Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Ultra violet light causes |
|
Answer» Sticky metaphases |
|
| 2. |
Which of the following shows electromeric effect? |
|
Answer» Alkanes |
|
| 3. |
The shape of sulphur hexafluoride is …………………. |
| Answer» SOLUTION :OCTAHEDRAL | |
| 4. |
Which of the following is most acidic? |
|
Answer» MgO |
|
| 5. |
Which of the following is dependen on temperature ? |
|
Answer» WEIGHT PERCENTAGE |
|
| 6. |
The relative reactivity of 1^(@)H, 2^(@)H and 3^(@)H in bromination reaction has been found to be 1:82:1600 respectively. In the reaction, The percentage yield of (A) and (B) are expected to be: |
|
Answer» a. `99.4% , 0.6%` |
|
| 7. |
What is indicated by the value of K_a ? |
| Answer» Solution :`K_a` indicate the ACIDIC strength and IONIZATION constant of WEAK ACID at definate temp. | |
| 8. |
What do you do to control the formation of photochemical smog ? |
|
Answer» SOLUTION :Many techniques are used to control the formation of photochemical smog. Some of the techniques are as follows: If we control the primary precursors of photochemical smog, such as `NO_2` and hydrocarbons, the secondary precursors such as ozone and PAN, the photochemical smog will automatically be reduced. Catalytic converters are used in the automobiles, which prevent the release of nitrogen oxide and hydrocarbons to the atmosphere. Certain plants e.g., Pinus, Juniparus, Quercus, Pyrus and Vitis can METABOLISE nitrogen oxide and THEREFORE, their plantation could HELP in this MATTER. |
|
| 9. |
Which of the following compounds contain all the carbon atoms in the same hybrisation state ? |
|
Answer» `H-C equiv C-C equiv C-H` (d) 'C' atoms are `sp^(2)` hybridised |
|
| 10. |
What is critical temperature ? What is value of critical temperature for CO_(2) gas ? |
|
Answer» Solution :At which temperature gas is CONVERTED into LIQUID that temperature is known as critical temperature. `T_(C )` for `CO_(2)` is `30.98^(@)C` (73 atm). |
|
| 11. |
What is the decreasing order of strength of the bases OH, NH_(2) , H-C=C and CH_(3),-CH_(2), |
|
Answer» `CH_(3)-CH_(2)^(-) gt NH_(2)^(-) gt H-C EQUIV C^(-) gt OH^(-)` |
|
| 12. |
Which is the correct order of decreasing basicity of halide ions? |
|
Answer» ` I^(-) gt Br^(-)gt Cl ^(-) gt F^(-) ` |
|
| 13. |
The substitution will mainly take place at position : |
|
Answer» |
|
| 14. |
The total number of optical isomers possible for 2, 3-dibromobutane is |
|
Answer» 2 |
|
| 15. |
Which of the following molecule/moleculas will have zero dipole moment ? CO_(2), H_(2)O C Cl_(4), CHCl_(3), BF_(3), BeF_(2), NH_(3). |
| Answer» SOLUTION :`CO_(2), C Cl_(4) BF_(3) and BeF_(2)`. | |
| 16. |
Which ions are present in MgO(s) Calculate the enthalpy change for the reaction Mg(s) + 1//2O_(2)(g) rarr MgO(s) What kind of enthalpy change is this? Standard enthalpy of formation of MgO. DeltaH_(atom(O))= +249 kJ mol^(-1) "" DeltaH_(atom(Mg))=+148 kJ mol^(-1) DeltaH_("1st ionisation energy" (Mg))=+738 KJ mol^(-1) "" DeltaH_(2nd "electron energy "(Mg))= +145 1 kJ mol^(-1) DeltaH _("1st electron affinity "(O))= -141 kJ mol^(-1) "" DeltaH_(2nd " electron affinity "(O)) =+798 kJ mol^(-1) DeltaH_("lattice energy "(MgO))= - 3791 kJ mol^(-1) |
