_((g))^(+) to X_((g))^(+) + e^(-) |
| Answer» Solution :Both reactions are indicated first and SECOND ionisation ENTHALPY RESPECTIVELY. | |
| 25. |
What volume of 4M HCl and 2M HCl should be mixed to get 500 mL of 2.5 M HCl ? |
|
Answer» Solution :Let the volume of 4M HCl required to prepare 500 mL of `2.5 mL of 2.5M HCl =x ` mL THEREFORE, the required volume of `2M HCl = (500-x)` mL We know from the equation `C _(1) V _(1) + C _(2) V_(2) = C_(3) V_(3)` `(4x ) + 2 ( 500 -x) = 2.5 xx 500` ` 4x + 1000 - 2x = 1250` `2x = 1250 - 1000` `x = (250)/(2) = 125 mL` Hence, volume of 4M HCl required = 125 mL Volume of 2M HCl required `= (500 - 125) mL = 375 mL` |
|
| 26. |
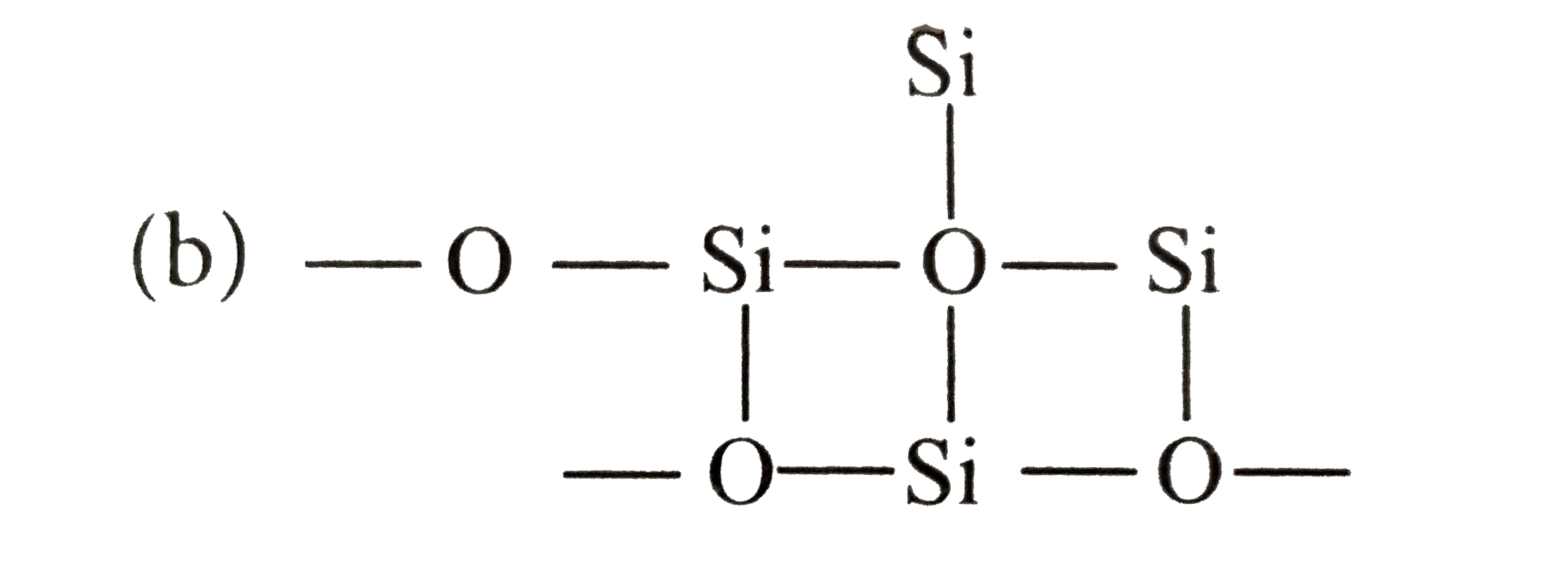
which of the following bonds is shown in silicones? |
|
Answer»
|
|
| 28. |
What is the maximum number of emission lines when the excited electron of a hydrogen atom in n = 6 drops to the ground state? |
| Answer» SOLUTION :Number of LINES produced when electron from the `n^"th"`ORBIT DROPS =n(n-1)/2=(6(6-1))/2`=15 lines | |
| 29. |
Which of the following statement(s) is (are) correct |
|
Answer» The electronic CONFIGURATION of Cris `[AR] 3d^5 4s^1` (Atomic Number of Cr =24) For magnetic quantum number (m), negative values are possible For s-subshell, l=0 , hence m=0 For p-subshell, l=1 , hence m=-1,0,+1 `._47Ag=1s^2 ,2s^2 2p^6 , 3s^2 3p^6 3d^10 , 4s^2 4p^6 4d^10 , 5s^1` Hence 23 electrons have a spin of one type and 24 of the opposite type. oxidation state of N in `NH_3` is -1/3 |
|
| 30. |
What is the weight percent of oxygen in Glucose ? |
|
Answer» SOLUTION :Weight percent of oxygen in GLUCOSE `=(6 XX 16)/(180) xx 100=53.33%` |
|
| 31. |
What makes a glass different from a soild such as quartz ?Under what conditons could quartz be converted into glass ? |
| Answer» SOLUTION :Glass is an AMORPHOUS solid in which the constituent particels `(SiO_(4)` tetrahdral) have only a short RANGE order and there is no long range order. In quartz, the constituent particles ` (SiO_(4)`tetrahdra) have short rangeas will as long range order. on melting quartz and then COOLING i repidly , it is converted into glass. | |
| 32. |
What will be the number of oxygen atoms in 1 mole of O_(2) ? |
| Answer» Solution :1 mole of `O_(2)`contains `6.022 xx 10^(23)`molecules of OXYGEN, and since the oxygen molecule is diatomic, i.e., 1 molecule contains 2 atoms, the no. of oxygen atoms in 1 mole of `O_(2)`is equal to `2 xx 6.022 xx 10^(23)` | |
| 33. |
What degree Fahrenheit will be equivalent to 293 K ? |
|
Answer» `273""^(@)F` `""^(@)F = 9/5 (""^(@)C) + 32 = 9/5(20)+32` `=36+32= 68""^(@)F` |
|
| 35. |
Two electrochemical cell Zn|Zn^(2+)||Cu^(2+)|CuFe |Fe^(2+)|Cu^(2+)|Cu are connected in series what will be the net e.m.f of the cell at 25^(@)C ? Give : Zn^(2+)|Zn=-0.76 V Cu^(2+)|Cu=+0.34 v,Fe^(2+)|Fe=-0.41V |
|
Answer» `+1.85 V` SINCE the two CELLS are connected in series therefore the NET EMF of the two cells =1.10 +0.75 =1.85 V |
