Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the two ions from the list given below that have the geometry that is explained by the same hybridization of orbitals NO_(2)^(-)and NO_(3)^(-),NH_(2)^(-)NH^(4+),SCN^(-)? |
|
Answer» `NO_(2)^(-)` and `NO_(3)^(-)` |
|
| 2. |
Which alkyl chloride would yield following pure alkene on reaction with alcoholic KOH ? (i)CH_3-oversetoverset(CH_3)(|)C=CH_2 " " (ii)CH_3-CH_2-CH_2-CH=CH_2 "" (iii)CH_3-CH_2-undersetunderset(CH_3)(|)C=CH_3 |
|
Answer» |
|
| 3. |
When1 mol of a gas is heated at constant volume, temperature is raisedfrom298 Kto 308 K.Heat supplied to the gas is500J. Then which statement is correct? |
|
Answer» `q=W=500J, DeltaU=0` As `W=0, DeltaU = q= 500J` |
|
| 4. |
Using molecular orbital theory, compare the bond energy and magnetic character ofO_(2)^(+) and O_(2)^(-) species. |
|
Answer» Solution :According to molecular ORBITAL theory electronic configurations of `O_(2)^(+) and O_(2)^(-)` species are as follows : `O_(2)^(+) : (sigma 1S)^(2) (sigma^(**) 1s)^(2) (sigma 2s)^(2) (sigma^(**)2s)^(2) (sigma 2p_(z))^(2) (pi 2p_(x)^(2) , pi 2p_(y)^(2)) (pi^(**) 2p_(x)^(2))` BOND ORDER of `O_(2)^(+) = (10 - 5)/(2) = (5)/(2) = 2.5 ` `O_(2)^(-) : (sigma 1s)^(2) (sigma^(**) 1s)^(2) (sigma 2s)^(2) (sigma^(**)2s)^(2) (sigma 2p_(z))^(2) (pi 2p_(x)^(2) , pi 2p_(y)^(2)) (pi^(**) 2p_(x)^(2), pi^(**) 2p_(y)^(2))` Bond order of `O_(2)^(-) = (10 -7)/(2) = (3)/(2) = 1.5 ` As Higher bond order of `O_(2)^(+)` shows that it is more stable than `O_(2)^(-)` . Both the species have UNPAIRED electrons. So, both are paramagnetic in nature. |
