Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Using curved arrow notation, show the formation of reactive intermediates when the following covalent bonds undergo heterolutic cleavage. (a) CH_(3)-SCH_(3) (b) CH_(3)-CN (iii) CH_(3)-Cu |
Answer» SOLUTION :(a) ALTHOUGH both C and S have same ELECTRONEGATIVITY (2-5) but the cleavage of the C-S BOND occurs towards S ATOM because S being bigger in size than C can disperse the -ve charge more effectively. 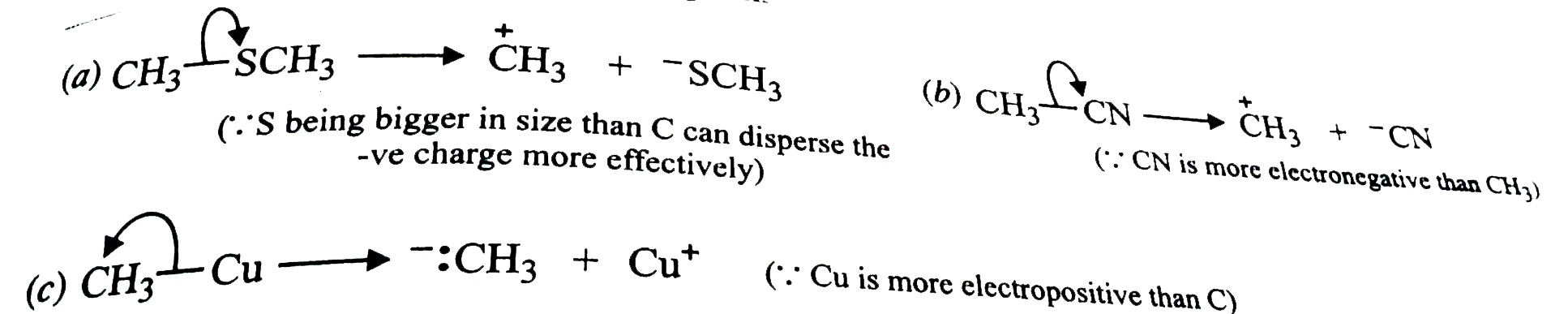
|
|
| 2. |
Which of the following concentration terms is/are independent of temperature ? |
|
Answer» MOLALITY |
|
| 3. |
Which of the following statements is/are correct for CO |
|
Answer» CO is an important fuel B) It forms carboxy haemoglobin with the haemoglobin of the blood C) `HCOOH+H_(2)SO_(4)toCO+H_(2)O` |
|
| 4. |
The volume of O_(2) required for the combustion of 10 lit of methane under the same condition is |
|
Answer» 10 lit |
|
| 5. |
What will be the change in pH by adding of 0.1 M NH_4Cl in 0.1 M NH_4OH (weak base) solution ? (K_b of NH_4OH = 1.77xx10^(-5) ) |
| Answer» SOLUTION :PH of `NH_4OH = 11.12` , pH of `NH_4Cl`= 9.251 DECREASE in pH = 1.87 | |
| 7. |
Which fo the following will release a gaseous product from Pb_3O_4 ? |
|
Answer» `HCl` `Pb_3O_4` is actually a stoichiometric mixture fo 2 mol fo `PBO` and ` 1 ` mol of ` PbO_2`. In `PbO_2`, lead is present in ` +4` oxidation STATE (an unstable state) while in ` PbO`, lead is present in state (an unstable state) while in `PbO`, lead is