Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following represents the correct IUPAC name for the compounds concerned? But-3-yn-1-ol or But-4-ol-1-yne |
|
Answer» |
|
| 2. |
Thevalue of plankconstantis 6.63 xx 10^(10^(34) kg thespeedslightis 3xx 10^(17)nms^(-1)whichvalue isclossedto the wavelengthin nanometerof a quantumof lightwithfrequencyof 6 xx 10^(15)s^(-1) ? |
|
Answer» 25 |
|
| 3. |
Which can be purified by crystalization? |
|
Answer» PHENOL |
|
| 4. |
Which of the following conditions are true about spontaneous process? |
|
Answer» `(DELTA G_("system"))_(T.P) LT 0` |
|
| 5. |
What volume of 95% sulphuric acid (density = 1.85 g cm^(-3)) and what mass of water must be taken to prepare 100 cm^3 of 15% solution of sulphuric acid (density = 1.10 g cm^(-3)) ? |
|
Answer» Solution :Calculation of mass of `H_2SO_4` required to PREPARE `100 cm^3` of 15% solution (d = `1.10 g cm^(-3)`) : 15% solution WOULD CONTAIN 15 g of `H_2SO_4` in 100 g of solution. Density `=("Mass.)/("Volume")` `therefore` Volume of 100 g of the solution `=("Mass")/("Density") = 100/1.10 = 90.91 cm^(3)` Thus, 90.91 cm of the given solution contain 15 g of `H_2SO_4`. `therefore`Mass of `H_2SO_4` required to prepare 100 cm of the solution `=15/90.01 xx 100 = 16.5 g` Calculation of volume of 95% `H_2SO_4` (d = `1.85 g cm^(-3)`) containing 16.5 g of `H_2SO_4` : SINCE, the given sample is 95%, the mass of the sample containing 16.5 g of `H_2SO_4` `=100/95 xx 16.5 = 17.37 g` `therefore` Volume `=("Mass")/("Density") = 17.37/1.85 = 9.39 cm^(3)` `therefore` Mass of water to be taken `=110 - 17.37 = 92.63 g` |
|
| 6. |
Which of the following statements are correct (A) Carbamates are nerve toxins (B) diethanol amine is an environment friendly herbicide (C) Fly ash is a contaminent in air due to thermal power plants and steel industry (D) Haemoglobin of the blood forms carboxy haemoglobin with CO The correct answer is |
|
Answer» A, B, C |
|
| 7. |
Which of the following statement(s) is(are) true? |
|
Answer» IONISATION energy `prop 1/"Screening effect"` |
|
| 8. |
Which ion gives green coloured bead during qualitative analysis by Borax bead test ? |
| Answer» Answer :D | |
| 9. |
You are given the following species C_(2)^(+), O_(2)^(2+), Be_(2), C_(2), O_(2)^(2-), C_(2)^(-) Arrange them in order of increasing bond strength giving reason. |
