Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Whichone fo the following is not a meta director |
|
Answer» `-NH_(2)` |
|
| 2. |
Draw the energy level diagramn of carbon molecule.Calculate its bond order also. |
Answer» Solution :Atomic number of carbon is `6:1s^(2),2s^(2),2P^(2)` 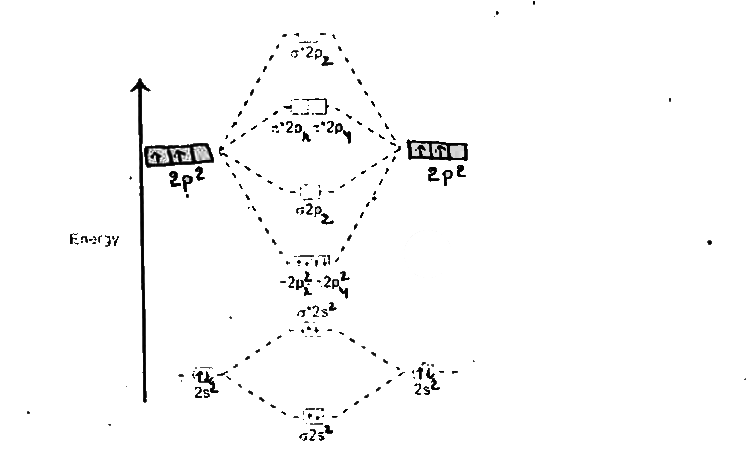 a) Electronic configuration of carbon molecule is `KKsigma2S^(2)SIGMA^(**)2S^(2)pi2p_(x)^(2)" "pi2p_(y)^(2)` b) Bond order `=(6-2)/(2)=2` c) MAGNETIC property = DIAMAGNETIC. |
|
| 3. |
Which of the following compounds gives same product on hydration through hydroborationfollowing by oxidation and hydration through sulphuric acid method? |
|
Answer»
|
|
| 4. |
Which of the following will give charcteristic colour with ferric chloride. |
|
Answer» Allyl alcohol |
|
| 6. |
Which one of the following has the smallest heat of hydrogenation per mole |
|
Answer» 1 - BUTENE |
|
| 7. |
When real gas shows ideal behaviour ? Why ? |
|
Answer» SOLUTION :At low pressure and high temperature PV = nRT Correction of pressure is `(an^(2))/(V^(2))=0` Correction of volume = NB = 0 `p_("REAL")=p_("ideal")` and `V_("real")=V_("Ideal")` |
|
| 8. |
Which of the following is least pollutant ? |
|
Answer» `NO_(X)` |
|
| 9. |
What do you understand by (i) electron deficient (ii) electron precise and (iii) electron rich compounds of hydrogen ? Provide justification with suitable examples. |
|
Answer» Solution :Dihydrogen forms molecular compounds with most of the p-block elements. Most familiar examples are `CH_4, NH_3, H_2O` and HF. For convenience hydrogen compounds of non metals have also been CONSIDERED as hydrides. Being covalent, they are volatile compounds. Molecular hydrides are further CLASSIFIED according to the relative numbers of electrons and bonds in their Lewis structure into : (i) electron-deficient, (ii) electron-precise, (iii) electron - RICH hydrides. An electron-deficient hydride, as the name suggests, has too few electrons for writing its conventional Lewis structure. Example : Diborane `(B_2H_6)` In FACT all elements of group 13 will form electron-deficient compounds. They act as Lewis acids i.e., electron acceptors. Electron-precise compounds have the required number of electrons to write their conventional Lewis structures. All elements of group 14 form such compounds (e.g., `CH_4`) which are tetrahedral in geometry. Electron-rich hydrides have EXCESS electrons which are present as lone pairs. Example : Elements of group 15-17 form such compounds. (`NH_3` has 1-lone pair, `H_2O` -2 and HF-3 lone pairs). They will behave as Lewis bases i.e., electron donors. The presence of lone pairs on highly electronegative atoms like N, O and F in hydrides results in hydrogen bond formation between the molecules. This leads to the association of molecules. |
