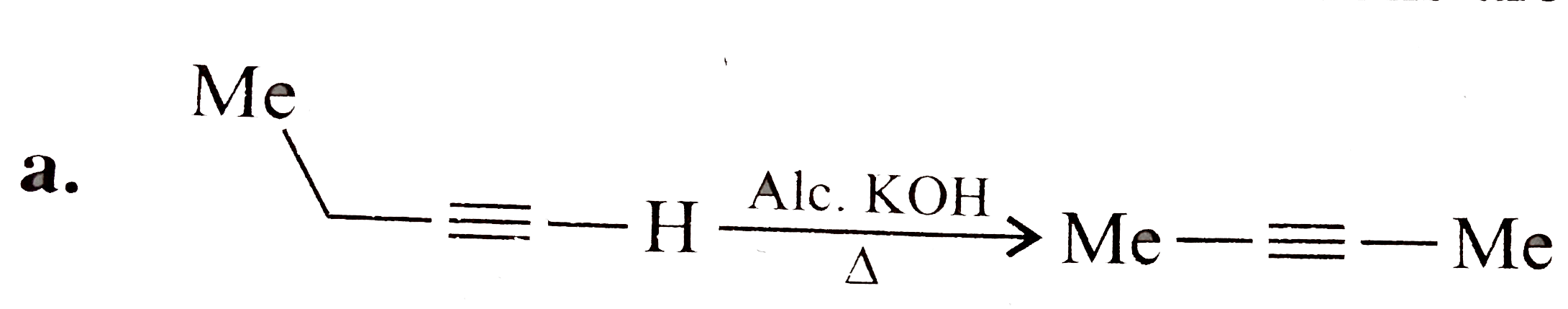Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which one of the following statements about water is false ? |
|
Answer» Watr is oxidized to oxygen during photo-synthesis intermolecular hydrogen bonding in water molecules but there is no intramolecular hydrogen bonding |
|
| 2. |
What is the difference between adsorbent and absorbate? |
Answer» SOLUTION :
|
|
| 3. |
Which one is present in stratosphere? |
|
Answer» `N_2` |
|
| 4. |
Write the significance of a plus and a minussign shown in representing the orbitals . |
|
Answer» Solution :As ORBITALS are respresented by weve functions, a PLUS sign in an orbital represents a +ve weve FOUNCTION and a MINUS sign represents a -ve weve founction . |
|
| 5. |
Which of the following will not give iodoform test? |
|
Answer» Ethanol |
|
| 6. |
Which of the following has both members from the same period of the periodic table |
| Answer» ANSWER :C | |
| 8. |
Which type of solvent is added in aqueous solution in differential extraction method? |
| Answer» Solution :(i) Solvent is easily VOLATILE (ii) Solvent should be insoluble in water (III) The SOLUBILITY of COMPOUND must be more in solvent compare to water | |
| 9. |
Which of the following can effectively remove all types of hardness of water |
| Answer» Answer :A | |
| 10. |
When 50 cm^(3) of 0.2N H_(2)SO_(4) is mixed with 50cm^(3) of 1N KOH, the heat liberated is |
|
Answer» `11.45 KJ` |
|
| 11. |
What is system ? Explain types of system. |
|
Answer» Solution :A very small part of universe in which experiments are done and observations are made, this small part of universe is CALLED SYSTEM. There are three types of the system. (1) Open System : In an open system, there is exchange of energy and matter between system and surroundings Figure (a). The presence of reactants in an open beaker is an example of an open system. (2) Closed System : In a closed system, there is no exchange of matter, but exchange of energy is possible between the system and the surroundings [Fig. (b)]. The presence of reactants in a closed vessel made of conducting MATERIAL. e.g., COPPER or steel is an example of a closed system.  (3) ISOLATED System : In an isolated system, there is no exchange of energy or matter between the system and the surroundings [Fig. (c)]. The presence of reactants in a thermos flask or any other closed insulated vessel is an example of an isolated system. |
|
| 12. |
Which will show geometrical isomerism |
|
Answer» I,ii,iii,iv |
|
| 13. |
Which are the name of (CH_(3))_(3)C- and (CH_(3))_(3) C CH - respectively? |
|
Answer» Tersary BUTYL and ethyl |
|
| 14. |
What is total number of sigma and pi bonds in the following molecules ? (a) C_(2) H_(2)(b) C_(2) H_(2) |
Answer» SOLUTION :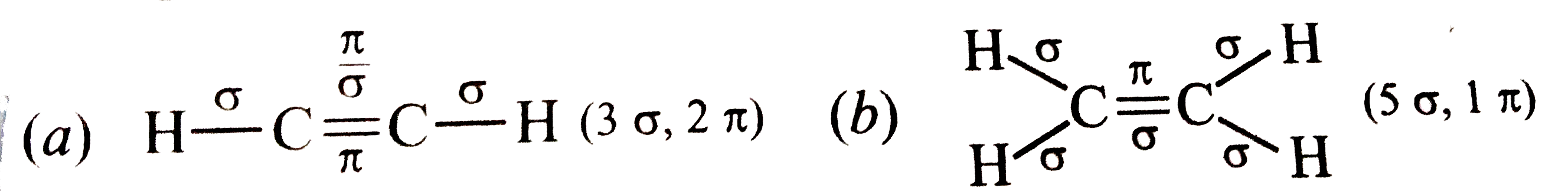
|
|
| 15. |
Which metals follows stock notation method ? |
| Answer» Solution :METALS which CONTAINS more than ONE OXIDATION states that metals FOLLOWS stock notation method. | |
| 16. |
Which of following elements can be involved in ppi-dpi bonding ? |
|
Answer» Carbon |
|
| 17. |
Which nomenclature is not according to IUPAC system ? |
|
Answer» `UNDERSET("4-Bromo,2,4-di-methylhexane")(CH_(3) - CH_(2) - undersetunderset(BR)(|)oversetoverset(CH_(3))(|)CH - CH_(2) - undersetunderset(CH_(3))(|)CHCH)` |