| Answer» SOLUTION : IONIC BOND : -548 kJ/mol | |
| 17. |
Which bond is responsible for solubility of beryllium halide in organic solvent ? |
|
Answer» IONIC |
|
| 18. |
Which of the following has the highest mass? |
|
Answer» 1 g-atorn of C |
|
| 19. |
Which is the set of amphiprotic species? |
|
Answer» ` H_3O^(+), HPO_4^(2-),HCO_3^(-)` |
|
| 20. |
Which of the following will be least reactive in nucleophilic substitution ? |
|
Answer» `CH_(3)CH_(2)CH_(2)Cl` |
|
| 21. |
Which of the following enter into human body and undergos various biochemical reactions and finallydemage DNA and cause cancer ? |
|
Answer» ETHYL ALCOHOL |
|
| 22. |
Write balanced chemical equation for each of the following chemical reactions. Lithium metal with nitrogen gas |
| Answer» SOLUTION :`6Li+N_(2)RARR 2LOi_(3)N` | |
| 24. |
What is the formulae of inorganic benzene? Write the structure of Diborane and explain the nature of bonding in it. |
|
Answer» SOLUTION :Formulae of inorganic benzene is `B_(3)N_(3)H_(6)` Structure of Diborane 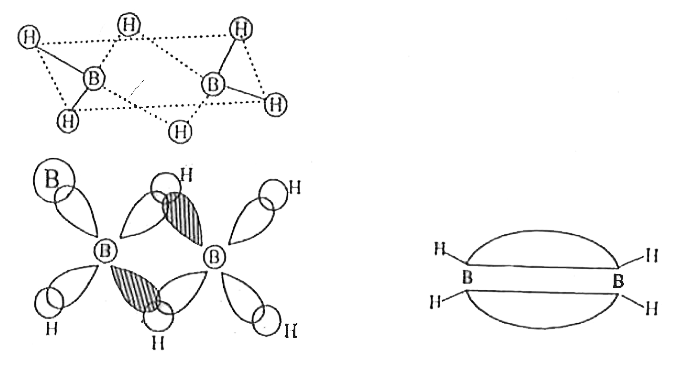 Bonding in diborane: Each B atom uses SP3 hybrides for boding. Out of the four sp3 hybrids one each B atom, one is without electrons shown in shaded region. The terminal B-H bonds are normal 2-centre-2electron bonds but the two bridge bonds adre 3-centre -2 -ELECTRON bridge bonds are also referreed to as banance bonds. |
|
| 25. |
What indicate by corner and end in the bond line structure method of organic compound? |
| Answer» Solution :(i) End MEANS methyl `-CH_(3)` group (ii) Each CORNER indicate carbon and hydrogen atom. By substracting no. of BOND with corner from 4 the value OBTAIN equal to no. of hydrogen with carbon in corner | |
| 26. |
What is proved by Boyle.s law in a quantitative manner ? Why ? |
| Answer» Solution :Experiments of Boyle, in a quantitative MANNER prove that GASES are highly COMPRESSIBLE because when a GIVEN mass of a GAS is compressed, the same number of molecules occupy a smaller space. This measns that gases become denser at high pressure. | |
| 27. |
Two substances when separated on the basis of partition coefficient between two liquid phase, then the technique is known as |
|
Answer» COLUMN CHROMATOGRAPHY |
|
| 29. |
When 15.68 litres of a gas mixture of methane and propane are fully combusted at 0^@C and 1 atmosphere, 32 litres of oxygen at the same temperature and pressure are consumed. The amount of heat released from this combustion in kJ is (DeltaH_C(CH_4)=-890 kJ mol^(-1) and DeltaH_C(C_3H_8)=-2220 kJ "mol"^(-1)) |
|
Answer» `-889 kJ` `DeltaH_C(CH_4)=-890 "kJ mol"^(-1)` `DeltaH_C(C_3H_8)=-"-2220 kJ mol"^(-1)` Let the mixture contain X lit of and lit of propane . `undersetx(CH_4)+underset(2X)(2O_2) to CO_2 + 2H_2O` `underset(15.68-x)(C_3H_8+5O_2) to underset"5(15.68-x)"(3CO_2 + 4H_2O)` Volume of oxygen consumed = 2x+5 (15.68-x)=32 lit 2x + 78.4 -5x =32 78.4 - 3X =32 3x=46.4 x=15.47 Given mixture contains 15.47 liters of methane and 0.213 liters of propane . Hence , `DeltaH_C=[((DeltaH_C(CH_4))/"22.4 lit")xx"lit"]+[((DeltaH_C(C_3H_8))/"22.4 lit")(15.68-x)"lit"]` `DeltaH_C=[((-890 "kJ mol"^(-1))/"22.4 lit")xx"15.47 lit "]+[((-2220)/"22.4 lit")"0.21 lit"]` `DeltaH_C=[-614.66 "kJ mol"^(-1)]+[-20.81 "kJ mol"^(-1)]` `DeltaH_C=635.47 "kJ mol"^(-1)` |
