Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which bond among the following is least ionic- |
|
Answer» `P-F` |
|
| 2. |
The temperature of O_(2) gas is changed from 25^(@)C to 50^(@)C then change in volume is ………….. (definite quantity of gas and constant pressure). |
|
Answer» LESS than TWICE |
|
| 3. |
What is the average velocity of oxygen gas molecules at 30^(@)C ? |
| Answer» SOLUTION :448 `MS^(-1 )` | |
| 4. |
What will be volume of 8 M solution containing 1 mole solute ? |
| Answer» Answer :A::B | |
| 5. |
What is meant by non hetero (polar) covalent bond? |
| Answer» Solution :In a COVALENT bond if the pair of ELECTRONS SHARED equally (symmetrically) between the ATOMS called non-polar covalent bond. | |
| 6. |
What is the effect of added inert gas on the reaction at equilibrium ? |
| Answer» Solution :When an inert gas (i.e , a gas which does not react with any other SPECIES involved in EQUILIBRIUM) is added to an equilibrium system at CONSTANT volume, the total number of moles of gas present in the container increases, that is, the total pressure of GASES increases. The partial pressure of the reactants and the products or themolar concentration of the substance involved in thereaction remains unchanged . Hence at constant volume, addition of inert gas has no effect on equilibrium. | |
| 7. |
Which one is incorrect match of reactions ? |
|
Answer»
|
|
| 8. |
What is wurtz fittingreaction |
|
Answer» SOLUTION :WHENA solution of bromobenzeneand iodonethane indry ether is treated withmetalicsodiumtoluene isformed `C_(6)H_(5)Br +2Na+ICH_(2) overset( "dry ether" ) (to)C_(6)H_(3) CH_(3) + NaBr +Nal` |
|
| 9. |
Write the name and atomic number of the following elements(i)The first halogen(ii)The third alkali metal (iii)The fourth alkaline earth metals (iv)The sixth element of the second transition series (v) The second inner transition element (vi)The fifth noble gas. |
|
Answer» Solution :(i) Fluorine ,F (Z=9) (ii) Potassium , K (Z=19) (iii)Strontium ,SR (Z=38) (iv)Ruthenium ,Ru (Z=44) (v)Praseodymium ,Pr (Z=59) (VI)XENON , XE (z=54) |
|
| 10. |
Titanium (Ti) crystallizes in fcc lattice. It reacts with C or H intersititally and these elements occupy the voids of host lattice (i.e., Ti) radius that can be occupies in TVs without Casing strain in the host lattice. Calculate the fromula fo titanium hydride and tianium carbide. |
|
Answer» 0.225 |
|
| 11. |
Titanium (Ti) crystallizes in fcc lattice. It reacts with C or H intersititally and these elements occupy the voids of host lattice (i.e., Ti) radius that can be occupies in TVs without Casing strain in the host lattice. What is the maximum ratio of "foreign" atom radius (i.e., H atom) to host atom (i.e ., Ti) radius that can be occupies in TVs without causing a strain in the host lattice. |
|
Answer» 0.225 |
|
| 12. |
Titanium (Ti) crystallizes in fcc lattice. It reacts with C or H intersititally and these elements occupy the voids of host lattice (i.e., Ti) radius that can be occupies in TVs without Casing strain in the host lattice. Calculate the formula of titanium hydride and titanium carbide. |
|
Answer» TiH,TIC Number of TVs=Number of H-atoms=8/unit cell (i) Formula of titanium hydride `=Ti_(4)H_(8)rArTiH_(2)` (ii) Number of Ovs=Number of C-atom=4/unit cell Formula of titanium carbide`=Ti_(4)C_(4)rArrTiC` |
|
| 13. |
The solubility of AgCl in water at 298 K is 1.06 xx 10^(-5) moleper litre. Calculateits solubility product at this temperature. |
|
Answer» Solution :The solubility equilibrium in the SATURATED solution is : `AgCI (s) overset(aq)(hArr) Ag^(+) (aq) + CI^(-) (aq)` The solubility of AgCi is `1.06 xx 10^(-5)`mole per litre. `[Ag^(+) (aq)] =1.06 xx 10^(-5) mol L^(-1)` `[CL^(-)(aq)] =1.06 xx 10^(-5) mol L^(-1)` `K_(SP) =[Ag^(+) (aq)] [ Cl^(-) (aq)]` `= (1.06 xx 10^(-5) mol L^(-1)) xx (1.06 xx 10^(-5) mol L^(-1))` `= 1.12 xx 10^(-10) mol^(2)L^(-2)` |