|
Answer» Solution :`C_(2)^(+)(11)=sigma_(1s)^(2)sigma_(1s)^(**2)sigma_(2s)^(2)sigma_(2s)^(**2) (pi_(2P_(x))^(2)= pi_(2p_(y))^(1))` Bond order = ` (1)/(2) (N_(b)- N_(a)) = (1)/(2) (7-4) = 1.5` `C_(2)^(2+)(14)=sigma_(1s)^(2)sigma_(1s)^(**2)sigma_(2s)^(2)sigma_(2s)^(**2)sigma_(2p_(Z))^(2) (pi_(2P_(x))^(2)= pi_(2p_(y))^(1))` Bond orger` = (1)/(2) (10 - 4) = 3 ` `Be_(2) (8) = sigma_(1s)^(2)sigma_(1s)^(**2)sigma_(2s)^(2)sigma_(2s)^(**2)` Bond order`= (1)/(2) (4-4) = 0 ` `C_(2)(12)=sigma_(1s)^(2)sigma_(1s)^(**2)sigma_(2s)^(2)sigma_(2s)^(**2)sigma_(2p_(z))^(2) (pi_(2P_(x))^(2)= pi_(2p_(y))^(2))` Bond order ` = (1)/(2) (8-4) = 2 ` `O_(2)^(2-) (18) = sigma_(1s)^(2)sigma_(1s)^(**2)sigma_(2s)^(2)sigma_(2s)^(**2)sigma_(2p_(z))^(**2) (pi_(2P_(x))^(2)= pi_(2p_(y))^(2)) (pi_(2P_(x))^(**2)= pi_(2p_(y))^(**2))` Bond order ` (1)/(2) (10 - 8 ) = 1 ` `C_(2)^(-)(13)=sigma_(1s)^(2)sigma_(1s)^(**2)sigma_(2s)^(2)sigma_(2s)^(**2) (pi_(2P_(x))^(2)= pi_(2p_(y))^(2))sigma_(2p_(z))^(1)` Bond order ` = (1)/(2) (9 - 4) = 2.5 ` Greater the bond order, greater is the bond strength . Hence, INCREASING order of bond strength will be `Be_(2) lt O_(2)^(2-) lt C_(2)^(+) lt C_(2) lt C_(2)^(-) lt O_(2)^(2+)` . |
|
| 10. |
Which of the following reactions are completed through free radical intermediate ? |
|
Answer» `(CH_3)_3CH+Br_2overset(hv)to (CH_3)_3 CBr+HBr`<BR>`(CH_3)_3C=CH_2+Br_2 to CH_3-undersetunderset(CH_3)(|)overset(Br)overset(|)C-oversetoverset(Br)(|)CH_2` |
|
| 11. |
Three pairs of compoundsare given below. Identify that compoundin each of the pairswhich hasgroup 13 elementin morestableoxidationstate. Given reasonfor your choice. State the nature ofbonding also. (i) TlCl_(3), TlCl, (ii) AlCl_(3), AlCl, (iii) InCl_(3), InCl |
|
Answer» Solution :(i) Due to stronginert pair effect ,+1oxidation state of Tl is more stable than `+3`. Since compoundsin lower oxidation state are ionic but covalent in higher oxidationstate,therefore `TlCl_(3)` is less stable and covalentin nature but `TlCl` is more stableand is ionic in nature. (ii) Due toabsence ofd-orbitals, Al does not show inert pair effect, Therefore its most stable oxidationstate is +3. Thus, `AlCl_(3)`is much more stable than `AlCl`. Furtherin the solid or the vapour