present in `+2` oxidation state (a stable state). Since lead hds a strong tendency to change from ` +4` state to` +2` state,`PbO_(2)` can act as an oxidizing agent (oxidant). THEREFORE, it oxidizes `Cl^(-)` ion fo ` HCl` into chlorine. On the other hand, `PbO` is a basic oxide. Therefore, it can react with hydrochloric acid `(HCl)` to FORM salt `(PbCl_2)` and water. Hence, the above reaction can be split into two reactions namely : (a) Redox reaction `overset(+4)(Pb) O_(2)+4Hoverset(-1)(Cl) rarr overset(+2)(Pb)Cl_(2)+overset(0)(C)l_(2)+2H_(2)O` (b) Acid-base reaction `2PbO + 4HCl rarr 2PbCl_2 +2H_2O` Since ` HNO_3` ( N is present in the highest oxidation state, +5) itself is an oxidizing agent, there is no reaction between ` PbO_2` and ` HNO_3`, However, an acid-base reaction takes place between ` PbO` and ` HNO_3` : ` 2PbO+4HNO_3 rarr 2Pb (NO_3)_2 +2 H_2O` Thus, it is the passive nature of `PbO_2` against ` HNO_3` that makes the reaction different from the one that follows with ` HCl`. |
|
| 8. |
What is dry ice ? Why is it so called ? |
| Answer» Solution :Solid `CO_(2)`, when it is KEPT in AIR under one atmosphere pressure,it sublimes. In otherwords, unlikeordinary ice, it does not MELT and hence does not wet the SURFACE on which it is kept.Therefore,it is CALLED dry ice. | |
| 9. |
Which kind of property shows CO and Al_(2)O_(3) respectively ? |
|
Answer» NEUTRAL and ACIDIC |
|
| 10. |
What is lithosphere ? |
| Answer» SOLUTION :Lithosphere includes SOIL, rocks and MOUNTAINS which are solid components of EARTH. | |
