Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following diagrams correctly describes behaviour of a fixed mass of an ideal gas? (T is measured in K) |
|
Answer»
|
|
| 2. |
What do you understand by the term 'auto protolysis of water'? What is the significance? |
| Answer» SOLUTION :For ANSWER, CONSULT Q.18 | |
| 3. |
Which of the following tests can be applied to distinguish 1^(@), 2^(@) and 3^(@) alcohols? |
| Answer» Answer :D | |
| 4. |
Which of the following is not an ideal solution ? |
|
Answer» Benzene & toluene |
|
| 5. |
Which structural factor favours the formation of stable hydratesfromaldehydes ande ketones? |
|
Answer» The presence of ELECTRON- WITHDRAWING groups on the ALKYL carbons |
|
| 6. |
Which of the following is the least stable ? |
|
Answer» `CH_(3)-CH_(2)-OVERSET(+)(C)H_(2)` |
|
| 7. |
Which of the following is least likely to behave as Lewis acid ? |
|
Answer» `OH^(-)` |
|
| 8. |
Which colour is produced when alkali metals dissolved in liquid ammonia? |
|
Answer» Red |
|
| 9. |
Which of the following conditions show the polluted environment. |
|
Answer» PH of rain WATER is 5.6 |
|
| 10. |
Which order are correct? |
|
Answer» Themal stability: `BeSO_(4) lt MgSO_(4) lt CaSO_(4) ltSrSO_(4) lt BaSO_(4 )` |
|
| 11. |
Which of the following hydrides of group 16 elements has the highest boiling point ? |
|
Answer» `H_(2)O` |
|
| 12. |
Which element has ns^(2)np^(5)type electronic configuration in its outermost shell |
|
Answer» CARBON |
|
| 13. |
Which of the following is the correct IUPAC name? |
|
Answer» 3-Ethyl-4,4-dimethylheptane |
|
| 14. |
Which of the following compounds is aromatic? |
|
Answer»
|
|
| 15. |
Which of the following is not present in portland cement? |
|
Answer» `Ca_(3)Al_(2)O_(6)` |
|
| 16. |
Which of the following does not exhibit hydrogen bonding in liquid phase |
|
Answer» Phenol |
|
| 17. |
Which of the following is not the effect of ozone bole. |
|
Answer» AGEING of skin |
|
| 18. |
What do you understand by linear combination of atomic orbitals in MO theory . |
|
Answer» SOLUTION :Linear combination of atomic orbitals : The WAVE functions for the molecular orbitals can be obtained by solving Schrodinger wave equation for the molecule. Since solving the Schrodinger equation is too complex , APPROXIMATION methods are used to obtain the wave function for molecular orbitals. The most common method is the linear combination of atomic orbitals (LCAO) . The atomicorbitals are represented by the wave function `Psi`. Let US consider two atomic orbitals represented by the wave function `Psi_(A)" and " Psi_(B)` with comparable energy, combines to form two molecular orbitals. One is bondingmolecular ORBITAL `(Psi_("bonding") )` and the other is antibonding molecular orbital `(Psi_("antibonding"))` . The wave functions for these two molecular orbitals can be obtained by the linear combination of the atomic orbitals `Psi_(A)" and "Psi_(B)` as below. `Psi_("bonding") = Psi_(A) + Psi_(B) ` `Psi_("antibonding") = Psi_(A) - Psi_(B) ` The formation of bonding molecular orbital can be considered as the result of constructive interference of the atomic orbitals and the formation of antibonding molecular orbital can be the result of the destructive interference of the atomic orbitals. The formation of the two molecular orbitals from two1s orbitals is shown below. Constructive interaction : The two 1s orbitals are in phase and have the same sign.  
