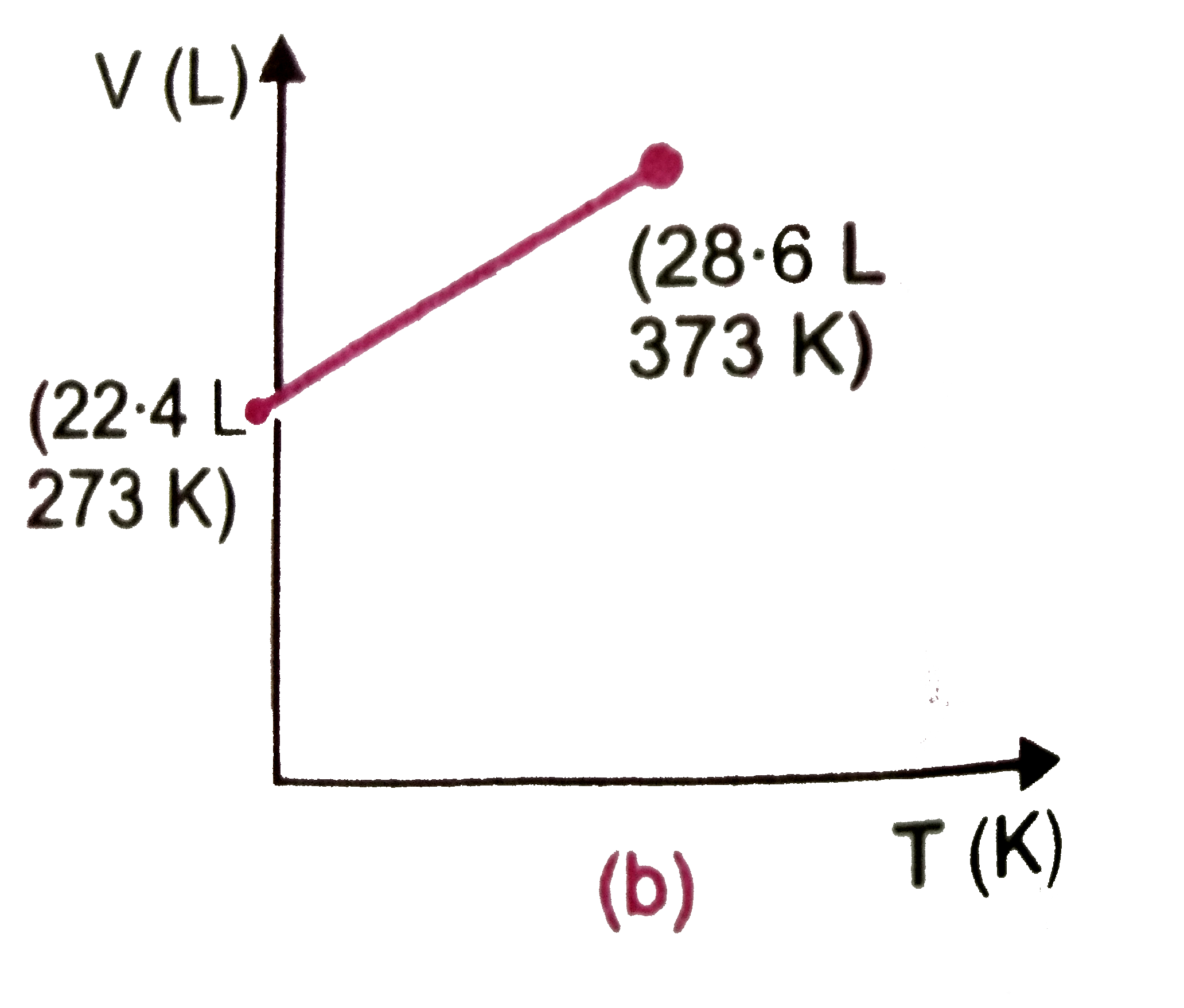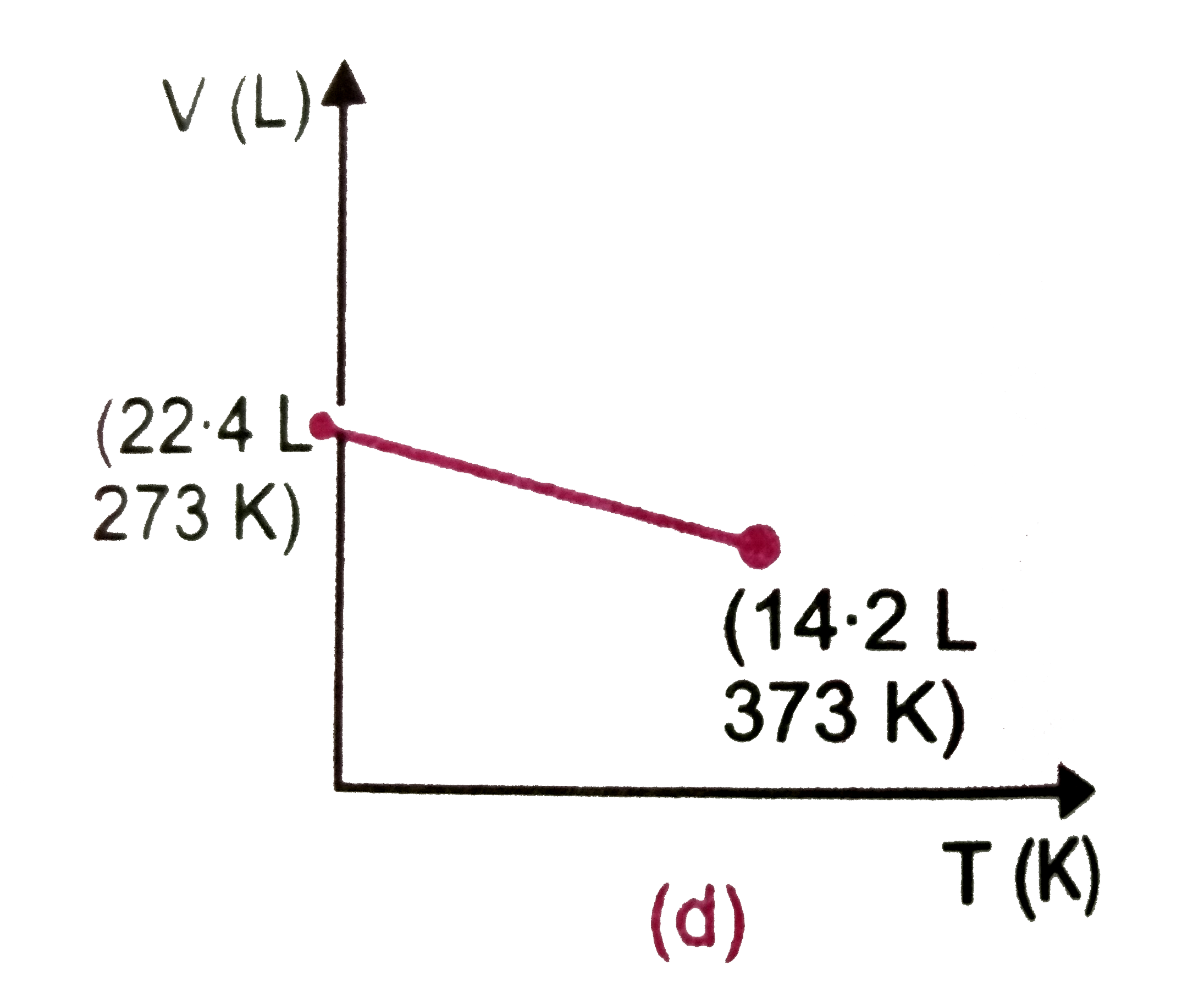Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
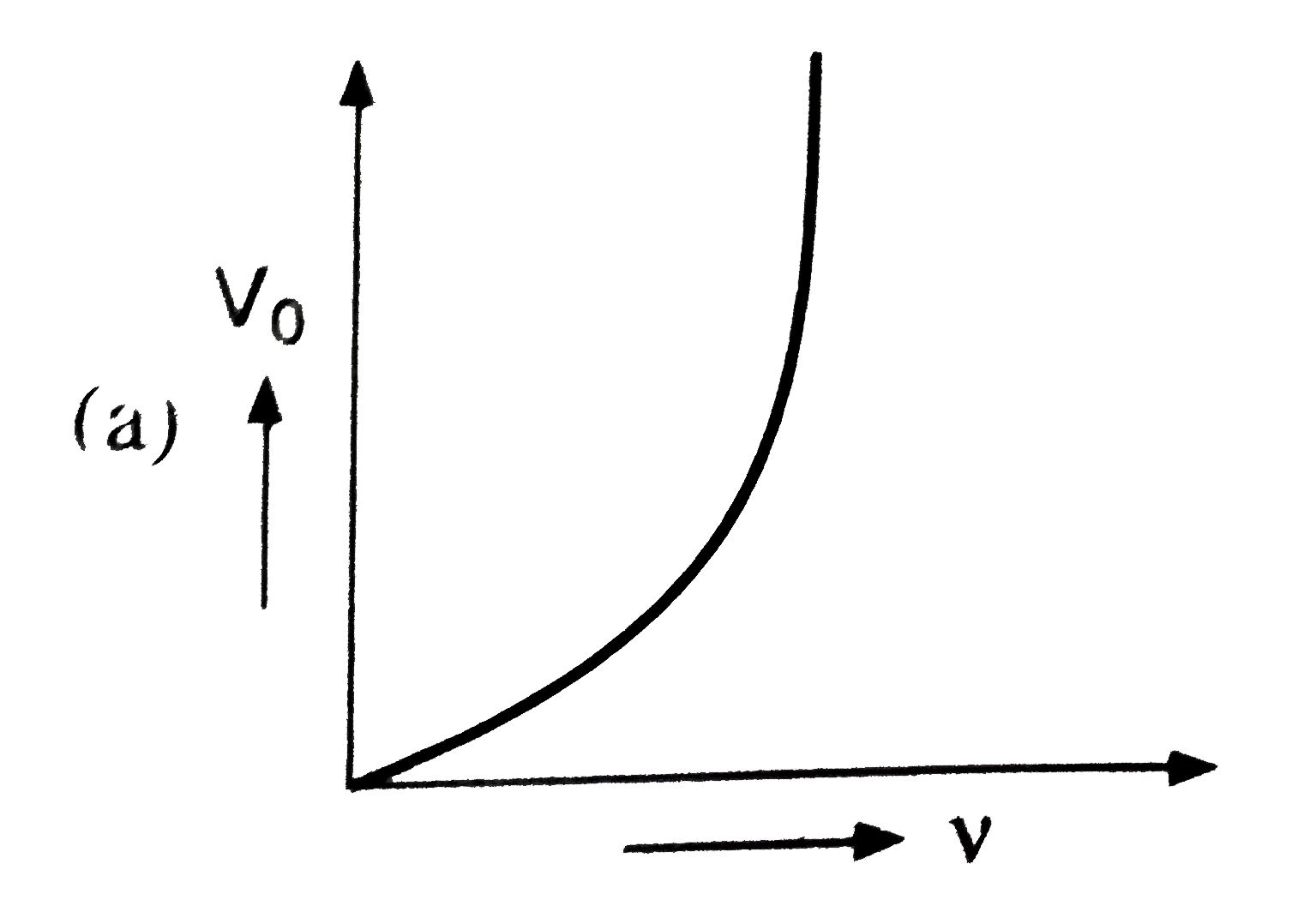
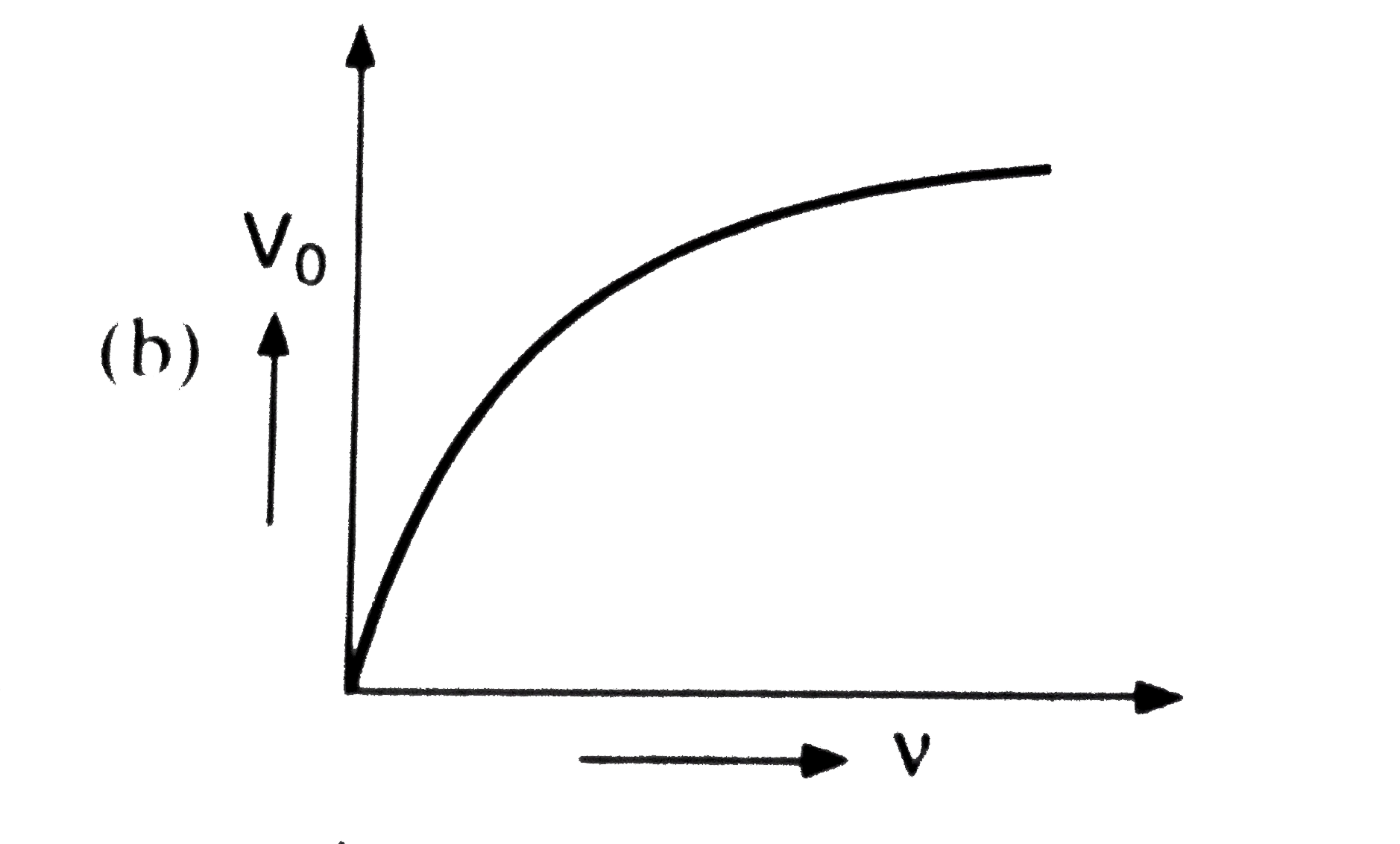
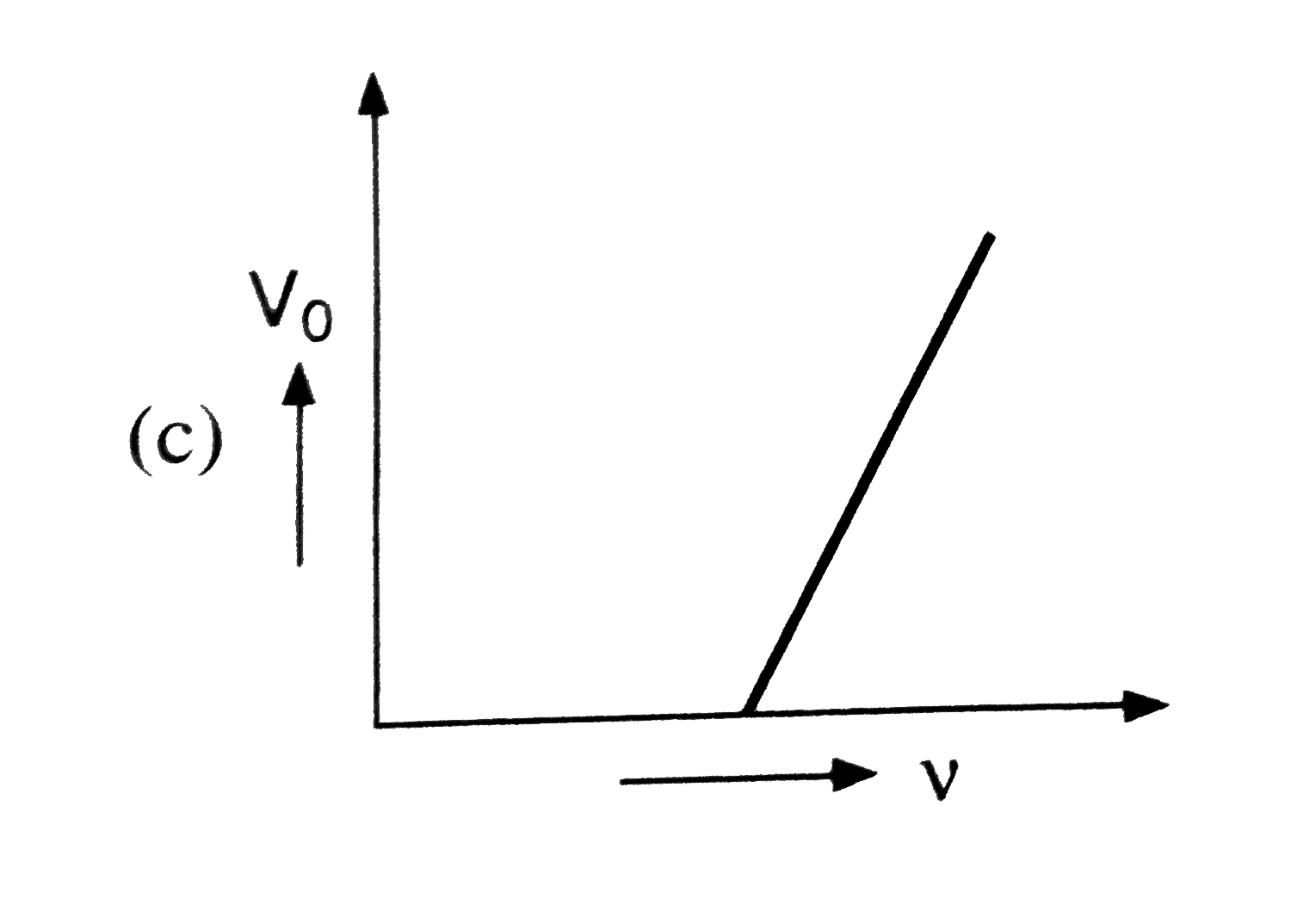
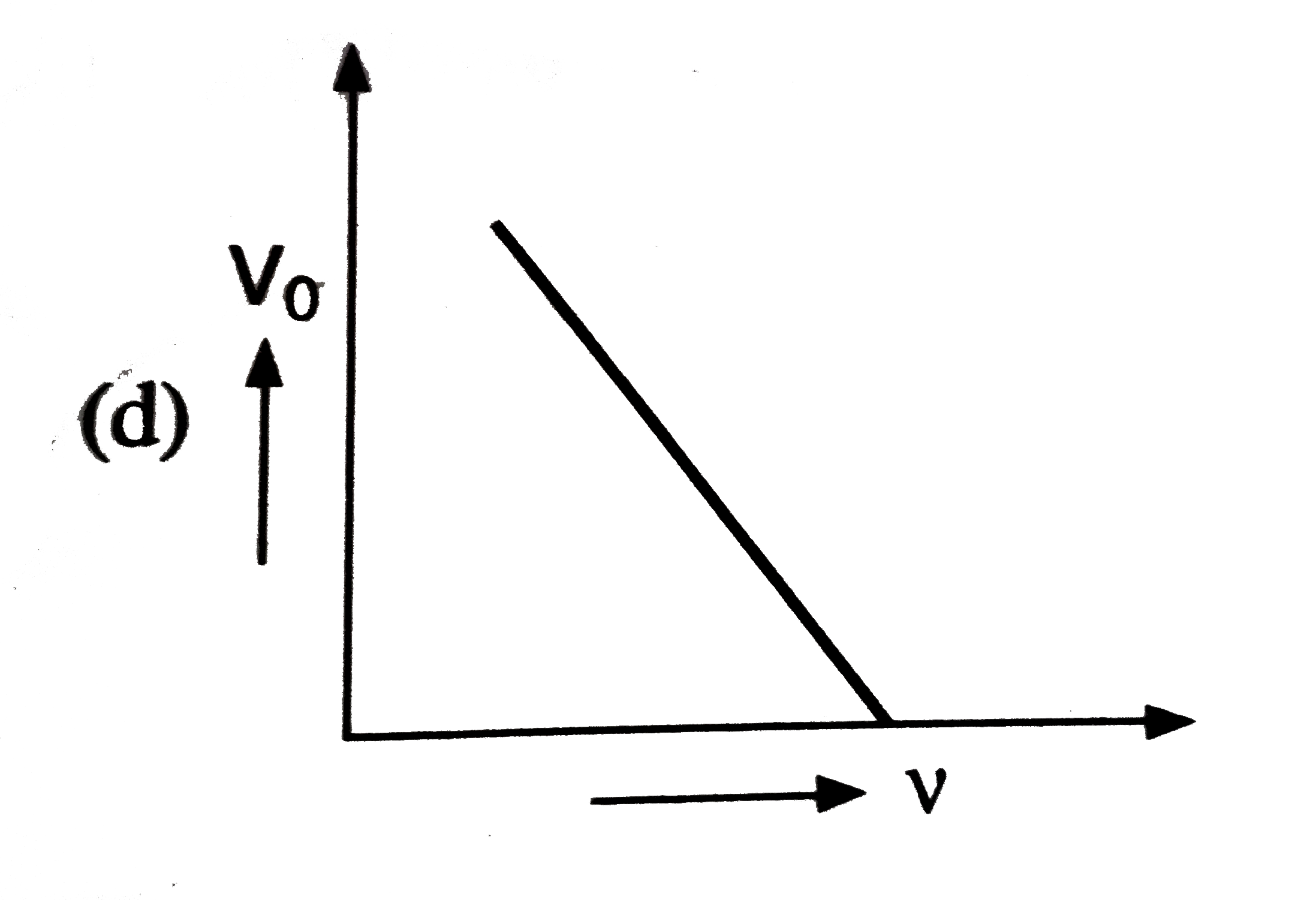
Which of the following is the graph between the frequency (v) of the incident radiations and the stopping potential ? |
|
Answer»
|
|
| 2. |
To make pastries like edible food light and porous ......... is added to it. |
|
Answer» `NaHCO_(3)` |
|
| 3. |
What is not correct at normal temperature and pressure ? |
|
Answer» `P_4O_10` is a WHITE solid |
|
| 4. |
Which of the following compounds is used to remove water hardens |
|
Answer» `Ca_(3)(PO_(4))_(2)` |
|
| 5. |
Which one of the folowing has highest bond order? N_(2),N_(2)^(+)orN_(2)^(-) ? |
|
Answer» Solution :Bond ORDER=`3,B.O=(N_(b)-N_(a))/(2)=(10-4)/(2)=3` `N_(2)^(+)` (13 electrons) Bond order=`2.5,B.O=(N_(b)-N_(a))/(2)=(9-4)/(2)=2.5` `N_(2)^(-)` (15 electrons) Bond order=`2.5,B.O=(N_(b)-N_(a))/(2)=(10-5)/(2)=2.5` So`N_(2)` has the HIGHEST bond order. |
|
| 6. |
Which is not responsible for environmental pollution |
