Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The most powerful oxidising agent among the following is: |
|
Answer» `H_(2)SO_(4)` |
|
| 2. |
The most insoluble salt of sodium is |
|
Answer» `Na_(4)[Fe(CN)_(6)]` |
|
| 3. |
The most important method of prepare lower hydrocarbon is |
|
Answer» Cracking |
|
| 4. |
The most important method of preparation of hydrocarbons of lower carbon number is |
|
Answer» PYROLYSIS of HIGHER carbon NUMBER hydrocarbons |
|
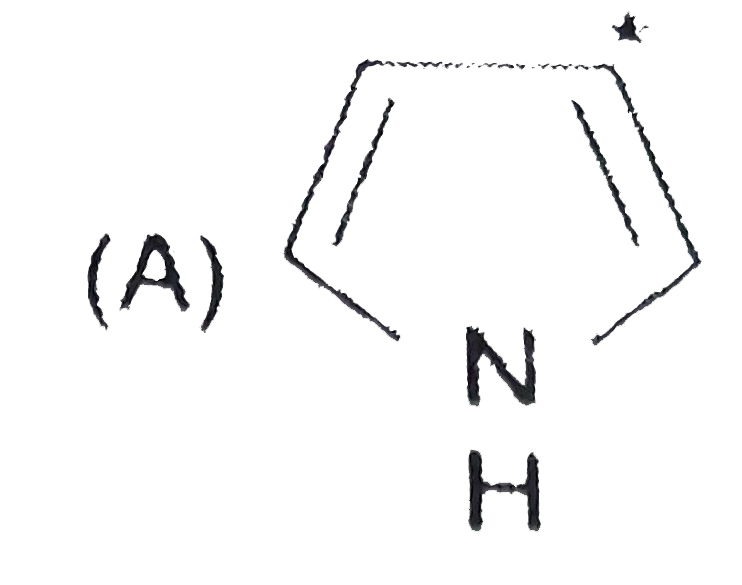
| 6. |
The most favourable position (indicated by) for an electrophilic attack is |
|
Answer»
|
|
| 7. |
The most fundamental Alkene which can show geometrical Isomerism (under proper substitutedconditions) contains ___Number of minimum carbon's |
|
Answer» 2 |
|
| 10. |
The most electronegative element possess the electronic configuration…......... |
|
Answer» `ns^(2)NP^(2)` |
|
| 11. |
The most electronegative element of the periodic table is _______ |
|
Answer» Iodine |
|
| 12. |
The most electronegative element..... |
|
Answer» oxygen |
|
| 13. |
The most electro positive element of the periodic table is __________ |
|
Answer» Gold |
|
| 14. |
The most easily hydrolysed molecule under SN_(1) condition is |
|
Answer» ALLYL CHLORIDE |
|
| 15. |
The most easily hydrolysed molecule under S_(N^2) reaction is …………………. . |
| Answer» SOLUTION :TERT. BUTYL CHLORIDE | |
| 16. |
The most easily hydrolysed molecule under S_(N^1)condition is ……………………. . |
|
Answer» ALLYL CHLORIDE |
|
| 17. |
The most commonly used reducing agent is …….. |
|
