Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The reaction CO(g) +3H_(2)(g) hArr CH_(4)(g)+H_(2)O(g) is at equilibrium at 1300 K in a 1K flask. It also contain 0.30 mol of CO, 0.10 mol of H_2 and 0.02 mol of H_2O and unknown amount of CH_(4) in the flask. Determine the cocentration of CH_(4) in the mixture . The equilbrium constant, K_C for the reaction at the given temperature is 3.90. |
|
Answer» Solution :`CO(G)+ 3H_(2)(g) HARR CH_(4) (g) +H_(2)O(g)` `K_(c)=([CH_4]xx[H_2O])/([CO] xx [H_2]^3)or 3.90 = ([CH_4]xx[0.02])/([0.30] xx[0.1]^3)` `[CH_4] = ((3.9)xx(0.30) xx (0.001))/((0.0.2 ))=5.85 xx 10^(-2) M` |
|
| 2. |
The reaction Cl_(2(g))+2OH_((aq))^(-)toClO_((aq))^(-)+Cl_((aq))^(-)+H_(2)O_((l)) represents the process of bleaching. Identify and name the species that bleaches the substances due to its oxidising action. |
|
Answer» Solution :`overset(0)(Cl_(2(g)))+overset(-2)(2O)overset(+1)(H_((AQ))^(-))tooverset(+1)(ClO_((aq))^(-))+overset(-1)(Cl_((aq))^(-))+H_(2)O_((l))` In the given reaction oxidation number of Cl is 0 to 1. and 0 to -1. So Cl is act as oxidising agent and reducing agnet. So this reaction is disproportionation reaction. So, `CLO^(-)` is dissociate. |
|
| 3. |
The reaction CI_(2)(g)+2OH^(-)(aq)rarrCIO^(-)(aq)+CI^(-)(aq)+H_(2)O(l) represents the process of bleaching identify and name the species that bleaches the substaces to its oxidisng action |
|
Answer» SOLUTION :Writing the O.N of each element above its symbol we have `CI_(2)(g)+2OH^(-)(AQ)rarrCIO^(-)+CI^(-)(q)+H_(2)O^(-)(L)` in this reaction O.N of CI increase from 0 in `CI_(2)` to + 1 in `CIO^(-)` therefore is both oxidised to `CIO^(-)`and reduced to `CI^(-)` since `CI^(-)` ion cannot ACT as an oxdising agent therefore `CI_(2)` bleaches substances dueto to oxidising ACTION of `CIO^(-)` ion |
|
| 4. |
The reaction CI_2(g) + 2OH^(-) (aq) rarrCIO^(-) (aq)+ CI^(-) (aq) + H_2O(l)represents the process of bleaching . Identify and name the species that bleaches . the substances due to oxidising action . |
|
Answer» SOLUTION :`overset(0)(Cl_2(G))+overset(-2+1)(2OH^(-))(aq)rarroverset(+1-2)("Cl O"^(-))(aq)+overset(-1)(Cl^(-))(aq)+overset(+1-2)(H_2O)(l)` In this reaction , O.N.of CI increases from O (in `Cl_2` ) to + 2 (in `CLO^(-)`) and decreases to - 1 (in `Cl^(-)`)Therefore , `CI^(-)`is both oxidised to `CIO^(-)`and reduced to CI. SINCE `CI^(-)` ION cannot act an oxidising agent ( because it cannot decrease its O.N. lower than - 1 ), therefore , `CI^-`bleaches substances due to oxidising action of hypochlorite, `CIO^(-)`,ion. |
|
| 5. |
The reaction CH_(3)COOC_(2)H_(5) + NaOH rarr CH_(3)COONa + C_(2)H_(5)OH is known as |
|
Answer» Esterification |
|
| 6. |
