Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The value of ebulliosepic constant depends upon |
|
Answer» ` Delta H_("SOLUTION ") ` |
|
| 2. |
Write structures and IUPAC names of different structural isomers of alkenes corresponding to C_(5)H_(10) |
|
Answer» SOLUTION :Structural isomers of `C_(5)H_(10)` molecular formula is as follows : (a) `underset("Pent-1-ene")(CH_(2)-CH-CH_(2)-CH_(2)=CH_(2))` (B) `underset("Pent-2-ene")(CH_(3)-CH=CH-CH_(2)-CH_(3))` (c) `underset("2-Methylbut-2-ene")(CH_(3)-underset(CH_(3))underset(|)(C)=CH-CH_(3))` (d) `underset("3-Methylbut-1-ene")(CH_(3)-underset(CH_(3))underset(|)(CH)=CH-CH_(2))` (e) `underset("2-Methylbut-1-ene")(CH_(2)-underset(CH_(3))underset(|)(C)=CH_(2)-CH_(3))` From the FOLLOWING (a) and (b) are position isomers of each other and (c), (d) , (e) are chain isomers of (a), (b). |
|
| 4. |
The value ofK_(p) " is " 1 xx10^(-3) " atm"^(-1) " at " 25^(@) C. " for the reaction " : 2 NO + Cl_(2)hArr2 NOCl.A flask contains NO at 0*02 "atm at " 25^(@)(C).Calculate the mol of Cl_(2)that must be added if 1 % of the NO is to be converted to NOCl at equilibrium. The volume of the flask is such that0*2mol of the gas produce 1 atm pressure at25^(@)C.( Ignore the probable association of NO toN_(2)O_(2)) |
|
Answer» <P> Solution :Suppose intial pressure of `Cl_(2)`added is p atm .`{:("Then ",2NO,+,Cl_(2),hArr,2 NOCl),("Intial",0*02 "atm",,p " atm",,),("A eqm. " ,0*02 - (0*02)/100,,(p-(0*01)/100),,(0*02)/100),(,=2 xx 10^(-2)-2 xx10^(-4),,=p-10^(-4),,-2 xx 10^(-4)"atm"),(,=2 xx 10^(-4)(100-1),,,,),(,=198 xx10^(-4) "atm",,,,):}` ` K_(p) = (p_(NOCl)^(2))/(p_(NO)^(2)xx p_(Cl_(2)) )` ` 10^(-3) = ( 2xx 10^(-4))^(2)/ ((198 xx10^(-4))^(2)xx(p-10^(-4))) ` or ` (p- 10^(-4)) = 4/(198)^(2) xx 1/ (10^(-3))= 0* 102 orp= 0* 102 + 0* 0001 = 0*1021 "atm "` Volume of the vessel can be calculated as follows : ` PV = NRT or V = (nRT)/P = (0* 2 xx 0* 082 xx 273 )/1 L = 4* 887 L ` To calculate the number of MOLES of `Cl_(2)`, again apply ` PV = nRT or n = (PV)/ (RT) = (0*1021 xx 4* 887 )/(0*082 xx 298) = 0* 204 "mol"` |
|
| 5. |
The shape of molecular orbital depends upon |
|
Answer» SIZE of the molecule |
|
| 6. |
The zeolite used to convert alcohols directly into gasoline is |
|
Answer» ZSM -5 |
|
| 7. |
