Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is Boyle temperature ? |
| Answer» Solution :The TEMPERATURE at which a REAL GAS behaves like an ideal gas over an appreciable pressure range is CALLED Boyle temperture or Boyle POINT. | |
| 2. |
Which pH value is more in the following ? (a)0.1 M HCl and 0.1 M NaOH (b) 0.1 M HCl and 0.01 M HCl (c) 0.1 M NaOH and 0.01 M NaOH |
| Answer» SOLUTION :(a)0.1 M NAOH ,(B)0.01 M HCL , (b)0.1 M NaOH | |
| 3. |
Which of the following cannot act as Lewis acid ? |
|
Answer» `C Cl_(4)` |
|
| 4. |
What is the oxidation state of CO in [CO(H_(2)O)_(5)CI]^(2+) ? |
|
Answer» `+2` `x+5xx0+(-1)=+2` `THEREFORE x=+2+1=+3` |
|
| 5. |
When copper is heated with conc. HNO_(3) it produces |
|
Answer» `Cu(NO_(3))_(2),NOandNO_(2)` |
|
| 6. |
Which of the following compunds exhibit gemetrical isomerism? Draw their cis-trans isomers (i) CH_3-CH=C Cl_2(ii) CH_3-CH=CHCl(iii)ClCH=CHCl(IV)CH_3-CH_2-CH=CH_2 |
Answer» Solution :Compound (II) and (III) show GEOMETRICAL ISOMERISM.
|
|
| 7. |
The solubilityof PbSO_4 "in"0.01 M Na_2SO_4 solution is (K_(sp)of PbSO_4 is 1.25 xx 10^(-9) ) |
|
Answer» ` 1.25 XX 10 ^(-7) M` ` S( 0.01 )= 1.25 xx 10 ^(-9), S =1 .25 xx 10 ^(-7) ` |
|
| 8. |
Which of the following compounds will give a secondary alcohol on reaction with Grignard reagent followed by acid hydrolysis ? (I) HCHO (II) C_(2)H_(5)CHO (III) CH_(3)COCH_(3) (IV) HCOOC_(2)H_(5) Select the correct answer using the codes given below. |
|
Answer» II only |
|
| 9. |
Which of the following arrangements represent increasingg oxidation number of the central atom? |
|
Answer» `CrO_(2)^(-),ClO_(3)^(-),CrO_(4)^(2-),MnO_(4)^(-)` |
|
| 10. |
What's the main difference between lone pair and bonded pair of electrons. |
| Answer» Solution :a valence set of two electrons that EXISTS WITHOUT bonding or SHARING with other atoms is CALLED lone pair. Pair of electrons involved in a covalent bond is called bonding pair. | |