|
| 5. |
Which of the following acids will give purple colour with ferric chloride solution ? |
|
Answer» `ALPHA-`Hydroxypropionic ACID |
|
| 6. |
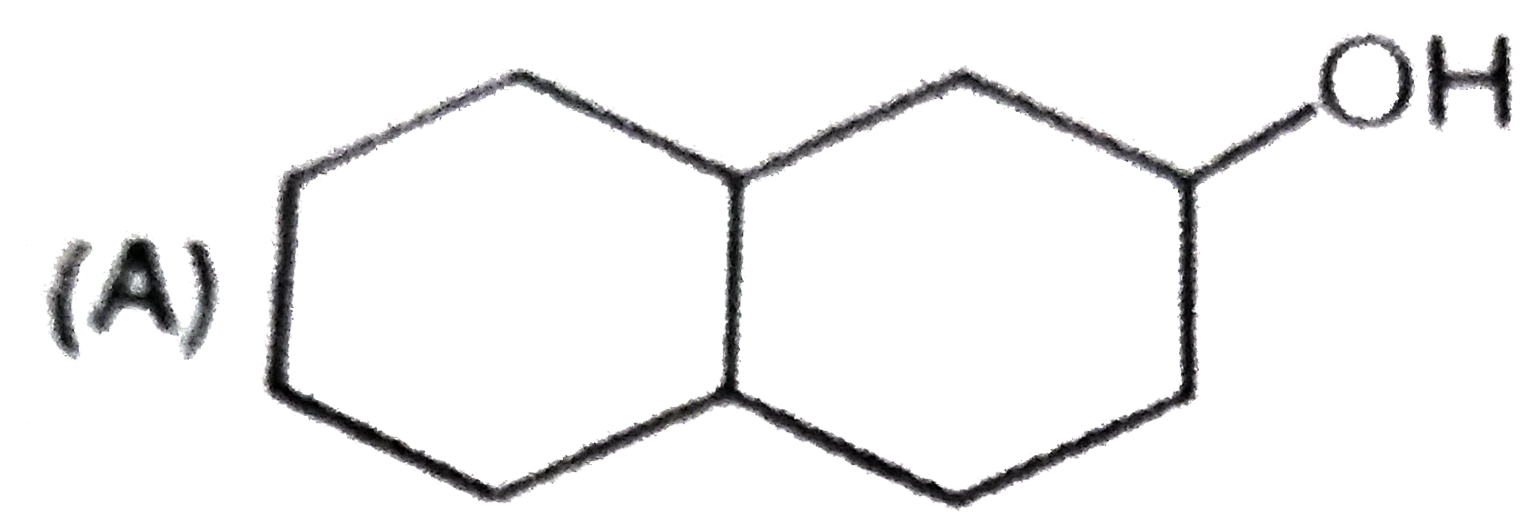
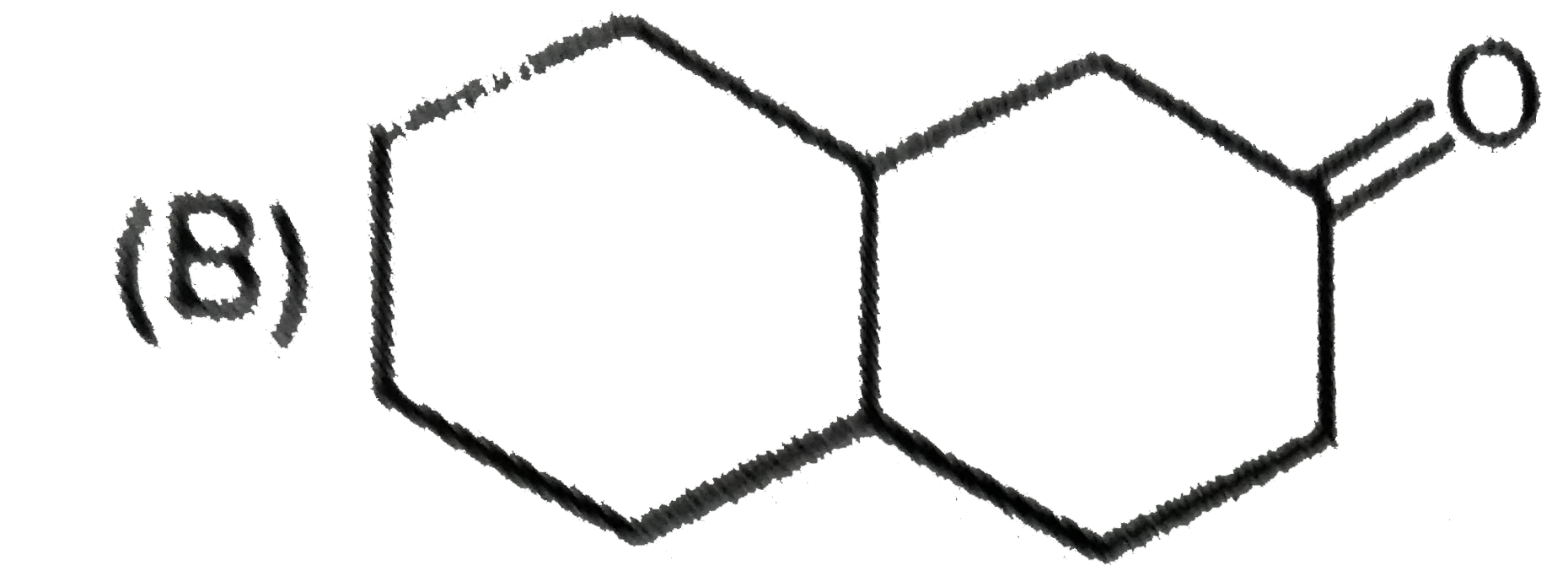
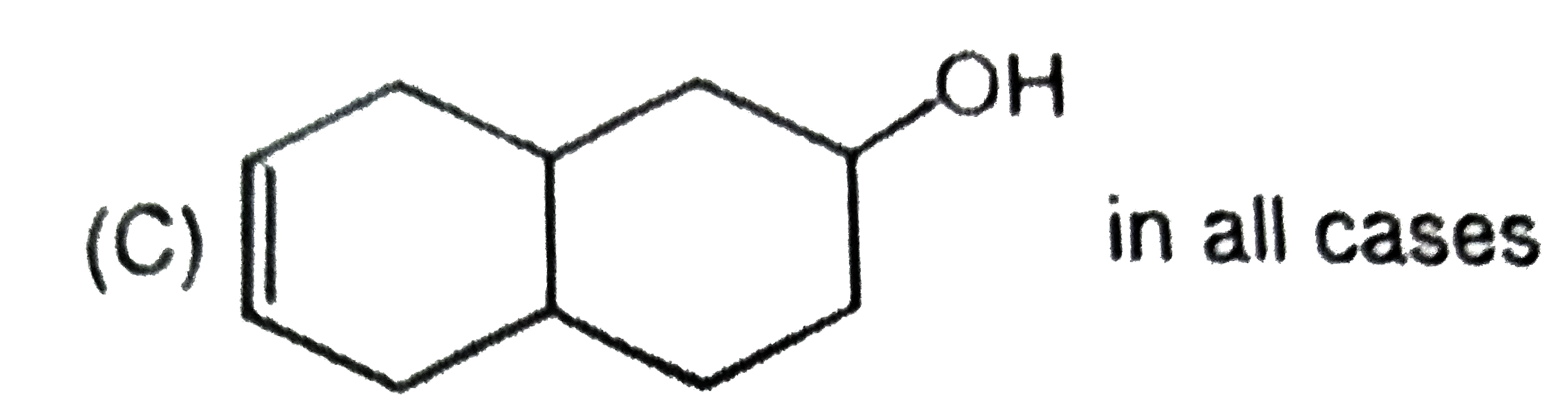
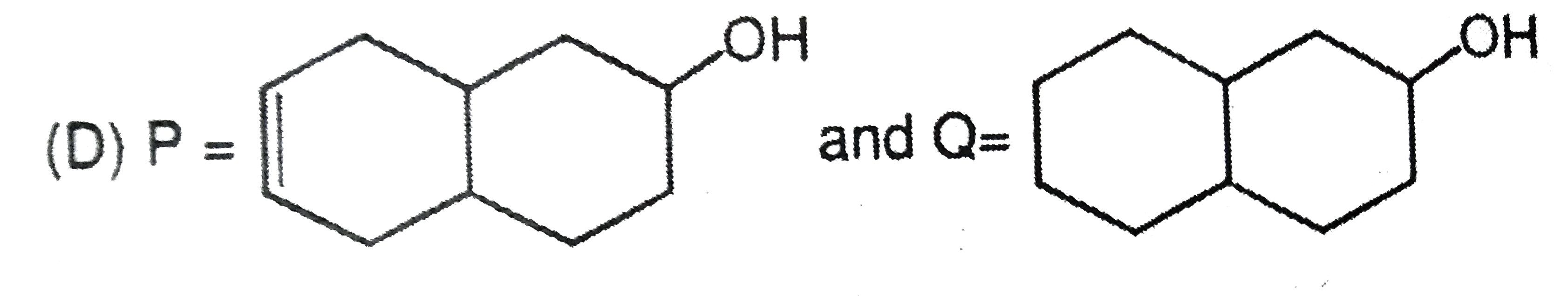
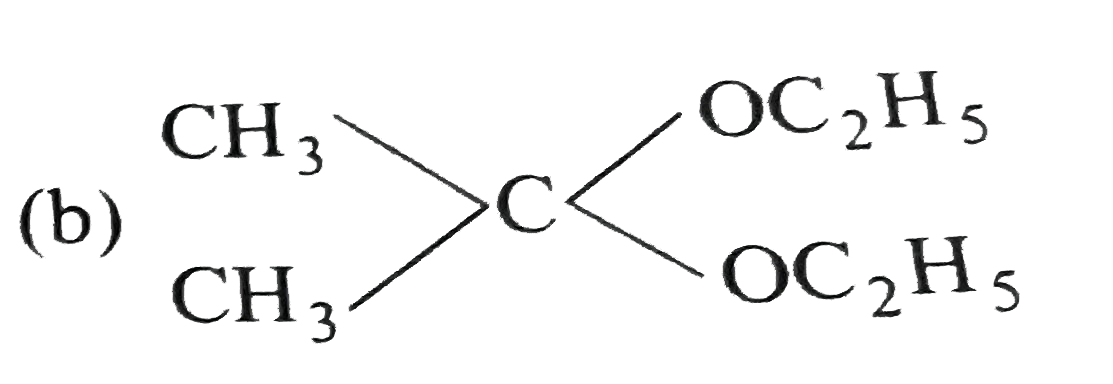
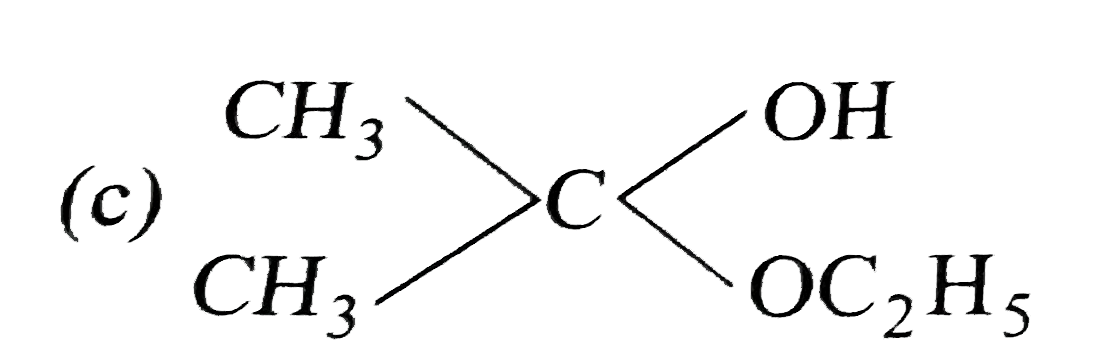
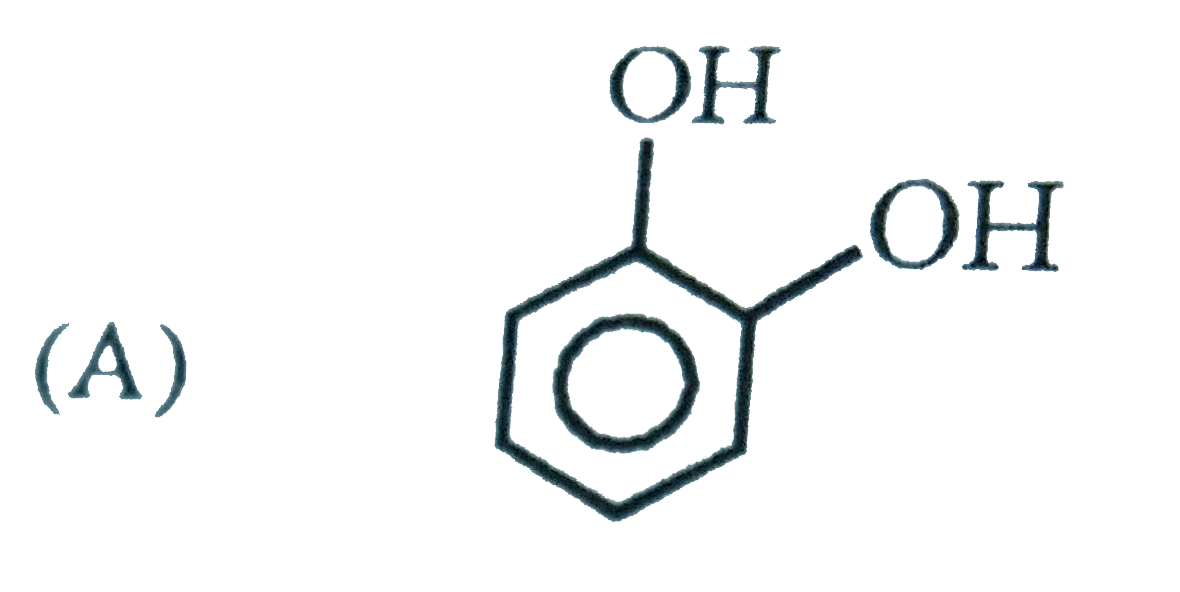
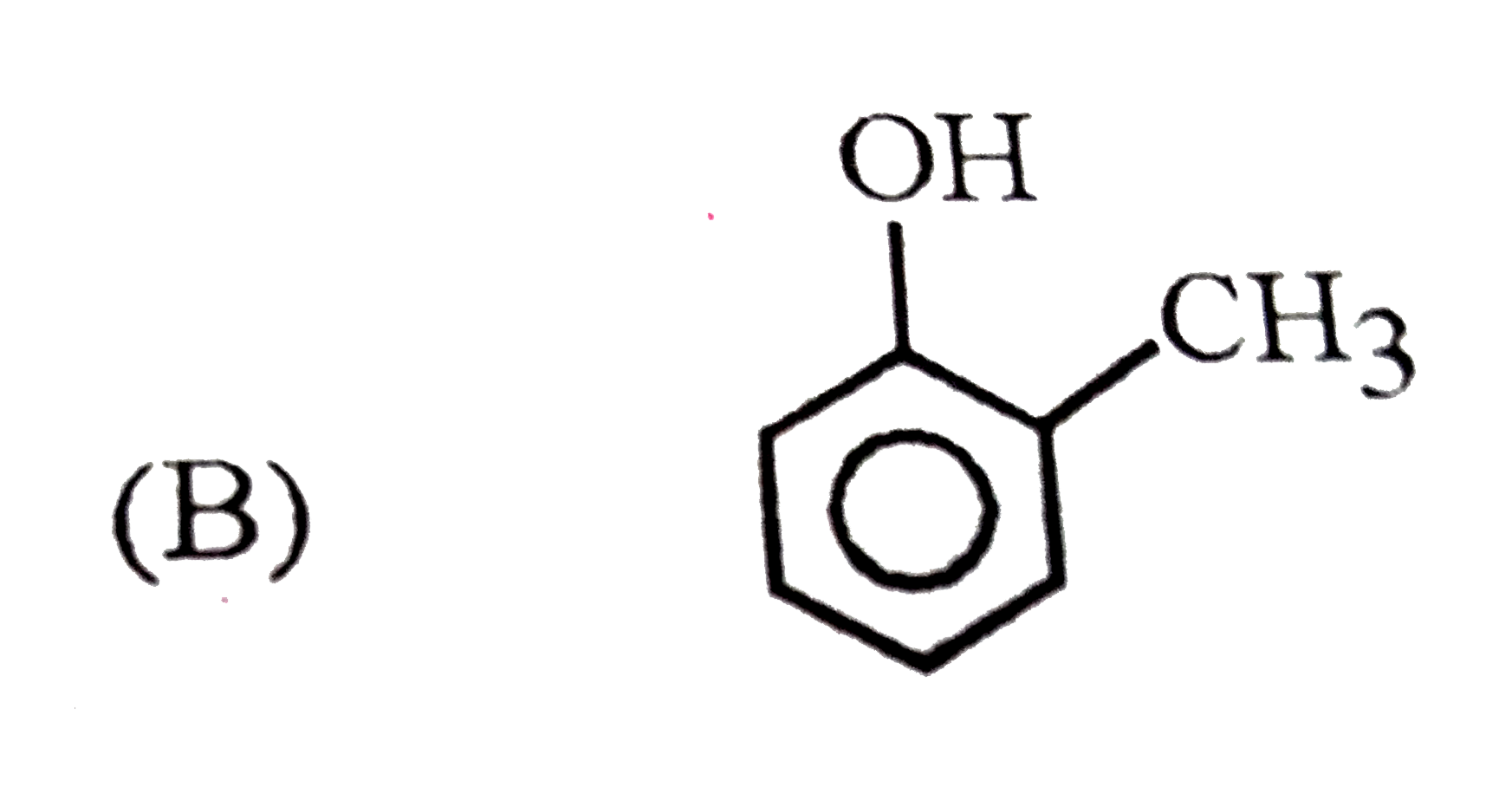
What are A and B in the following ? |
|
Answer»
|
|
| 7. |
What is photovoltaic cell ? |
| Answer» Solution :The DEVICE USED to CONVERT sunlight into electrifcity is CALLED a photovoltaic CELL. | |
| 8. |
Write the important conditions required for the linear combination of atomic orbitals to formn molecular orbitals. |
|
Answer» Solution :The linear combination of atomic orbitals to form molecular orbitals take place only if the following conditions are satisfied. The COMBINING atomic orbitals must have the same or nearly the same energy. This MEANS that LS orbital can combine with another 1s orbital but not with 2s orbital because the energy of 2s orbital is appreciably HIGHER than that of 1s orbital. This is not true if the atoms