The reagent which will be suitable to distinguish 1-methoxy-3-methyl-2-butene from isomeric 4-methyl-3-pentene -1-ol is |
|
Answer» BROMINE in chloroform |
|
| 8. |
Which of the following decreases the dissolved oxygen in water |
| Answer» Answer :A | |
| 9. |
To prepare 3-Ethylpentane-3-ol, the reagents needed are |
|
Answer» `CH_(3)CH_(2)MgBr+CH_(3)COCH_(2)CH_(3)` 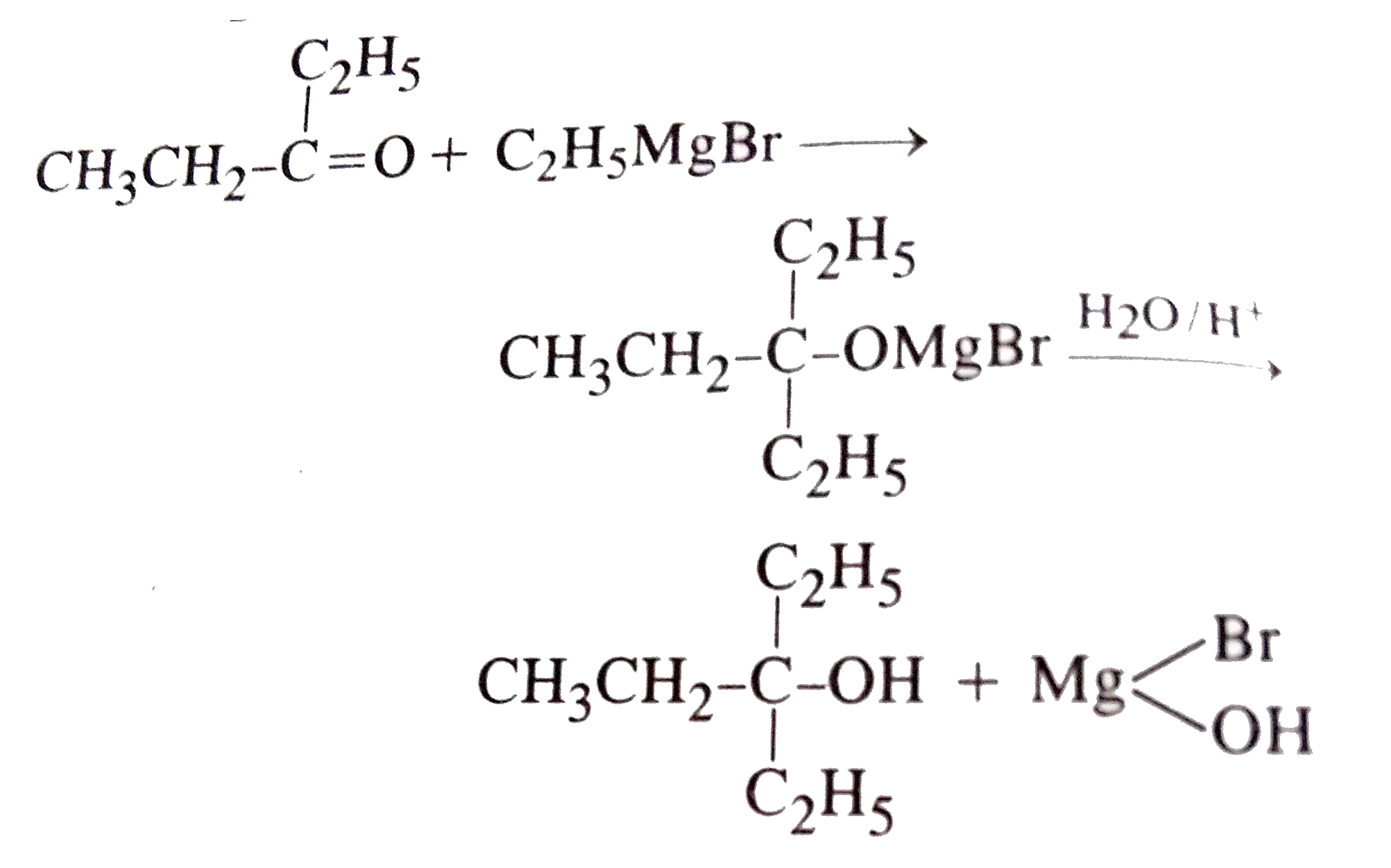
|
|
| 10. |
Which of the following expression correctly represents the relationship between the average kinetic energy of CO and N_(2) molecules at the same temperature? |
|
Answer» `vecE(CO)gtvecE(N_(3))` |
|
| 11. |
What will be the values of psi_(2_(p_(x))),phi_(2_(p_(y))) and phi_(2_(p_(z))) when the value of r=0? |
| Answer» SOLUTION :When r=0, the value of `phi_(2_(px)),phi_(2_(py)) and phi_(2_(pz))` is ZERO (0). | |
| 12. |
The wavelength of a certain line in Balmer Series is observed to be 4329 Å. To what value of .n. does this correspond ? (R_(H) = 109678cm^(-1)) (Z=1) |
|
Answer» `implies1/(4329xx10^(-8)) = 1.09678 XX 10^5 [1/4 - 1/(n_2^2)]` `THEREFORE n_2 = 5` |
|
| 13. |
Which of the following is a nonaromatic. |
|
Answer»

|
|
| 14. |
The stability of alkyl free radicals is due to |
|
Answer» HYPERCONJUGATION |
|
| 15. |
Which type of reactions are easily performed by alkane and alkene ? |
| Answer» SOLUTION :Free radical SUBSTITUTION REACTION and ELECTROPHILIC ADDITION reactions respectively. | |
| 16. |
What is the normality of 250 mL aqueous solution of H_(2)SO_(4) having pH=0.00. |
|
Answer» 0.50 M |
|
| 17. |
Which of the following can give nucleophilic addition on alkyne |
|
Answer» `Cl_(2)` |
|
| 18. |
What amount of ice (in g) will remain when 52 g ice is added to 100 g of water at 40^(@)C? Specific hate of water is 1 cal//g and latent heat of fusion of ice is 80 cal//g Round off your answer to next interger. |
|
Answer» |
|
| 19. |
What is the wave length of H_(b) line Balmer series of hydrogen spectrum ? (R = Rydberg constant) |
|
Answer» 36/5R |
|
| 20. |
What is the best description of the change that occurs when Na_(2)O,(s) is dissolved in water? |
|
Answer» Oxidation number of OXYGEN increase 
|
|
| 21. |
Which of the following is an amorphous solid ? |
|
Answer» Graphite (C ) |
|
| 22. |
Which of the following has maximum % of s-character- |
|
Answer» `N_(2)H_(2)`  . .