|
| 18. |
What is Z in the following reactions ? BCl_3 + H_2 underset(450^@ C)overset(Cu-Al)to X + HClX overset("methylation")to Z |
|
Answer» `(CH_3)BH_2` |
|
| 19. |
Upon mixing 45.0 mL of 0.25 M lead nitrate solution with 25 mL of 0.1 M chromic sulphate solution, precipitation of lead sulphate takes place. How many moles of lead sulphate are formed ? Also calculate the molar concentrations of the species left behind in the final solution. Assume that lead sulphate is completely insoluble. |
|
Answer» `underset(3"mole")(3Pb(NO_(3))_(2))+underset(1"mole")(Cr_(2)(SO_(4))_(3)) to underset(3"mole")(3PbSO_(4))+underset(2"mole")(2Cr(NO_(3))_(3))` No. of moles of `Pb(NO_(3))_(2)=45xx10^(-3)xx0.25` `=11.25xx10^(-3)` mole No. of moles of `Cr_(2)(SO_(4))_(3)=25xx10^(-3)xx0.1=2.5xx10^(-3)` mole Thus, `Cr_(2)(SO_(4))_(3)` has limiting concentration. It shall be consumed fully and the number of moles of lead sulphate produced will be `=3xx2.5xx10^(-3)=7.5xx10^(-3)` mole No. of moles of lead nitrate left `=11.25xx10^(-3)-7.5xx10^(-3)` `=3.75xx10^(-3)` mole Total volume =(45.0+25.0)=70 ML of `70xx10^(-3)` litre Molarity `=(3.75xx10^(-3))/(70xx10^(-3))=0.0536 M` No. of moles of `Cr(NO_(3))_(3)` formed`=2xx2.5xx10^(-3)=5xx10^(-3)` mole Molarity `=(5xx10^(-3))/(70xx10^(-3))=0.0714 M` `Pb(NO_(3))_(2) " and " Cr(NO_(3))_(3)` will be PRESENT in solution in IONIC form. Thus, `[Pb^(2+)]=0.0536 M` `[Cr^(3+)]=0.0714 M` `[NO_(3)^(-)]=(2xx0.0536)+(3xx0.0714)` =0.3214 M |
|
| 20. |
Which of the following statement is wrong ? |
|
Answer» Single N-N bond is stronger than the single P-P bond |
|
| 21. |
What is electrical conductivity due to in (a) metals, (b) ionic solids , and (c )semiconductors ? |
|
Answer» SOLUTION :(a)Due to FLOW of electrons. (B)Due to flow of ions in solution or melt and DEFECTS in the solid state (C )Due to the presence of impurities and defects. |
|
| 22. |
What do you understand by (i) electron-deficient ,(ii) electron-precise , and (iii) electron-rich compounds of hydrogen ? Provide justification with suitable examples. |
|
Answer» Solution :Hydrides of group 13 (i.e, `BH_(3)` , `AlH_(3)` etc.) donot have SUFFICIENT number of electrons to FORM normal covalant bonds and hence are called elctron-deficient hydrides. To make up this deficieny ,they generally exit in polymeric forms such as `B_(2)H_(6)`, `B_(4)H_(10)` , `(AlH_(3))` etc. (ii) Hydrides of gropu 14 (i.e, `CH_(4)` , `SiH_(4)`, `GeH_(4)`, `SnH_(4)`, `PbH_(4)`) have exact number of electrons to form covalent bonds and hence are called electron-precise hydrides. All these hydrides have tetrahedral shapes. (iii) Hydrides of group 15,16 and 17 (i.e, `NH_(3)` ,`PH_(3)`,`H_(2)O`,`H_(2)S`,HF,HCL, etc.) have more electrons than required to form normal covalent bonds and hence are called electron-rich hydrides. The excess electrons in these hydrides are present as lone pairs of electrons. |
|
| 23. |
Write the chemical equations for the reactions involved inSolvay process of preparation of sodium carbonate. |
|