The reaction : CH_(3)CH_(2)I + KOH_((aq)) rarr CH_(3)CH_(2)OH + KI is classified as……… |
|
Answer» ELECTROPHILIC SUBSTITUTION |
|
| 7. |
The reaction: CH_(3)CH_(2)I + KOH (aq) rarr CH_(3)CH_(2)OH + KI is classified as : (a) electrophilic substitution (b) nucleophilic substitution (c) elimination (d) addition. |
| Answer» SOLUTION :This is an example of nucleophilic substitution REACTION since the nucleophile `I^(-)` is replaced by the nucleophile `OH^(-)` ion. THUS, option (b) is correct. | |
| 8. |
The reaction CH_(3)CH = CH_(2) underset(H^(+))overset(CO + H_(2)O)rarr {:(CH_(3) - CH - CH_(3)),(""|),(""COOH):} is known as |
|
Answer» WURTZ REACTION |
|
| 9. |
The reaction, (CH_(3))_(3)C-Br overset(H_(2)O)rarr (CH_(3))_(3)C-OH is |
|
Answer» ELIMINATION REACTION |
|
| 10. |
The reaction CH_(3)-CH_(2)-CH_(2)-CH_3 overset(HCl " Gas")underset(AlCl_3)rarr CH_(3) - underset(CH_(3))underset(|)(CH)-CH_(3) , is an example of |
|
Answer» ISOMERISATION |
|
| 11. |
The reaction C_(8)H_(18) rarr H_(4)H_(10) +C_2H_(4) +CH_(4)+Crepresents |
|
Answer» SYNTHESIS |
|
| 12. |
The reaction Br_(2),Cl_(2), I_(2), P_(4) with NaOH involves |
|
Answer» DECOMPOSITION |
|
| 13. |
The reaction between sodium and water can be made less vigorous by |
|
Answer» Lowering the temperature |
|
| 14. |
The reaction between phenols and benzene-diazonium chloride in slightly alkaline medium at 273-278 K to give azo dyes is called |
|
Answer» Reimer-Teimann REACTION 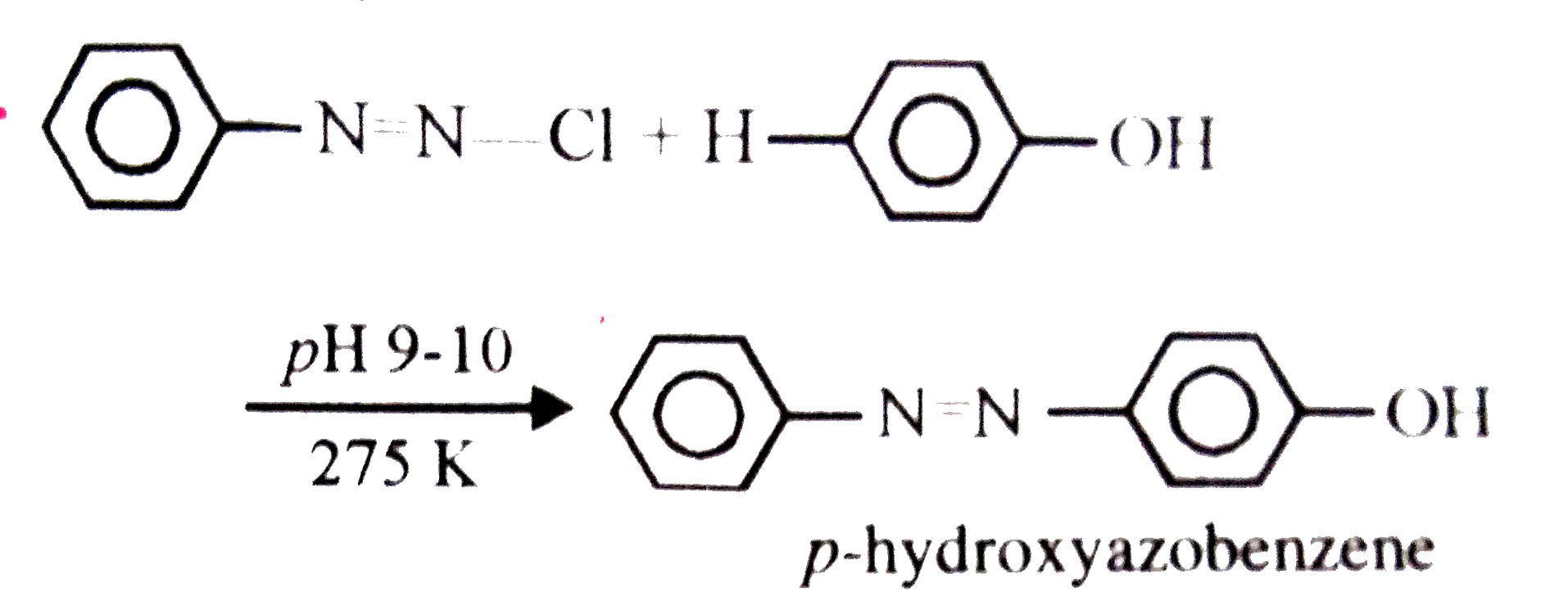 This reaction is CALLED coupling reaction. |
|
| 15. |
The reaction between NH^(-)andN_(2)O gives |
|
Answer» NO |
|
| 16. |
From the given reaction 2KMnO_(4) + 3H_(2)SO_(4) + 5H_(2)O_(2) rarr K_(2)SO_(4) + 2MnSO_(4) + 8H_(2)O + 5O_(2) Find the normality of H_(2)O_(2) solution, if 20 mL of it is required to react completely with 16 mL of 0.02M KMnO_(4) solution (Molar mass of KMnO_(4) = 158 g mol^(-1)) |
|
Answer» `0.025`M |
|
| 17. |
The reaction between H_2O_2 and KMnO_4is 2KMnO_4 + 3H_2SO_4 + 5H_2O_2to K_2SO_4 + 2MnSO_4 + 8H_2O + 5O_2.In a reaction excess of H_2O_2is added to 0.1 mole of acidified KMnO_4solution. Then the volume of 02 gas liberated at STP is |
|
Answer» 5.6lit |
|
| 18. |
The reaction between gaseous NH_3 and HBr produces a white solid NH_4Br. Suppose that NH_3 and HBr are introduced simultaneously into the opposite ends of an open tube of 1 metre length. Where would you expect the white solid to form?s |
|
Answer» At a DISTANCE of 34.45 cm from `NH_3` END |
|
| 19. |
The reaction between gaseous NH_3 and HBr produces a white solid NH_4Br. Suppose that NH_3 and HBr are introduced simultaneously into the opposite ends of an open tube of 1 metre length. Where would you expect the white solid to form? |