|
|
| 19. |
Which of the following strategy is not a correct approach to reduce global warming |
|
Answer» Reducing the green house has EMISSION by limiting the use of fossil fules. |
|
| 20. |
Which of the following lattices has the highest packing efficiency ? (i) simple cubic (ii) body-centred cubic (iii) hexagonal close-packed lattice. |
| Answer» SOLUTION :Packing EFFICIENCIES are : Simple cubic= 52.4%, Body-centred cubic=68%, Hexagonal close-packed = 74%. Thus, hexagonal close-packed LATTICE has the highest packing EFFICIENCY | |
| 21. |
When BiCl_3is dissolved in excess of D_2O,the product formed is |
|
Answer» `Bi(OD)_3` |
|
| 22. |
Which one of the following has the smalJes, heat of hydrogenation per mole of the compound ? |
|
Answer» 1-Butene |
|
| 23. |
Which of the following does not contains -COOH groups ? |
|
Answer» Aspirin 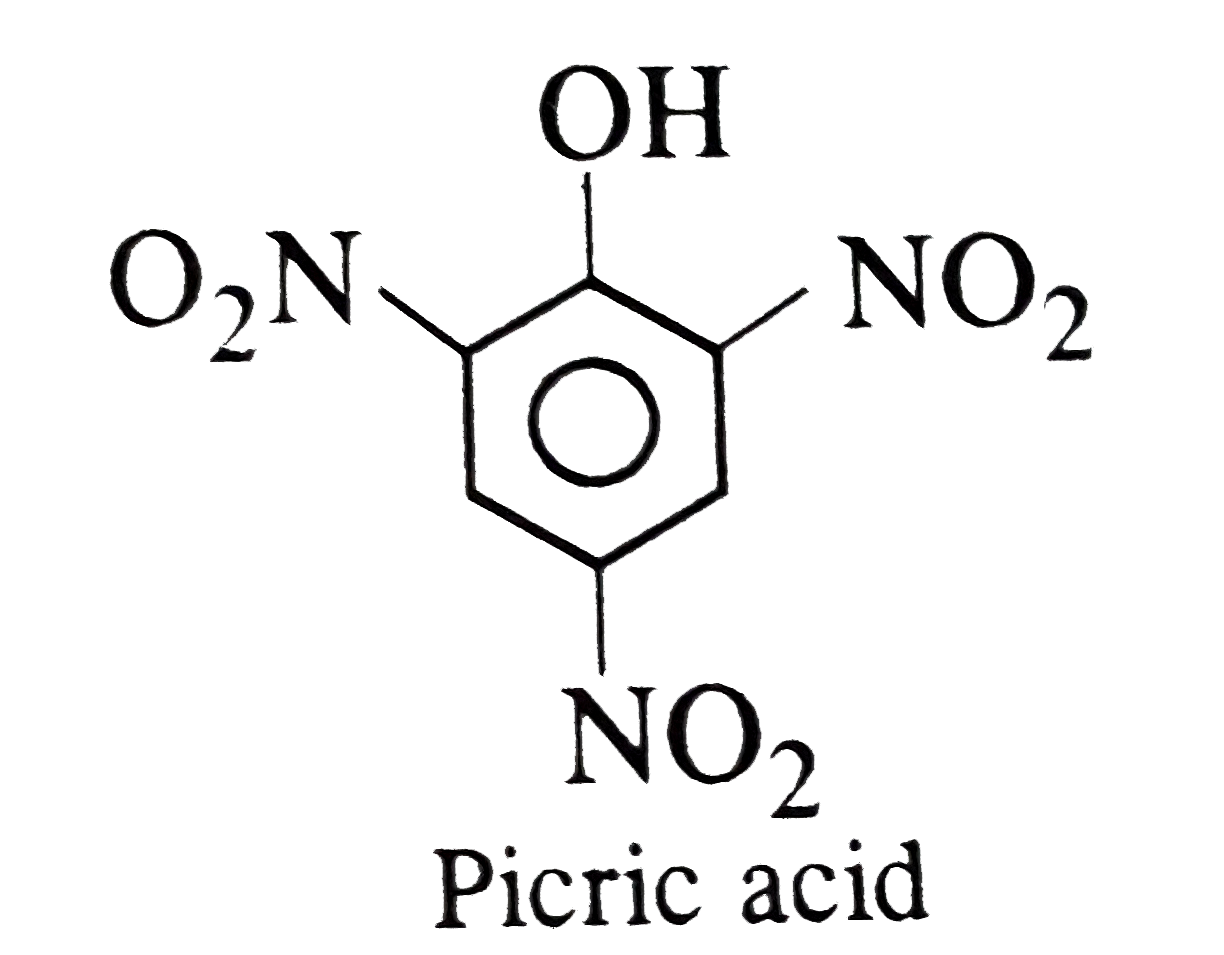
|
|
| 24. |
Which of the following is relatively stronger acid? K, values are given in brackets |
|