Answer» Solution :Solvary proces: The Solvay PROCESS is represented by the below chemical equations: (i) `2NH_(3) + H_(2)O + CO_(2) rarr underset("Ammonium carbonate")((CH_(4))_(2)CO_(3))` (ii) `(NH_(4))_(2)CO_(3) + H_(2)O + CO_(2) rarr underset("Ammonium bicarbonate")(2NH_(4) HCO_(3))` (iii) `NH_(4)HCO_(3) + NaCl rarr underset("Ammonium chloride")(NH_(4)Cl) + NaHCO_(3)` (iv) `2NaHCO_(3) rarr underset("Sodium carbonate")(Na_(2)CO_(3) + ) CO_(2) + H_(2)O` |
|
| 24. |
Why are the following reactions not redox reactions ? AgNO_(3)(aq)+HCl(aq)toHNO_(3)(aq)+Ag(Cl)(s) |
| Answer» SOLUTION :Given reactions do not involve any change in OXIDATION NUMBER. | |
| 25. |
What is BOD ? |
| Answer» Solution :The AMOUNT of oxygen consumed by microorganisms in decomposing waste in a sample of SEWAGE WATER is called BOD (Biochemical Oxygen Demand). | |
| 26. |
The sharing of valence electrons between the atoms will lead to the formation of _________ |
|
Answer» IONIC BOND |
|
| 27. |
Which of the following does not represent the mathematical expression for the Heisenberg uncertainty principle? |
|
Answer» `Deltax.Deltap GE H/(4PI)` |
|
| 28. |
Which type of value are [H^+],[OH^-], pH and pOH in acidic solution ? |
|
Answer» Solution :In ACIDIC solution `[H^+] gt [OH^-]` Therefore `[H^+] gt 1 xx 10^(-7)` M and `[OH^-] lt1 xx 10^(-7)` M So, pH `LT` 7.0 and pOH `gt` 7.0 |
|
| 29. |
The separation of the constituents of a mixture by column chromatography is based on the………….of the various components in the mixture. |
|
Answer» differential extraction |
|
| 30. |
Which of the following show amphoteric behaviour |
|
Answer» `Zn(OH)_2` |
|
| 31. |
Which of the following compounds on boiling will KMnO_(4) (alk) and subsequent acidification will not give benzoic acid ? |
|
Answer» BENZYL alochol 
|
|
| 32. |
When do we use a find paper or hot water funnel for filtration ? |
| Answer» Solution :To AVOID CRYSTALLIZATION during FILTRATION, flutes filter paper is used when the VOLUME of the solution to be FILTERED is small and hot water funnel when the volume is large. | |
| 33. |
The total num ber of orbitals present for principle quantum number n = 4 i s ..... |
|
Answer» 12 |
|
| 34. |
The starting element and last element in the largest period in modern periodic table are |
|
Answer» RB and Xe |
|
| 35. |
Two elements 'X' and 'Y' have the following configurationX= 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 Y=1s^2 2s^2 2p^6 3s^2 3p^5 The compound formed by the combination of 'X' and 'Y' will be |
|
Answer» `XY_(2)` |
|
| 36. |
What is the maximum mass of H_(2)O (in gm) which can be obtained if total 42 gm of propyne and oxygen are subjected to combustion? |
|
Answer» |
|
| 37. |
Which of the following C-H bond has the lowest bond dissociation energy? |
|
Answer» PRIMARY `(1^(@))C-H` bond |
|
| 38. |
To Whichblock(s,p , d or f ) doesthe elementwith atomicnumber50 belong ? |
|
Answer» <P> Solution :E.C . Of Z= 50 is`1s^(2)2s^(2)2p^(6)3s^(2) 3p^(6)3d^(10)4S^(2)4p^(6)4D^(10)5S^(2)2p^(2).` Sincehere the lastelectronenters thep - orbital , thereforeit a p- ELEMENT. |
|
| 39. |
What are the necessary conditions for any system to be aromatic ? |
|
Answer» SOLUTION :The necessary conditions for a molecule to be aromatic are : (i)It should have a single CYCLIC cloud of delocalised `pi`-electrons above and below the plane of the molecule. (ii)It should be planar . This is because complete delocalization of `pi`-electrons is possible only if the ring is planar to allow cyclic overlap of p-orbials. (III)It should contain Huckel number of electrons, i.e., (4n+2) `pi`-ELECTRONSWHERE n=0,1,2,3.... etc. A molecule which does not satisfy any one or more of the above conditions is said to be non-aromatic. |
