Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Write the resonance structures for (i) Ozone molecule (ii) N_(2)O |
Answer» SOLUTION :(i) OZONE molecule. `O_(3)`  (ii) NITROUS oxide, `N_(2)O` 
|
|
| 2. |
When a molecule is formed by the combinaiton of two atoms having small difference in the electronegativeties, ________. |
|
Answer» only FORCES of ATTRACTION between them OPERATE |
|
| 3. |
Which of the following equilibrium is favored by a temperature increase? |
|
Answer» C (graphite) `HARR` C (diamond) |
|
| 4. |
Which of the following statement is/are incorrect? |
|
Answer» Solubility of AgCl is LESS in 0.5 M KCl solutionn than in PURE WATER |
|
| 5. |
the secreenning constant of last electron of which element will ne maximum : |
|
Answer» 24 ` sigma=1xx(18)=18` |
|
| 6. |
Write the structuresand names of all plossible isomersof trimethyl benzenes. |
Answer» SOLUTION :
|
|
| 7. |
Which one of the following is notan isomer of 3-methylbut-1-yne ? |
|
Answer» Pent-1-yne |
|
| 8. |
Whatare thefrequencyand wavelengthofaphoton emittedduringa transitionfromn=5stateto the n=2statein the hydrogenatom ? |
|
Answer» Solution :Sinceinitialenergystate `n_(i)=5 ` calculateof `Delta E ` `Delta E = 2.18 xx 10^(18)((1)/(n_(1)^(2)) - (1)/( n_(f ) ^(2)))` =`2.18 xx 10^(18)((1)/(5^(2)) - (1)/(2^(2)))J` Calculationof FREQUENCYOF photon( v) : `v= (Delta E)/(h ) ` ` (4.578 xx 10^(18))/( 6.625 xx 10^(34) J s) ` Calculationof waveleng `(lambda)` `= (3.0 xx 10^(18) ms^(-1))/(6.9091 xx 10^(14) s^(1))` `lambda=434.21 NM` |
|
| 9. |