are very different. The combining atomic orbitals must have the same symmetry about the molecular axis. By convention Z-axis is TAKEN as the molecular axis. It is important to note that atomic orbitals having same or nearly the same energy will not combine if they do not have the same symmetry. e.g., `2p_(z)` orbital of one atom can combine with `2p_(z)` orbital of the other atom but not with the `2p_(x) " or " 2p_(y)` orbitals because of their different symmetries. The combining atomic orbitals must overlap to the maximum extent Greater the extent of overlap, the greater will be the electron density between the nuclei of a molecular orbital. |
|
| 9. |
The sink for dead plants and animals is |
|
Answer» SEA water |
|
| 10. |
Which of the following molecules of alkanes will give only one mono halogenated product onreaction with halogen in presence of sunlight? |
|
Answer» `CH_3- CH_3 ` 
|
|
| 12. |
What is the wavelength of light emitted when the electron in a hydrogen atom undergoes transition from an energy level with n = 4 to an energy level with n = 2 |
|
Answer» Solution :`"Given "=n_(1)=2, n_(2)=4`, we have `bar(v)=(1)/(lambda)=R_(H)[(1)/(n_(1)^(2))-(1)/(n_(2)^(2))]rArr barv=109677[(1)/(2^(2))-(1)/(4^(2))]` `bar=20564.5cm^(-1)rArr lambda=4.862xx10^(-5)cm` |
|
| 13. |
The spontaneity means having the potential to proceeds without the assistance of exertnal agency. The processes which occur spontaneously are |