| 11. |
Which of the following is secondary alkyl halide? |
|
Answer» ISO BUTYL CHLORIDE |
|
| 12. |
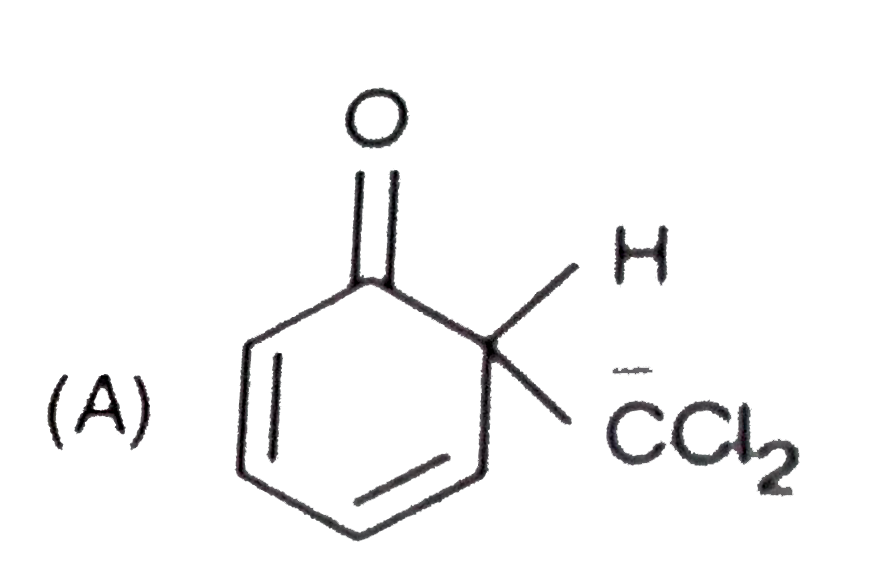
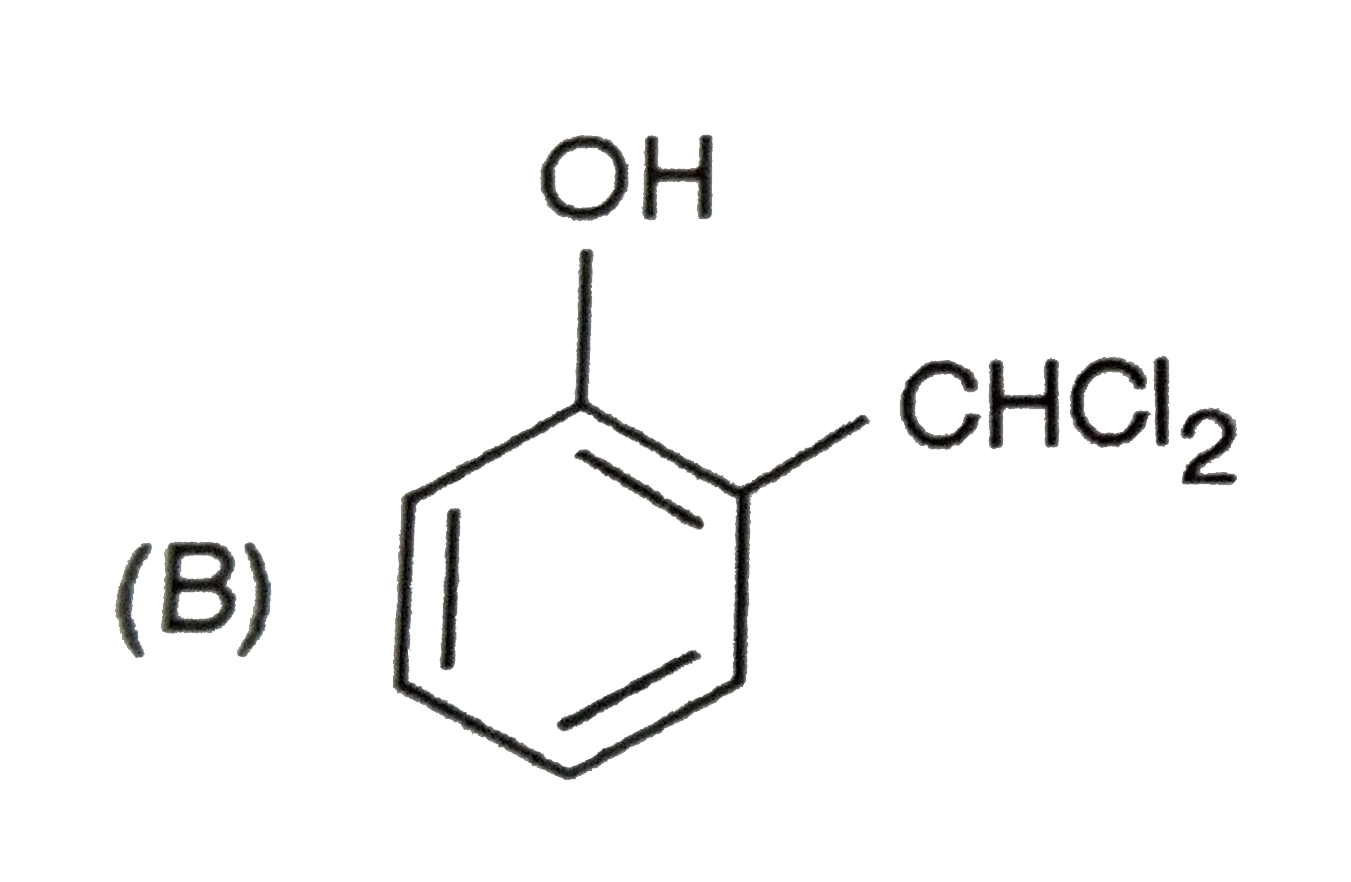
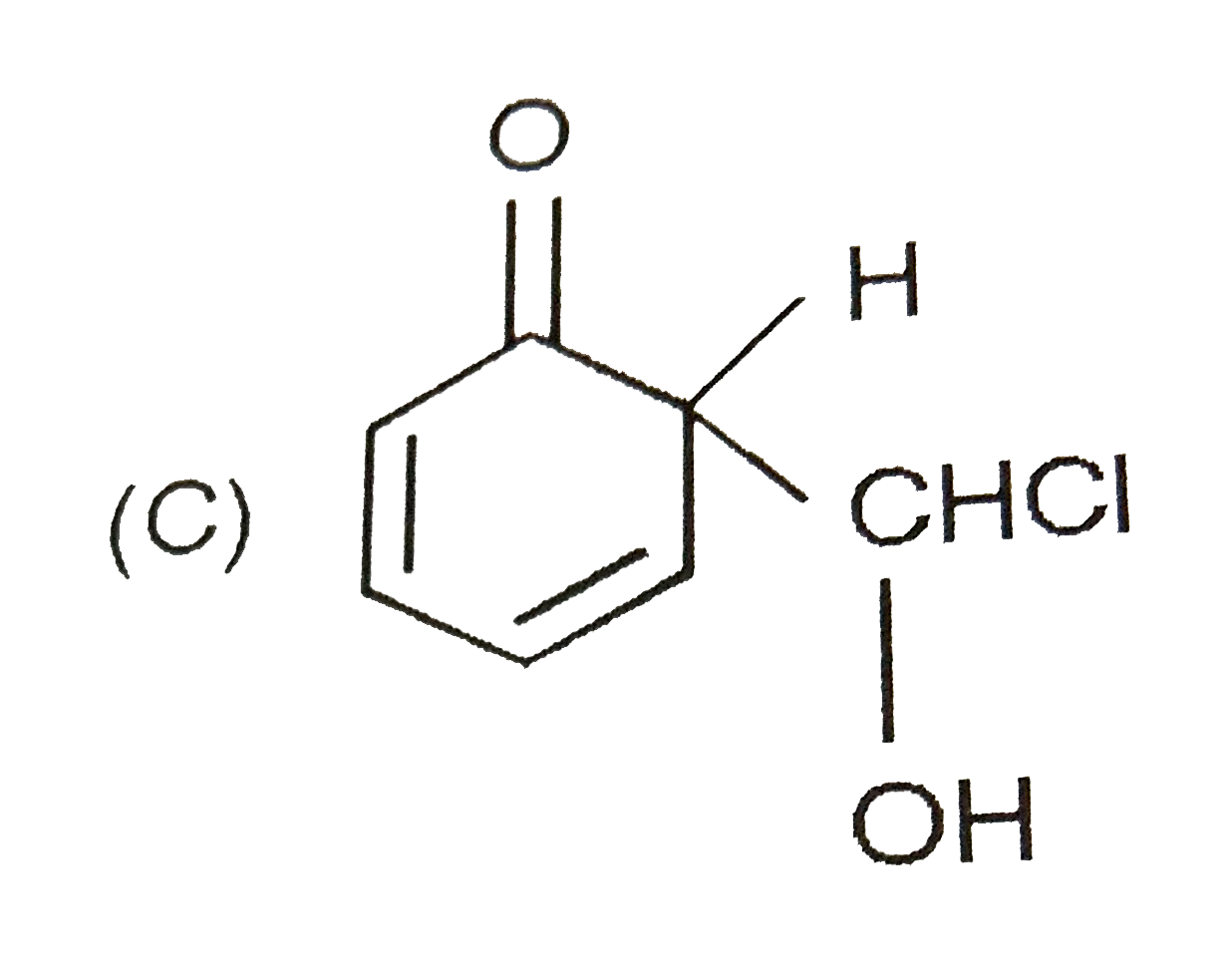
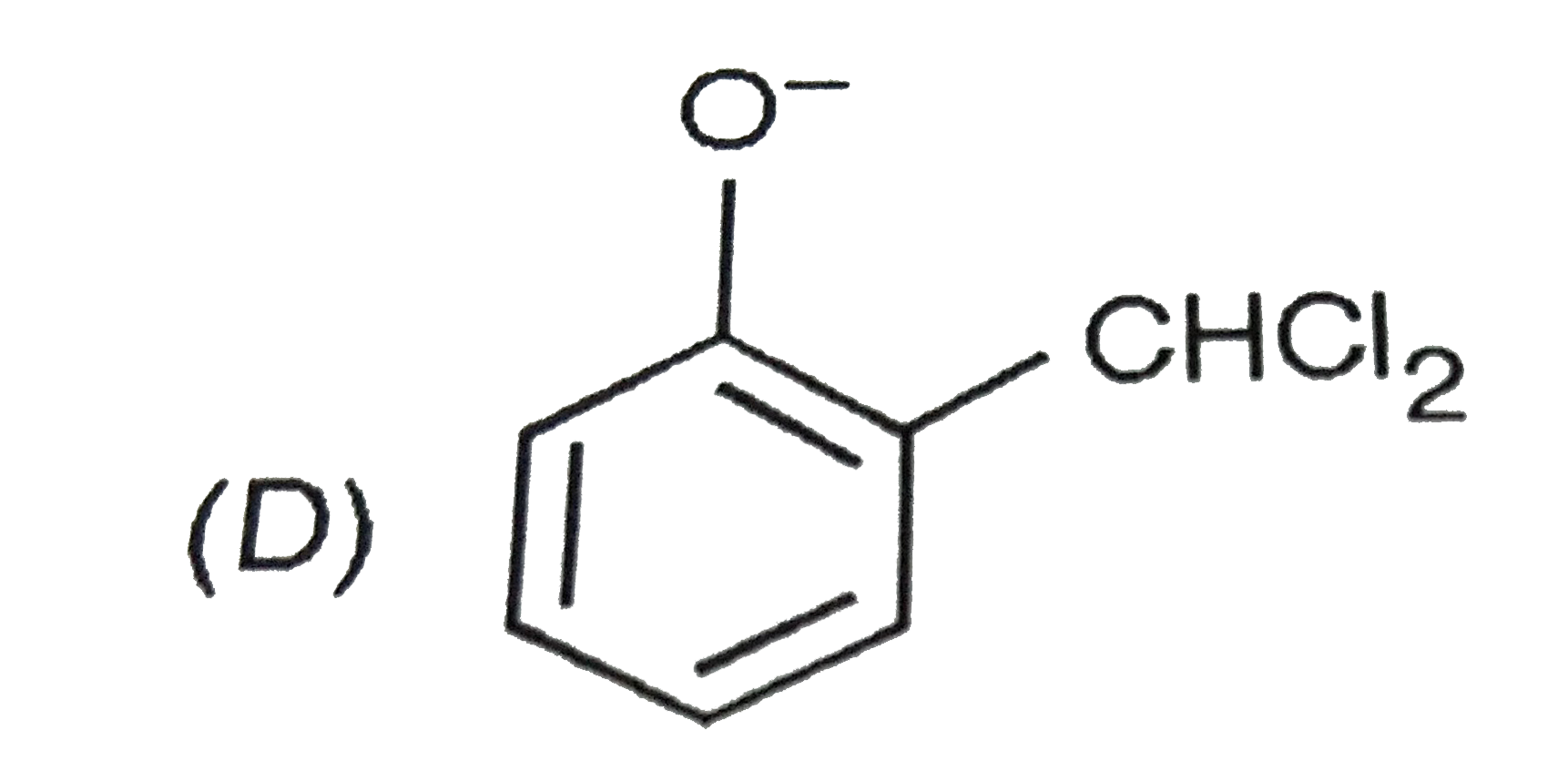
When phenol is treated with CHCl_(3) and NaOH followed by acidification, salicyladehyde is obtained. Which of the following species are involved in the above mentioned reaction as intermediates? |
|
Answer»
|
|
| 13. |
What iselectronconfigurationof atom?Explainitsmethodby example. |
|
Answer» Solution :Thedistributionof electronintoorbitalsof an atmin calleditselectronicconfiguration method-1 :Thesubshellisrepresentedby therespectiveylettersymboland thenumber ofelectronspresentin thesubshellisdepicted assubshellrepresentedfor differentshellsisnumberbeforethe respectivesubshell. Example : Electronconfigurationexpressbyorbitalnotationof B (z=5)(z= 6)are asunder  Method -2:(Electron configurationby orbitaleachorbitalof thesubshellisrepresentedbyboxis as under  the electrons isrepresentedby NARROW (i) a POSITIVE`(+(1)/(2))`SPINOR anarrow`(to)`1A negativespin `(-(1)/(2))` Example: theorbitalpictureof theseelementscan berepresentedas follows : 
|
|
| 14. |
Thesubstance usedpurefatsandoilsis _______. |
|
Answer» SODIUMCARBONATE |
|
| 15. |
Which one of the following reactions is not associated with the Solvay process of manufacture of sodium carbonate ? |
|
Answer» `NH_(3)+H_(2)O+CO_(2)toNH_(4)HCO_(3) ` |
|
| 16. |
What is the product of each reaction |
|
Answer» |
|
| 17. |
What is the main difference between ionic bond and covalent bond ? |
|