state, `AlCl_(3)` is covalent in nature but in aqueous solution , it IONIZES to FORM `Al^(3+) (AQ)` and `CL^(-) (aq)` ions. (iii) Due to inert pair effect, indium exists in both +1and +3oxidationstates out of which +3 oxidation state is more stable than +1 oxidationstate.In other WORDS,`InCl_(3)`is more stable than `InCl`. Being unstable, IClundergoesdisproportionation reaction. `3ICl (aq) rarr 2 In (s) + In^(3+) (aq) +3Cl^(-) (aq)` , |
|
| 12. |
Which is more reactive, C_(6)H_(6) or C_(6)D_(6) towards electrophilic substitution? |
|
Answer» Solution :The mechanism involves two STEPS in which scission of C-H or C-D is a fast step. HENCE both substances `C_(6)H_(6)-C_(6)D_(6)` REACT at the same rate. |
|
| 13. |
Which quantum number reveal information about the shape, energy, orientation and size of orbitals? |
| Answer» SOLUTION :Magnetic quantum NUMBER reveal information about the shape, ENERGY orientation and SIZE of orbitals. | |
| 14. |
What is heterogeneous equilibria ? Give its types with examples. |
|
Answer» Solution :Equilibrium in a system having more than one phase is called heterogeneous equilibrium. Type-1: Liquid `hArr` Gas e.g. `H_2O_((L)) hArr H_2O_((g))` Type -2: Solid `hArr` Aqueous solution SATURATED solution : `Ca(OH)_(2(s) + (aq)) hArr Ca_((aq))^(2+) + 2OH_((aq))^(-)` `C_12H_12O_(11(s) + (aq)) hArr C_12H_22O_(11(aq))` Type -3: Solid `hArr` Gas `CaCO_(3(s)) hArr CaO_((s)) + CO_(2(g))` `3C_((s)) + 5/2 O_(2(g)) hArr CO_((g)) + 2CO_(2(g))` `NH_4Cl_((s)) hArr NH_(3(g)) + HCl_((g))` `C_((s)) + CO_(2(g)) hArr 2CO_((g))` `Ni_((s)) + 4CO_((g)) hArr Ni(CO)_(4(aq))` |
|
| 15. |
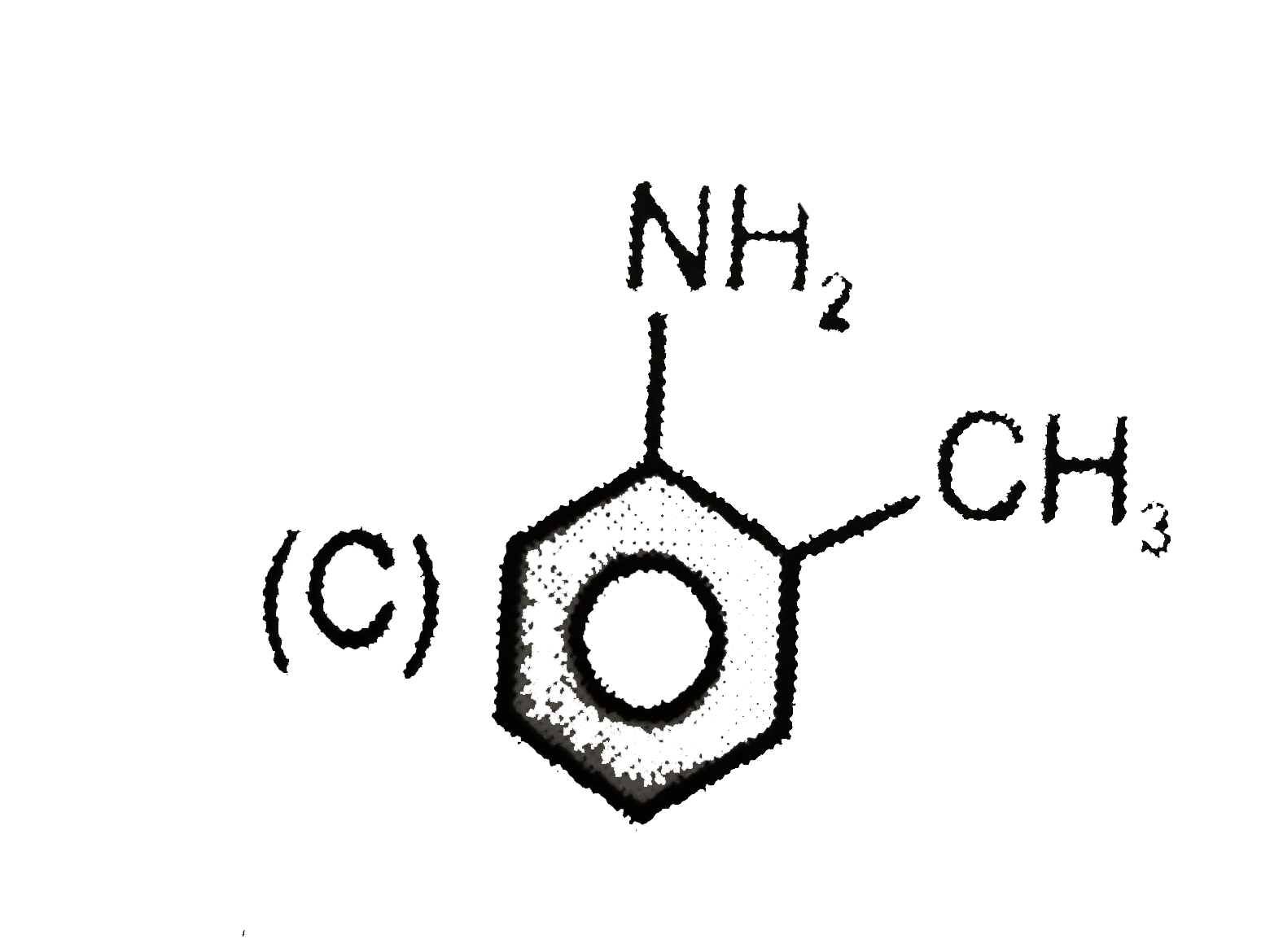
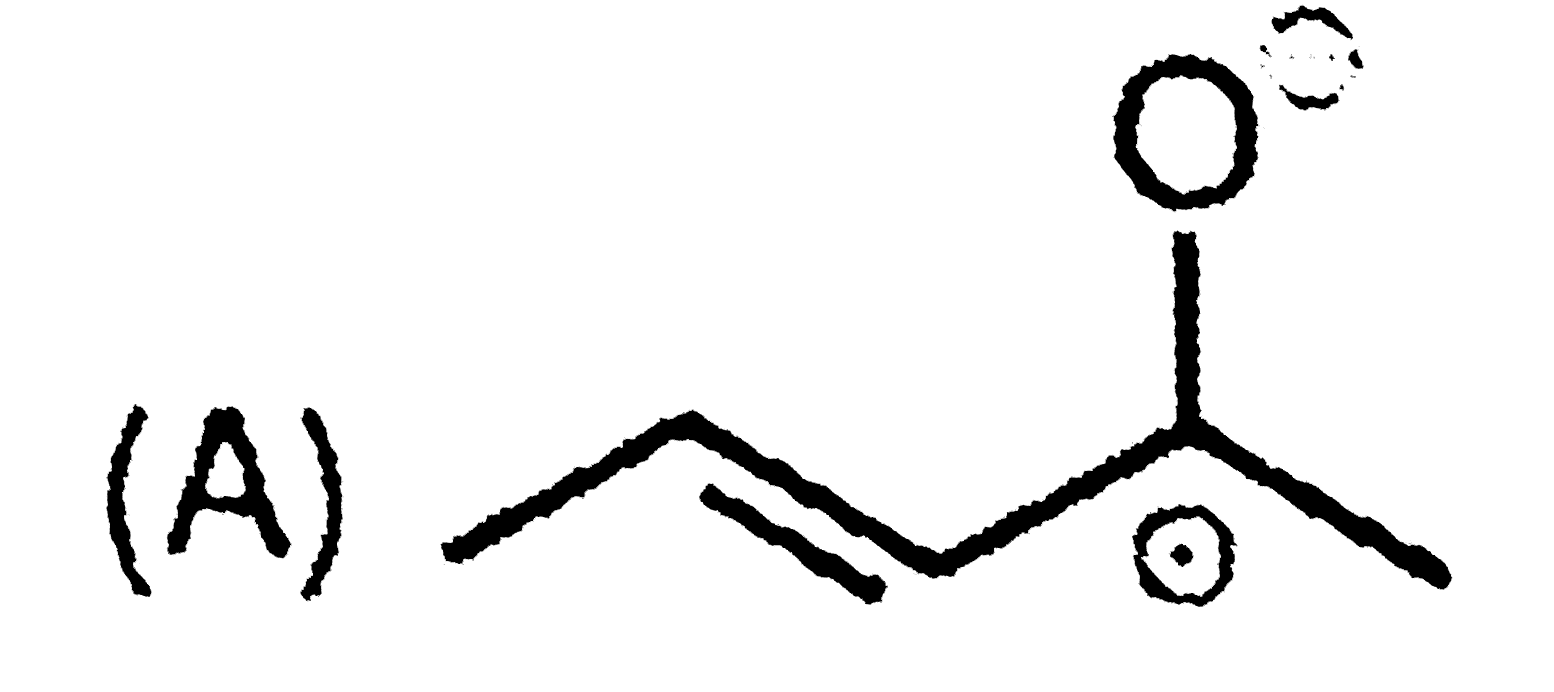
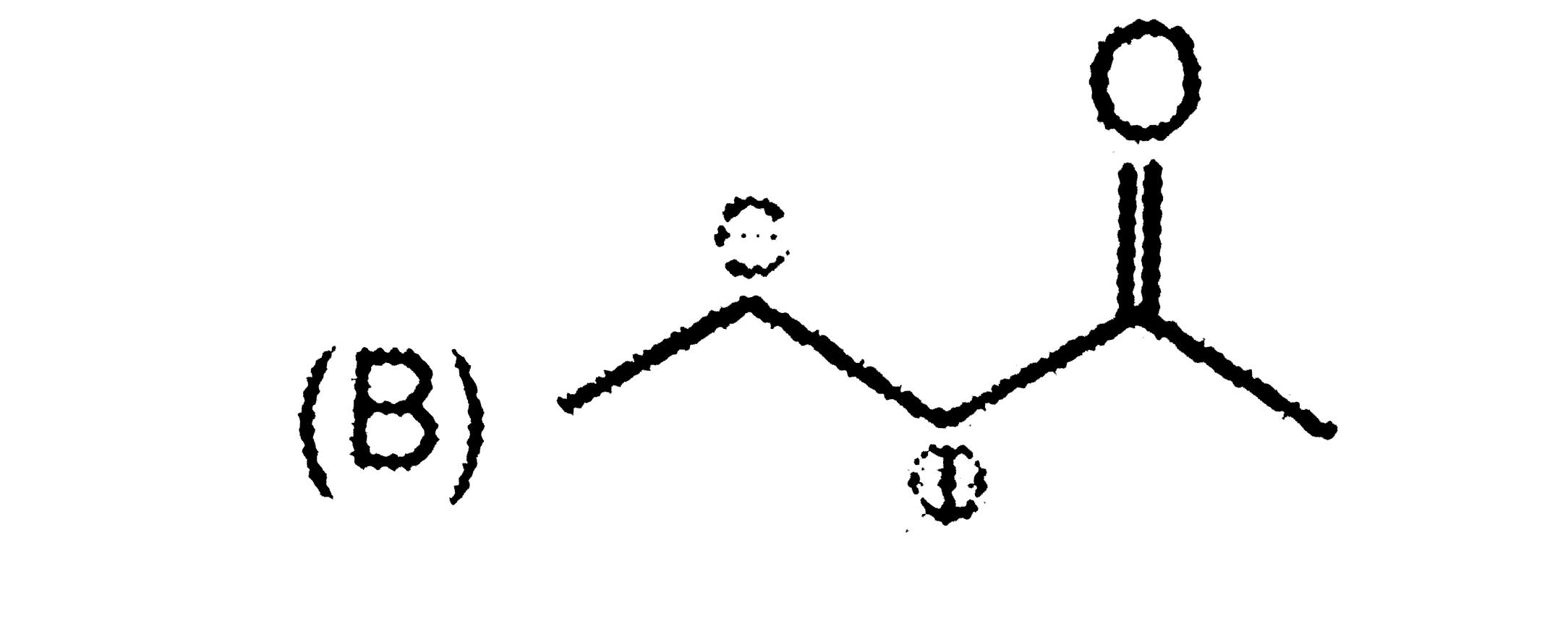
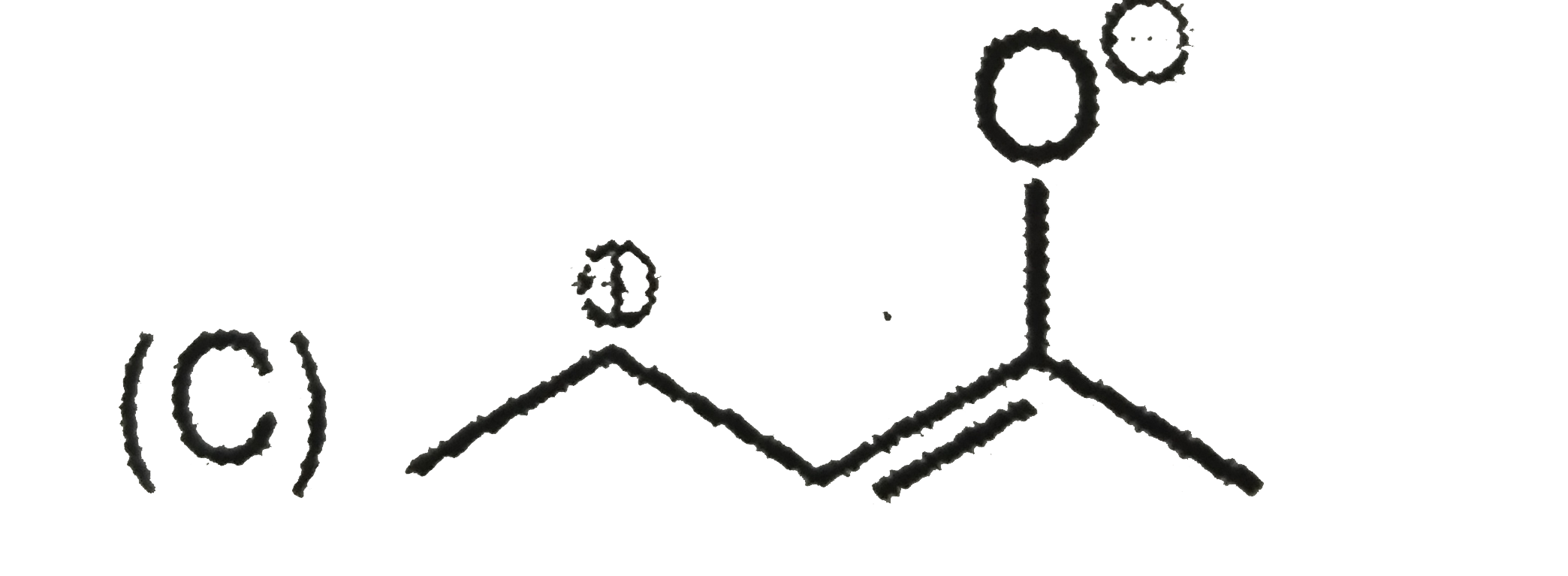
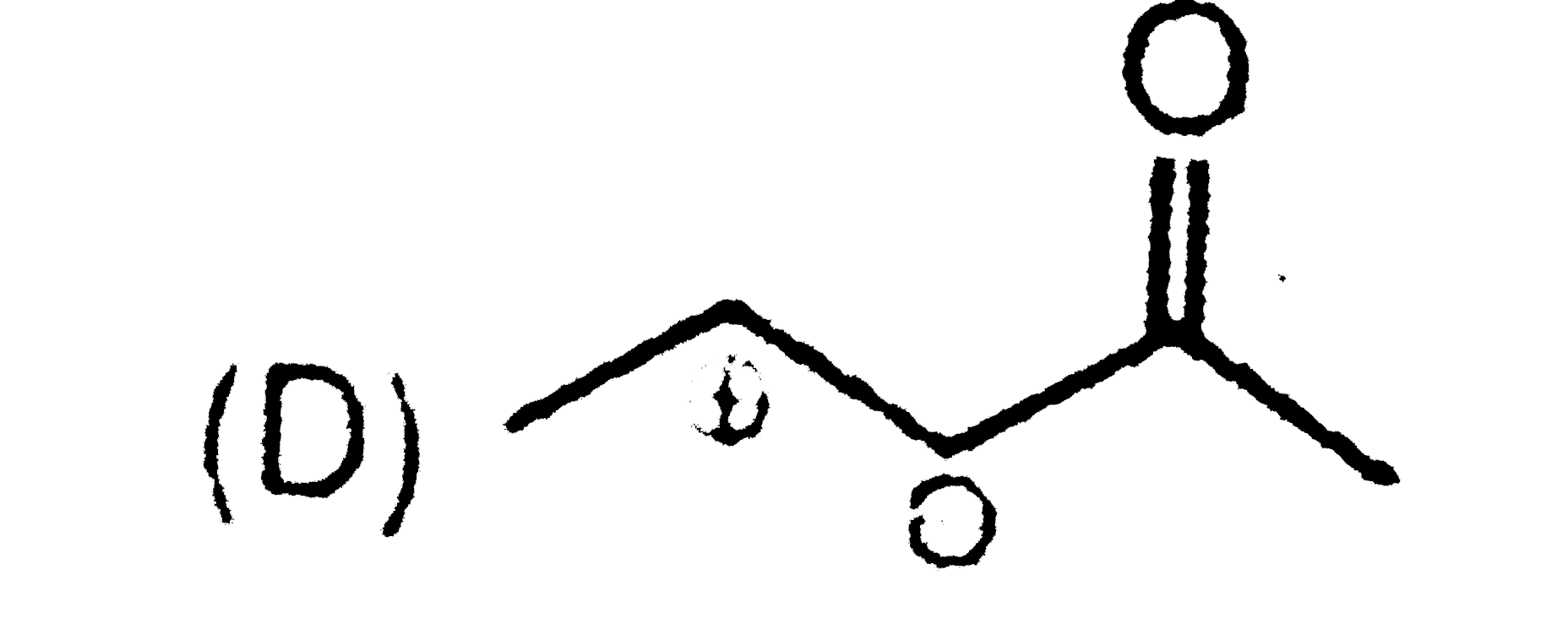
Write the IUPAC names of the following : |
Answer» SOLUTION :
|
|
| 17. |
The weight of H_2O_2present in 70 ml of 6% (w/v) H_2O_2solution is |
|
Answer» 6g |
|
| 18. |
Tonics in general contains |
|
Answer» Ether |
|
| 19. |
Write the Henderson Hasselbalch equation for acidic buffer |
|
Answer» |
|
| 20. |
Thomas Andrew measured variation of the volume with pressure of "____________" at different constant temperatures. |
|
Answer» `CO_(2)` |
|
| 21. |