|
| 14. |
Which of the following is reduced by H_(2)I_(2) in acid medium |
|
Answer» `KMnO_(4)` |
|
| 15. |
What volume of CO_(2) is obtained at STP by heating 4 g of CaCO_(3) ? |
| Answer» SOLUTION :0.896 LIT | |
| 16. |
Write the energy level diagram of oxygen molecule. |
|
Answer» Solution :Atomic number of oxygen =8. Electronic configuration of oxygen `=1s^(2)2s^(2)2P^(4)`. When two oxygen atoms COMBINES, the molecular orbital energy level diagram is as shown in the figure. From the diagram, the molecular electronic configuration of oxygen is `sigma_(1s^(2)),sigma_(1s^(2))^(***),sigma_(2s^(2))^(***),sigma_(2p_(2)^(2)),{pi_(2p_(x))^(2)=pi_(2p_(y))^(2)}` `{pi_(2p_(x)^(1))^(***)=pi_(2p_(y)^(1))^(***)}` Bond order `=("no. of BMO"e^(-)-"no. of"ABMOe(-))/2` `=(N_(be)-N_(ABE))/2` 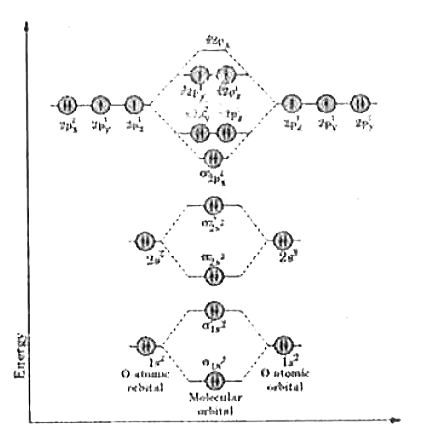 Here BMO electrons `sigma_(1s)^(2),sigma_(2s^(2)),sigma_(2p_(x)^(2)),{pi_(2p_(x)^(2)=pi_(2p_(y)^(2))}` i.e. `N_(be)=10` ABMO electrons `sigma_(1s^(2))^(***),sigma_(2s^(2))^(***),sigma_(2p_(x)^(1))^(***){pi_(2p_(x)^(1))^(***)=pi_(2p_(y)^(1))^(***)}`, i.e. `N_(abe)=6` `BO=(10-6)/2=4/2=2` i.e. between oxygen there exists double bond. Magnetic property: Therefore are two UNPAIRED electrons `{pi_(2p_(x)^(1))^(***)=pi_(2p_(y)^(1))^(***)}` therefore, oxygen molecule is paramagnetic in nature. |
|
| 17. |
Two vessels of equal volume contain O_2 and CO_2 gases separately at the same temperature and pressure. Then, which gas contains (a) more number of molecules ? (b) more number of atoms? (c) more kinetic energy ? (d) high RMS velocity ? |
| Answer» SOLUTION : both have EQUAL K.E. | |
| 18. |
Two vessels of equal volume contain O_2 and CO_2 gases separately at the same temperature and pressure. Then, which gas contains more number of molecules ? |
| Answer» SOLUTION :Both have same NUMBER of MOLECULES | |
| 19. |
The strength of H_(2)O_(2) (wg/litre) in 11*2 volume solution of H_(2)O_(2) is |
|
Answer» 17 |
|
| 20. |
What volume of nitrogen at S.T.R can be obtained from a mixture of 10 g each of NH_4CI and NaNO_2. |
|
Answer» `underset(53.492 g)(NH_(4)Cl) + underset(69 g)(NaNO_(2)) to underset(64.052 g)(NH_(4)NO_(2)) + NaCl` `underset(64.052 g)(NH_(4)NO_(2)) to underset(22.4 "L at S.T.P.")(N_(2)) + 2H_(2)O` According to these equations, 53.492 g of `NH_4CI` react with 69 g of `NaNO_2` to FORM 22.4 L of `N_2` at S.T.R Since, given mixture contains 10 g each of `NH_4CI` and `NaNO_2`, it is obvious that `NH_4CI` is in excess and some PART of it will be left behind. Hence, `NaNO_2` is the limiting reagent in this case. `therefore 69 g` of `NaNO_(2)` from `N_(2) = 22.4 L` at S.T.P `therefore 10 g` of `NaNO_(2)` will form `N_(2) = (22.4)/69 xx 10 = 3.25 L` THUS, the given mixture will GIVE 3.25 L at S.T.P. |
|
| 21. |
What is the common property observed in naturally existing solution? Explain it. |
|
Answer» SOLUTION :(i) Sea water, air are the naturally EXISTING homogeneous mixture. The common property observed in THESR is homogeneity. (II) The homogeneity implies uniform distribution of their CONSTITUENTS or components through out the mixture. |
|
| 22. |
The sodium frame test has a characteristic yellow colour due to the emissions of wavelength 589nm. What is the mass equivalence of one photon of this wavelength? |
|
Answer» |
|
| 23. |
When epsom salt is heated at 150^@C, the compound obtained is |
|
Answer» `MgSO_4` |
|
| 24. |
Which of the following statement (s) are incorrect? |
|
Answer» Photons having energy 400 kJ will BREAK 4 mole bonds of a molecule `A_(2)` where A-A bond dissociation energy is `100kJ//"mol".` |
|
| 25. |
Which of the following is true regarding the hydroboration oxidation of alkene ? |
|
Answer» It leads to ANTI`-` hydraction of alkene and occurs with a REGIOSELECTIVITY opposite to Markovnikov's rule. |
|
| 26. |