|
Answer» INDUSTRIALISATION |
|
| 7. |
Which of the following compounds is efflorescent ? |
|
Answer» A. `Na_2CO_3` |
|
| 10. |
What is the de Broglie wavelength (in cum) of a 160g cricket ball travelling at 140 Km hr^(-1). |
|
Answer» Solution :160g=.0.16kg. v=velocity of the criket ball=`140Km HR^(-10)` `=(140xx5)/(18)=38.88ms^(-1)` de Broglie equation=gamma=`h/(MV)` h=planck.s constant=`6.626xx10^(34)kgm^(2)s^(-1)` de Broglie wavelength `=6.626xx10^(-34)cancel(kg m^(2) s^(-1))/(cancel(0.16kgxx38.88)cancel(ms^(-1)))` `gamma=6.626xx(10^(-34))/(6.2208)` `gamma=1.065xx10^(-34)m` wavelength in cum`=1.065xx10^(-34)XX100` `=1.065xx10^(32)cm(or) 1.065xx10^(-34)m` |
|
| 11. |
Which of the following is not a general characteristic of equilibria involving physical processes? |
|
Answer» EQUILIBRIUM is POSSIBLE only in a close system at a given temperature |
|
| 12. |
Which product is given by sulphur trioxide on dissolution in to a sulphuric acid ? |
|
Answer» `H_2SO_3` |
|
| 13. |
Which of the following compounds are not arranged in correct order as indicated? |
|
Answer» `SrCL_(2) lt CaCl_(2) lt MgCl_(2) lt BeCl_(2)` (INCREASING of HYDROLYSIS) |
|
| 14. |