|
| 10. |
The standard Gibbs energy change for the reaction N_(2)(g) + 3H_(2)(g) iff 2NH_(3)(g) is -33.2 kJ"mol"^(-1) at 298 K. (a) Calculate the equilibrium constant for the above reaction. (b) What would be the equilibrium constant if the reaction is written as 1/2N_(2)(g) + 3/2H_(2)(g) iff NH_(3)(g) (c) What will be the equilibrium constant if the reaction is NH_(3)(g) iff 1/2N_(2)(g) + 3/2 H_(2)(g). |
|
Answer» Solution :(a) For the reaction : `N_(2)(G) + 3H_(2)(g) to 2NH_(3)(g)` `DeltaG^(@) = -33.2 kJ"mol"^(-1)` `log K = -(DeltaG^(@))/(2.303 RT)` `R = 8.314 J K^(-1) "mol"^(-1),T = 298 K` `therefore log K = -(-33.2xx 10^(3)J "mol"^(-1))/(2.3030 xx (8.314 J K^(-1) "mol"^(-1)) xx(298 K))` ` = 5.82` or K = `6.6 xx 10^(5)`. (b) For the reaction : `1/2N_(2)(g) + 3/2H_(2)(g) to NH_(3)(g)` `DeltaG^(@) = 1/2 xx (-33.2) = -16.6 kJ"mol"^(-1)` or `logK = (-16.6 xx 10^(3)J mol^(-1))/(2.303 xx (8.314 J K^(-1) "mol"^(-1)) xx (298 K)) = 2.91` or `K = 8.1 xx 10^(2)`. (c) For the reaction : `NH_(3)(g) to 1/2N_(2)(g) + 2/3 H_(2)(g)` `DeltaG^(@) = -[-16.6 kJ"mol"^(-1)] = 16.6 kJ mol^(-1)` `logK = -(16.6 xx 10^(3)J"mol"^(-1))/(2.303 xx (8.314 J K^(-1) "mol"^(-1))xx (298 K)) = -2.91` or `K = 1.23 xx 10^(-3)`. |
|
| 11. |
Which one of the following orders is not in accordance withthe property stated against it- |
|
Answer» `F GT Cl gt Br gt I`, Oxidising POWER |
|
| 12. |
Which one of the following will not give flame test? |
| Answer» Solution :Be and mg don’t give FLAME test because flame energy is not sufficient to EXCITE the electrons. | |
| 13. |
The uncertainty in position and velocity of a particle are: 10^(-10)mand 5.27 xx 10^(-24) m sec^(-1) respectively. Calculate the mass of the particle (h = 6.625 xx 10^(-34) J sec) |
|
Answer» |
|
| 14. |
Write the functional group of the following compounds: (i) carboxylic acid (ii) Acid anhydride (iii) Acylchloride (iv) Amide (v) Imines (vi) Nitroso compound |
Answer» SOLUTION : 
|
|
| 15. |
Write t he IUPAC name for thefollowing compoundCH_(3)- underset(CH_(3))underset(|)(CH)-CH_(2)-CH_(3) |
|
Answer» SOLUTION :IUPAC names 2 - METHYLBUTANE |
|
| 16. |
Which of the following optoins are correctly matched? |
|
Answer» TFTF 
|
|
| 17. |
Which of the following atom has the lowest ionization enthalpy? |
|
Answer» `1s^(2)2s^(2)2P^(3)` |
|
| 18. |
What are interstitials in a crystal ? |
| Answer» Solution :Atoms or ions that FILL the NORMAL VACANT INTERSTITIAL VOIDS in a crystal are called interstitials | |
| 19. |
Which halogen does not appreciably react with methane in a free-radical substitution reaction? |
|
Answer» Chlorine |
|
| 20. |