The trans-alkenes are formed by the reduction of alkynes with |
|
Answer» `H_2-Pd//C ,BaSO_4` |
|
| 10. |
The volume of phosgene formed at STP when 11.2 lit of chlorine reacts with carbon monoxide is |
|
Answer» `11.2` lit |
|
| 11. |
Write general characteristics of equilibria involving physical processes. |
|
Answer» Solution :For the physical processes FOLLOWING characte ristics are COMMON to the system at EQUILIBRIUM. (i) Equilibrium is possible only in a closed system at a given temperature. (ii) Both the OPPOSING processes occur at the same rate and there is a dynamic but stable condition. (iii) All measurable properties of the system remain constant. (iv) When equilibrium is attained for a physical process, it is characterised by constant value of one of its PARAMETERS at a given temperature. (v) The magnitude of such quantities at any stage indicates the extent to which the physical process has proceeded before reaching equilibrium. |
|
| 12. |
Upon treatment with ammoniacal H_(2)S solution, the metal ion precipitating as sulphide is: |
|
Answer» `Fe (III)` |
|
| 13. |
The set of quantum numbers for the outermost electron for copper in its ground state is …. |
|
Answer» 4,1,1,+1/2 `._29Cu to 1s^2 2s^2 2p^6 3s^2 3p^6 3d^10 4s^1` OUTERMOST electron is in 4s sub-shell For 4s n=4, l=0, m=0, s=+1/2 or -1/2 |
|
| 14. |
Which of the following reactions is not correct according to the law of conservation of mass ? |
|
Answer» `2 Mg_((s)) + O_(2(g)) rarr 2MgO_((s))` `{:(C_(3)H_(8(g)),+,O_(2(g)),rarr,CO_(2(g)),+,H_(2)O_((g))),(44g,,32G,,44g,,18g):}` The mass of REACTANTS `!=` mass of product Hence, LAW of CONSERVATION of mass is not followed. |
