Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The molecules BF_3 and NF_3are covalent compounds. But BF_3is non-polar and NF_3is polar. The reason is |
|
Answer» Boron is a metal and nitrogen is a gas in uncombined state |
|
| 2. |
The molecule with maximum number of lone pairs on central atom is |
|
Answer» `XeO_3` |
|
| 3. |
The molecule which contains sigma_(sp^3-sp^3) and sigma_(sp^3-p) bonds in it is |
|
Answer» `CH_3 CH_3` |
|
| 4. |
The molecule orbital shown in the diagram can be described as |
| Answer» ANSWER :D | |
| 5. |
The molecule in which the distance between the two adjacent carbon atom is largest in….. |
|
Answer» ethane |
|
| 7. |
The molecule having smallest bond angle is |
|
Answer» `AsCl_(3)` As we move down the group the size of atom increases and as size of central atom increases, lone pair-bond pair repulsion also increases. Thus bond angle decreases. Increasing order of ATOMIC radius : `N lt P lt As lt Sb ` Decreasing order of bond angle : `NCl_(3) GT PCl_(3) gt AsCl_(3) gt SbCl_(3)` |
|
| 8. |
The molecule havaing smallest bond angle is |
|
Answer» `AsCI_(3)` |
|
| 9. |
The molecule has...........structure |
|
Answer» |
|
| 10. |
The molecule H_(3)C-CH=CH-underset(CH_(3))underset(|)CH-COOH can exhibit |
|
Answer» Geomelical isomeronly |
|
| 11. |
The molecule H_3C - CH = CH - underetoverset(|)(CH_3)(C )H - COOHcan exhibit |
|
Answer» GEOMETRICAL ISOMERISM only |
|
| 12. |
The molecular weights of two ideal gases A and B are respectively 100 and 200. One gram of A occupies V litres of volume at STP. What is the volume (in litres) occupied by one gram of B at STP ? |
|
Answer» V/2 |
|
| 13. |
The molecular weight of low density polythene ranges between .......and..... |
| Answer» SOLUTION :50,000, 3,00,000 | |
| 14. |
The molecular weight of haemoglobin is about 65.000 g/mol. Haemoglobin contains 0.35% Fe by mass. How many iron atoms are there in a haemoglobin molecule? |
|
Answer» Solution :100 g of haemoglobin contain 0.35 g of IRON. or, `100/65000` mole of haemoglobin CONTAINS `0.35/56` mole of Fe. `THEREFORE 1` mole of haemoglobin contains `0.35/56 xx 65000/100` MOLES of Fe. =4.06 moles of Fe Thus, one molecules of haemoglobin contains four iron atoms. |
|
| 15. |
The molecular weight of benzoic acid in benzene as determined by depression in freezing point method correcponds to |
|
Answer» IONIZATION of benzoic ACID 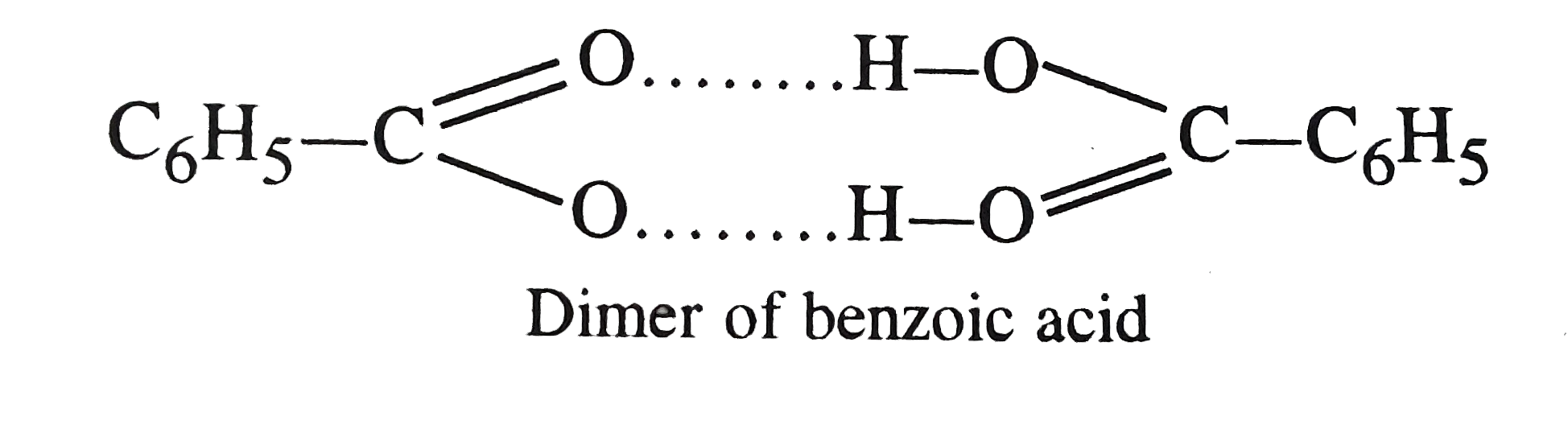
|
|
| 16. |
The molecular weight of an organic compound is 180. Its empirical formula is CH_(2)O. Its molecular formula is |
|
Answer» `CP_(6)H_(12)O_(6)` |
|
| 17. |
The Molecular weight of a gas is 40. At 400K if 120 g of this gas has a volume of 20 litres, the pressure of the gas in atm is |
|
Answer» `4.92` |