| 11. |
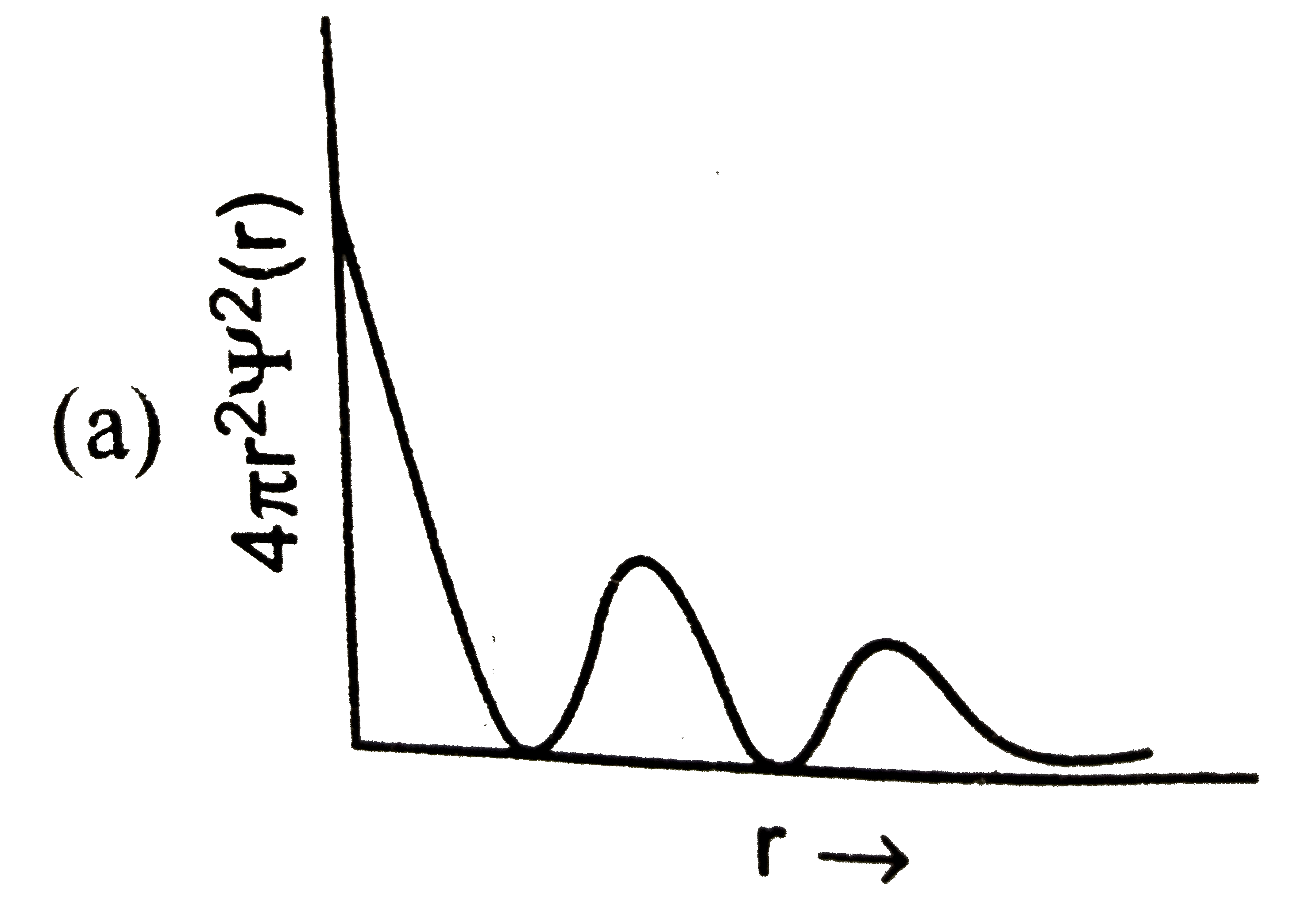
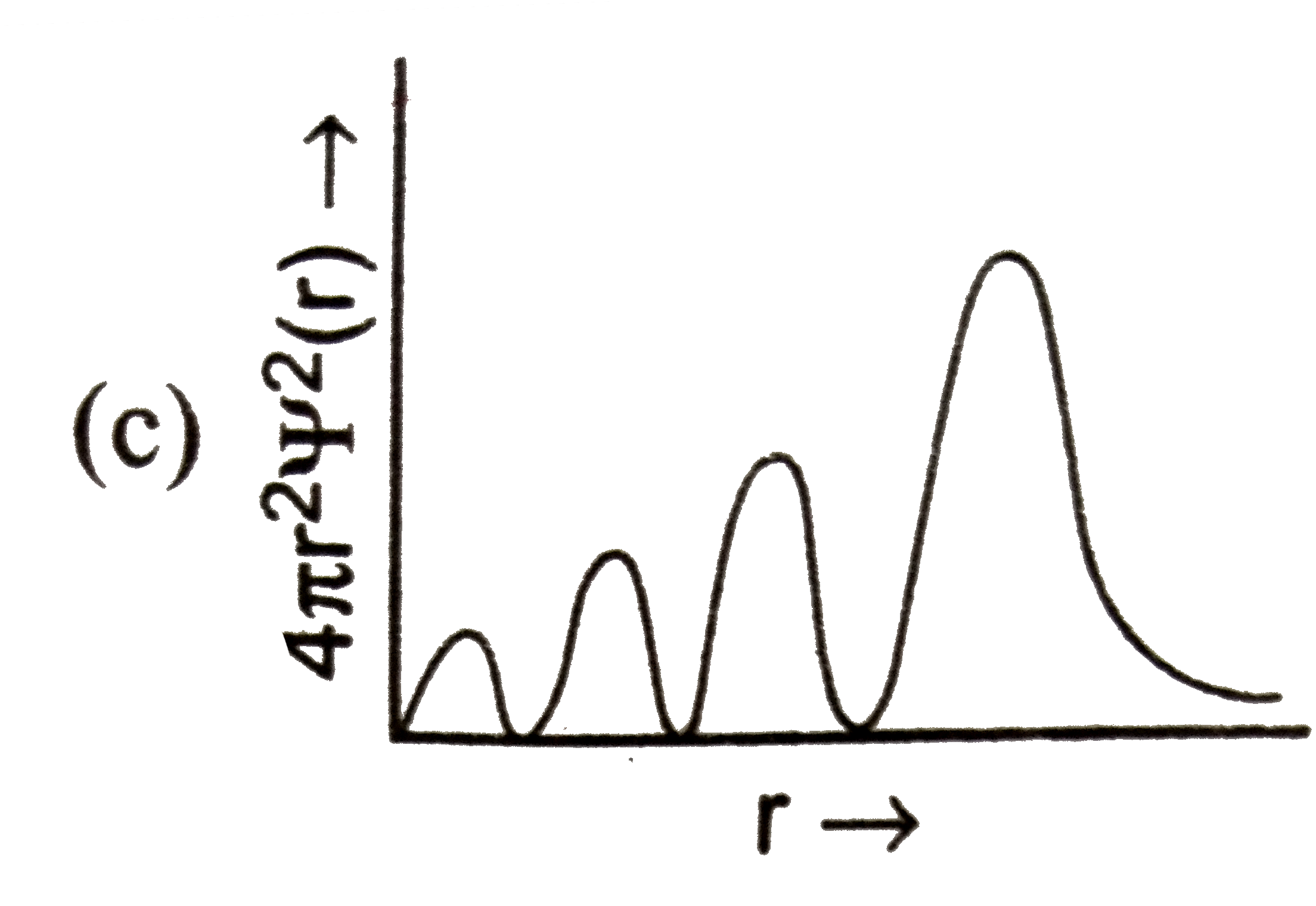
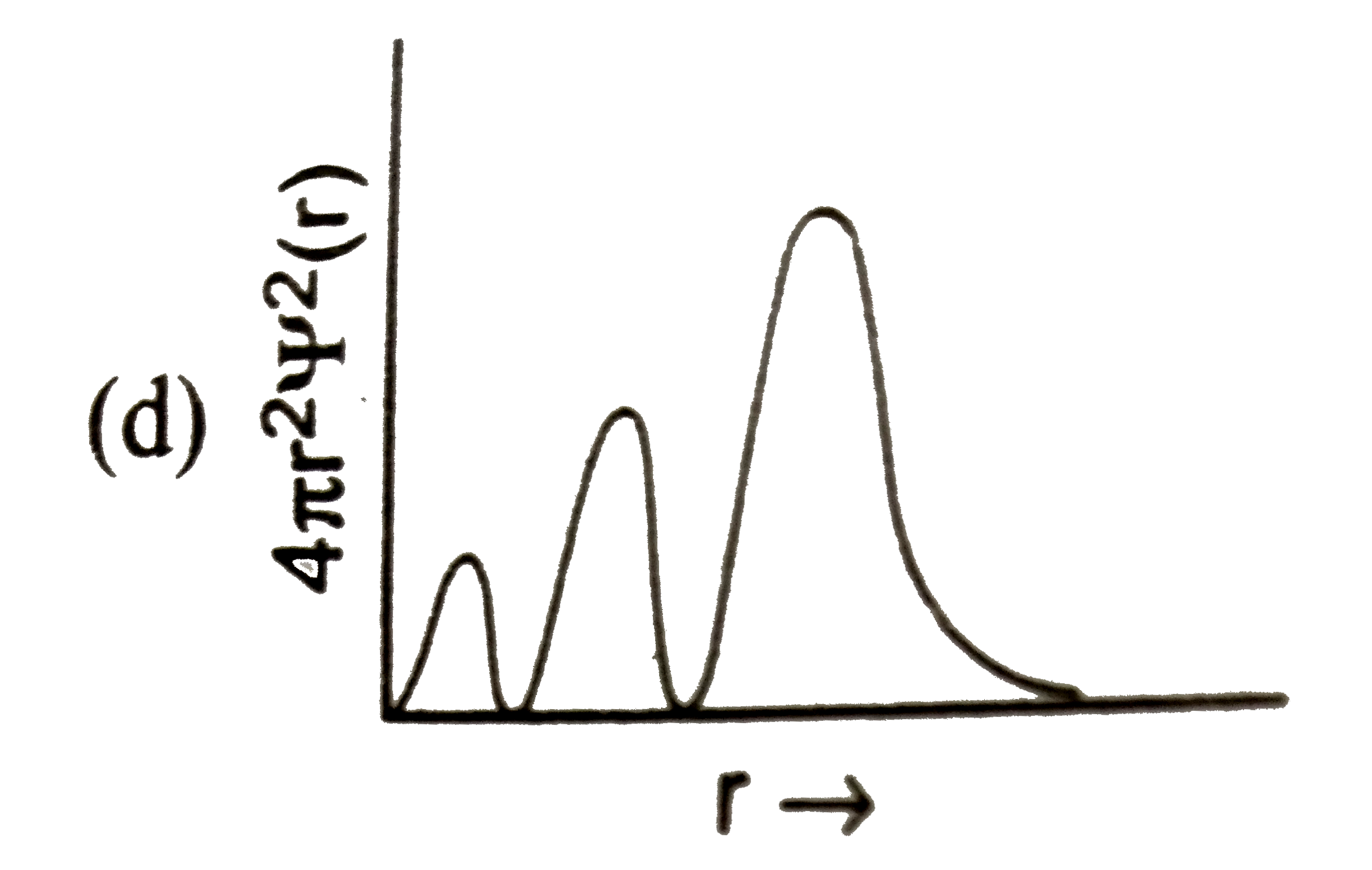
Which of the following graphs between radial probability distribution and radius of atom corresponding to 4s-orbital (n=4,l=0) is correct? |
|
Answer»
|
|
| 12. |
What is the basic difference in approach between the Mendeleev's Periodic Law and the Modern Periodic Law ? |
| Answer» Solution :According to Mendeleev.s PERIODIC law, the physical and CHEMICAL properties of the lements are periodic function of their ATOMIC weight. On the other HAND, according to modem periodic law the properties are periodic function of their atomic umbers. | |
| 13. |
What will be the value of pH of 0.01 mol dm^(-3) CH_3COOH (K_a=1.74xx10^(-5) ) ? |
|
Answer» Solution :If `K_a=1.74xx10^(-5)` Concentration of `CH_3COOH`= 0.01 MOL `dm^(-3)` `[H^+]=sqrt(K_a. C)` `=sqrt(1.74xx10^(-5)xx0.01)=4.17xx10^(-4)` `pH=-LOG[H^+]` `=-log(4.17xx10^(-4))`=3.4 |
|
| 14. |
Which of the following are formed when toluene is chlorinated in presence of FeCl_3 ? |
|
Answer»

|
|
| 15. |
Which will be the proper alternative in place of A in the following equation ? 2Fe^(+3)+Sn^(+2)to2Fe^(+2)+A |
|
Answer» Solution :`2Fe^(+3)+Sn^(+2)to2Fe^(+2)+A` Total charge = `2(+3)+(+2)TO2(+2)+A` = `(+6)+(+2)to(+4)+A` = `(+8)to(+4)+A` So, A = +4 which is in `Sn^(+4)` |
|
| 16. |
Write the electronic configuration of a divalent ion of a coinage metal. |
| Answer» Solution :`CU^(2+) = 1S^(2) 2S^(2) 2p^(6) 3s^(2) 3p^(6) 3D^(9)` | |