Two solid compounds A "and" B dissociate into gaseous products at 20^(@)C as (i) A(S)hARrA'(g)+H_(2)S(g) (ii) B(S)hArrB'(g)+H_(2)S(g) At 20^(@)C pressure over excess solid A "is" 50mm and that over excess solid B "is" 68mm find, (a) The dissociation constant of A "and" B (B) Relative no. of moles ofA "and" B' in the vapour phase over a mixture of solid A "and" B. (c) Show that the total pressure of the gas over the solid mixture would be 84.4mm |
|
Answer» `A(S)hArrA'(g)+H_(2)S(g) P=50mm rArr P_(H_(2)S=P_(A).=P//2=25mm` `B(S)hArrB'(g)+H_(2)S(g) P=68mm rArr P_(H_(2)S=P_(B')=P//2=34mm` (i) `K_(P1)=(25)^(2)=625MM^(2)` `K_(P2)=(34)^(2)=1156mm^(2)` (ii) Ratio of moles is same as that of PARTIAL pressure so A`(S) hArr A,(g) + H_(2)S(g)` `P_(1) P_(1)+P_(2)` `B(S) hArr B'(g) + H_(2)S(g)` `P_(2) P_(2)+P_(1)` `K_(p1)=P_(A.)xxP_(H_(2)S=P_(1)(P_(1)+P_(2))` .....(i) `K_(p2)=P_(B.)xxP_(H_(2)S=P_(2)(P_(1)+P_(2))` .....(ii) `(K_(p1))/(K_(P2))=(P_(1))/(P_(2))=(625)/(1156)` (iii) TOTAL pressure `=P_(1)+P_(2)+(P_(1)+P_(2))=P_(1)+P_(2))` (i)+(ii)`=(p_(1)+P_(2))^(2)` `sqrtK_(P1)+K_(P2)=P_(1)+P_(2)` `P_(T)=2xx(sqrtK_(P1)+K_(P2))=84.4mm`. |
|
| 22. |
Which of the following pairs of ions is isoelectronic and isostructural ? |
|
Answer» `CO_(3)^(2-) , NO_(3)^(-)` Shape = TRIGONAL planar In `NO_(3)^(-)`, no . of electrons= ` 7 + 3 xx 8 + 1= 32` Shape = trigonal planer In `SO_(3)^(2-) `, no of electrons = ` 16 + 3 xx 8 + 2 = 42 ` Shape = pyramidal In `CIO_(3)^(-)`, no. of electrons ` 17 + 3 xx 8 + 1= 42` Shape = pyramidal |
|
| 23. |
What is hydrogen bond ? Explain its characteristics with example. |
|
Answer» Solution :Attraction force present between partially positive `n^(+)` ion and partially negative molecules like N - H, O - H or H - F is Known as HYDROGEN Bond. Example :(i) N - H of `NH_(3)`, (ii) O - H of `H_(2)O, C_(2)H_(5)OH, C_(6)H_(5)OH`(iii) H - F of H - F,(iv) O - chloro phenol formation of H - Bond in H - F. `overset(delta+)(H)-overset(delta-)(F).....overset(delta+)(H)-overset(delta-)(F)` Characteristics : Although hydrogen bonding is regarded as being limited to N, O, F , but species such as `Cl^(-)` may also participate in hydrogen bonding. ENERGY of hydrogen bond varies between 10 to 100 KJ `mol^(-1)`. This is quite a significant amount of energy , therefore