|
| 15. |
What is the difference between molecular mass and molar mass? Calculate the molecular mass and molar mass for carbon monoxide. |
|
Answer» Solution :Molecular mass: (i) Relative molecular mass is defined as the ratio of the mass of the molecule to the unified atomic mass unit. (ii) It can be calculated by adding the relative atomic masses of its constituent atoms. (iii) For carbon monoxide (CO) Molecular mass = Atomic mass of carbon + Atomic mass of oxygen 12+16 = 28u. MOLAR mass: (i) It is defined as the mass of one mole of a substance. (ii) The molar mass of a COMPOUND is equal to the sum of the relative atomic masses of its constituent expressed in g `mol^(-1)` . (iii) For carbon monoxide (CO) 12+ 16 = 28 g `mol^(-1)` . Both molecular mass and molar mass are NUMERICALLY same but the units are different. |
|
| 16. |
When an atom of an electronegative element becomes anion, which of the following occurs? |
|
Answer» It acts as a REDUCING agent |
|
| 17. |
x' on ozonolysis gives a dial while 'y' reacts with Baeyer's reagent to give a diol then 'x' and 'y' respectively are |
|
Answer» `C_(2)H_(2)&C_(6)H_(6)` |
|
| 18. |
When sodium thisolphate is treated with dilute hydrochlorixc acid sulphut dioxide gas is evolved along with simultaneous deposition of elemental sulhur what does this reaction indicate about the oxidation number of the two suplhur atoms ? Alsowrite the structure of thiossulphate ion |
|