Which molecules represented by the bold atoms show their highest oxidation state? |
|
Answer» `F_2ulO` |
|
| 15. |
Which one of the following is positively charged electrophiles? |
|
Answer» `CO_2` |
|
| 16. |
The value of n in the molecular formula Be_(n)I_(2)Si_(6)O_(18 is |
|
Answer» `n(+2)+2(+3)+6(+4)+18(-2)=0` or 2n+6+24-36=0 or 2n=6 n=3 |
|
| 17. |
What is the half life of Fr? |
|
Answer» 21 minute |
|
| 18. |
Write the conditions which proves exceptional stability of bezene. |
|
Answer» Solution :The following conditions proves the excepetional stability of BENZENE. (i) Benzene resist the addition reaction with respect to ALKENE. (ii) Although having LESS amount of `PI` -bond and Hydrogen atom, benzene easily gives substitution reaction. (iii) Benzene possess neither 154 pm length for C-C single bond length nor 133 pm length for C=C but it possess 139 pm intermediate bond length. (iv) Benzene has least hydrogenation ENTHALPY, only `36 K Cal mol^(-1)` respect to cyclohexatriene. Resonance energy of benzene is `36 K cal mol^(-1)`. (v) Benzene is stable due to its aromatic character. |
|
| 19. |
Which one of the following does not have sp^(2) hybridized carbon ? |
|