| 17. |
Which of the following is/are incorrect for the Humphrey lines of hydrongen spectrum? |
|
Answer» `n_(2)=7ton_(1)=2` |
|
| 18. |
Which is not an oxidising agent ? |
|
Answer» `KClO_3` |
|
| 19. |
The standard enthalpies of formation of SO_2and SO_3 are -297 "kJ mol"^(-1) 396 "kJ mol"^(-1) respectively. Calculate the standard enthalpy of reaction for the reaction:SO_2 + 1//2O_2 to SO_3 |
|
Answer» SOLUTION :`DeltaH_(f)^(@)(SO_(2))=-297 "KJ mol"^(-1)` `DeltaH_(f)^(@)(SO_(3))=-396 "kJ mol"^(-1)` `SO_(2)+1/2O_(2)rarrSO_(3)DeltaH_(r)^(@)=?` `DeltaH_(r)^(@)=(DeltaH_(r)^(@))_("COMPOUND")-sum(DeltaH_(f))_("elements")` `DeltaH_(r)^(@)=DeltaH_(f)^(@)(SO_(3))-(DeltaH_(f)^(@)(SO_(2))+1/2DeltaH_(f)^(@)(O_(2)))` `DeltaH_(r)^(@)=-396" kJ mol"^(-1)-(-297" kJ mol "^(-1)+0)` `DeltaH_(r)^(@)=-396" kJ mol"^(-1)+297=-99 " kJ mol "^(-1)` |
|
| 20. |
Which of the following statement are correct? (A) Ocean is sink for CO_(2) (B) Green house effect causes lowering of temperature of earth's surface (C) To control CO emission by automobiles usually catalytic convertor are fitted into exhaust pipes (D) H_(2)SO_(4) herbicids and insecticides from mist. |
|
Answer» C & B |
|
| 21. |
Which one of the following is covalent in character? |
|
Answer» `BeCl_(2)` |
|
| 22. |
Which of the following reagent not convert compound into alcohol ? |
|
Answer» DiBAL-H 
|
|
| 23. |
The representative elements get the nearest inert gas configuration |
|
Answer» By LOSING ELECTRONS |
|
| 24. |