Answer» Solution :In Ionic bond POSSESSING molecule ions have - opposite change and because of opposite charge they are stabilize by electrostatic force of attraction and POSSESS low energy. While the molecules having covalent bond the atoms combine by joint sharing of their valence ELECTRON and attain stable electron configuration like inert gas so energy decreases and molecule is stable. The atoms having more difference in electronegativity form ionic bond. The atom having LESS difference in electronegativity form covalent bond. |
|
| 18. |
What is one amu or u ? |
|
Answer»
1 amu `=1.66056xx10^(-24) GM` |
|
| 19. |
Which colour is formed in the Lassaigne's test for nitrogen? |
|
Answer» PRUSSIAN BLUE colour |
|
| 20. |
The values of Kp_(1) and Kp_(2) for the reaction given below are in the ratio 2: 1. If degree of dissociation of X and A are equal, then total pressure of equilibrium (1) and cquilibrium (2) are in the ratio X hArr Y + Z , Kp_(1) : A hArr 2B : Kp_(2) |
|
Answer» `Kp_(1)=(((alpha_(1))/(1+alpha_(1)).p_(1)) xx ((alpha_(1))/(1+alpha_(1)).p_(1)))/(((1-alpha_(1))/(1+alpha_(1)) xx p_(1)))` `KP=(alpha_(1)^(2) xx p_(1))/(1-alpha_(1)^(2))` ...(1) `underset(1-alpha_(1))(A) harr underset(2alpha_(1))(2B), Kp_(2)` `Kp_(2)=(((2alpha_(1))/(1+alpha_(1)) xx p_(2))^(2))/((1-alpha_(1))/(1+alpha_(1)) xx p_(2)), Kp_(2)=(4alpha_(1)^(2) xx p_(2))/(1-alpha_(1)^(2))` ...(2) `((1))/((2))(Kp_(1))/(Kp_(2))=(1)/(4) xx (p_(1))/(p_(2)) IMPLIES (p_(1))/(p_(2))=4 xx (Kp_(1))/(Kp_(2))=4 xx 2=8` |
|
| 21. |
Which of the following equation depicts reducing nature of H_2O_2 ? |
|
Answer» `2[Fe(CN)_6]^(4-) + 2H^(+) + H_2O_2 to 2[Fe(CN)_6]^(3-) + 2H_2O` `I_2 + H_2O_2+ 2OH^(-) to 2I^(-) + 2H_2O + O_2` `2MnO_4^(-) + 3H_2O_2 to 2MnO_2 + 3O_2 + 2H_2O + 2OH^(-)` |
|
| 22. |
What happens when slaked lime is treated with dry chlorine? |
| Answer» SOLUTION :`2CA(OH)_2Cl_2 RARR CaCl_2+CaSiO_3` | |
| 23. |
What are the elements present in group 2? Give their general electronic configuration. |
| Answer» SOLUTION :Group 2 contains beryllium, magnesium, calcium, STRONTIUM, BARIUM and radium. Their general ELECTRONIC configuration is (noble gas] `ns^(2)` | |