Answer» `HA (2xx10 ^(-4) ) ` |
|
| 25. |
The single plant nutrient mainly responsible for eutrophication is ________. |
|
Answer» |
|
| 26. |
Which of the two structures A and B given below is more stabilised by resonance? Explain . {:( CH_(3) COOH ," and ", CH_(3) COO), (A,"" ,B):} |
Answer» Solution :Resonating structures of A and B are as follows ` square ` II is more stable than structure I. because later carriesseparation of positive and NEGATIVE charges . `square ` The contribution of structure II is LESS than that of I . ` square ` On the contrary , structure III and IV are of equal energy and HENCE contribute equally towards the RESONANCE hybrid of compound B . ` squaretherefore ` Structure (B) is more stable than (A) . |
|
| 27. |
Two reactionshaving their energy of activation E_(1) and E_(2) temperature coefficients T_(c_(1)) and T_(c_(2)) respectively within the temperature 300 and 310K. The ratio of their temperature coefficient is: |
|
Answer» `e^(E_(1)//E_(2))` `2.303 log T_(c_(2)) = (E_(2))/(R) [(T_(2) - T_(1))/(T_(1) T_(2))]` `2.303 [log T_(C_(1)) - log T_(C_(2))]` `= ((E_(1) - E_(2)))/(R) xx [(310 - 300)/(300xx310)]` `:. 2.303 log ((T_(C_(1)))/(T_(C_(2)))) = [(E_(1) - E_(2))/(R)] xx 1.07xx10^(-4)` or In `(T_(c_(1)))/(T_(C_(2))) = [(E_(1) - E_(2))/R] xx 10^(-4)` `:. (T_(c_(1)))/(T_(c_(2))) = e^([(E_(1) - E_(2))/(R)]xx10^(-4))` |
|
| 28. |
When an ideal gas expands in vaccum, there is neither absorption nor evolution of work ? Why ? |
|
Answer» <P> SOLUTION :It is because no WORK is done.i.e., `w = 0 ""w = -p_(ext) XX Delta V = 0 xx Delta V = 0`. In `Delta U = q + w` q = 0 because the gas CHAMBER is insulated.`Delta U = 0 + 0 = 0`. |
|
| 29. |
What is the -O-O-H bond angle in solid form of H_2O_2 ? |
| Answer» SOLUTION :In SOLID FORM of `H_2O_2 -O-O-H` BOND ANGLE is `101.9^@`. | |
| 30. |
What is the maximum numbers of electrons that can be associated with the following set of quantum numbers ? n=3, I=1 and m=-1 |
|
Answer» 4 |
|
| 31. |