Answer» Acetone `underset("Acetonitrile")(overset(sp^(3))(CH_(3))-overset(sp)(C)-=)N,""underset("Acetamide")(overset(sp^(3))(CH_(3))-overset(sp^(2))(C)ONH_(2))` |
|
| 20. |
Which of the following represents the given sequence of hybridisation of carbon atoms from left to right sp^(2),sp^(2),sp,sp? |
|
Answer» `CH -= C - C -= CH` |
|
| 21. |
What effect does branching of an alkane chain has on its melting point? |
| Answer» Solution :As branching increases AMONG isomeric alkanes, BOILING point decreases. As branching increases, the molecule attains the shape of a sphere. This decreases the surface AREA and RESULTS in weaker VAN der Waal's forces. This reduce the boiling poin. | |
| 22. |
Which expression gives the fraction by mass of nitrogen in ammonium dihydrogen phosphate? |
|
Answer» `(11/115)` |
|
| 23. |
'X' overset(Ag//Delta)to 'Y' where if 'Y' is ethyne, X is. |
|
Answer» `CH_3Cl` 
|
|
| 24. |
What type of defect can arise when a solid is heated ? Which physical property is affected by it and in what way? |
| Answer» Solution :When a solid is heated, vacancy defect is produced in the CRYSTAL. This is because on HEATING, some atoms or ions leave the lattice SITE completely, ie, some lattice sites become vacant As a result of this defect, the density of the substance DECREASES because some atoms/ions leave the crystal completely | |
| 25. |
Which of the following ion is paramagnetic |
|
Answer» `LA^(3+)(Z=57)` |
|
| 26. |
Which of the following has maximum number of hyper conjugative structures? |
|
Answer» ISOPROPYL CARBONIUM ION |
|
| 27. |
What will be the heat change at constant volume for the reaction whose heat change at constant pressure is -560 kcal at 27^(@)C? The reaction is C_(8)H_(16) + 12O_(2) to 8CO_(2) + 8H_(2)O (Given R = 2 cal mol^(-1)K^(-1)) |
|
Answer» `-557600` calories `C_(8)H_(16)(l) + 12O_(2)(g) to 8CO_(2)(g) + 8H_(2)O(l)` `DeltaH = DeltaU + Deltan_(g) RT` `Deltan_(g) = 8 -12 = -4` `-560000 = DeltaU + (-4) xx 2 xx 300` = `-557600`. |
|
| 28. |
Using stock notation represent HAuCI_(4). |
| Answer» Solution :The oxidation NUMBER of the metallic element Au in `HAuCl_(4)` is `+3`. HENCE, the COMPOUND is represented as `HAU(III)Cl_(4)` | |
| 29. |
Which of the following compound(s) fail in Friedel-Crafts alkylation reaction? |
|
Answer»
|
|
| 30. |
What are diathermic and adiabaticcontainers? |