hydrogen bonds are powerful force in determining the STRUCTURE and properties of many compounds, for example proteins and nucleic acids. Strength of the H-bond is determined by coulombic interaction between the lone pair electrons of the electronegative atom of one molecule and the hydrogen atom of other molecule. Energy of hydrogen Bond varies between 10 to 100 kJ `mol^(-1)` This is quite a significant amount of energy , therefore, hydrogen bonds are powerful force in determining the structure and properties of many compounds, for example proteins and nucleic acids. Distance between molecules in same substance increases, strength of H - Bond decreases. ex. : Solid `to` Liquid `to` Gas |
|
| 25. |
Tolerable limits of lead and fluorides drinking water according to international standard are respectively |
|
Answer» 50 PPM and 3 ppm |
|
| 26. |
Writr note on the necessity for Hund's rule. |
| Answer» Solution :Aufbau.s principle does not DEAL with the filling of ELECTRONS in the DEGENERATE orbitals (i.e. orbitals having same energy) such as px, PY and pz. | |
| 27. |
The valuse of K_(p_(1)) and K_(p_(2)) for the reactions XhArrY+Z AhArr2B are in the ratio 9:1 if degree of dissociation and initial concentration of X and A be equal then total pressure at equilibrium P_(1), and P_(2) are in the ratio |
|
Answer» `36:1` |
|
| 28. |
Which of the following compound will give smell of NH_(3) with conc. NaOH |
|
Answer» `CH_(3)-CH_(2)-underset(O)underset(||)(C)-NH_(2)` |
|
| 29. |
Triethylamine is reacted with a peracid to obtain. The nitrogen atom in X has formal charge. |
| Answer» SOLUTION :N//A | |
| 30. |
What is the reaction of concentration of solution and degree of hydrolysis ? |
|
Answer» Solution :No RELATION. The VALUE of hydrolysis is FREE from the concentration of solution. (For SALT of weak acid + weak base) |
|
| 31. |
Which one of the following is not identified by Lassaigne's test? |
|
Answer» a)nitrogen |
|
| 32. |
Which oxide of carbon is an anhydride of carbonic acid ? |
| Answer» SOLUTION :`CO_(2)` ( CARBON DIOXIDE) | |
| 33. |
What is correct about isothermal expansion of the ideal gas ? |
|
Answer» `W _(rev) = W _(IR R)` |
|
| 34. |
Write the acidity and calculate equivalent weight of following base. a. Ca(OH)_(2) b. Fe(OH)_(3) |