|
Answer» a flow of HEAT from colder to warmer body. |
|
| 14. |
Write non metal redox reaction in alkali and alklaine earth metals? |
| Answer» Solution :Active metals LIKE alkali and alkaline earth metals are GOOD reducing agents and DISPLACE HYDROGEN from cold water. `2Na(s)+2H_2O(I)to2NaOH(aq)+H_2(g)` | |
| 15. |
Which of the following compound give negative test with Tollen's reagent ? |
|
Answer» `Ph-overset(O)overset(||)(C)-H` |
|
| 16. |
What is the origin of geometrical isomerism in alkenes ? |
| Answer» Solution :Hindered rotation of CARBON ATOMS about a DOUBLE BOND. | |
| 17. |
What are the properties of calcium oxide? |
|
Answer» Solution :(a) It is a WHITE crystalline solid (B) When CaO is exposed to ATMOSPHERE, if absorbs moisture and `CO_(2)`. `CaO + H_(2) O to Ca(OH)_(2)` `CaO + CO_(2) to CaCO_(3)` |
|
| 18. |
Which one of the following is the standard for atomic mass? |
|
Answer» `""_(1) H^(1)` hint: standardelementusedto deteremineatomicmassis ` ""_(6)C^(12)` |
|
| 19. |
Whichof the followingstatementisINCORRECTregardingelectron movementinorganicreactions ? |
|
Answer» Thecurvedarrowendsat a location TOWHICH the electron moves. |
|
| 20. |
Which ion has the stable electronic configuration?Ni^(2+) or Fe^(3+). |
|