|
Answer» At a DISTANCE of 34.45 cm from `NH_3` END |
|
| 20. |
The reaction between diethyl cadminum and acetyl chloride gives |
|
Answer» DIMETHYL kentone |
|
| 21. |
The reaction between benzene and ICI in presence of anhyd. AICI_3 yields _____ |
|
Answer» |
|
| 22. |
The reaction between ammonia and boron trifluoride is :: NH_(3) + BF_(3) rarr H_(3)N : BF_(3) Identify the acid and base in this reaction. Which theory explains it ? What is the hybridisation of B and N in the reactants ? |
| Answer» Solution ::`NH_(3)` is Lewis base and `BF_(3)` is Lewis acid. Lewis electronic THEORY of acids of acids and bases can EXPLAIN it. Boron in `BF_(3)` is `SP^(2)` hybridized whereas N in `NH_(3)` is `sp^(3)` hybridised (Refer to chapter on Chemical Bonding ). | |
| 23. |
The reaction between alcohol and carboxylic acids is called |
|
Answer» Esterification |
|
| 24. |
The reaction between 4-chlorobutanoic acid with an alkali yields |
|
Answer» 2-Butenoic acid, `CH_(3)CH = CH - CO_(2)H`  In 4-chlorobutanoic acid, `Cl` and `COOH` groups are apart and HENCE each group REACTS IMMEDIATELY |
|
| 25. |
The reaction A_((s)) harr B_((s))+C_((g)) attains equilibrium in closed container in such a way that vapour pressure of C_((g)) is equal to 1 atm. The incorrect relationstipal at equilibrium is |
|
Answer» `DeltaG^(@)=0` `Delta G=Delta G^(@)+RT lnk=0` |
|
| 26. |
The reaction and condition which obey bimolecular nucleophilic substitution reaction with respect to tetrahedral intermediate is - |
|
Answer» <P>(IV) (i) P |
|
| 27. |
The reaction An+CuSO_4toZnSO_4 goes completely to the right. In an experiment, 10.0 g of Zn was added to 200 " mL of " CuSO_4 solution. After all the Cu was precipitated, it was found that not all the Zn had dissolved. After filtration, the total weight of the solid at the end of the reaction was 9.810 g. Calculate. (a). The weight of copper deposited and, (b). Molarity of CuSO_4 in the original solution. |
|
Answer» Solution :LET the WEIGHT of COPPER deposited be x g . Weight of usused `Zn=9.81-x g` and the weight of Zn USED`=10-(9.81-x)=0.19+x g` `Zn+CuSO_4toZnSO_4+Cu` `therefore(0.19+x)g of Zn=(63.5(0.19+x))/(65.4)g of Cu` `therefore(63.5(0.19+x))/(65.4)=x` `x=0.185+0.97x=6.37g` `Mkk=(W_2xx100)/(Mw_2xx"volume of solution in ML")` `=(6.37xx1000)/(6.35xx200)=0.501` |
|
| 28. |
The reaction A to B is impossible, if.….... . |
|
Answer» `Delta H` positive, `Delta S` positive but `Delta H LT T Delta S` |
|
| 29. |
The reactionA+B Leftrightarrow C has the value of DeltaH and value of DeltaS respectively -12.52kJ and 313Jmol^(-1)K^(-1).AtWhat temperature the reaction attains equilibrium state. |