|
| 18. |
The molecular velocity of any gas is |
|
Answer» inversely PROPORTIONAL to the SQUARE ROOT of temperature |
|
| 19. |
The molecular speeds of gasesous molecules are analogous to those of rifle bullets. Then why is the odour of a gas not detected so fast ? |
| Answer» Solution :Though the molecules of a gas travel at HIGH speeds but they do not travel in straight lines in one direction like bullets. As they travel, they collide with the molecules of the gases PRESENT in the air. As a result, they are deflected. Thus, they follow a zig-zag PATH, i.e., the net distance travelled in a PARTICULAR direction is quite small in a given TIME. That is why the odour is not detected so fast. | |
| 20. |
The molecular orbital electronic configuration is (sigma_(1s))^(2),(sigma_(1s)^(**))^(1). It corresponding to |
| Answer» Answer :D | |
| 21. |
The molecular orbital shown below is described as |
|
Answer» `sigma^(**)` |
|
| 22. |
The molecular mass of Mohr's salt, FeSO_(4)(NH_(4))_(2)SO_(4). 6H_(2)O, is 392. Its equivalent mass is : |
|
Answer» 196 |
|
| 23. |
The molecular mass of K_(2)Cr_(2)O_(7) is 294 amu. It acts as oxidising agent in a redox titration. Its equivalent mass in acid medium will be : |
|
Answer» 294 Change in oxidation number = 6 EQUIVALENT mass `=("Molecular mass")/(6)=(294)/(6)=49` |
|
| 24. |
The molecular mass of H_(3)PO_(3) is 82. Its equivalent mass, if it is completely neutralised, is: |
|
Answer» 82 |
|
| 25. |
The molecular mass of KMnO_(4) is M.Its equivalent mass in acidic medium will be : |
|
Answer» M |
|
| 26. |
The molecular mass of hydrogen peroxide is 34. What is the unit of molecular mass ? |
|
Answer» mole |
|
| 27. |
The molecular mass of benzene is 78 amu. Its percentage composition is 92.31 % C and 7.69 % H. Determine the molecular formula of benzene. |
|
Answer» `{:("Element","Percentage","Atomic mass","Gram ATOMS (Moles)","Atomic ratio (Molar ratio)","Simplest WHOLE no. ratio"),("C",92.31,12,(93.31)/(12)=7.69,(7.69)/(7.69)=1,1),("H",7.69,1,(7.69)/(1)=7.69,(7.69)/(7.69)=1,1):}` Empirical formula of benzene = CH Step II. Molecular formula of benzene Empirical formula mass `= 12 +1 =13u` Molecular mass `= 78 u` `N=("Molecular mass")/("Empirical formula mass")=((78u))/((13u))=6` Molecular formula `= n xx` Empirical formula `=6xx CH = C_(6)H_(6)`. |
|
| 28. |
The molecular mass of an organic acid was determined by the study of its barium salt. 4.290g of salt was quantitiatively converted to free acid by the reaction with 21.64ml of 0.477 MH_(2)SO_(4). The barium salt was found to have two moles of water of hydration per Ba^(+2) ion and the acid is mono basic. what is molecular weight of anhydrous acid. |
|