Answer» Solution :`Ni(Z=28).1S^(2)2s^(2)2p^(6)3s^(2)3p^(6)4s^(2)3d^(8)` `Ni^(2+)` electronic configuration =`1s^(2)2s^(2)2p^(6)3s^(2)3p^(6)3d^(8)` `Fe(Z=26).1s^(2)2s^(2)2p^(6)3s^(6)4s^(2)3d^(6)` `Fe^(3+)1s^(2)2s^(2)2p^(6)3s^(6)3d^(5)` If d ORBITAL is half FILLED,ACCORDING to Aufbau PRINCIPLE,it is more stable.So `Fe^(3+)` is more stable than `Ni^(2+)` |
|
| 21. |
What is the volume of O_2 liberatedat N.T.P. by complete decomposition of 100mL of 2m solution of H_2O_2? |
|
Answer» SOLUTION :Volume strength `=11.2 XX M =22.4` `100mL=0.1L i.e. 22.4 xx 0.1=2.24 L O_(2)"released"` |
|
| 23. |
Write the molecular and possible structural formula of the first four members of homologous series of carboxylic acids. |
Answer» SOLUTION :
|
|
| 24. |
Which of the following is not a use of baking soda? |
|
Answer» In MEDICINES as antacid |
|
| 25. |
Which of the following reactions require largest amount of energy? |
|
Answer» `Ga_((G))^(2+) to GA _((g))^(3+) + e^(-)` (B) `Ga_((g)) + to Ga_((g))^(+) + e^(-) Delta_(i)H_(1)` (First ionization Energy) (C ) `Ga_((g))^(+) to Ga_((g))^(2+) + e^(-) Delta_(i)H_(2)` (Second ionization Energy) (A) `Ga_((g))^(2+) to Ga_((g))^(3+) + e^(-) Delta_(i)H_(3)` (Third ionization Energy) The value of next ionization ENTHALPY INCREASES graduaely as `Delta H_(1) lt Delta_(i)H_(2) lt Delta_(i)H_(3) `.... According to this reaction requires largest amount of energy. |
|
| 26. |
The two bonds N=O and N--O in H_(3) CNO_(2) are of same bond length due to |
|
Answer» INDUCTIVE effect |
|
| 27. |