|
| 40. |
Which is the first element to have 4d-electron in its electronic configurations |
|
Answer» Ca |
|
| 41. |
When a mixture of Cu_(2)S and CuSis titrated with Al(MnO_4)_3in acidic medium, the oxidation product of Cu_(2)S and CuSare Cu^(+2) and SO_2 . If the molecular weight of Cu_(2)S, CuS and Al(MnO_(4))_3 , be M_1 ,M_2 and M_3respectively, then which of the following statement are correct? |
|
Answer» EQUIVALENT weight of `Cu_2S` is `M_1//8` n - factor for `CuS RARR 6` n-factor for `Al(MnO_4)_3 rarr 15` |
|
| 42. |
Which of the following can be measured by the Oswald-Walker dynamic method ? |
|
Answer» VAPOUR PRESSURE of the solvent |
|
| 43. |
Which of the following expression regarding the unit of coefficient of viscosity is not true? |
|
Answer» DYNES `CM^(-2)`s |
|
| 45. |
What are addition reactions? Give an example. |
| Answer» Solution :All organic COMPOUNDS having double or triple bond adopt ADDITION reactions in which two substances unite to form a single compound.During the addition reaction the hybridization ofthe substrate CHANGES as only one bond breaks and two new bonds are FORMED. <BR> Example :`CH -= underset("Acetylene")(CH_(2)) + HBr to underset("bromoethane")(CH_(2) =CH oplus Br)` | |
| 46. |
What will be the conjugate acid-base pair in HCl_((aq)) + H_2O_((aq)) hArr H_3O_((aq))^(+) +Cl_((aq))^(-) |
| Answer» SOLUTION :HCL and `CL^(-) , H_3O^(+)` and `H_2O` | |
| 47. |
What is the weight of calcium carbonate required for the production of 1 L of carbon dioxide at 27^(@)C and 750 mm, by the action of dilute hydrochloric acid? |
|
Answer» Solution :Givne conditions STP conditions `{:(P_(1)="750 mm",P_(2)="760 mm"),(T_(1)="300 K",T_(2)="273 K"),(V_(1)="1 Lit",V_(2)=?):}` From the equation of state, the unknown volume is obtained `V_(2)=(P_(1)V_(1))/(T_(1))xx(T_(2))/(P_(2))=(750xx1xx273)/(300xx760)=0.898L` Calcium carbonate on reaction with hydrochloric acid gives CARBONDIOXIDE. `CaCO_(3)+2HClrarrCaCl_(2)+H_(2)O+CO_(2)` `"1 mole of "CO_(2)-="1 mole of "CaCO_(3)` `"22.4L of "CO_(2)"at STP"="100 grams of "CaCO_(3)` `"0.898 L of "CO_(2)" at STP = ?"` The weight of calcium carbonate required = `(100xx0.989)/(*22.4)="4.009 grams"` |
|
| 48. |
Write the various definition of first law of thermodynamics. |
|
Answer» SOLUTION :First law of thermodyanmics (i)The total energy of an isolated SYSTEM remains constant though it MAY change from one form to another (ii) Whenever energy of a particular type disappears equivalent amount of another type must be produced . (iii)Total energy of a system and surroundings remains constant. (iv) Energy can neither be CREATED nor destroyed, but may be converted from one form to another. (v)The change in the internal energy of a closed system is equal to the energy that PASSES through its boundary as heat or work. (vi) Heat and work are equivalent ways of changing a system.s internal energy. |
|
| 49. |
The reagent used in the conversion of Chlorobenzene to Benzene is …………………… . |
| Answer» SOLUTION :Ni-Al/NaOH | |