| 24. |
Which of the following statements is notcorrect ? |
|
Answer» The fraction of the total VOLUME OCCUPIED by the atoms in a PRIMITIVE cell is 0.48. |
|
| 25. |
The values of x and y in the following redox reaction xCI_(2)+6 OH^(-) rarr CIO_(3)^(3) rarr CIO_(3)^(-)+y CI^(-)+3 H_(2)O are |
|
Answer» x=2, y=4 |
|
| 26. |
The study of diple moment of a molecule is useful to explain the shape of a molecule and also to predict a The net dipole moment of a polyatomic molecule is the resltant of the different bond moments present in that molecule . The values are generalluy expressed in Debye or in the S.I. units in terms of Coulomb meter (C m) A covalent molecule , X-Y is found to have a dipole moment of1.5 xx10^(-29) C m and a bond length of 150 pm . The per cent ionic character of the bond will be |
|
Answer» `50%` ` = 2.4 xx 10^(-29) `C m `mu_("calculated")=1.5 xx10^(-29) C m` `therefore % `ionic CHARACTER`=(1.5 xx10^(-29) ) / (2.4xx 10^(-29)) XX100 = 62.5%` |
|
| 27. |
What is borax glass? |
| Answer» SOLUTION :`Na_2 B_4 O_7` | |
| 28. |
The study of diple moment of a molecule is useful to explain the shape of a molecule and also to predict a The net dipole moment of a polyatomic molecule is the resltant of the different bond moments present in that molecule . The values are generalluy expressed in Debye or in the S.I. units in terms of Coulomb meter (C m) Which our of the following will have maximum dipolemoment ? |
|
Answer» `NF_(3)`  F is more electronegative than N ,N-F bond OPPOSES the bond moment of N with the LONE pair . In all order cases , it favours but electronegativity differenceis maximum between N and H . Hence, `NH_(3)` has maximum DIPOLE moment . (Electronegativity of Cl = N = 3.0) |
|
| 29. |
Which of the following statement(s) is (are) correct? |
|
Answer» The `p^(H)` of `10^(-8)` M SOLUTION of `HCl` is `8` |
|