The standard state Gibbs free energies of formation of C ( graphite) and C( diamond) at T=298 K are Delta_(f) G^(@) [C ( graphite) = 0 kJmol^(-1) Delta_(f) G^(@) [ C ( diamond) = 2. 9 kJ mol^(-1) The standard state means that the pressure should be 1 bar,and substance should be pure ata given temperature. The conversionofgraphite [C(graphite ) ]to diamond [ C ( diamond)] reducesits volumeby 2 xx 10^(-6) m^(2) mol^(-1). If C( graphite0 is convertedto C( diamond) isothermally at T= 298 K , the pressure at which C( graphtie) is in equilibriumwith C ( diamond) is [ Useful information : 1 J = 1kgm^(2) s^(-2), 1 Pa=1 kg m^(-1) s ^(-2), 1 "bar" = 10^(5) Pa] |
|
Answer» 58001 bar For isothermal process at 298 K, `DeltaT= 0 :.S d T =0` `:. dG =VdP` Starting from initial pressureof1 bar, if required pressure is P bar, then `int_(1)^(P)dG =int_(1)^(P) VdP `or`DELTAG= V [P]_(1)^(P)` `[V=` constant becausesolids are involved] `= V(P-1)` or`G-G^(@) = V( P-1)` `:. `For the process , C ( graphite)`rarr C( `DIAMOND) `Delta_(r)G= [G_("diamond")^(@) +V_(d) (P-1)]-[G_("graphite")^(@) + V_(g)(P-1)]` `= [ G_("diamond")^(@)-G_("graphite")^(@)]+ (P-1)(V_(d) -V_(g))` WhenC ( graphite) and C ( diamond) are in equilibrium, `Delta_(r)G=0` `0= ( 2.9 xx10^(3)-0)+( P-1) 10^(5) ( - 2 xx 10^(-6))` or`P -1 = ( 2.9 xx 10^(3))/( 10^(5) xx ( 2 xx 10^(-6)))=14500 ` bar or `P =14501 ` bar |
|
| 28. |