The threshold frequency for a certain metal is 3.3xx10^(14) cycle/sec, calculate the cut-off potential for the photoelectron. |
|
Answer» |
|
| 21. |
Which of the following is reduced by H_(2)O_(2) in acid medium |
|
Answer» `KMnO_(4)` |
|
| 22. |
Two separate bulbs contain ideal gas A and B. The density of a gas A is twice that of a gas B. The molecular mass of A is half that of gas B. The two gases are at the same temperature. The ratio of the pressure of A to that gas B is |
| Answer» ANSWER :C | |
| 23. |
Which following are in correct |
|
Answer» `BeCO_(3) gt mgCO_(3) gt CaCO_(3) gt SrCO_(3) gt BaCO_(3)` (solubility) `BaO gt SrO gt CaO gt mgO gt BeO` Decreasing hydration ENERGY `Be gt mg gt Ca gt Sr gt Ba` |
|
| 24. |
The total number of monohalogenated products formed byhalogenation of 2,2,4-trimethylhexane is |
|
Answer» 5 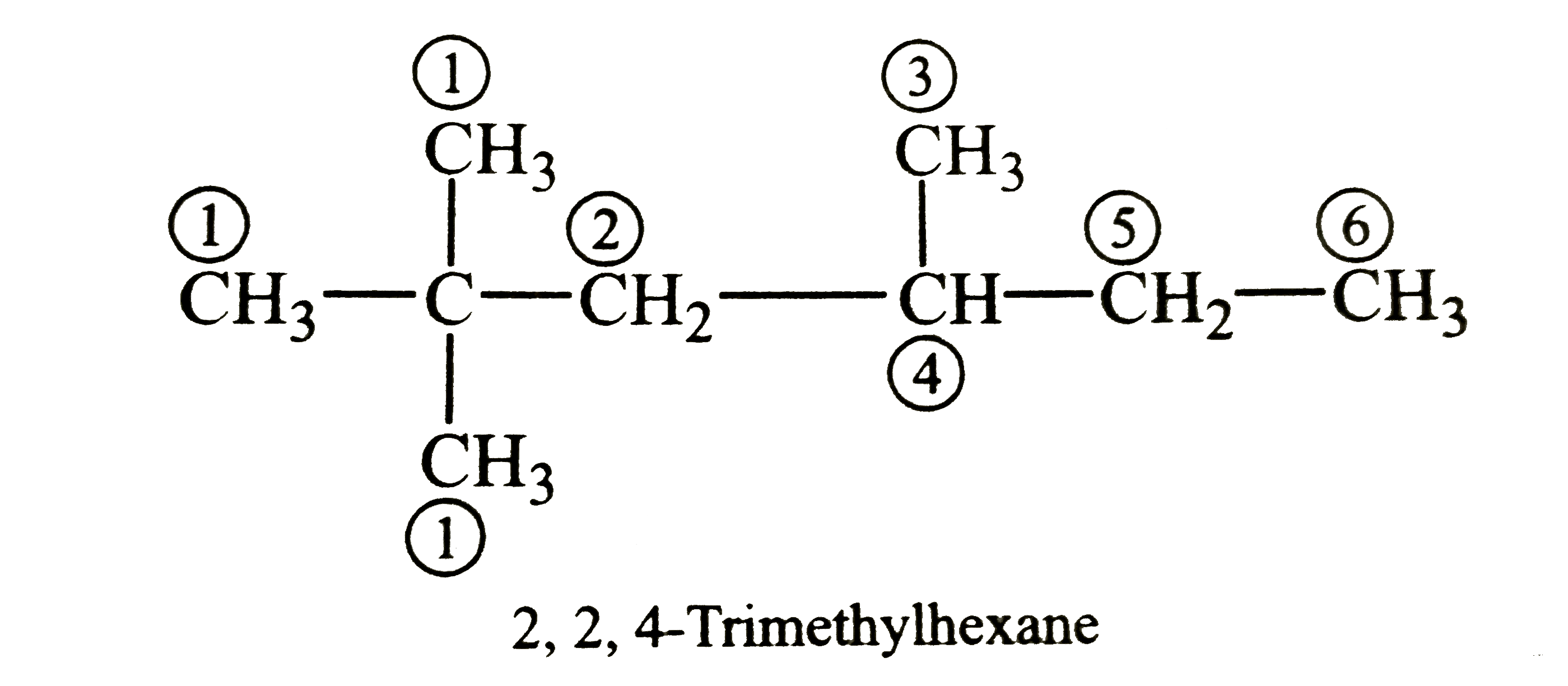 2,2,4-Trimethylhexane has six sets of DIFFERENT HYDROGENS and hence gives six monohalogenated products on HALOGENATION. |
|
| 25. |
Which is a wrong statement ? |
|
Answer» Total ORBITAL ANGULAR MOMENTUM of electron in 's' orbital is equal to zero |
|
| 26. |
Which of the following statements is correct regarding the following reaction ? |
|
Answer» Diasetereomers are formed . |
|
| 27. |
Tri alkyl aluminium molecules exists as dimer which contains 3 centered 2e bonds. The coordination number of bridged carbon atoms is |
Answer»  co.ordeination NUMBER = 5 |
|
| 28. |
Which type of reaction is occur in Kolbe's electrolysis ? Why ? |
| Answer» Solution :Kolbr.s electrolysis is of redox KIND reaction because on anode the DEELECTRONATION gives oxidation and on CATHODE ELECTRONATION gives reduction reaction. | |
| 29. |
What is correct for bond order ? |
|
Answer» BO = ABMO no. of ELECTRONS - BMO no. of electronsno. of `e^(-)` INNO. of `e^(-)` in |
|
| 30. |
Which of the following molecules acts as aLewis acid ? |
|
Answer» `(CH_3)_2O` |
|
| 31. |
What is the relationship between the atomic mass and actual mass of one atom of an element ? |
| Answer» SOLUTION :The actual MASS of an ATOM in grams is equal to its atomic mass in grams divided by `6.022 XX 10^(23)` | |
| 32. |
What do you understand by the terms empirical formula and molecular formula? |
Answer» SOLUTION :
|
|
| 33. |
What happens if we compressed and expand CO_(2) at 30.98^(@)C is taken constant ? |
| Answer» Solution :In both condition CURVE will BECOME WIDE but if its up to limit of compressible it will become liquid but there is no effect of COMPRESSED. | |
| 34. |
When the principal quantum number (n=3) , the possible values of azimuthal quantum number (l) is |
|
Answer» 0,1,2,3 |
|
| 35. |