| Answer» Solution :The CONTAINERS that permit exchange of energy with the SURROUNDINGS are called DIATHERMIC while those which do not allow are called ADIABATIC. | |
| 31. |
Which of the following molecules is least resonance stabilised ? |
|
Answer»

|
|
| 32. |
Which one has highest pH from the following ? |
|
Answer» `NaNO_3`  `Na_2CO_3` is comperatively most BASIC . The POH of `Na_2CO_3` is MINIMUM and `p^H` is MAXIMUM. |
|
| 33. |
What type of compounds are purified by sublimation ? |
| Answer» Solution :Substances whose vapour PRESSURE become EQUAL to the atmospheric pressure much below their melting points. For EXAMPLE, NAPHTHALENE, benzoic ACID, camphor, iodine, etc. | |
| 34. |
What is meant bythe conjugate acid-base pair ? Find the conjugate acid/base for the secies : HNO_(2), CN^(-), HClO_(4),F^(-),OH^(-),CO_(3)^(2-) and S^(2-) |
|
Answer» SOLUTION :An acid-BASE pair which differ by a proton is calledconjugate acid base pair . `NO_(2)^(-), HCN, ClO_(4)^(-) , HF, H_(2)O ` (acid) or `O^(2-) ` (base), `HCO_(3)^(-) and HS^(-)`. |
|
| 35. |
Which of the following represent correct product formation. |
|
Answer»
 is FORMED DUE to RING EXPANSION. is FORMED DUE to RING EXPANSION.