| 30. |
The study of diple moment of a molecule is useful to explain the shape of a molecule and also to predict a The net dipole moment of a polyatomic molecule is the resltant of the different bond moments present in that molecule . The values are generalluy expressed in Debye or in the S.I. units in terms of Coulomb meter (C m) 1 Debye is equivalent to |
|
Answer» `3.33 xx10^(-30) ` C m |
|
| 31. |
When 50 gm of a sample of sulphur was burnt in air 4% of the sample was left over. Calculate the volume of air required at STP containing 21% oxygen by volume. |
|
Answer» Solution :Weight of sample of sulphur TAKEN =50g PERCENTAGE of impurity =4% Weight of impurity in sample `=(4 xx 50)/(100)=2gr` Weight of sulphur in sample =50-2=49grams Combustion of sulphur GIVES sulphurdioxide `S+O_(2) to SO_(2)` 1 mole of S=1 mole of `O_(2)` 32grams of S=22.4 L of `O_(2)` at STP 48 GRAMS of S=? Volume of oxygen required at STP `=(48)/(32) xx 22.4=33.6 L` 21 L of `O_(2)` is present in 100L of air 33.6 L of `O_(2)` present in ? Volume of air required at STP `=(33.6)/(21) xx 100=160L` |
|
| 32. |
The value of Delta_(f) H^( Theta ) for NH_(3) is -91.8 "kJ mol"^(-1). Calculate enthalpy change for the following reaction: 2NH_(3(g)) to N_(2(g)) + 3H_(2(g)) |
|
Answer» SOLUTION :`(1)/(2) N_(2(g)) + (3)/(2) H_(2(g)) to NH_(3(g)) , Delta_(f) H^( Theta ) = -91.8 "kJ mol"^(-1)` (`Delta_(f) H^( Theta )` means ENTHALPY of formation of 1 mole of `NH_(3)` ) `therefore` Enthalpy change for the formation of 2 moles of `NH_(3)` `N_(2(g)) + 3H_(2(g)) to 2NH_(3(g)) , Delta_(f) H^( Theta )= 2 xx -91.8 =-183.6 "kJ mol"^(-1)` for the reverse REACTION, `2NH_(3(g)) to N_(2(g)) + 3H_(2(g)) , Delta_(f) H^( Theta ) = + 183.6 "kJ mol"^(-1)` Hence, the value of `Delta_(f) H^( Theta ) ` for `NH_(3)` is `+183.6 "kJ mol"^(-1)`. |
|
| 33. |
Which is the principle of paper chromatography? |
|
Answer» Adsorption |
|