Answer» SOLUTION : 
|
|
| 35. |
What quantity (in mL) of a 45% acid solution of a mono-protic strong acid must be mixed with a 20% solution of the same acid to produce 800 mL of a 29.875 % acid solution ? |
|
Answer» 330 |
|
| 36. |
What are the maximum number of emission lines when the excitedelectron of a H atom in n = 4 drops to the ground state ? |
|
Answer» |
|
| 37. |
The vapour pressure of C Cl_(4)" at "30^(@)C is 143 mm of Hg.0.5 gm of a nonvolatile non electrolyte substance with molar mass 65 is dissolved in 100 ml of C Cl_(4). What will be the vapour pressure of the solution. Density of C Cl_(4)" at "30^(@)C = "1.58 gm per cc." |
|
Answer» Solution :Vapour pressure of pure solvent, `P^(@)=143`mm of HG. Vapour pressure of solution, `p = ?` Weight of solute, `W_(2) = 0.5 gm` Mol. wt of solute, `M_(2) = 65` Mol. wt of solvent `(C Cl_(4)), M_(1) = 154` Weight of solvent, `W_(1) = 100xx1.58 =158 gm` `("MASS" =" DENSITY"xx "volume")` `(p^(@)-p)/(p^(@))=(W_(2))/(M_(2)).(M_(1))/(W_(1))"(By Raoult.s law)"` `therefore (143-p)/(143)=(0.5)/(65)xx(154)/(158)` `therefore"p = 141.93 mm of Hg."` |
|
| 38. |
Which of the followng substances undergo disprotionation reaction under basic medium ? |
|
Answer» `F_(2)` |
|
| 39. |
What is chloropicrin? How is it obtained? Mention its uses. |
|
Answer» Solution :(i) Chloropicrin is `C Cl_3 NO_2`. (II) Chloroform reacts with NITRIC acid to form chloropicrin (trichloro-nitromethane): `UNDERSET("Chloroform")(CHCl_3) + HNO_3 overset(Delta)to underset("Chloropierin")(C Cl_3 NO_2) + H_2O` (iii) Chloropicrin is used as an insecticide and soil sterilising AGENT. |
|
| 40. |
Which of the following statements are not correct? Rectify them. (a) sodium when heated in excess of air forms sodium oxide. (b) Solvay ammonia process is employed for the manufacture of both Na_(2)CO_(3) and K_(2)CO_(3). (c ) Rock salt is chemically NaCI. (d) Water glass is sodium silicate. (e) NaHCO_(3) is less soluble in water then KHCO_(3). (f) Potassium is used in photoelectric cells. (g) The solution of Na_(2)CO_(3) in water is basic due to cationic hydrolysis. Microcosmic salt has formula Na(NH_(4)) HPO_(4).4H_(2)O (i) Glaubar's salt is Na_(2)SO_(4).5H_(2)O. (j) Sodium carbonate upon heating forms CO_(2). |
|