Which of the following reactions will get affected by increasing the pressure ? Also mention whether change will cause the reaction to go into forward or backward direction ? (i)COCL_(2) (g) hArr CO(g) + Cl_(2) (g)(ii) CH_(4) (g) + 2 S_(2) (g) hArr CS_(2) (g) + 2 H_(2) S (g) (iii) CO_(2) (g) + C (s) hArr 2 CO (g)(iv) 2 H_(2) (g) + CO (g) hArr CH_(3) OH (v)CaCO_(3) (s) hArr CaO (s) + CO_(2) (g)(vi)4 NH_(3) (g) + 5 O_(2) (g) hArr 4 NO (g) + 6 H_(2) O (g) |
|
Answer» Solution :Reactionsaffected will be those in which `(n_(p) != n_(r))_(gaseous)`. Hence , reactions (i), (III), (iv), (V) and (vi) will be affected . By applying Le Chetelier's principle , we can PREDICT the direction . Increase of pressurewill SHIFT the equilibrium to the side in whichthe number of moles is less. (i) `n_(p)=2, n_(r) = 1 , i.e, n_(p) gt n_(r) ,` reaction will go in the backward direction (ii)`n_(p)=3, n_(r) = 3 , i.e, n_(p) gt n_(r) ,`reaction will not be affected by pressure . (iii)`n_(p)=2, n_(r) = 1 , i.e, n_(p) gt n_(r) ,` reaction will go in the backward direction . (iv)`n_(p)=1, n_(r) = 3 , i.e, n_(p) gt n_(r) ,` reaction will go in the fiorward direction . (v) `n_(p)=1, n_(r) = 0 , i.e, n_(p) gt n_(r) ,` reaction WIL go in the backward direction. (vi) `n_(p)=10, n_(r) = 9 , i.e, n_(p) gt n_(r) ,`reaction will goo in the backward direction |
|
| 25. |
When NaCl dissolve in water than what change observe in electrostatic force ? Why ? |
| Answer» Solution :When NACL is dissolve than electrostatic attraction decrease in multiple of 80. As DIELECTRIC constant of water is 80 (it is HIGH) | |