Answer» SOLUTION :`Na_(2)SO_(3)+2HCIrarr2NaCI+SO_(2)+S+H_(2)O` This reaction INDICATES that the two S atoms have different OXIDATION number APPLYING the gernal rules of determing oxidation numbr the structure of `S_(2)O_(3)^(2-)` ion is 
|
|
| 19. |
Which has the highest boiling point ? |
|
Answer» HF |
|
| 20. |
Which of the following molecules has maximum carbon atoms in the longest chain? |
|
Answer»
|
|
| 21. |
Total number of moelcules which are antiaromatic ? |
|
Answer» `"AROMATIC -a,C,g,h,i,j,k.;Antiaromatic - b, d,E;Nonaromatic-t"` |
|
| 22. |
The wave function of 3s electron is given by psi_(3s) = (1)/(81sqrt(3 pi))((1)/(a_(0)))^(3//2)[27 - 18((r )/(a_(0))) + 2((r )/(a_(0)))^(3)]e^(-r//3a0) It has anode at r = r_(p) .Find the radiationbetween r_(0) and a_(p) |
|
Answer» Solution :At nodal point `V = 0` fromthe given wave function we find that `psi = 0 `at following values of r `[27 - 18 ((r )/(a_(0))) + 2 ((r )/(a_(0)))^(3)] = 0 ` SOLVING `r_(0)//a_(p)` we get `(r_(0))/(a_(0)) = (18 +- sqrt(10^(2) - 216))/(4) = (16 +- 10.4)/(2)` Hence `s = 14.2a_(0) `and `r_(0) = 3 9a_(0)` Besides there is a node at `r = oo` |
|
| 23. |
Which equation follows Boyle.s Law ? |
|
Answer» <P>`(p_(1))/(p_(2))=(V_(1))/(V_(2))` |
|
| 24. |
What is the relationship among the magnitudes of the enthalpies of combustion (DeltaH_(comb)), fusion (DeltaH_(fus)) and vaporization (DeltaH_(vap)) for a hydrocarbon such as hexane,C_(6)H_(14)? |
|
Answer» `DeltaH_(FUS)ltDeltaH_(fus)lt DeltaH_(vap)` |
|
| 25. |
What are spontaneous reaction? Give three examples for spontaneous reaction. |