Answer» Solution :(a) Sodium upon heating in excess of air forms sodium peroxide. `2Na + O+_(2) to Na_(2)O_(2)` (b) Solvay Ammonia process is employed for the manufacture of only `Na_(2)CO_(3)` because in case of `K_(2)CO_(3)`, the hydrogen carbonate `(KHCO_(3))` expected to be initially formed in the reaction is HIGHLY soluble and can not be precipitated. ltbgt (g) The solution of `Na_(2)CO_(3)` in WATER is basic due to anionic hydrolysis and not cationic hydrolysis. Only `CO_(3)^(2-)` IONS (anions) have reacted with `H^(+)` ions while `Na^(+)` ions do not COMBINE with `OH^(-)` ions. `Na_(2)CO_(3) to 2Na^(+) + CO_(3)^(2-)` `2HOH hArr 2H^(+) + 2OH^(-)` `2H^(+) + CO_(3)^(2-) hArr H_(2)CO_(3)` (i) Glauber's salt is `Na_(2)SO_(4).10H_(2)O` Sodium carbonate does not DECOMPOSE upon heating. |
|
| 41. |
Which of the following gas causes green house effect to maximum extent? |
|
Answer» `CH_4` |
|
| 42. |
Write the IUPAC names of the following compounds : |
|
Answer» Solution :(i) 3,3-Dimethylbutan-1-ol (ii) 3,4-Dibromo-4-methylhexan-1-ol (iii) 4-Methoxynitrobenzene (iv) N-Ethyl-N-methylpropan-1-amine (v) 4-Hydroxy-2-methylbutanamide (vi) Methy-2-amimobutanoate (VII) 2-Methylbutanoyl chloride (viii) 4-Nitropentanenitrile (ix) 2,5-Dinitro-3-methylhexane (X) 2,3-Dimethylbutanal (xii) 3-Methylpent-2-endioic acid (xiii) 2-Bromo-5-hydroxy-3-nitrobenzoic acid (xiv) 2-Chloroethoxybenzene (xv) 2-Nitro-N-methylpropanamine (xvi) Pent-3-ynal (xvii) 1,3-Dibromobutane (xviii) 4-Bromo-2-methyl-3-oxopentanoic acid (xix) Methyl-3-chlorobut-2-enoate (xx) 3-Methylcyclohexyne (xxi) 3-Chloro-2-methylbutanoic acid (XXII) 2,5-Dimethylhexane-1,3-doil (xxiii) Chlorophenylmethane or benzylchloride (xxiv) 4-Chloropentan-2-one (xxv) 1-Methoxy-2-methylbutane (xxvi) 4,4'-Diphenylmethanone (xxvii) N-Ethyl-N-methylbenzamide (xxix) 2-Bromo-3-methylbut-2-en-1-ol (xxx) 1,2-Diphenylmethanone (xxxi) But-3-en-2-ol (xxxii) 2,2-Dimethy-1-bromopropane (xxxiii) 2,5-Dimethylphenol (XXXIV) 2-phenylprop-2-en-1-ol (XXXV) 2-chloro-4-methoxypentan-3-one (xxxiv) 3-3 Dimethylpentan-2-ol. |
|
| 43. |
Which of the following is stabilised by mesomeric effect ? |
|
Answer» `CH_(3)-OVERSET(OPLUS)(C)H_(2)` |
|
| 44. |
The slag consists of molten impurities, generally , in the form of |
|
Answer» METAL carbonate |
|
| 45. |