| Answer» SOLUTION :`127^(@)C` | |
| 30. |
The reaction, 3CIO_((aq))^(-)toCIO_(3(aq))^(-)+2CI_((aq))^(-) is an example of _____ |
|
Answer» OXIDATION reaction |
|
| 31. |
The reaction 2NO_((2)) g hArr N_2O_4(g) DeltaH= negative occurring in a closed vessel attains equilibrium. If this vessel is kept in ice then which of the following change will be observed ? |
|
Answer» Equilibrium state will remain constant. For this reaction `DeltaH=(-) ve` `therefore` This is a ENDOTHERMIC reaction. When the vessel is kept in ice, its temperature decreases. So, according to Le-chatelier.s principal to minimise the effect of decrease temp. The temp. should be increase and hence the reaction take place in forward direction and HEAT is release and new equilibrium is established. When the new equilibrium is established then, (1) The amount of product `N_2O_4` will increases (II) The amount of reactant `NO_2` (brown) will decreases. i.e. Intensity of brown colour decreases. |
|
| 32. |
The reactant which is entirely consumend in reaction is known as limiting reagent. In the reaction2 A + 4 B to 3C + 4D , when 5 moles of A react with 6moles of B , then(i)Which is the limiting reagent(ii)Calculate the amount of Cformed. |
|
Answer» According to the above equation, 2 mols of 'A' require 4 mols of 'B' for the reaction. Hence, for 5 mols of 'A' , the MOLES of 'B' required ` 5"MOL of " A xx (4 " mol of "B)/(2 "mol of "A)` = 10 mol B Butwe haveonly 6 mols of 'B' hence ,'B' is the limiting reagent . So amount of 'C' formedis detemined by amount of 'B'. Since 4 mols of 'B' GIVE 3 mols of 'C' . Hence 6 mols of 'B'will give ` 6 "mol of" B xx (3 "mol of"C)/(4 "mol of "B)= 4 . 5 ` mol of C . |
|
| 33. |
The reactant which is entirely consumed in reaction is known as limiting reagent, in the reaction 2 A + 4 B rarr 3 C + 4D, when 5 moles of A react with 6 moles of B, then (i) which is the limiting reagent ? (ii) calculate the amount of C formed ? 2A + 4 B rarr 3 C + 4D |
|
Answer» Solution :ACCORDING to the GIVEN reaction, 2 moles of A react with 4 moles of B hence, 5 moles of A will react with 10 moles of B (`(5xx4)/(2)=10` moles) (i) In the first case reactant B is limiting reagent as it will CONSUME first in reaction because we have only 6 moles of B. (ii) Limiting reagent decide the amount of product produced. According to the reaction, 4 moles of B produces 3 moles of C `:.` 6 moles of B willproduce `=(3xx6)/(4) = 4.5` moles of C. Note : Limiting reagent limits the amount of product formed because it is present in lesser amount and gets consumed first. |
|
| 34. |
The raw materials used in ammonia-soda process are |
|
Answer» Brine, `NH_(3), CO_(2)` |
|
| 35. |