|
|
| 23. |
Which of the following metal is not in liquid state? |
|
Answer» GALLIUM |
|
| 24. |
What do you mean by critical volume (Vc)? Give the unit of coefficient of viscosity. |
| Answer» SOLUTION :The VOLUME of 1 MOLE of GAS at critical temperature is called critical volume. Poise or 1 g `cm^(-1)s^(-1)` | |
| 25. |
Wrong statement regarding 'London smog' is |
|
Answer» Observed in WINTER season |
|
| 26. |
When alcohol reacts with concentrated H_(2)SO_(4) intermediate compound formed is |
|
Answer» CARBONIUM ion 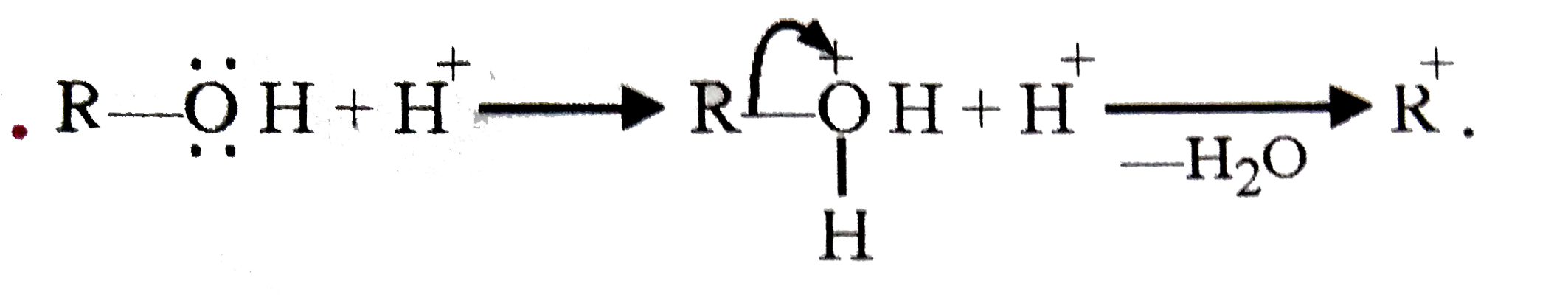
|
|
| 27. |
Which is soluble in water |
|
Answer» SODIUM PALMITATE |
|
| 28. |
Which of the following property of water can be used to explain the sperical shape of rain droplets ? |
|
Answer» Viscosity |
|
| 29. |
What isthe atomic number of element whose outer most electrons are represented by 3s^1 |
| Answer» Solution :COMPLETE ELECTRONIC configuration is `1S^(2)2s^(2)2p^(6)3^(1)therefore`Atomic NUMBER of the element = 11 | |
| 30. |
Which one of the following has the lowest melting point |
|
Answer» B |
|
| 31. |
Which of the following isomer of butanol has a chiral structure? |
|
Answer» `(CH_(3))_(3)COOH` `CH_(3)-underset("2-Butanol")underset(H)underset(|)overset(OH)overset(|**)(C)-CH_(2)-CH_(3)` |
|
| 32. |
Which of the following atoms and ions are isoelectronic i.e. have the same numbe of electrons with the neon atom |
| Answer» Solution :`F^-` have the same number of ELECTRONS with the neon atom | |
| 33. |
When pressure is increased upon a gas at constant temperature |
|
Answer» the R.M.S. velocity DECREASES |
|
| 34. |
When Na_2CO_3 . 10 H_2O is kept open in air, it(a)absorbs moisture(b)loses water to form anhydrous salt(c) loses water to form a monohydrate(d) decomposes to give CO_2 and Na_2O. |
|
Answer» ABSORBS moisture |
|
| 35. |
Which of the following are correct statements ? |
|
Answer» Van der WAAL constant a is a MEASURE of attractive force |
|
| 36. |
Which one of the following has highestdipole moment ? |
|
Answer» `NH_(3)` |
|
| 37. |
What is the minimum number of carbon atoms of an alkane must have to form an isomer? |
|
Answer» 4 |
|
| 38. |
Which of the following have the highest value of electronegativity? |
|
Answer» Halogens |
|
| 39. |
Which of the following will not give a white pt. when AgNO_(3) is added to its solution? |
|
Answer» `C Cl_(4)` |
|
| 40. |
What is the amount of work done when two moles of an ideal gas is comoressed from a volume of 1 m^(3) to 10 dm^(3) at 300 K against a pressure of 100 kPa? |
|