Answer» `AlCl_3` e.g., `SnCl_2 + 2FeCl_3 to SnCl_4 + 2FeCl_2` `SnCl_2+ 2FeCl_3 to 2FeCl_2+ SnCl_4` `SnCl_2 +2CuCl_2 to 2CuCl_2 + SnCl_4` |
|
| 18. |
Why do alkenes prefer to undergo electrophilec addition reaction while arenes prefer electrophilic substitution reactions ? Explain. |
|
Answer» |
|
| 19. |
The most common oxidation state of an element is -2. The number of electrons present in its outermost shell is |
|
Answer» 2 |
|
| 20. |
The most common oxidation state of an element is -2 The number of electrons present in its outermost shell is |
|
Answer» 2 `O(Z= 8)1s^(2) 2S^(2)2p^(4)` . VALENCY electrons 6 |
|
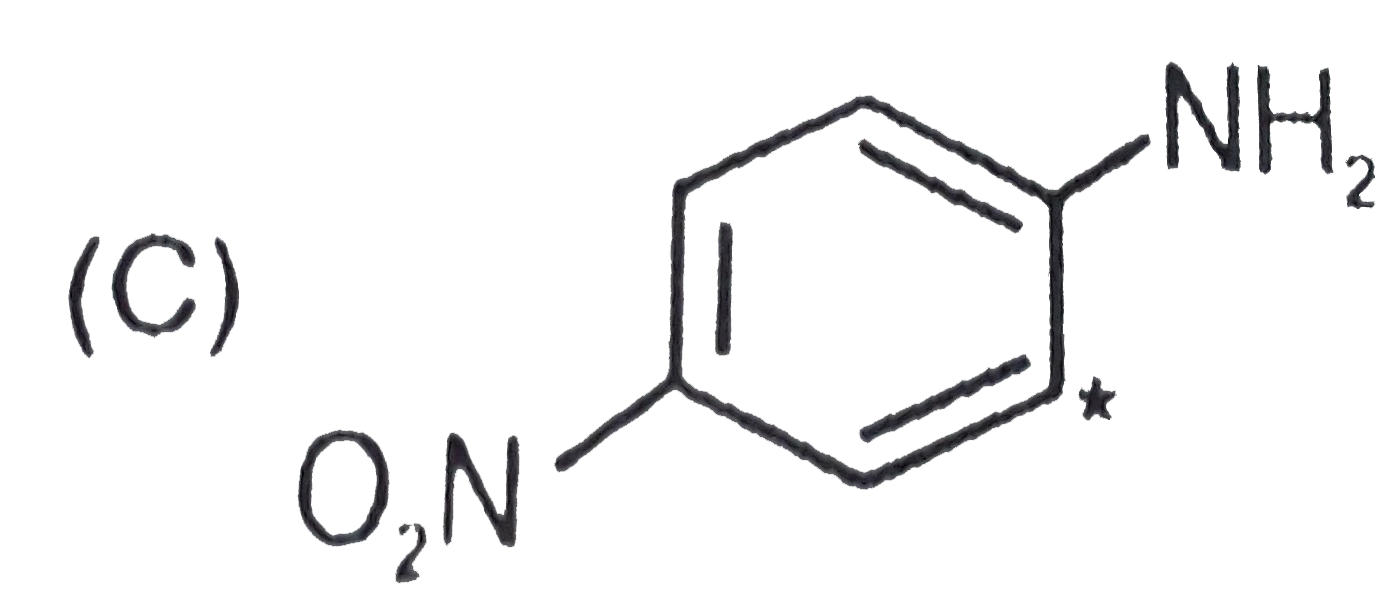
| 22. |
The most basic one is |
|
Answer»
|
|
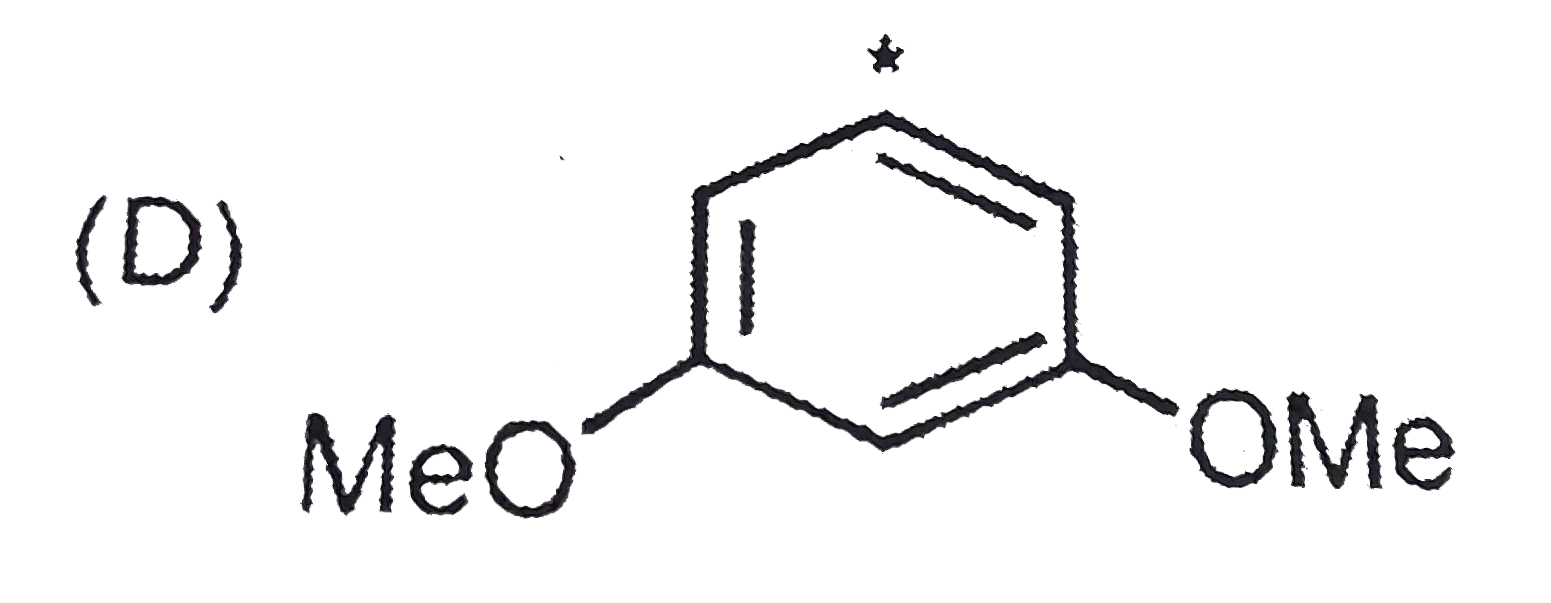
| 24. |
The most basic one is : |
|
Answer»
|
|
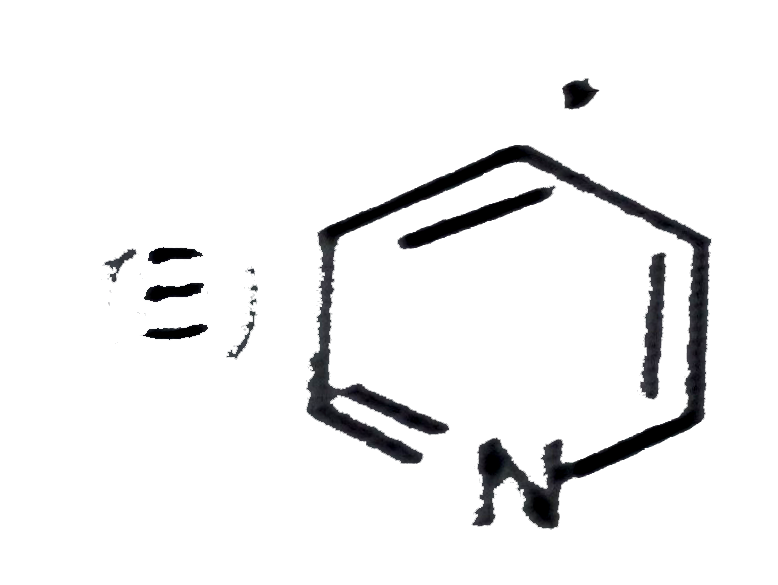
| 25. |
The most basic nitrogen in the following compound is |
|
Answer» I |
|
| 26. |
The most appropriate reagent for converting pentan-2-one to butanoic acid is |
|
Answer» Sodium HYPOCHLORITE followed by acidic hydrolysis |
|
| 27. |
The most adsorbed gas on activated charcoal is |
|
Answer» `N_(2)` |
|
| 28. |
The most acidic hydrogen atoms are present in |
|
Answer» Ethane |
|
| 29. |
The most abundant metal on the surface of the earth is |
|
Answer» Fe |
|
| 31. |
The most abundant elements by mass in the body of a healthy human adult are : Oxygen (61.4 %): Carbon (22.9 %): Hydrogen (10.0 %) and Nitrogen (2.6 %). The weight which a 75 g person would gain if all ""^(1)H atoms are replaced by ""^(2)H atoms is ..... |
|
Answer» 15 KG |
|
| 34. |
The most abundant and least abundant isotopes of hydrogen respectivley are |
|
Answer» <P>P,T |