|
| 30. |
Which of the following is not a disproportionation reaction ? |
|
Answer» <P>`P_(4)(s)+3OH^(-)(aq)+3H_(2)O(l)toPH_(3)(g)+3H_(2)PO_(2)^(-)(aq)` |
|
| 31. |
Which of the following are isoelectronic species? Na^+, K^+, Mg^(2+), S^(2-), Ar. |
|
Answer» Solution :Isoelectronic - species having some NUMBER of electrons `._(11)Na^(+) = 11 - 1 = 10 e^(-) , ._(19)K^(+) = 19-1 = 18e^(-)` `._(12)Mg^(2+) = 12 - 2 = 10 e^(-) , ._(20)Ca^(2+) = 20 - 2 = 18e^(-)` `._(16)S^(2-) = 16 + 2 = 18 e^(-) , ._(18)Ar = 18 e^(-)` HENCE the isoelectronic species are (i) `Na^(+) and Mg^(2+) ""(ii) K^(+) , Ca^(2+), S^(2-) and Ar`. |
|
| 32. |
Which kind of defect is found in KCl crystal? |
|
Answer» Frenkel |
|
| 33. |
Which of the following liquid will exhibit highest vapour pressure ? |
|
Answer» `C_(2)H_(5)OH (l)` |
|
| 34. |
Which orhital Is more negative form sp, sp^(2) and sp^(3) ? Why ? |
Answer» SOLUTION :sp is more negative than `sp^(2) and sp_(2)` is more negative than `sp^(3)`. Because s CHARACTER is more than it is more negative. 
|
|
| 35. |
Which of the following is a component of most didney stones? |
|
Answer» POTASSIUM OXALATE |
|
| 36. |
Which reaction is correct of oxidation half reaction in basic medium P_(4)toH_(2)PO_(2)^(-) ? |
|
Answer» <P>`P_(4)+8OH^(-)to4H_(2)PO_(2)^(-)+4OH^(-)` |
|
| 37. |
What is the Gibbss Helmhotzequation ? |
| Answer» SOLUTION :`DeltaG = DeltaH-T DeltaS` where `DeltaG , DeltaH` and`DeltaS` are FREE ENERGY CHANGE and ENTROPYCHANGE respectively. | |
| 38. |
Which of the following statements are correct? |
|
Answer» Transition elements exhibit higher ENTHALPIES of atomizaton as they have STRONGER interatomic interation. |
|
| 39. |
Thiosulphate reacts differently with iodine and bromine in the reactions given below : S_(2)O_(3)^(2-)+I_(2) rarr S_(4)O_(6)^(2-) + 2I^(-) S_(2)O_(3)^(2-)+2Br_(2)+5H_(2)O rarr 2SO_(4)^(2-)+2Br^(-)+10H^(+) Which of the following statements justifies the above dual behaviour of thiosulphate ? |
|
Answer» Bromine is a STRONGER oxidant than iodine.<BR>Bromine is a weaker oxidant than iodine. |
|
| 40. |
Which of the following are antiaromatic ? |
|
Answer»
1) `4n pi`electrons. 2) PLANAR STRUCTURE 3) CONJUGATION |
|
| 41. |
Whast is the oxidation state of Fe in the product formed when acidified potassium ferrocyanide K_(4)[Fe(CN)_(6)] is treated with hydrogen peroxide? |
| Answer» ANSWER :B | |
| 42. |
Why BF_(3) forms as an adduct with ammonia ? |
Answer» Solution :`BF_(3)` is an electronsdeficientcompounds sincethe VALENCE shell of Bhas only six electrons. It needs two moreelectronsto complete its octet.On the other HAND, NIN `NH_(3)` has a lone pairof electronswhich it can donate to B to forma coordinate bond. Thus, `BF_(3)` FORMS an adduct with ammonia. 