|
|
| 36. |
Three different gases x, y, z of molecular masses 2, 16 and 64 were enclosed in a vessel at a constant temperature is correct |
|
Answer» Gas `z` will be at the top of the vessel Thus, pressure due to the depth of the lake = 7 atm = 70 m of water. |
|
| 37. |
Which of the following hydids are generally nonstoichiometric in nature |
|
Answer» IONIC hydrogen |
|
| 38. |
Whichof the following species have same shape/same bond order ? N_(3)^(-) , NO_(2)^(-), CO_(2), O_(3) |
|
Answer» SOLUTION :Isoelectonic species have same shape/same BOND order. The NUMBER of OUTER electrons is same in `CO_(2)` and ` N_(3)^(-)`and same in `O_(3) and NO_(2)^(-)`. |
|
| 39. |
Which of the following is NOT a postulate of the kinetic molecular theory of gases ? |
|
Answer» The MOLECULES possess a volume that is negligibly samll compared o the container |
|
| 40. |
The total number of cyclic isomers possible for a hydrocarbon with the molecular formula C_(4)H_6 is |
|
Answer» 2 |
|
| 41. |
Which one of the following statements is incorrect in the case of Heterogenous catalysis |
|
Answer» The CATALYST LOWERS the energy of activation |
|
| 42. |
Which compound is formed when calcium carbide reacts with heavy water? |
|
Answer» `C_(2)D_(2)` |
|
| 43. |
When rain is accompanied by a thunderstorm, the collected rain water will have pH value |
|
Answer» slightlyhiger than thatwhen thethunder stormis not there |
|
| 44. |
Units of K_c for xA_((g)) harr yB_((g)) is lit^2-mol^(-2), then the values of x & y cannot be |
|
Answer» 1,2 |
|
| 45. |
When washing soda is heated |
|
Answer» `CO` is RELEASED |
|
| 46. |
Which oxide cannot be reduced by H_(2)? |
|
Answer» `Al_2O_3` |
|
| 47. |
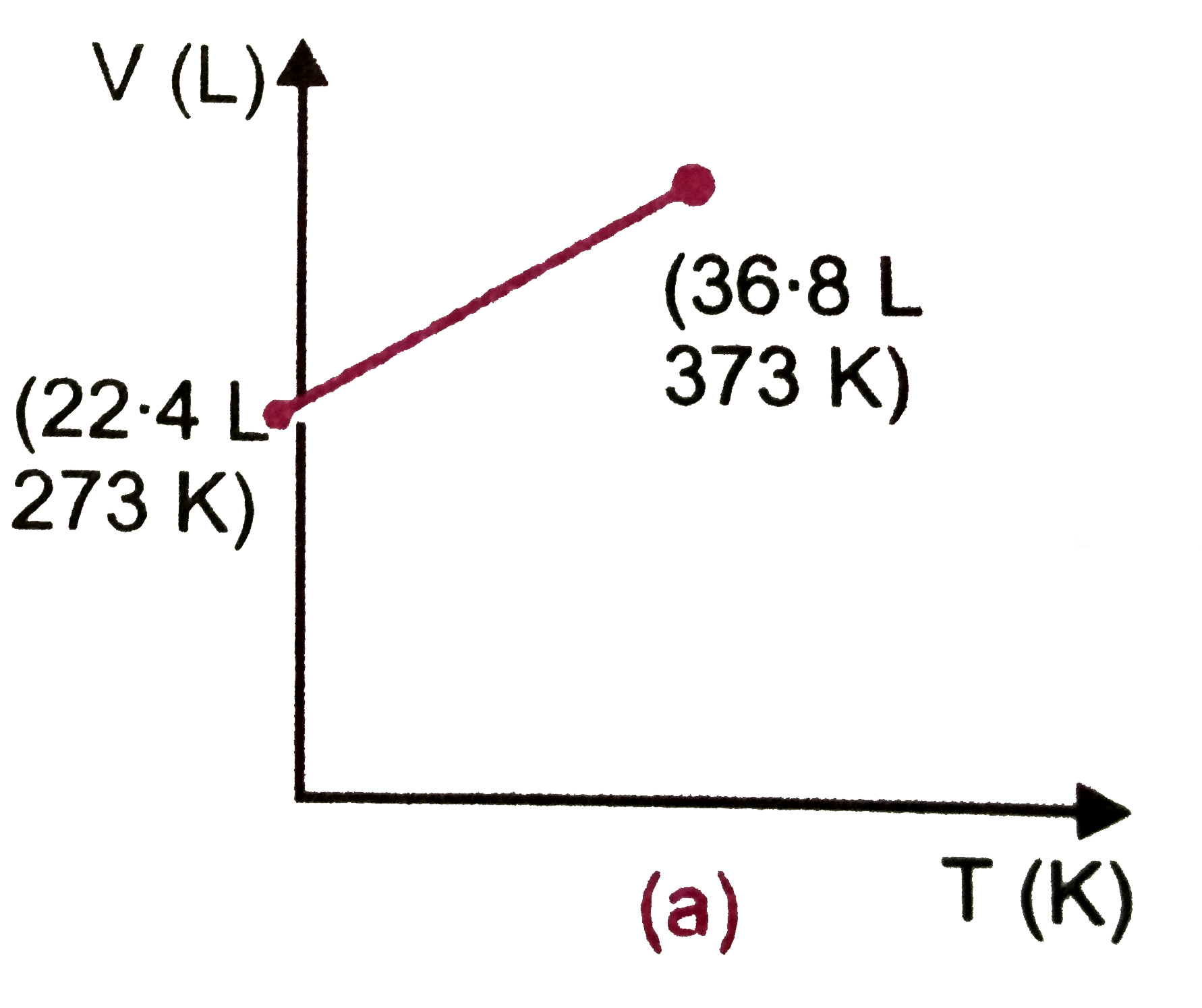
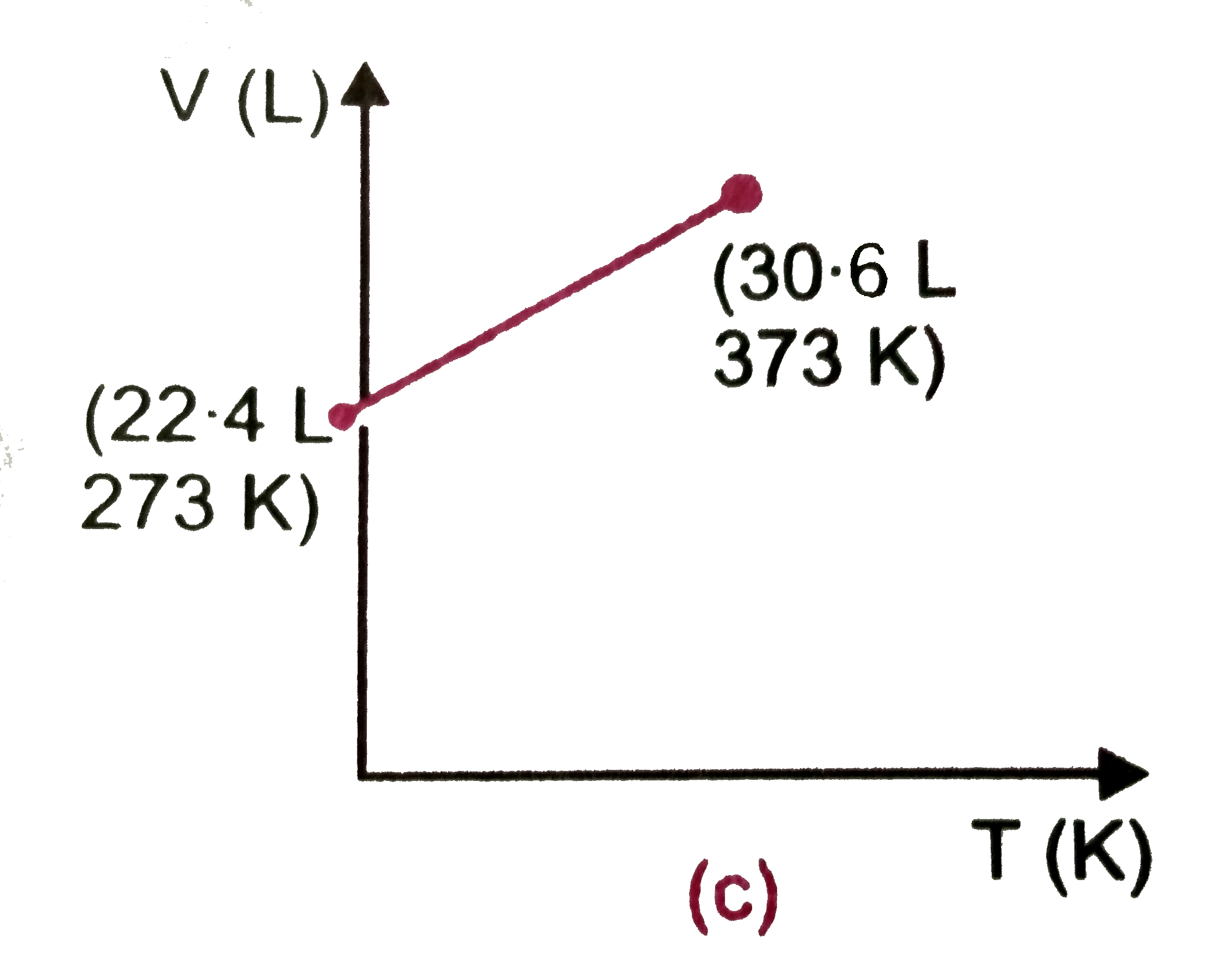
Which of the following volume (V)-temperature (T) plotsrepresents the behaviour of one mole of an ideal gas at one atmospheric pressure ? |
|
Answer»
`V=(RT)/(P)=(0.082xx373)/(1)=30.58" L"~~30.6" L"` |
|
| 48. |
Which of the following reaction do you think will result in the absorption of heat ? |
|
Answer» CARBON burning in air |
|
| 49. |
Which of the following is more stable |
|
Answer» `PB^(4+)` |
|