| 34. |
Which of the following compound does not undergo addition reaction? |
|
Answer» Ethyne |
|
| 35. |
What happens to the probability of finding the electron in the m.o.'s after the combination of two atomic orbitals ? |
|
Answer» SOLUTION :Probability (electron DENSITY) increases in the INTERNUCLEAR REGION of bonding m.o., WHEREAS it decreases in the antibonding m.o. |
|
| 36. |
Which of the following disproportionation reaction |
|
Answer» `2Cu^(+) rarrCu^(2+) + Cu` |
|
| 37. |
When 30 ml of 5.93 millimolar solution of AgNO_(3) was added to 2.0 ml of 8.89 millimolar solution of KCl, the mixture turns turbid. The solubility product of AgCl is |
|
Answer» `1.96xx10^(-6) "mol"^(2)L^(-2)` `M_(1)V_(1)=M_(2)V_(2), i.e., 30xx5.93xx10^(-13)=32xxM_(2) "or" M_(2)=5.56xx10^(-3)M` `[Cl^(-)] : 2.0xx8.89xx10^(-3)=32xxM_(2) " or" M_(2)=5.56xx10^(-4)M` `:.` Ionic product of AGCL `=[Ag^(+)][Cl^(-)]` `=(5.56xx10^(-3) "mol" L^(-1))(5.56xx10^(-4) "mol" L^(-1))` `=3.09xx10^(-6) "mol"^(2) L^(-2)` which exceeds (a) but not(b). UNITS in (C) and (d) are not correct. |
|
| 38. |
Two oxides of copper contain respectively 88.8% and 79.91 % of copper. Calculate equivalent masses of copper in each of the oxide. |
|
Answer» |
|
| 39. |
Which of the following is incorrect statement ? |
|
Answer» `SnF_4` is ionic in NATURE |
|
| 40. |
What is the molarity of H_2SO_4 solution which contains 98% H_2SO_4 by weight and whose specific gravity is 1.84 ? |
|
Answer» 1.84 |
|
| 41. |
Which of the following is the main product in the ozonolysis of 1,4-dimethylcyclohexene followedby a reductive workup with Zn and ethanoic acid? |
|
Answer»
|
|
| 42. |
When 1 mole of N_2 and 1 mole of H_2 is enclosed in a 5 L vessel and reaction is allowed to attain equilibrium . It is found that at equilibrium there is x mole of H_2 . The number of moles of NH_(3) formed would be |
|
Answer» `(2X)/(3)` |
|
| 43. |