The work done by the liberated gas when 55.85 g of iron (molar mass 55.85 g "mol"^(-1)) reacts with hydrochloric acid in an open beaker at 25^@C |
|
Answer» `-2.48` kJ 1 mole of IRON LIBERATES 1 mole of hydrogen gas 55.85 g Iron = 1 mole Iron `therefore` n=1 `T=25^@C`=298 K `w=-PDeltaV` `w=-P((NRT)/P)` w=-nRT w=-1 x 8.314 x 298 J w=-2477.57 J w=-2.48 kJ |
|
| 36. |
What is the effect of increasing temperature on the entropy ofa substance? |
| Answer» SOLUTION :ENTROPY INCREASES with INCREASE of TEMPERATURE. | |
| 37. |
What is the molecular type of T-shaped molecule ? |
|
Answer» `AB_(4) E_(2)` |
|
| 38. |
What happens when chlorine reacts with slaked lime |
|
Answer» Solution :When chloride is added to slaked LIME, it GIVES BLEACHING powder. `Ca(OH)_(2)+Cl OVERSET(Delta)to underset("Bleaching powder")(CaOCl_(2)+H_(2)O)` |
|
| 39. |
What is the degree of hardness of a sample of water containing 6 mg of MgSO_4 (molecular mass 120) per kg of water ? |
|
Answer» `10^6` g water will contain ` =6000 mg` of `MgSO_4` `120 g "of " MgSO_4`= 100 g of `CaCO_3` So, 6g of `MgSO_4 = 100/120 XX 6 = 5G " of " CaCO_3`, i.e., Hardness= 5 PPM `LiH + H_2O to LIOH + H_2` |
|
| 41. |
Which of the following is the least stable carbanion? |
|
Answer» `HC=-BARC` |
|
| 42. |
Using CO_(2), NH_(3),NH_(4)NO_(3), and K_(2)CrO_(4) as the only reagents, devise a qualitative analysis scheme for separating and identifying the following ions, which might all be present in the same mixture: Ba^(2+), Ca^(2+), Mg^(2+), Na^(o+), Pb^(2+). Assume that each cation present is 0.10M. Sate the conditions of pH and the reagent concentration which are required in each step. |
|
Answer» Solution :`{:(Ba^(2+)),(Ca^(2+)),(MG^(2+)),(Na^(o+)),(Pb^(2+)):}}underset(NH_(3)+NH_(4)^(o+))(_)underset(Pb(OH)_(2)(s))({{:(Ba^(2+)),(Ca^(2+)),(Mg^(2+)),(Na^(o+)):}})underset(NH_(3))(_){{:(Ba^(2+)),(Ca^(2+)),(Na^(o+)),(Mg(OH)_(2)(s)):}}underset(NH_(3))overset(CO_(2))(_){{:(BaCO_(3)(s)),(CaCO_(3)(s)),(Na^(o+)):}` `{:(BaCP_(3),,,,),(CaCO_(3),,,,):}}underset("acidity")rarrunderset(CrO_(4)^(2-))rarr{:(BaCrO_(4)(s),,,,),(Ca^(2+)(aq),,,,):}` The `pB^(2+)` IONS is identified by acidified followed by treatement with chromate ion, upon which it forms a yellow SOLID precitate. The `Na^(o+)` is identifed by the intense yeellow flame it yields. |
|
| 43. |
What are aqueous and non-aqueous solution? Give example. |
|
Answer» Solution : If the solute is dissolved in the solvent WATER, the resultant solution is called as an aqueous solution. e.g., salt in water. (ii) If the solute is dissolved in the solvent other than water such as BENZENE, ether, `C CI_(4)` etc,the resultant solution is called a non aqueous solution, e.g.,` Br_(2) ` in ` C CI_(4)` |
|
| 44. |