|
| 35. |
The most abundant amounts by mass in the body of a healthy human adult are oxygen (61.4%), carbon (22.96%) hydrogen (10.0%) and nitrogen (2.6%). The weight which a 75 kg person would gain if all ""^(1)Hatoms are replaced by ""^(2)Hatoms is |
| Answer» ANSWER :C | |
| 36. |
The more positive the value of E^(@) the greater is the tendency of the species to get reduced using the standard electrode potential of redox couples given below find out which of the following is the strongest oxidising agent E^(@) "values" : (F^(3+))/(Fe^(2+))=+0.77, I_(2)(s)//I^(-)=+0.54,(Cu^(2+))/(Cu^(2+))=+0.34, (Ag^(+))/(Ag(s))=+0.80,Cu^(2+)//Cu(s)=+0.34 (I_(s))/(I)^(-)=+0.54 |
| Answer» Solution :SINCE `E^(@)` of the redox couple `Ag^(+)//Ag` is the most positive i.e 0.80 V therefore `Ag^(+)` is the STRONGEST OXIDISING agent | |
| 37. |
The more positive the value ofE^(Ө), the the greater is the tendency of the species to get reduced. Using the standard electrode potential of redox couples given below find out which of the following is the strongest oxidising agent ? E_(Fe^(+3)//Fe^(+2))^(Ө)=+0.77"volt",E_(I^(2)//I^(-))^(Ө)=+0.54"volt" E_(Cu^(+2)//Cu)^(Ө)=+0.34"volt",E_(Ag^(+)//Ag)^(Ө)=+0.80"volt" |
|
Answer» `FE^(+3)` |
|
| 38. |
The most abundant alkali metal in the earth's crust is |
|
Answer» Lithium |
|
| 41. |
The monomers used in the manufacture of nylon-6,6 are |
|
Answer» sebacic ACID and hexamethylene DIAMINE |
|
| 43. |
The molecules that can exhibit tautomerism are |
|
Answer» I, IV |
|
| 44. |
The molecules that will have dipole moment |
|
Answer» 2,2-dimethylpropane |
|
| 45. |