| 26. |
Which of the following is elimination reaction? |
|
Answer» `CH_(3)CH_(2) Cl + KOH rarr CH_(3)CH_(2)OH + KCl` |
|
| 27. |
Write the chemical formula of Inorganic benzene. |
|
Answer» |
|
| 28. |
Which element of group 3 to 13 shows anomalous behaviour ? |
|
Answer» Solution :First ELEMENT of every GROUP shows anomalous BEHAVIOUR. Ex. : Li: first element of group-I Be : first element of group-II Group 13 to 17 like B of F. |
|
| 29. |
Which ions are present in aluminosilicates ? |
| Answer» SOLUTION :Partial displacement of `SI^(4+)` by `Al^(3+)` in `SiO_2` can PRODUCE alumino silicates (Feldspar). | |
| 30. |
Which of the following statements best explain (s) the relationship between two stereoisomers of CH_(3)CH=CHCH_(3) ? |
|
Answer» They are diastereomers  CIS isomerTrans ISOMER Cis and TRANS isomers `IMPLIES` diastereomers |
|
| 31. |
The volume of a given mass of a gas is 100ml at 100^@C. If pressure is kept constant at what temperature will the sample have the volume of 200ml? |
|
Answer» `50^@C` |
|
| 32. |
What happens when chlorine reacts with slaked lime? |
| Answer» Solution :`underset("Slanked lime")(2CA(OH)_2) + 2Cl_2 to underset("Bleaching POWDER")ubrace(CaCl_2 + Ca(OCl)_2) + 2H_2O` | |
| 33. |
Write structures of all the alkenes which on hydrogenation give 2-methylbutane. |
|