|
|
| 43. |
Which is not true about borax ? |
|
Answer» It is a USEFUL primary STANDARD for titrating against acids |
|
| 44. |
Two radio-isotopes P and Q of atomic weight 10 and 20 respetively are mixedratio is found to be 1:m 4 Isotope P has a half-life of 10 day The half of isotope Q is: |
|
Answer» Zero Initial mole of `P = (w)/(10)`, mole of `Q = (w)/(20)` Final mole of `P = (w_(1))/(5xx10)` Final mole of `Q = (4w_(1))/(20xx5)` For P, `(P_(N_(0)))/(P_(N))=e^(lambda_(1)t)` For `Q, (Q_(N_(0)))/(Q_(N))=e^(lambda_(2)t)` For `P :. (w xx 5 xx10)/(10xx w_(1)) = e^(lambda_(1) xx20)` ...(i) For `Q :. (w xx 20xx5)/(20XX w_(1) xx4) = e^(lambda_(1) xx20)` ...(ii) By (i) and (ii), `4 = e^(lambda_(1) - lambda_(2)xx20)` `:. 20(lambda_(1) - lambda_(2)) = log_(e) 4` `:. t = oo` |
|
| 45. |
What is the density (in "g lit"^(-1)) of CO_2, at 400 K and exerting a pressure of 0.0821 atm (R = 0.0821 "lit atm mol"^(-1) k^(-1)) |
|
Answer» 0.01 |
|
| 46. |
Which of the following is most abundant in the earth crust ? |
|
Answer» Boron |
|
| 47. |
Which of the following compounds will not exist as resonance hybrid? Give reason for your answer. (a) CH_(3)OH (b) R- CONH_(2) (c ) CH_(3)CH= CHCH_(2)NH_(2) |
|
Answer» SOLUTION :(a) `CH_(3)OH` does not contain `pi`-electron and hence it cannot be regarded as a resonance hybrid. (b) The COMPLETE structure of `R- CONH_(2)` is as under  In this 1-(lone pair), 2- (`pi`-electron), 3-single bond and 4 nonbonding p electron on N and alternate `pi` bond system is PRESENT so following rsonance structure can be expressed  `therefore` (b) `RCONH_(2)` has POSSIBLE resonance structure. (c ) In this alteranate `pi//p` ELECTRONS are not there and C=C has more than one singma bond. So in C resonance is not possible. |
|
| 48. |
Which of the following process makes the one porous? |
|
Answer» EVAPORATION of LIQUID into vapour. |
|
| 49. |
Write structure for each of the following compounds. Why are the given names incorrect ? Write correct IUPAC names. (i) 2-Ethylpentane (ii) 5-Ethyl-3-methylheptane |
|
Answer» Solution :(i) The STRUCTURE of 2-Ethylpentane : `CH_(3)-underset(C_(2)H_(5))underset(|)(CH)-CH_(2)-CH_(2)-CH_(3)` Longest chain is of six carbon atoms and not that of five. Hence, correct name is 3-Mehylhexane. (ii) `overset(7)CH_(3)-overset(6)(CH_(2))-underset(CH_(3))underset(|)overset(5)(CH)-overset(4)(CH_(2))-underset(C_(2)H_(5))underset(|)overset(3)(CH)-overset(2)CH_(2)-overset(1)(CH_(3))` NUmbering is to be started from the end which gives LOWER number to ETHYL GROUP. Hence, correct name is 3-ethyl-5-methylheptane. |
|