The molecules of PCl_(5) in the solied state is ………..in nature consisting of tetrahedral…………cation and octahedral……………anion. |
|
Answer» |
|
| 46. |
The molecules or ions in which the central atom has only bond pairs of electrons are |
|
Answer» `NH_(4)^(+)` |
|
| 47. |
The molecules having same hybridisation, shape and number of lone pairs of electrons are. . . . . . . . . |
|
Answer» `SeF_(4),XeO_(2)F_(2)` |
|
| 48. |
The molecules having same hybridisation , shape and number of lone pairs of electons are |
|
Answer» `SeF_(4), XeO_(2) F_(2)` |
|
| 49. |
The molecules having dipole moment are _______ |
|
Answer» 2,2-Dimethylpropane 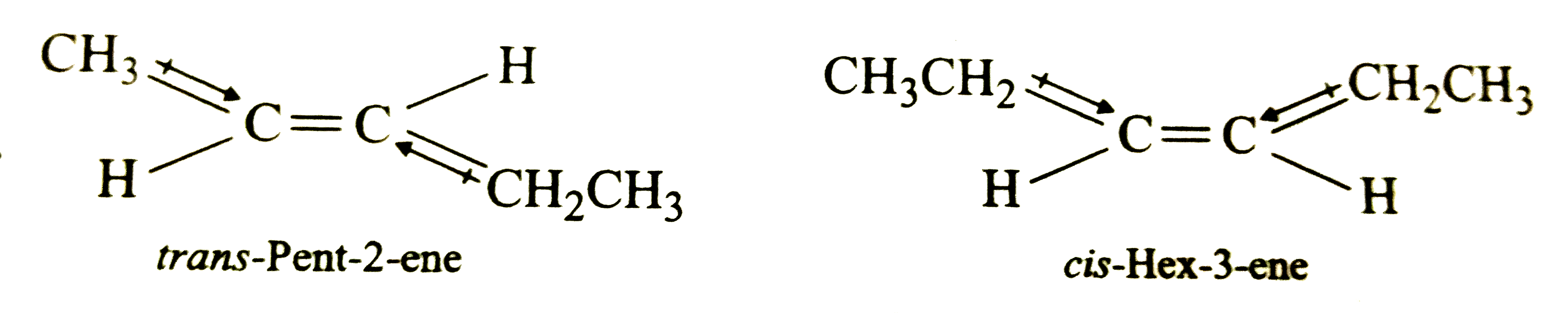 Since the +I-effect of `CH_3CH_2` group is higher than that of `CH_3` group, therefore , the dipole moments of `C-CH_3` and `C-CH_2CH_3` BONDS are unequal . Although these two dipoles oppose each other, YET they do not exactly cancel out each other and HENCE trans-2-pentene has small but finite dipole moment.In cis-hex-3-ene , although the dipole moment of the two `C-CH_2CH_3` bonds are equal , they are inclined to each other at angle of `60^@` and hence have a finite resultant. Further, 2,2-dimethylpropane and 2,2,3,3-tetramethylbutane have zero dipole moments because of the following two reasons : (i)C-C and C-H bonds in alkanes are almost non-polar and have no FIXED orientations in SPACE , therefore, their resultant dipole moment, if any , are almost negligible . (ii)Both these are symmetricalmolecules and hence their dipole moments is zero. |
|
| 50. |
The molecules having dipole moment are ……. |
|
Answer» 2,2-Dimethylpropane |
|