|
Answer» Solution :A reaction that does occur under the given set of conditions is called a spontaneous reaction EXAMPLE: 1. waterfall RUNS downhill, but never up, spontaneously. 2. Heat FLOWS from hotter OBJECT to a colder one. 3. AGEING process |
|
| 26. |
Which statement is proper for water ? |
|
Answer» VOLUME of `H_2O` is SMALLER than HYDRIDE of 16th GROUP elements. |
|
| 27. |
Which of the following is the most stable carbocation ? |
|
Answer»
|
|
| 28. |
Trans isomer is more stable than cis isomer. Justify this statement. |
| Answer» SOLUTION :Trans isomer is more stable than cis isomer. This is because in the cis isomer, the bulky GROUPS are on the same side of the DOUBLE BOND. The STERIC repulsion of the groups makes the cis isomers less stable than the trans isomers in which bulky groups are on the opposite side. | |
| 29. |
What do you mean by Co-ordinate Bond ? Explain with example. |
|
Answer» Solution :The BOND formed by donation of electron from one of the constitunets present in the MOLECULE is called co-ordinate bond. It is only directional bond & indicated by `..to..` `H_(3)toBF_(3)` [Nitrogen donates PAIR of ELECTRONS to form bond] `NH_(3)BF_(3)` |
|
| 30. |
Which of the following indicates Kinetic gas equation ? a) PV = (3M)/(C^2) "" b) PV = 1/3 mnC^2 "" c) P = 1/3 dC^3 "" d) KE = 3/2 RT |
|
Answer» `PV = (3M)/(C^2)` |
|
| 31. |
The relationship between P_(c), V_(c) and T_(c)is |
|
Answer» `P_cV_c = RT` |
|
| 32. |
What is the total number of sigma and pi bonds in the following molecules? (a) C_(2)H_(2) (b) C_(2)H_(4) |