Answer» `137.32` Molecular weight of salt `=415.7` MW of ANHYDROUS salt `=379.7` Molecular weight of anion `=397.7 - 137.3 = 242.4` Molecular weight of acid `= 244.4` |
|
| 29. |
The molecular mass of an organic acid was determined by the study of its barium salt. 4.290 g of salt was quantitatively converted to free acid by the reaction with 21.64 mL of 0.477 M H_(2)SO_(4). The barium salt was found to have two mole of water of hydration per Ba^(2+) ion and the acid is mono-basic. What is molecular weight of anhydrous acid? |
|
Answer» |
|
| 30. |
The molecular mass of an organic acid was determind by the study of its barium salt. 4.290 g of salt was converted to free acid by the reaction with 21.64 mL of water of hydration per Ba^(2+) ion and the acid is monobasic. What is molecular weight of anhydrous acidgt |
|
Answer» Solution :MEQ of Braium SALT = mEq of acid `(4.290)/(Mw..2) XX 1000 = 21.64 xx 0.477 xx 2` MOLECULAR weight of salf = 415.60 Molecular weight of anion `= (("Mw of salt" - Mw of BA^(2+) - Mw of 2 H_(2) O))/(2)` `= (415.60 - 137 - 36)/(2) = 121.16` `:.` Molecular weight of acid `= 121.6 + 1 = 122.6` |
|
| 31. |
The molecular interaction responsible for hydrogen bonding in HF. |
|
Answer» ion-induced DIPOLE |
|
| 32. |
The molecular formular of a commerical resin used for exchanging ions in water softening is C_(8)H_(7)SO_(3)Na(Mol . Wt. of 206). What would be the maximum uptake of Ca^(2+) ions by the resin when expressed in mole per gram resin ? |
|
Answer» `2//3009` `(C_(8)H_(7)SO_(3)^(-))_(2)Ca^(2+)+Na^(+)` `therefore ` 1 mole of `Ca^(2+)=412` g of resin or maximum uptake of `Ca^(2+)` ions by the resin `=(1 mol)/(412g)=(1)/(412) ` mol/g |
|
| 33. |
The molecular formula of the compound formed from B and C will be : |
|
Answer» BC |
|
| 34. |
The molecular formula of Napthacene is …….. |
|
Answer» `C_(18)H_(10)` |
|
| 35. |
The molecular formula of Iron (III) Sulphate is ____ . |
|
Answer» `Fe_(3)(SO_(4))_(2)` They can FORM the neutral compound `Fe_(2)(SO_(4))_(3)` |
|
| 36. |
The molecular formula of inorganic benzene is "………….." and is obtained by heating the adduct of "………….." with "…………..". |
|
Answer» |
|
| 37. |
The molecular formula of an organic compound is C_(4)H_(9)N. The volume of N_(2) that will be given by 0.2 g of the above compound at STP is …….. (ml) |
|
Answer» 31.5 71 grams LIBERATE 11,200 ml `N_(2)` 0.2 gram liberate …….. ? …….. `N_(2)` |
|
| 38. |
The molecular formula of an organic compound is C_(4)H_(9)N. The volume of N_(2) that will be given by 0.2g of the above compound at STP is…(ml) |
|
Answer» 31.5 |
|
| 39. |
The molecular formula of a commercial resin used for exchanging ions in wter softening is C_(8)H_(7)SO_(3Na (mol.wt.206), What would be the maximum uptake of Ca^(2+) ions by resin when expressed in moe per gram resin? |