Answer» 99 KJ `p_(ext = 100 KP = 10^(5)Pa` `therefore w = 10^(5)Pa(1 - 0.001)m^(3)` ` = 10^(5) xx 99.9 xx 10^(-2) Pam^(3)` ` = 99.9 xx 10^(3)J (because Pa m^(3) = J)` = 99.9 kJ |
|
| 41. |
Which is the equation showing the relation between K_p and K_c ? |
|
Answer» `K_p=K_c(RT)^(DELTAN)` |
|
| 42. |
Volume of a molecule is related to Vander Waal's constant 'b' and Avagadro Number 'N_(0)' by the equation : |
|
Answer» `V = b/(N_0)` |
|
| 43. |
What would bethe IUPACname andsymbol for theelementwithatomicnumber120 ? |
|
Answer» SOLUTION :Fromtable3.6gage `3//12` the rootsfor 1,2and o are un , BI and NIL respectively. `:. ` the IUPAC name of theelement =un + bi + nil + ium = UNBINILIUM and symbolof the element = ubn. |
|
| 44. |
What happen when calcium oixde is treated with carbondioxide? |
|
Answer» SOLUTION :Calcium oixde on TREATING with `CO_(2)` give calcium CARBONATE. `CAO + CO_(2) to CaCO_(3)` |
|
| 45. |
The wavelength of the electron emitted, when in a hydrogen atom, electron falls from infinity to stationary state 1, would be (Rydberg constant = 1.097 xx 10^(7) m^(-1)) |
|
Answer» 91 nm `= 1.097 xx 10^(7) m^(-1) ((1)/(1^(2)) - (1)/(OO^(2)))` `= 1.097 xx 10^(7) m^(-1)` or `lamda = (1)/(1.097 xx 10^(7)) m = 0.91 xx 10^(-7) m = 91 nm` |
|
| 46. |
Which of the following is named as perhydrol and used as an antiseptic? |
| Answer» Answer :b | |
| 47. |
Write the equilibrium expression and equilibrium constant for the given reverse reaction. |
|
Answer» Solution :`N_(2)(g)+3H_(2)(g)hArr2NH_(3)(g),K=0*50` at 6734 `K_(c)=([N_(2)][H_(2)]^(3))/([NH_(3)]^(2))`. For the reverse reaction, `K_(c)=0.50rArrK_(E)'=1/K_(e)=1/0.50=2`. |
|
| 48. |
Write the general electronic configuration of group 14 elements. |
| Answer» SOLUTION :[Rare GAS ] `NS^(2) np^(2)` | |
| 49. |
What volume of 5M Na_2SO_4 must be addedto 25 mL of 1M BaCl_2 to produce 10 g of BaSO_4 ? |
|
Answer» 8.58 mL No. MOLES of `BaSO_4=w/M=10/233=0.0429` `THEREFORE " No. of moles of " Na_2SO_4 " NEEDED " = (MxxV)/(1000)` or `0.0429=(5xxV)/(1000) RARR V=8.58mL` |
|
| 50. |
What volume of hydrogen at N.T.P would be liberated by the action of 50 mL of dilute H_(2)SO_(4) of 40 % purity and having a specific gravity of 1.3 g mL^(-1) on 65 g of zinc ? (Atomic mass of Zn = 65) ? |
|
Answer» `=(50 mL )xx ("1.3 g mL"^(-1))=65g` Mass of pure `H_(2)SO_(4)` in the sample `= ((65g)xx40)/(100)=26g` The balanced chemical equation is : `Zn+underset(underset(98g)("1 mol"))(H_(2)SO_(4))rarrZnSO_(4)+underset(underset(22.4L)("1 mol"))(H_(2))` 98 g of `H_(2)SO_(4)` EVOLVE `H_(2)` at N.T.P = 22.4 L 26 g `H_(2)SO_(4)` evolve `H_(2)` at N.T.P `= ((22.4L))/((98.0g))xx(26.0g)=5.94L`. |
|