|
Answer» Solution :`H - C -= C - H` Sigma BOND = 3 `pi` bond = 2 (b)  Sigma bond = 5 `pi` bond = 1 |
|
| 33. |
Which hybrid orbitals are used by carbon atoms in the following molecules? (1) CH_(3)-CH_(3): (2) CH_(3)-CH=CH_(2), (3) CH_(3)-CH_(2)-OH (4) CH_(3)-CHO (5) CH_(3),COOH |
Answer» SOLUTION :
|
|
| 34. |
Which one of the following statements is NOT true about the effect of an increase in temperature on the distribution of molecular speeds in a gas ? |
|
Answer» The most probable speed increases. |
|
| 35. |
Which oxide of nitrogen is not a common pollutant introduced into the atmospheres both due to natural and human activity ? |
|
Answer» `N_(2)O_(5)` |
|
| 36. |
What happens to the disociation of PCl_(5) ina closed vassel if helium gas is introduced into it at the same temperature ? |
| Answer» SOLUTION :No EFFECT. | |
| 37. |
Which of the following metal oxides are reduced by CO ? |
|
Answer» ZnO |
|
| 38. |
Which of the following is the use of H_(2)O_(2) (i) hair bleach, disinfectant and antiseptic. (ii) In the synthesis of hydroquinone, paper pulp, (iii) Bleaching agent for textile, paper pulp, leather, oils, fats, etc. (iv) Treatment of domestic and industrial effluents. |
| Answer» Answer :D | |
| 39. |
Total number of possible isomers (configuartional only ) for 2,3,4- trichlorohexane are |
|
Answer» 2 |
|
| 40. |
Which of the two : O_(2)NCH_(2)CH_(2)O^(-) or CH_(3)CH_(2)O^(-) is expected to be more stable and why? |
|