Answer» Solution :The basic skeleton of 2-methylbutane is `overset1Coverset2-oversetoversetC|C overset3-C overset4-C` PUTTING double bond at various different positions and satisfying the tetracovalency of each carbon, the STRUCTURES of various alkenes which give 2-methylbutane on hydrogenations are : `underset"3-Methylbut-1-ene"(overset4CH_3-.^(3)oversetoverset(CH_3)|CH-overset2CH=overset1CH_2)` `underset"2-Methylbut-2-ene"(overset1CH_3-.^(2)oversetoverset(CH_3)|C-.^(3)CH-overset4CH_3)` `underset"2-Methylbut-1-ene"(overset1CH=.^(2)oversetoverset(CH_3)|C-overset(3)CH_2overset4CH_3)` |
|
| 34. |
Which of the following is an example of heterocylic aromatic compound? |
|
Answer» THF |
|
| 35. |
Which of the following chemical equation represents the formation of colloidal solution? |
|
Answer» `Cu+CuCl_(2) RARR Cu_(2)Cl_(2)` |
|
| 36. |
Whichofthefollowinggstructurereoresentsaketone ? |
|
Answer» `CH_(3) CH_(2) -O-CH_(2) CH_(3)` |
|
| 37. |
Which one of the following statements is correct in reversible reaction A catalyst |
|
Answer» Increases the RATE of forward reaction |
|
| 38. |
Write formulas for the following compounds : Chromium (III) oxide |
| Answer» SOLUTION :`Cr_(2)O_(3)` | |
| 39. |
What is used in ion exchange method ? |
| Answer» Solution :Both (A) and (B) | |
| 40. |
Which compound is formed when sodium is heated in oxygen ? |
| Answer» SOLUTION :`Na_2O_2` | |
| 42. |
Write oxidation number of .C. in acetic acid. |
|
Answer» Solution :`CH_(3)COOH:CH_(3)-overset(O)overset(||)(C)-O-H` Oxidatio number of ALKYL group which is attached to CARBONYL group will be zero. `(CH_(3))+1(C)+2(O)+1(H)=0` `therefore0+C+2(-2)+(+1)=0` `thereforeC=+3` |