Which of the following compounds undergo disproportionation in aqueoussolution ? |
|
Answer» `TlCl_(3)` |
|
| 32. |
Which of the following statements is INCORRECT for a real gas ? |
|
Answer» At Boyl TEMPERATURE, compressibility FACTOR, Z = 1 |
|
| 33. |
Which pump is important in biological reaction in human body? |
| Answer» Answer :B | |
| 34. |
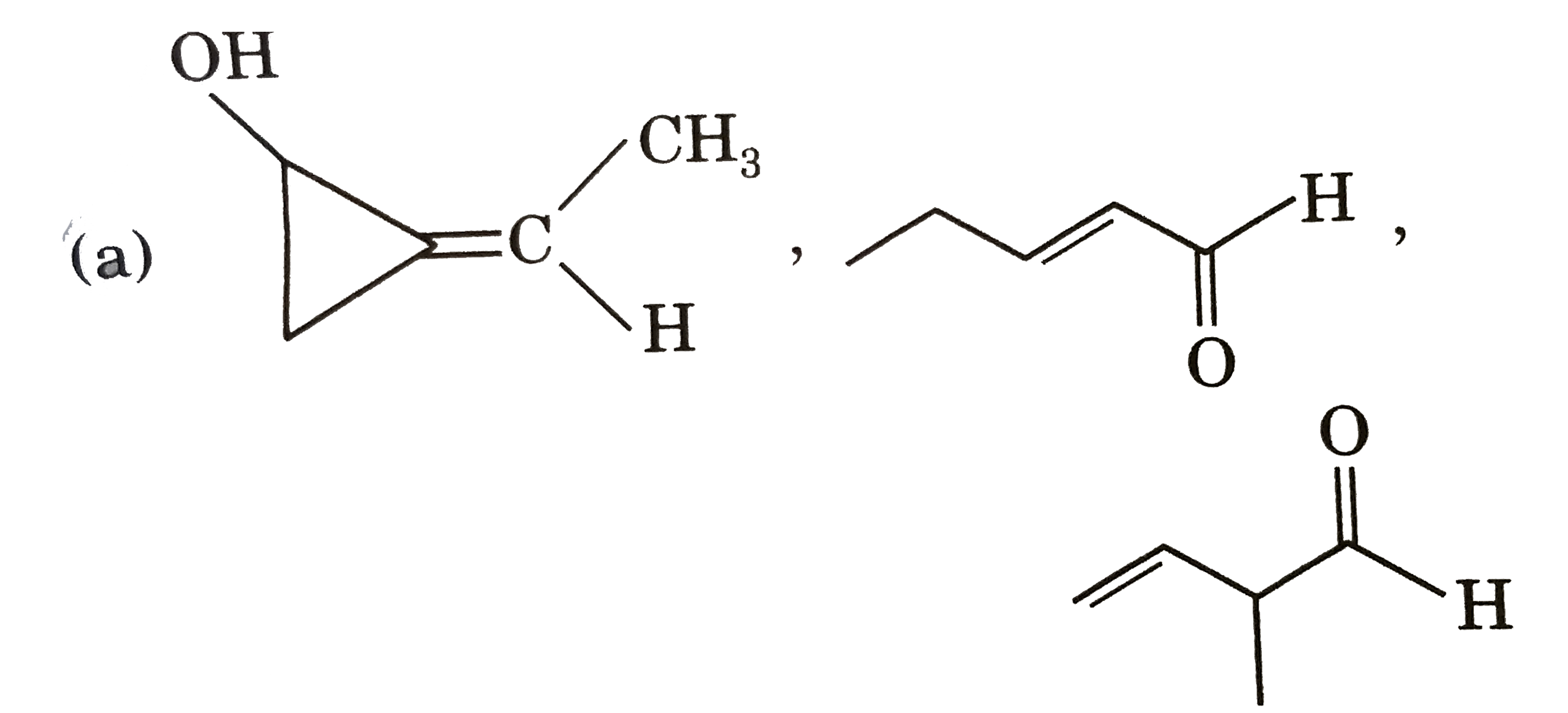
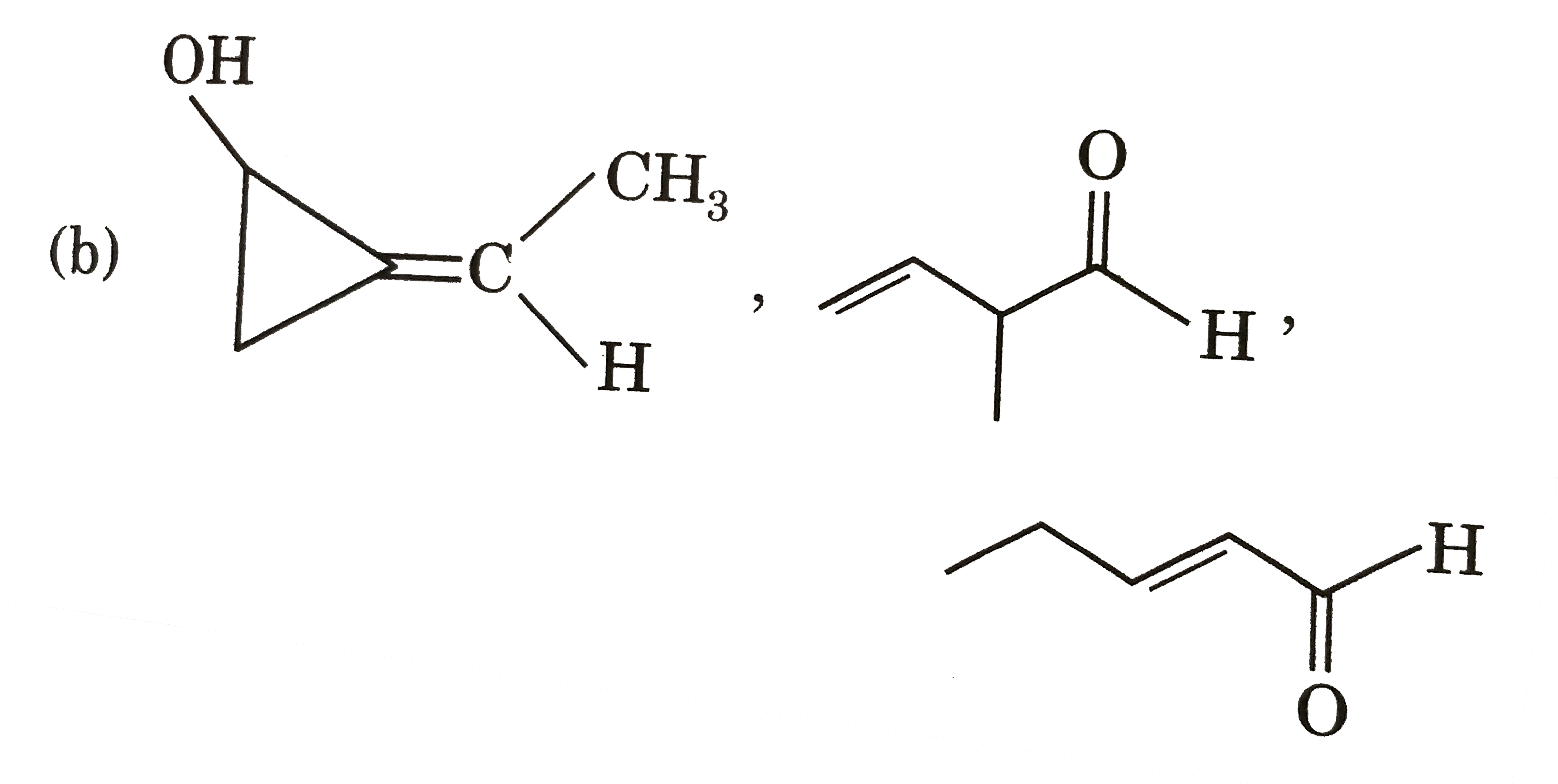
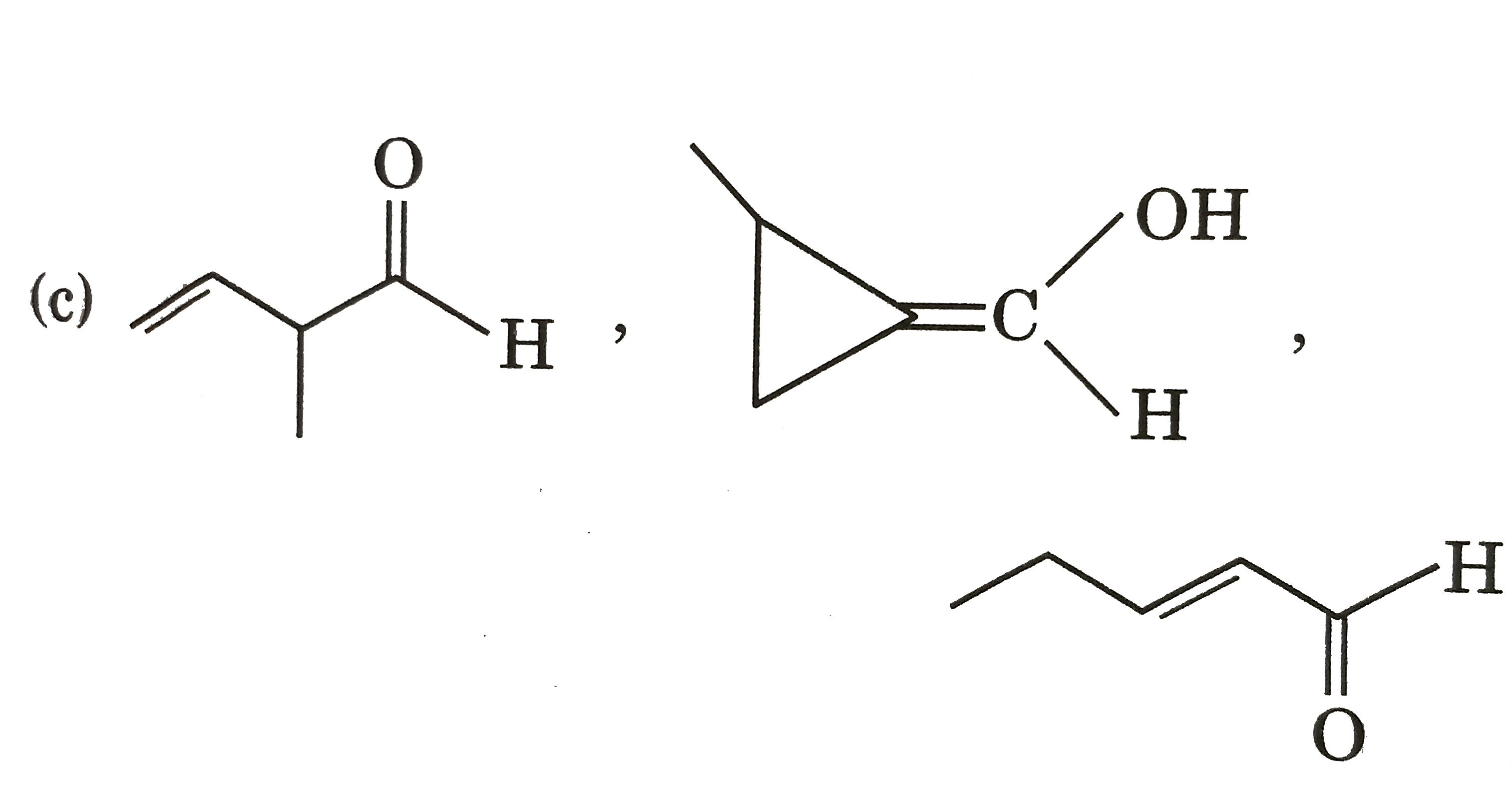
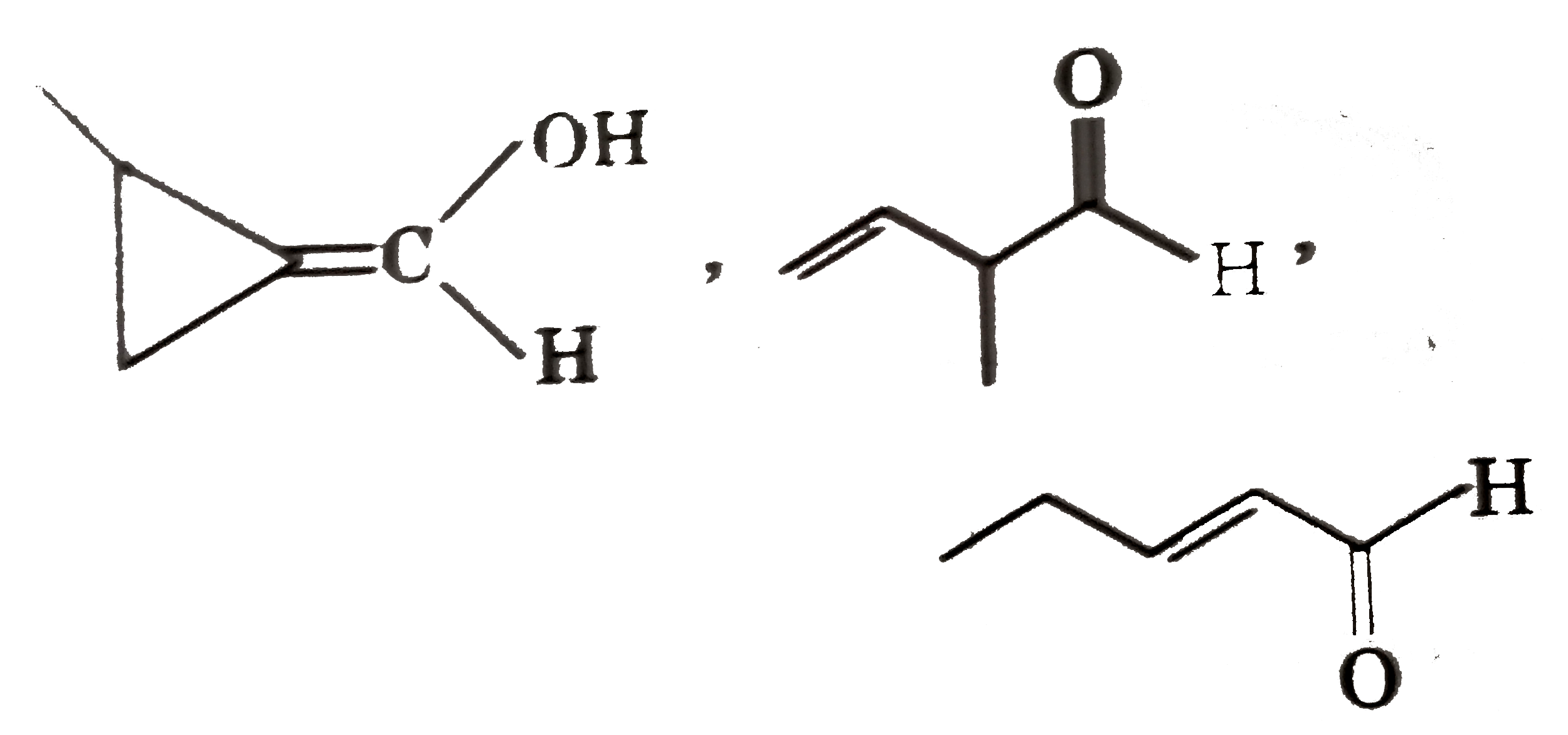
Three structures P,Q and R having seme molecular formula, i.e., C_(5)H_(8)O. Structure (P) shows geometrical isomerism and optical isomerism but not tautomerism. Structure (Q) shows optical isomerism and toutomerism but not gemetrical isomerism. tautomerism but not optical isomerism. The structure of P, Q and R are respectively : |
|
Answer»
|
|
| 35. |
Which of the following chemical reaction depicts the oxidising behaviour of H_(2)SO_(4)? |
|
Answer» (a)NaCI`H_(2)SO_(4) rarrNaHSO_(4)+HCI` |
|
| 36. |
Which of the following is not exhibited by |