Answer» Solution :In these `O_(2)NCH_(2)CH_(2)O^(-)` is more stable because, (a) RESONANCE stability: `O_(2)NCH_(2)CH_(2)O^(-)` MEANS  Its resonance structure are as under  (b) Stability by INDUCTIVE effect:  [NEGATIVE charge of `O^(-)` is transfer by the `-I` effect of `-NO_(2)`] [NEGATIVE charge of `O^(-)` is transfer by the `-I` effect of `-NO_(2)`]  [The transfer of `O^(-)` is decrease by the +I effect of `CH_(3)` and the stability decrease] [The transfer of `O^(-)` is decrease by the +I effect of `CH_(3)` and the stability decrease]
|
|
| 41. |
Which of the following reacts with water to give ethane ? |
|
Answer» `CH_4` |
|
| 42. |
What is compression factor? |
|
Answer» Solution : Thedeviationof real gasesfrom idealbehavioris measuredin TERMSOF a ratioof PVto nRT. Thisis TERMED ascompresssionfactor . compressionfactor`= Z = (PV )/( nRT )` |
|
| 43. |
Which of the following is true about the value of refractive index of quartz glass ? |
|
Answer» Same in all DIRECTIONS |
|
| 44. |
A piece of magnesium is burned in air.What do you observe ? |
| Answer» SOLUTION :`2Mg+O_2 RARR 2MgO | |
| 45. |
Which of the following will not be soluble in sodium carbonate soluton? |
|
Answer»
|
|
| 46. |
The root mean square velocity of a gas is doubled when the temperature is |
|
Answer» reduced to one-half |
|
| 47. |
What is photoelectric effect? State the result of photoelectric effect experiment. |