The reducing power of divalent species decreases in the order |
|
Answer» `GegtSngtPb` `underset("more stable")(Pb^(+2))tounderset("less stable")(Pb^(+4))` `underset("more stable")(Sn^(+2))tounderset("less stable")(Sn^(+4))` |
|
| 30. |
What is disproportionation reactions? Give example. |
|
Answer» Solution :The reactions in which an element undergoes SIMULTANEOUSLY both oxidation and reduction are called as disproportionation reactions. Example: `P_(4)` + 3NaOH + `3H_(2)O rarr PH_(3) + 3NaH_(2)PO_(2)` 2HCHO + `H_(2)O rarr CH_(3)OH` + HCOOH |
|
| 31. |
What is the maximum concentration of equimolar solutions of ferrous sulphate and sodium sulphide so that when mixed in equal volumes, there is no precipitation of iron sulphide ? For iron sulphide, K_(sp)=6.3xx10^(-18). |
|
Answer» Solution :Suppose the CONCENTRATION of each of `FeSO_(4) and Na_(2)S ` is xmol `L^(-1)` . Then after mixing equal volumes, `[FeSO_(4)]=[Na_(2)S]=(x)/(2) M, i.e., [Fe^(2+)]=[S^(2-)]=(x)/(2)M` `F_(sp) "for" =[Fe^(2+)][S^(2-)] , i.e., 6.3xx10^(-18) = (x)/(2)xx(x)/(2)orx^(2) = 25.2 xx 10^(-18), or x =5.02xx10^(-9)M`. |
|
| 32. |
Whichof thefollowingCORRECTLYrepresentschain isomersconsideringthatallthe valenciesof carbonatomsare satisfiedwithhydrogen? |
|
Answer»
|
|
| 33. |
What happens when isobutly lene is treated with acidified potassium permanganate |
Answer» SOLUTION :Isobutyleneis TREATEDWITH ACIDIFIED`KMnO_(4)` to giveacetone.