Write the IUPAC names of the following compounds : |
|
Answer» Solution :(i) Prop-2-enal (ii) But-1-en-3-yne (iii) 2,3-Dichloro-2-methylbutan-1-ol (iv) 2-Ethoxy-4-methoxypentan-3-one (v) 2-Methylpropan-2-ol (vi) But-2-en-1-amine (vii) Propenitrile (viii) 2,3-Dihydroxy-4-methoxypentanol (ix) 4-Hydroxypentan-2-one (x) 3-Bromo-5-chloro-3,5-dimethylheptane (xi) 2,4-Dimethylpentan-2-one (XII) 2-Methylbutanoic ACID (xiii) 3-Methyl-3-(N-methyl)-pentananmine (xiv) 2-Methoxy-2,3-dimethylbutanal (xv) 1-Methoxybutane-2,3-dione (xiv) N-Chloro-N-ethymethanamide (xvii) 3-Carbomylpent-3-enoic acid (xviii) Propane-1,2,3-carbonitrile (xix) 3-(Carboxymethyl) heptane-1,7-dioic acid (xx) 2-Ethoxy-4-methoxypentan-3-one (xxi) But-3-en-1-amine (xxii) Ethylethanote ltb rgt (xxiii) 4-Methylhexanoic acid (xxiv) 3-Bromo-2-methylprop-1-ene. (xxv) Pent-2-enal (xxvi) Hex-4-en-3-ol (xxvii) 3-Chloro-2,2-dimethylbutane (xxviii) 2-Bromo-4-chlorohexane (xxix) 4-Chloropent-1-ene (xxx) 2-Methylpropanal (xxxi) 3-Hydroxybutanoic acid (xxxii) 3-Methylbut-2-en-1-ol (XXXIII) 2-Methylpropan-1,3-diol (xxxiv)3-Methylpropan-1,3-diol (xxxv) 2Methylprop-2-en-1-ol (xxxvi) N,N-diethylpropanamine (XXXVII) 2,2-Dimethylpropan-1-ol (xxxviii) 2-(N,N-dimethyl)-butane (xxxix) 4-Methylpentan-2-one (xxxx) 2-Bromo-3-methybutan-1-ol (xxxxi) Ethanedioic acid. |
|
| 46. |
What are the applications of Hess's Law of constant heat summation? |
|
Answer» Solution :(i) It helps to CALCULATE the enthalpies of formation of those compounds which cannot be determined EXPERIMENTALLY. (ii) It helps to DETERMINE the ENTHALPY of allotropic transformations like C (graphite) `rarr` C (diamond). (iii) It helps to calculate the enthalpy of hydration. |
|
| 47. |
Which statement are correct for the reaction, NH_(4)NO_(2)overset(Delta)rarrN_(2)+2H_(2)O |
|
Answer» The reaction is disproportionation in nitrogen `n-f=n-f" of "NH_(4)^(+)=n-f" of "NO_(2)^(-)=3` implies `E=(M)/(3)` |
|
| 48. |
Which of the following resonating structure is the least contributing structure ? |
|
Answer»
|
|
| 49. |
Two samples of lead oxide were separately reduced to metallic lead by heating in a current of hydrogen. The weight of lead from one oxide was half the weight of lead obtained from the other oxide. The data illustrates. |
|
Answer» LAW of RECIPROCAL proportions |
|