Answer» Solution :Photoelectric effect when radiation with certain minimum frequency `(V_(0))` strke the surface of a metal. The elec trons are ejected from the surface of the metal. This phenomenon is called photoelectric effect. The electrons emitted arecalled photoelectrons. 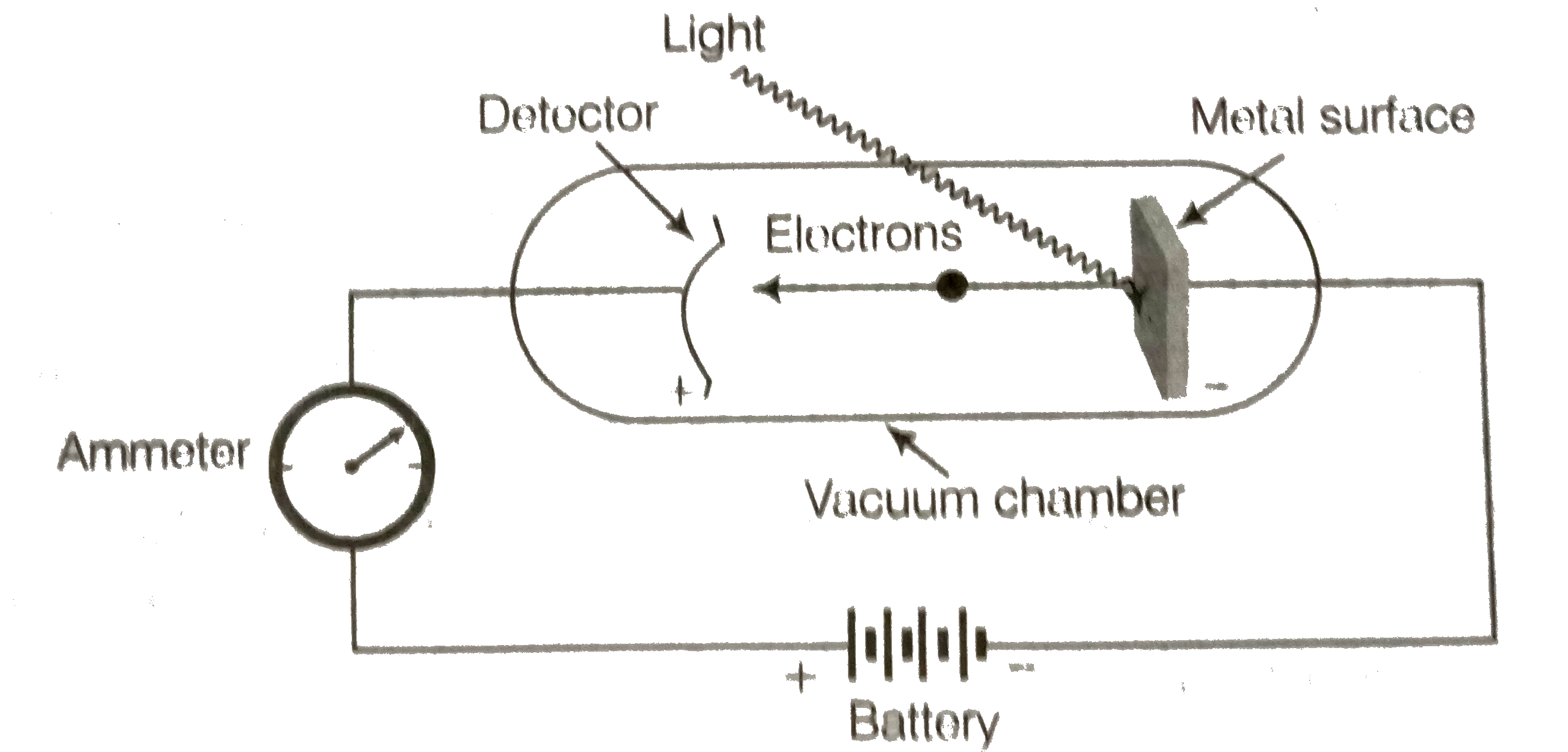 Equipment for studying the photoelectric effect Light of a particular frequency strikes a clean metal surface inside a vacuum chamber. Electrons are ejected from the metal and are counted by a detector that measures their kinetic ENERGY The result observed in this experiment were (i) The electrons are ejected from the metal surface as SOON as the beam of light strikes the surface. i.e. there is no TIKE lag between the striking of light beam and the ejection of electrons from the metal surface. (ii) The number of electorons ejected is proportional to the intensity or brightness of light. (III) For each metal, there is a characteristic minimum frequency, `v_(0)` (also known as threshold frequency) below which photoelectric effect is not observed. At a f requency `vgtv_(0)` the ejected electrons come out with certain kinetic energy. the kinetic ENERGIES of these ejections increases withthe increase of frequency of the light used |
|
| 48. |
When sodium peroxide is treated with dilute sulphuric acid, we get ……… . |
|
Answer» SODIUM SULPHATE and water |
|
| 49. |
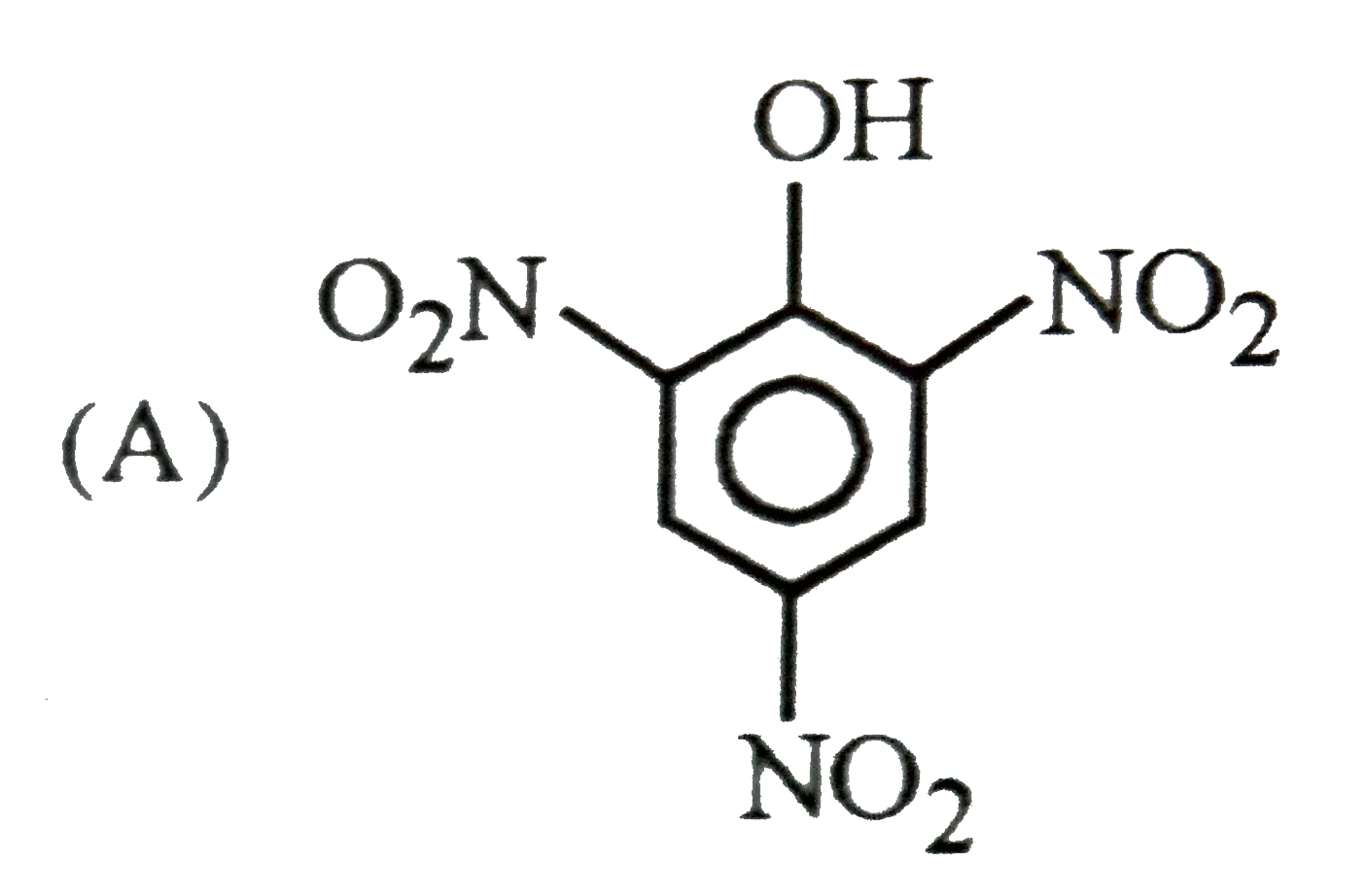
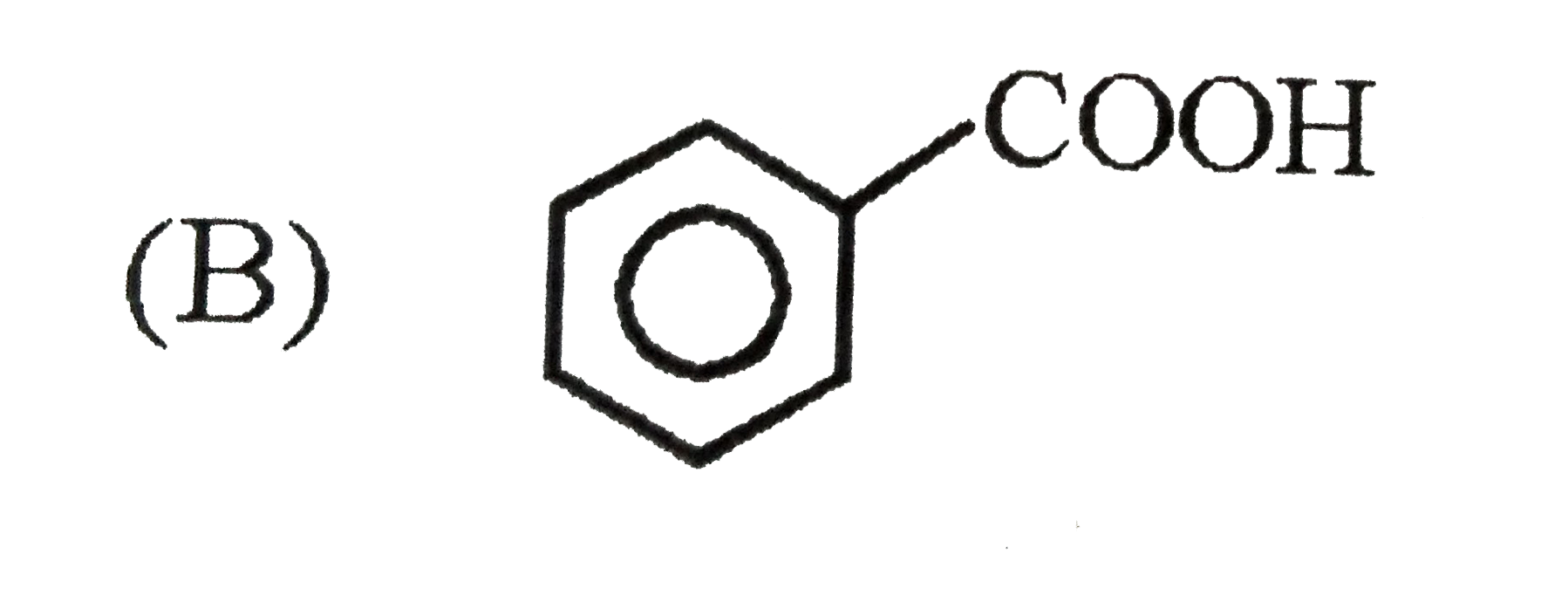
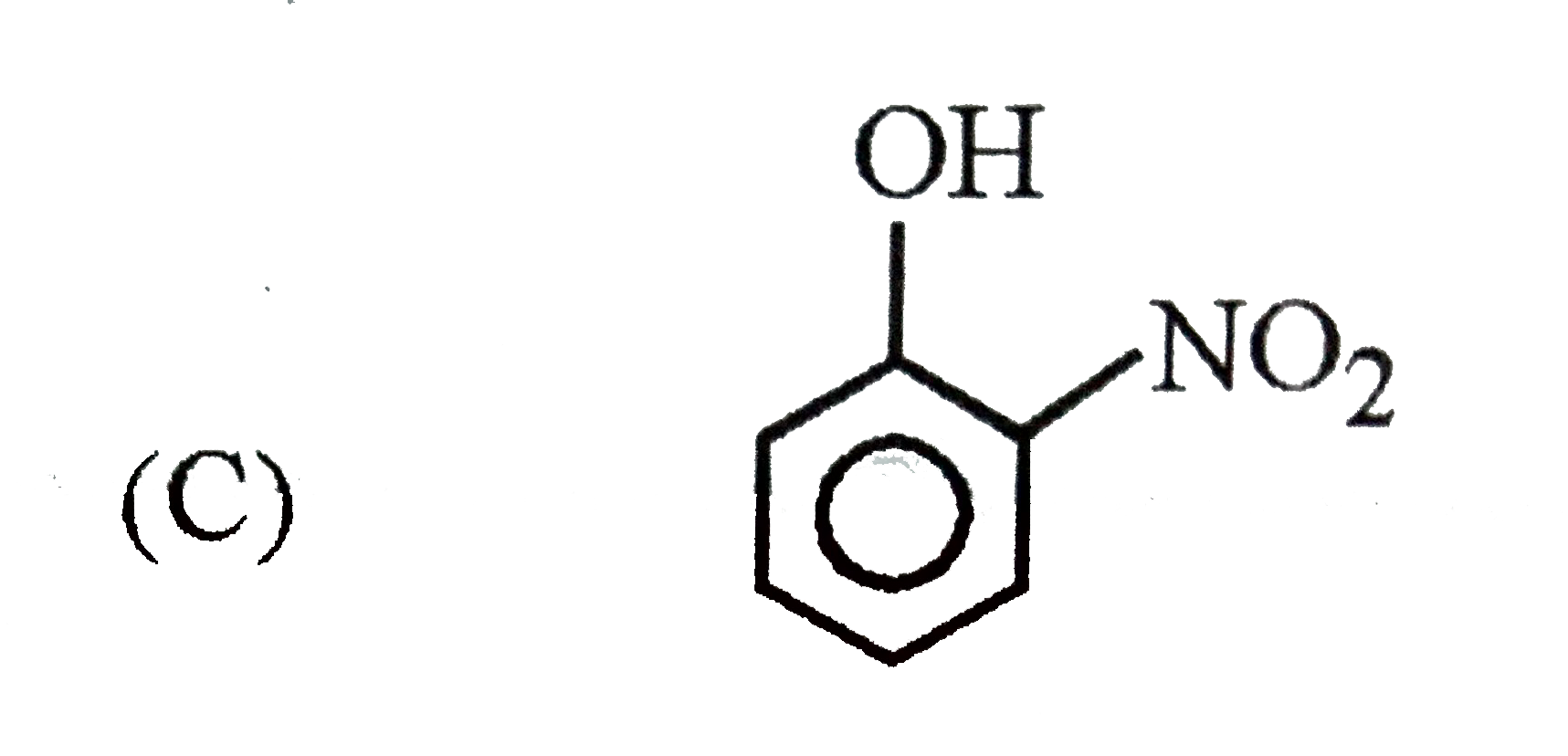
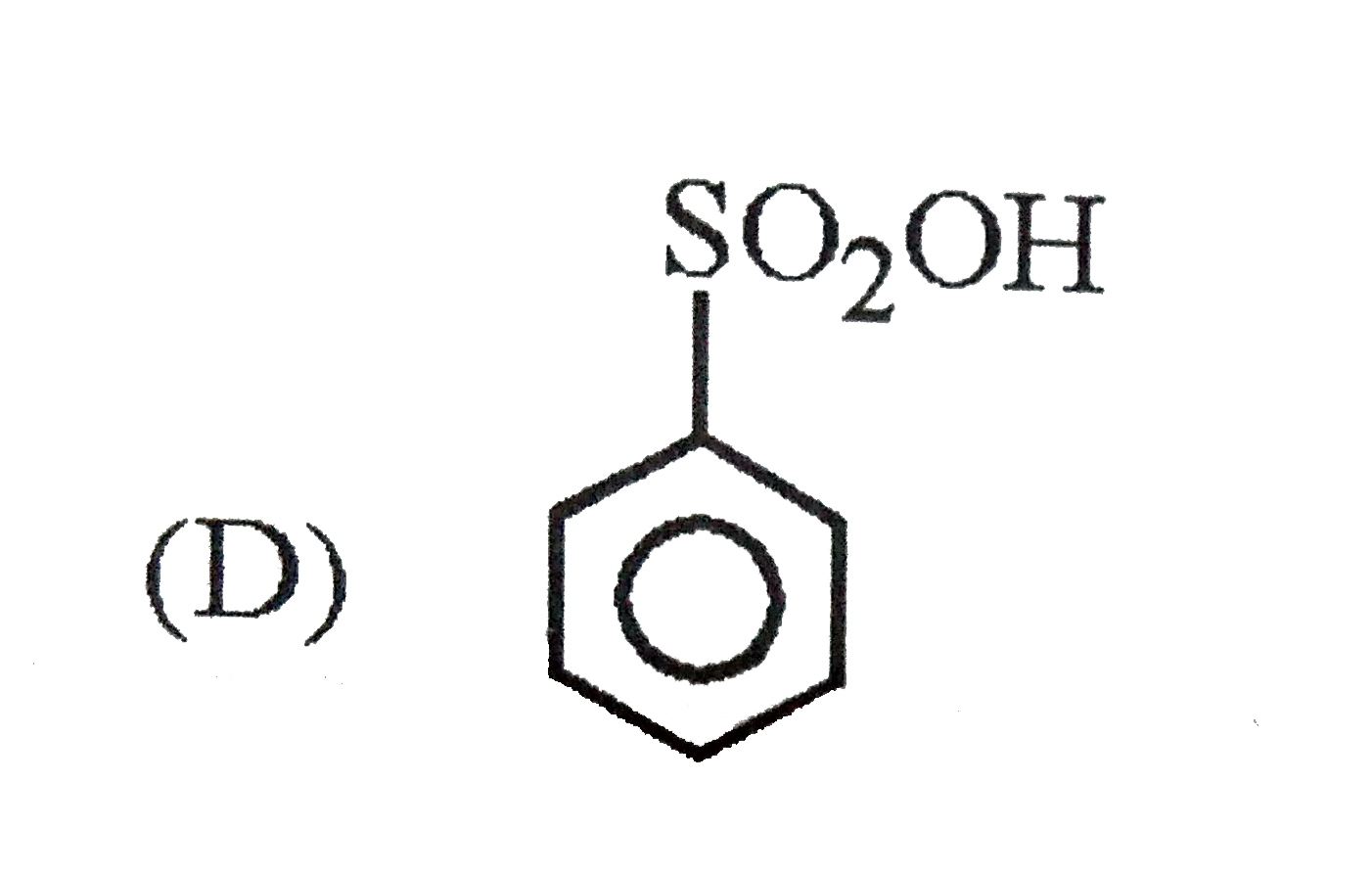
Which of the following substituent in the benzene is deactivating but o,p-directing ? |
|
Answer» SOLUTION :`-Cl , -N -= O` are moderately deactivating but O, P-DIRECTING. `-NO2 ,- OCH3` are meta directing . |
|