|
Answer» MILK |
|
| 37. |
Which of the following statement is incorrect. |
|
Answer» `H_(2)O_(2)` can acts as an OXIDISING agent |
|
| 38. |
Write the significance of a plus and a minus sign shown in representing the orbitals. |
| Answer» SOLUTION :Plus and minus sign CORRESPONDS to sign of electron wave. The CRESTS of a wave are usually GIVEN `+ve` sign and troughs of a wave given `-ve` sign. | |
| 39. |
The total number of orbitals associated with the principal quantum number n = 3 is………………… . |
|
Answer» 9 n=3 ,1 , `m_1 =-1,0,1 -three p ORBITALS n=3, 1=2, `m_1 =-2, -1 , 0, 1,2` - five d orbitials , OVERALL nine orbitals are possible. |
|
| 40. |
What would be the molality of a solution obtained by mixing equal volumes of 30% by weight H_(2)SO_(4) ( d= 1.218 g mL^(-1)) and 70% by weight H_(2)SO_(4) ( d= 1.610g mL^(-1)) ? If the resulting solution has density 1.425g//mL, calculate its molarity. |
|
Answer» |
|
| 41. |
Which of the following is the smallest ring that can accommodate a triple bond? |
|
Answer» Cyclohexyne |
|
| 42. |
Water pollution does not cause |
|
Answer» CHANGE in COLOUR and salinity of water |
|
| 43. |
What is the number of significant figures in each of the following measurement ? (a) 4.425 g (b) 5.6 xx 10^(3) cm (c) "0.000124 m"^(3) |
|
Answer» |
|
| 44. |
Two sparnigly soluble salts AX and BX_(2) have their solubility product constant equal . Which of the following is (are) correct deduction(s) ? |
|
Answer» Solubility of `AX` ISW greater than solubility of `BX_(2)`. `K_(sp) = (BX)_(2) = 4S_(2)^(3)` If `S_(1)^(2) = 4S_(2)^(3)`, then `S_(1) = sqrt(4S_(2)^(3)) = 2(S_(2))^(3//2)` `S_(1) gt S_(2)` |
|
| 45. |
Which of the statements given below are true for the structure of water molecule ? (i) Oxygen undergoes sp^(3) hybridisation. (ii) Due to presence of two lone pairs of electrons on oxygen the H - O - H bond angle is 118.4^(@). (iii) Due to angular geometry the net dipole moment of water is not zero, mu = 1.84 D. |
|
Answer» (i) and (ii) |
|
| 46. |
Which of the following reaction conditions are used for the conversion of ethanol to ethylene? |
|
Answer» con `H_(2)SO_(4)//70^(@)C` |
|
| 47. |
Which gas has maximum kinetic energy between N_(2) and O_(2) at 300 K ? |
| Answer» SOLUTION :KINETIC energy of both GAS have equal because TEMPERATURE is same of both. KE `prop` T. | |
| 48. |
What happened with halogen when AgNO_(3) is added in lassaigne solution of acidify with HNO_(3) ? |
| Answer» Solution :Precipitate of silver HALIDE is FORMED with HALOGEN. `Ag^(+) + X^(-) RARR AGX darr` | |
| 49. |
When the hybridisation state of carbon atom changes from sp^(3) to sp^(2) and finaly to sp, the angle between the hybrid orbitals |
|
Answer» Decreases gradually |
|
| 50. |
What is Empirical formula? Give an example for a compound whose Empirical formula and molecular formula are the same. |
| Answer» Solution :It is SIMPLEST formula of a compound which REPRESENTS RATIO of ATOMS of DIFFERENT atoms in a compound. Eg. `CH_(4)` | |