|
| 36. |
Who proposed the valence bond theory ? |
|
Answer» Powling |
|
| 37. |
Which one of the following molecules will form a linear polymeric structure due to hydrogen bonding ? |
| Answer» ANSWER :D | |
| 38. |
When an ideal gas undergoes unrestrained expansion, on cooling occurs because the molecules |
|
Answer» are above the inversion temperature |
|
| 39. |
Using the standard electrode potentials given in the Table 8.2, predict if the reaction between the following is feasible: Ag(s) and Fe^(3+)(aq) |
| Answer» SOLUTION :Not FEASIBLE. | |
| 40. |
Using the standard electrode potentials given, predict if the reaction between the following is feasible: Fe^(3+)(aq) and l^(-)(aq) Fe3+/Fe2+ : +0.77 V I-/I2 : -0.54 V |
| Answer» Solution :Feasible : `2Fe^(3+)(aq)+2L^(-)(aq)to2Fe^(2+)(aq)+l_(2)(s)` | |
| 41. |
Using the standard electrode potentials given in the Table 8.2, predict if the reaction between the following is feasible: Ag^(+)(aq) and Cu(s) |
| Answer» Solution :Feasible : `CU(s)+2Ag^(+)(AQ)toCu^(2+)(aq)+2Ag(s)` | |
| 42. |
Using the standard electrode potential, find out the pair between which redox reaction is not feasible. E^(θ) values: Fe^(3+)//Fe^(2+)=0.77, I_(2)//I^(-)=+0.54, Cu^(2+)//Cu=+0.34, Ag^(+)//Ag=+0.80V |
| Answer» SOLUTION :Feasible : `Cu(s)+2FE^(3+)(AQ)toCu^(2+)(aq)+2Fe^(2+)(aq)` | |
| 43. |
Using the standard electrode potentials , predict if the reaction between the following is feasible: Br_(2)(aq) and Fe^(2+)(aq) |
| Answer» Solution :Feasible : `Br_(2)(aq)+2FE^(2+)(aq)to2Br^(-)(aq)+2Fe^(3+)(aq)` | |
| 44. |
The van der Waal's constant 'b' for water vapour is 0.03 litre mol^(-1). The radius of water vapour molecule is : |
|
Answer» `1448 CM` |
|
| 45. |
When solid NaF is treated with conc. H_(2)SO_(4) in a test tube, followed by warming, then dimeric form of a gas is evolved, by which the test tube acquires a greasy appearance on its walls. Subsequently, the gas liberated when brought in contact with moistened glass rod forms a deposit which contains: |
|
Answer» `SiF_(4)` |
|
| 46. |
Vapour density of equilibrium PCI_(5)(g)hArrPCI_(3)(g)+CI_(2)(g) is decresed by |
|
Answer» increasing temperature |
|
| 47. |
Thermite is a mixture of |
|
Answer» 3 parts of powdered AL and 1 part of `Fe_(2)O_(3)` |
|
| 49. |
The volume respectively of 10 N and 4 N HCl required to make 1 L of 7 N HCl respectively are |
|
Answer» 0.75 L of 10 N HCl and 0.25 L of 4N HCl As we KNOW that, `N_(1)V_(1)+ N_(2)V_(2) = NV` 10V + 4(1-V) = 7 xx1 10 V + 4-4V = 7, 6V = 7-4, V=3/6 = 0.50 L Volume of 10 N HCl = 0.50 L Volume of 4N HCl = 1-0.50 =0.50 L |
|
| 50. |
When BaCl_(2) is added to an aqueous salt solution, a while precipitate is obtained. The anion among CO_(3)^(2-), SO_(3)^(2-) and SO_(4)^(2-) that was present in the solubtion can be : |
|
Answer» `CO_(3)^(2-)` but not any of the other two |
|