The raw material of Raschig process is |
|
Answer» chlorobenzene  
|
|
| 36. |
The raw material for Rasching process is................. |
| Answer» Solution :benzene | |
| 37. |
The raw material for Rasching process is ......... |
|
Answer» CHLOROBENZENE |
|
| 38. |
The raw material for Rasching process is …………………… . |
|
Answer» CHLOROBENZENE |
|
| 39. |
The radius and charge of each of six ions are shown in the table below. The ionic solids JX, LY and MZ arc of the same Lattice type. What is the correct order of Lattice energies? |
|
Answer» JX =LY=MA |
|
| 40. |
The raw material for Rasching process |
| Answer» Solution :benzene | |
| 41. |
The ration of pure orbitals to hybridized orbitals in ethylene is |
|
Answer» `2:3` |
|
| 42. |
The ratio ofC_(p) value of a triatomicgas to the C_(v) value of a monoatomic gas is |
|
Answer» |
|
| 43. |
The ratio rate of diffusion of gases A and B is 1:4 If the ratio of their masses oresent in the initial mixture is 2:3 calculate the ratio of their mole fraction . |
|
Answer» |
|
| 44. |
The ration of de Broglie wavelentghs of a deuterium atom to that of an a-particicle,when the velocity of the former is five times greater than that of later is…………. |
|
Answer» 4 |
|
| 45. |
The ratio of wavelength values of series limit liens (n_(2) = oo) of Balmer series and Paschen series are |
| Answer» ANSWER :A | |
| 46. |
The ratio of wavelength values of series limit lines (n_(2) =oo) of Balmer series and Paschen serie are |
|
Answer» `4:9` |
|
| 47. |
The ratio of volumes of CH_(3)C O O H, 0.1 N to CH_(3) C O O Na, 0.1 N requiredto prepare a buffer solution of pH 5.74is (given : pK_(a) of CH_(3) C O O H is 4+74 ) |
|
Answer» `10:1` = V mL and volume of 0.1 N `CH_(3)CO OnN`a solution = X V mL `CH_(3)CO OH` present in the final solution `=(0.1)/(1000)xxV ` mol = `Vxx10^(-4)` mol `CH_(3)CO ON`apresent in the final solution `=(0.1)/(1000)XX x V "mol" = x V xx 10^(-4)` mol `pH = pK_(a) + log .(["Salt"])/(["Acid"])` `:. 5.74=4.74+log. (x V xx10^(-4))/(V xx 10^(-4))` orlog x = 1or x = 10 `:. ` Ratio = V mL : x V mL = 1 : x = 1 : 10 |
|
| 48. |
The ratio of van der Waals constant (a/b) has the dimensions : |
| Answer» Answer :A::B | |
| 49. |
The ratio of the wave lengths of the first line in the Lyman series of the spectrum of Hydrogen atom and the first line in the Balmer series of the spectrum of He^(+) is |
|
Answer» `20//27` |
|
| 50. |
The ratio of the wave lengths of the first line in the Lyman series of the spectrum of Hydorgen atom and the first line in the Balmer series of the spectrum of He^(+) is |
|
Answer» `20//27` `(lambda_H)/(lambda_(He^+)) = ((1)^2 (1/((1)^2) - 1/((2)^2)))/((2^2) (1/((2)^2) - 1/((3)^2))) = (27)/(5)` |
|