|
|
| 34. |
Which of the following reactions does not show the correct products of the reaction? |
|
Answer» `CH_(3)-CH=CH_(2) underset("peroxide") overset(HBr)to CH_(3)-CH_(2)-CH_(2)Br` |
|
| 35. |
The strength of H_(2)O_(2) (in g/litre) in 11.2 volume solution of H_(2)O_(2) is |
|
Answer» 17 or `11.2 = 5.6xx("Strength in " gL^(-))/(17)` or Strength in `gL^(-1)=(11.2xx17)/(5.6)=34` |
|
| 36. |
Which among the following is not correctly matched with their colour? |
|
Answer» `{:("COMPOUND","Colour"),(Na_(4)[FE(CN)_(5)NOS],"PURPLE"):}` |
|
| 37. |
Which of the following contains Avagadro number of atoms? |
|
Answer» one mole of HELIUM GAS 22.4 litres of `CO_(2)` at STP contains Avogadro number of molecules `(11.2)/(22.4)xx6.023xx10^(23)xx2` atom of hydrogen `(3.2)/(16)xx6.023xx10^(23)xx5` atoms |
|
| 38. |
Which of the following will not give pure propane? |
|
Answer» `CH_3CH_2CI+2Na+CICH_3overset("ETHER")to` |
|
| 39. |
When KMnO_(4) is reduced with oxalic acid in acidic medium, the oxidation number of Mn changes from : |
|
Answer» 7 to 4 |
|
| 40. |
Which of the following statement is//are correct regarding H_(2)O_(2) ? |
|
Answer» It oxiides FE(II) to Fe(III) in acidic medium |
|
| 42. |
Which of the following is most toxic ? |
|
Answer» `Hg_2^(2+)` |
|
| 43. |
Which ion when enters into soil act as fertilizer? |
|
Answer» `NO_3^-` |
|
| 44. |
Which is te wrong pair (i) starch solution:sol (ii) Aq NaCl: True solution (iii). Milk: emulsion (iv). Aq BaSO_(4):true solution |
|
Answer» (i) |
|
| 45. |
Which of the following figures does not represent 1 mole of dioxygen gas at STP ? |
|
Answer» 16 G of GAS Thus, 1 mole of `O_(2)` gas = 32 g of `O_(2)=6.02xx10^(23)` molecules = 22.4 L of `O_(2)` at STP |
|
| 46. |
What ishydrogen spectrumgive its characteristic? |
|
Answer» Solution :Hydrogen spectrum : Whenanelectric dischargeis passedthroughgaseoushydrogenthe `H_(2)` moleculesdissociateand theenergeticallyexcitedhydrogenatomsproducedemitelectronmagneticknownas hydrogenspectrum Characteristics: Hydrogenspectrum is linespectrumemissionspectrumanddiscontinuesnumber oflineappearingin differentregion of wavelengths. Each beingnamedafterthenameof itsdiscovered. |
|
| 47. |
Which of the following will weigh less in presence of external magnetic field ? |
| Answer» Answer :D | |
| 48. |
Which of the following alkylbromide will react with sodium to form 4,5-diethyloctane? |
|
Answer» 1-Bromobutane |
|
| 49. |
What is BOD? |
| Answer» Solution :The total amount of oxygen (in MILLIGRAMS) consumed by micro0organisms in decomposing the waste in one litre of water at `20^@C` for a period of 5 days is called biochemical oxygen demand (BOD) and its value is expressed in PPM | |
| 50. |
Which situation deviation shows between ideal gas and real gas ? |
|
Answer» Solution :At high pressure : ATTRACTION forces becomes effective between molecules then change in pressure is `(an^(2))/(V^(2))`. `p_("ideal")=p_("real")+(an^(2))/(V^(2))` At high pressure : Repulsive forces becomes effective between molecules then change in volume in nb. `V_("ideal")=(V_("real")-nb)` At very low temperature : Intermolecular forces become significant. As the molecules travel with low average SPEED, these can be CAPTURED by one another due to attractive forces. |
|