Which one of the following is not an extensive property ? |
|
Answer» MOLE |
|
| 27. |
Water is considered as pure if it has BOD less than______ ppm |
|
Answer» |
|
| 28. |
Why boron compounds such as BF_(3) are calledelectron deficient compounds ? |
| Answer» SOLUTION :BECAUSETHE valenceshell of boron in `BF_(3)`has only sixelectrons and needs TWO more electronsto COMPLETE its octet. | |
| 29. |
Three students namely A, B, C have done an experiment two times individually, for which the correct value is 2.00g. The results are given below {:(,"Experiment - 1","Experiment - 2"),("Student A",1.95,1.93),("Student B",1.94,2.05),("Student C",2.01,1.99):} Whose results are accurate and precise? |
|
Answer» C |
|
| 30. |
Tropospheric pollution is caused by gaseous air pollutants as well as particulate polutants. Among the gaseous air pollutants, oxides of sulphur (SO_(x)) oxides of nitrogen (NO_(x)) and carbon monoxide play an important role towards pollution. Formation of acid rain and photochemical smog. green house effect and global warming all are the result of various chemical and photochemical reactions taking place in the environment. Various particulates including viable and non-viable are also responsible for causing serious air pollution. Which of the following has largest concentration in acid rain ? |
|
Answer» `HNO_(3)` |
|
| 31. |
Which of the following atoms has the highest first ionization energy? |
|
Answer» Sc |
|
| 33. |
Which of the following paris can be easily separated by using hydrochgloric acid(HCl, aq.) as the solvent for crystallization ? (i) Napthalene and oxalic acid (ii) Napthalene and p-toluidine (iii)Aniline and nitrobenzene (iv) Benzene acid and napthalene |
|
Answer» (i), (iv) |
|
| 34. |
Tropospheric pollution is caused by gaseous air pollutants as well as particulate polutants. Among the gaseous air pollutants, oxides of sulphur (SO_(x)) oxides of nitrogen (NO_(x)) and carbon monoxide play an important role towards pollution. Formation of acid rain and photochemical smog. green house effect and global warming all are the result of various chemical and photochemical reactions taking place in the environment. Various particulates including viable and non-viable are also responsible for causing serious air pollution. The aromatic compounds present as pariculates are |
|
Answer» Benzene |
|
| 35. |
Tropospheric pollution is caused by gaseous air pollutants as well as particulate polutants. Among the gaseous air pollutants, oxides of sulphur (SO_(x)) oxides of nitrogen (NO_(x)) and carbon monoxide play an important role towards pollution. Formation of acid rain and photochemical smog. green house effect and global warming all are the result of various chemical and photochemical reactions taking place in the environment. Various particulates including viable and non-viable are also responsible for causing serious air pollution. Which of the following does not contribute towards the formation of photochemical smog ? |
|
Answer» NO |
|
| 36. |
The temperature at which Methane molecules have the same average Kinetic energy as that of oxygen molecules at 27°C is |
|
Answer» `327^@ C` |
|
| 37. |
Which element shows less ionisation enthalpy? |
|
Answer» NOBLE gases |
|
| 38. |
When there are two electrons in the same orbital they have ……. Spins |
|
Answer» `+(1)/(2),+(1)/(2)` |
|
| 39. |
Which one of the following is not a redox reaction? |
|
Answer» RUSTING of iron |
|
| 40. |
What is the pH of 10^(-8) "mol L"^(-1) HCl form 8, 6.5 and 7.5 ? Why ? |
| Answer» SOLUTION :6.5 because the solution is ACIDIC | |
| 41. |
The spectrum of while light ranging from red to violet is called a continuous spectrum because |
|
Answer» different colours are seen as different bands in the spectrum |
|
| 42. |
What is the arrangement of atoms in the lattice structure of diamond and give contribution of each C atom ? |