What mass of NHO_3 is needed to convert 5g of iodine into iodic acid according to the reaction ? |
|
Answer» 12.4 g eq. `HNO_3=I_2,OVERSET(+5)(NO_3^(-))overset((1))RARROVERSET(+4)(NO_2)` `w_1/E_1"" =w_2/E_2,overset(0)I_2rarroverset(5)(2IO_3^(-))+5` `w_1 = (5xx10xx63)/(254) = 12.4 ` GM `1 + 14 + 48 = 63` |
|
| 44. |
Which of the following reactions will yield 2,2-dibromopropane ? |
|
Answer» `CH_3-CH=CH_2+HBr to ` |
|
| 45. |
Which of the following compound is known as oil of winter gree ? |
|
Answer» PHENYL benzoate |
|
| 46. |
Which is not a s- block element ? |
|
Answer» `[Ar]4s^(2)3d^(10)4p^(6)5s^(1)` |
|
| 47. |
Which one of the following orders presents the correct sequence of the increasing basic nature of the given oxides ? |
|
Answer» `MgO LT K_(2)O lt Al_(2)O_(3) lt Na_(2)O` `Al_(2)O_(3) lt MgO lt Na_(2)O lt K_(2)O` Group 1 oxides are more basic than group 2 oxides. Basic character of oxides increases down the group. |
|
| 48. |
What is theheat of formation of C_(6)H_(6), given that the heats of combustion of benzene, carbon and hydrogen are 782, 94 and 68K. Cal respectively |
|
Answer» `+14` KCAL `DELTA H_("comb") = 6 Delta H_(f_(CO_(2))) + 3 Delta H_(f_(H_(2)O)) - Delta H_(f_(C_(6)H_(6)))` `-782 = 6 XX (-94) + 3 xx (-68) - Delta H_(f_(C_(6)H_(6)))` `rArr Delta H_(f_(C_(6)H_(6))) = + 14` Kcal |
|
| 49. |
What are hybridisation states of each carbon atom in the following compounds? (i) CH_(2) = C = O (ii) CH_(3)CH = CH_(2) (iii) (CH_(3))_(2)CO (iv) CH_(2) = CHCN (v) C_(6)H_(6) |
Answer» SOLUTION :
|
|
| 50. |
When alpha-D glucose is dissolved in water , it undergoes mutarotation to form an equilibrium mixture of alpha - D glucose and beta - D glucose containing 63*6 % of the latter. Calculate K_(c) for the mutarotation. |
|
Answer» Solution :`{:(,alpha -" D GLUCOSE ",hArr,beta - " D glucose "),(" At EQUILIBRIUM " ,36*4%,,63*6 %):}` ` K_(c)(63*6)/(36*4) = 1* 747` |
|