|
| 43. |
What is thebasicdifference inbetweenthe termselectrons gainenthalpyandelectronegativity . |
| Answer» Solution :Bothelectrongainenthalpy and electronegativityrefer to the tendency of theatom OFAN element toattractelectrons. Whereaselectrongainenthalpy REFERS to THETENDENCY of an isolatedgaseousatom to acceptanadditionalelectronto FORMA gaseousnegativeion, electronegativityrefers to the tendencyof the atomof an elementto attractthe sharedpair ofelectrons towardsit in a COVALENTBOND. | |
| 44. |
Which of the following fumes in moisture in air ? |
|
Answer» `BeCl_(2)` |
|
| 45. |
Which d - orbitals are involved in sp^(3)d^(2) hybridisation in SF_(6)? |
| Answer» SOLUTION :`d_(X^(2)-y^(2)) and d_(Z^(2))`. | |
| 46. |
Whichof thefollowingstatementis incorrectregardingcathoderays ? |
|
Answer» a) Characteristicscathode raysdo notdependsupon thematerialof ELECTRODE. |
|
| 47. |
When propyne is treated with aqueous H_2SO_4 in the presence of HgSO_4. the major product is |
|
Answer» Propanol |
|
| 48. |
What is the formula of sodium pyro phosphate ? |
|
Answer» `Na_4P_2O_7` |
|
| 49. |
Which of the following properties is not shown by NO ? |
|
Answer» Its BOND order is 2.5 |
|
| 50. |
What are enantiomers ? Draw the structures of the possible enantiomers of 3-methylpent-1-ene. |
Answer» SOLUTION :STEREOISOMERS which are non-superimposable mirror IMAGES of each other are called enantiomers. The enantiomers of 3-methylpent-1-ene are SHOWN on R.H.S. 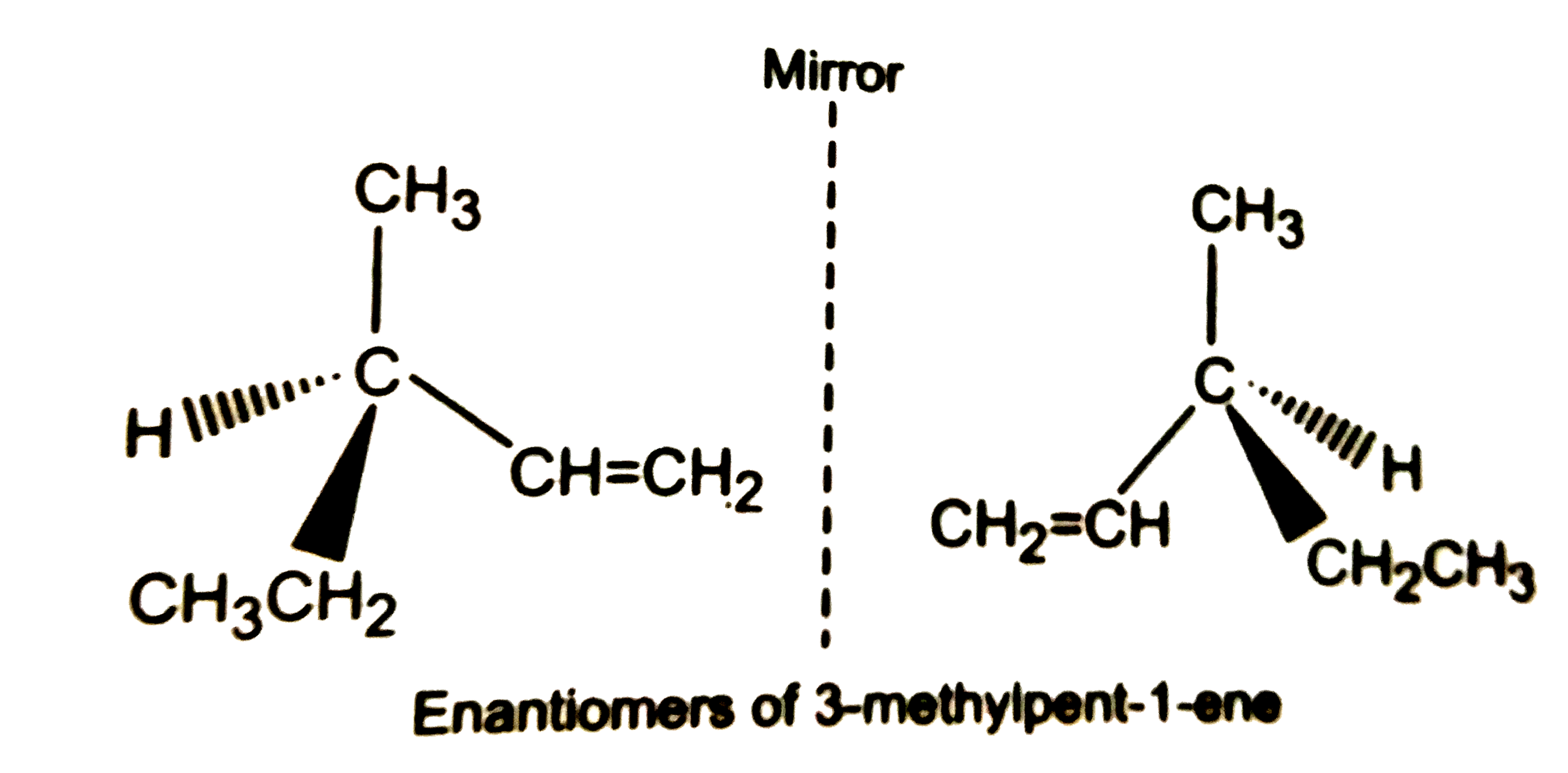
|
|