|
Answer» SOLUTION :Diamond is a covalent ctystal in which each C -atom is` sp^(3)` hybridized. THUS, each C-atom is covalently bonded to four othertetrahedrally. These tetrahedra are linked together into a three dimesionalgiant molecule. The structureof diamond is similar to that of ZnS ( in which `S^(2-)`ions form fcc lattice and `ZN^(2+)` ions are present on the body diagonals and occupy alternate tetrahedral voids). In diamond, we have C-atoms is place of ` Zn^(2+) and S^(2-)`ions. Thus, diamond has face - centred cubic STRUTURE in which C -atoms are present at the corners as well as face- centres and alternatetetrahedral voids. Contribution of C- atoms at the corners = ` 8 xx 1/8=1` Contribution of C-atoms at face-centres = ` 6 xx 1/2 =3` As four C -atoms are present on the body diagonals i.e, in the alternate tetrahedral voids, their contribution = 4. Total no. of C -atomsin the unit cell =1+3+ 4= 8 . |
|
| 43. |
Which of the following compounds is covalent ? . |
| Answer» Solution :`H_(2) H-H` | |
| 44. |
Which one Fe^(3+), Fe^(2+) is more paramagnetic and why ? |
| Answer» Solution :As `Fe^(3+)` contains 5 unpaired electrons while `Fe^(2+)` contains only 4 unpaired electrons, `Fe^(3+)` is more PARAMAGNETIC. | |
| 45. |
Which one of the following is an example of chemical equilibrium ? |
|
Answer» `2 NO(g) + O_(2) (g) HARR 2NO_(2) (g)` |
|
| 46. |
The standard electrode potentials at 298 k are given below E_(Zn^(2+)//Zn)^(@)=-0.76 V, E_(Fe^(2+)//Fe)^(@)=-0.44V E_(H^(+)//H_(2))^(@)=0.0V and E_(Cu^(2+)//Cu)^(@)=+0.34V Which of the two electrodes should be combined to form a cell having highest EMF? Identify the cahode and the anode and write the cell reaction also mention the direction flow of electrons in the external as well as the internal circuit |
|
Answer» SOLUTION :To have maximum EMF the anode should have theminimum and cathode should have the maximum `E^(@)` THEREFORE `Zn^(2+)//Zn` couple should be made as the anode while `1Cu^(2+)//Cu` should be made as the cathode the cell reaction `Zn(S)+Cu^(2+)(AQ)rarrrZn^(2+)(aq)+Cu(s)` EMF`=E_(cu^(2+)//Cu)^(@)0-E_(Zn^(2+)//Zn)^(2)=+0.34 -(-0.76)=+1.1 V` the direaction of flow of electrons is from Zn to cu in the external circuit and from cu to zn in the internal CIRCUT the direction of flow of current is howver in the reverse direction i.e from cu to zn in the external circuit and from zn to cu in the internal circuit |
|
| 47. |
The simplest aromatic hydrocarbon C_6H_6 reacts B on treatment with sodium hydroxide will (C_6H_5OH), Phenol, C as the product. Also Cl_2 to give A which on reaction with sodium hydroxide gives B. B of molecular formula C_6H_6O. B on treatment with ammonia will give C_6H_7N as D. Identify A, B, C and explain the reactions involved. |
|
Answer» Solution :(i) The aromatic hydrocarbon A is Benzene, `C_6H_6`. (II) Chlorobenzene B reacts with SODIUM hydroxide to give `(C_6H_5OH)` phenol, C as the product `underset("Benezene A")(C_6H_6 + Cl_2) overset(FeCl_3)to underset("Chloro benzene B")(C_6H_5Cl) + HCl` (ii) Chlorobenzene B reacts with sodium hydroxide to give `(C_6H_5OH)` phenol, C as the product. `underset(R)(C_6H_5Cl_2) + NAOH underset(300atm)overset(350^@C)to underset("PhenolC")(C_6H_5OH + NaCl)` (IV) Chlorobenzene reacts with ammonia to give Aniline, `C_6H_5NH_2` D as the product. `C_6H_5Cl + 2NH_3 underset(50 atm) overset(250^@C)to underset("Aniline D")(C_6H_5NH_2) + NH_4Cl` |
|
| 48. |
Which of the following statement is (are) not correct ? |
|
Answer» Weak electrolytes are `100%` dissociated at infinite dilution |
|
| 49. |
What happens when (i) Quicklime is heated with silica. (ii) Sodium burns vigorously in oxygen. |
|
Answer» Solution :(i)Calcium Silicate is formed `CaO+SiO_(2)toCaSiO_(3)` (II) SODIUM BURNS vigorously in oxygen `2Na+O_(2)overset(575K)(to)Na_(2)O_(2)` |
|
 <
<