Which of the following gcompounds has a stereoisomer that is a meso compund? |
|
Answer» 2,-4-dibromohexne (C ) has plane of symmetry and no INTERNAL compensation `therefore` It is meso. |
|
| 45. |
Which of the following have a planner structure ? |
|
Answer» `XeO_2F_2` |
|
| 46. |
What is lewis symbols. |
| Answer» Solution :The number of valence electrons are represented as .dots. written around the symbol of the ELEMENT such NOTATION are CALLED .LEWIS SYMBOLS. Ex : Li | |
| 47. |
Which of the following will give 2,2-dibromopropane on reaction with HBr? |
|
Answer» `CH_(2)-CH=CH_(2)` |
|
| 48. |
When half-filled valence orbital of one atom overlaps with half-filled valence orbital of other atom, a covalent bond is fonned. Head on or axial overlap leads to the fonnation of sigma bond. Sidewise or lateral overlap of atomic orbitals (p) give rise to the formation of pi-bond. Similarly side wise overlap between d-orbitals can lead to the fomation of delta-bond. In case of pi -bond, the probability of finding the shared electron cloud is zero along the line joining two nuclei while ithe probability of finding the shared electron cloud is maximum along the inter nuclear axis. The strength of bonds by s-s, p-p, s-p overlap is in the order : |
|
Answer» `s-s LT s-p lt p-p ` |
|
| 49. |
When half-filled valence orbital of one atom overlaps with half-filled valence orbital of other atom, a covalent bond is fonned. Head on or axial overlap leads to the fonnation of sigma bond. Sidewise or lateral overlap of atomic orbitals (p) give rise to the formation of pi-bond. Similarly side wise overlap between d-orbitals can lead to the fomation of delta-bond. In case of pi -bond, the probability of finding the shared electron cloud is zero along the line joining two nuclei while ithe probability of finding the shared electron cloud is maximum along the inter nuclear axis. If lhe internuclear axis is x-axis, which of the following will not give non bonding combination? |
|
Answer»
|
|
| 50. |
When half-filled valence orbital of one atom overlaps with half-filled valence orbital of other atom, a covalent bond is fonned. Head on or axial overlap leads to the fonnation of sigma bond. Sidewise or lateral overlap of atomic orbitals (p) give rise to the formation of pi-bond. Similarly side wise overlap between d-orbitals can lead to the fomation of delta-bond. In case of pi -bond, the probability of finding the shared electron cloud is zero along the line joining two nuclei while ithe probability of finding the shared electron cloud is maximum along the inter nuclear axis. lf x-axis is the internuclear axis then which of the following combination will result into the formation of pi-bond? |
|
Answer» `s+2p_(X)` |
|