|
Answer» `(2)/(309)` `"1 mole of" Ca^(2+)=412"g of RESIN"` `"Maximum uptake of" Ca^(2+) "ions by the resin" =(1"mol")/(412g)` |
|
| 40. |
The molecular fo rmula of the compound formed from 8 and C will be |
|
Answer» BC In the electronic CONFIGURATION SHOW that B represents PHOSPHORUS and C represents chlorine. The stable compound formed is `PCl_(3)`i.e., `BC_(3)` |
|
| 41. |
The molecule electronic configuration of B_(2) is |
|
Answer» `KK ( SIGMA 2s)^2 ( sigma^(**)2s)^2 ( PI 2p)_(x)^(1) ( PIP)_(y)^(1)` |
|
| 42. |
The mole ratio of products H_(2), NaOH, CI_(2) formed in a Nelson cell method is |
|
Answer» `1:2:1` |
|
| 43. |
What is the mole percentage of O_(2) in a mixture of 7g of N_(2) and 8g of O_(2)? |
|
Answer» 25% |
|
| 44. |
The mole fraction of water in a solution of HCI is 0.78. Calculate the molality of the solution. |
|
Answer» SOLUTION :Mole fraction of WATER = 0.78 Mole fraction of HCI = `1 - 0.78 = 0.22`. SUPPOSE, the solution contains `n_(1)`moles of HCI and `n_2` moles of water. Therefore, `n_(1)/(n_(1) + n_(2)) = 0.22`……..(i) and `n_(2)/(n_(1) + n_(2)) = 0.78`……...........(ii) DIVIDING eq. (i) by eq. (ii), we have `n_(1)/n_(2) = 0.22/0.78 = 0.28`.............(iii) Since, molality refers to the number of moles of solute dissolved in 1000 g of solvent (water in the present case), `n_(2) = 1000/18 = 55.55` Substituting the value in eq. (iii), we have `n_(1)/55.55 =0.28` or `n_(1) = 0.28 xx 55.55 = 15.55` Hence, the molality of the given solution is 15.55 m. |
|
| 45. |
The mole fraction of solute in an aqueous solution is (1)/(112). The molality (m) of the solution is 0.1x. What is x? |
|
Answer» |
|
| 46. |
The mole fraction of benzene in a solution in toluene is 0.40. Calculate the weight per cent of benzene in the solution. |
|
Answer» |
|
| 47. |
The mole concept helps in handling a large number of atoms and molecules in stoichiometric calculations. Calculate the amount of CO_2 formed by the complete combustion of 80 g of methane as per the reaction CH_(4(g))+2O_(2(g))rarrCO_(2(g))2H_2O_((g)) (Atomic masses: C = 12.01u, H = 1.008u, O=16u).. |
| Answer» SOLUTION :`CH_(4g)+2O_2toCO_2+2H_2O16g` of `CH_4` GIVEN 44 G of `CO_2` | |
| 48. |
What is the number of hydrogen atoms in one mole of methane (CH_4)? |
| Answer» Solution :Number of HYDROGEN ATOMS =No. of mole `xxN_Axxn=4N_A=1xx6.023xx10^23xx10^24xx4=24.092xx10^23"atoms"=2.409xx10^24`ATOM | |
| 49. |
Define 1 mole |
| Answer» Solution :1 MOLE is defined as the AMOUNT of any substance which contains one Avogadro NUMBER of particles. (i.e” atoms, ions or molecules) | |
| 50. |
The molarity of the solution containing 2.8%(mass/volume) solution of KOH is : (Given atomic mass of K = 39) |
|
Answer» `0.1` M |
|