Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is the value of root(3)(a/(P_(c )b^(2))) ? |
| Answer» SOLUTION :`SQRT((a)/((a//27b^2)b^2)) = sqrt(27) = 3`. | |
| 2. |
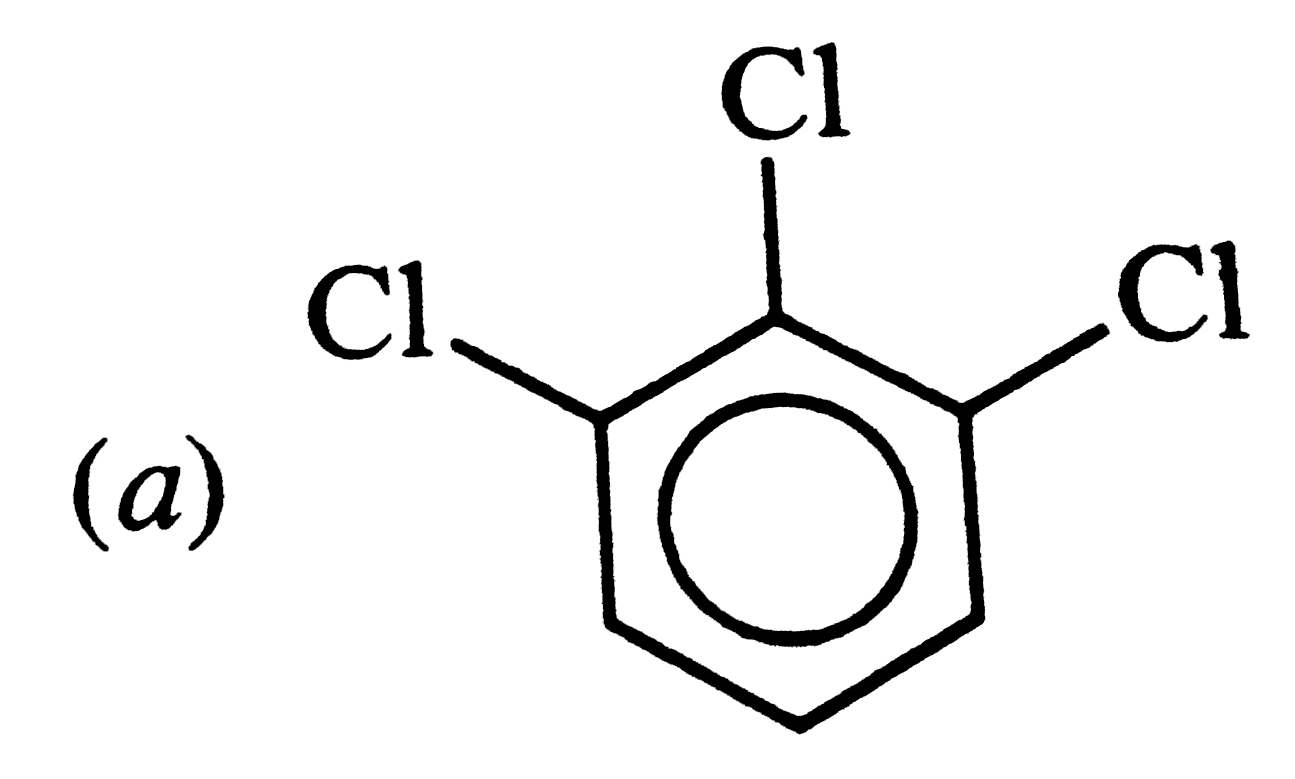
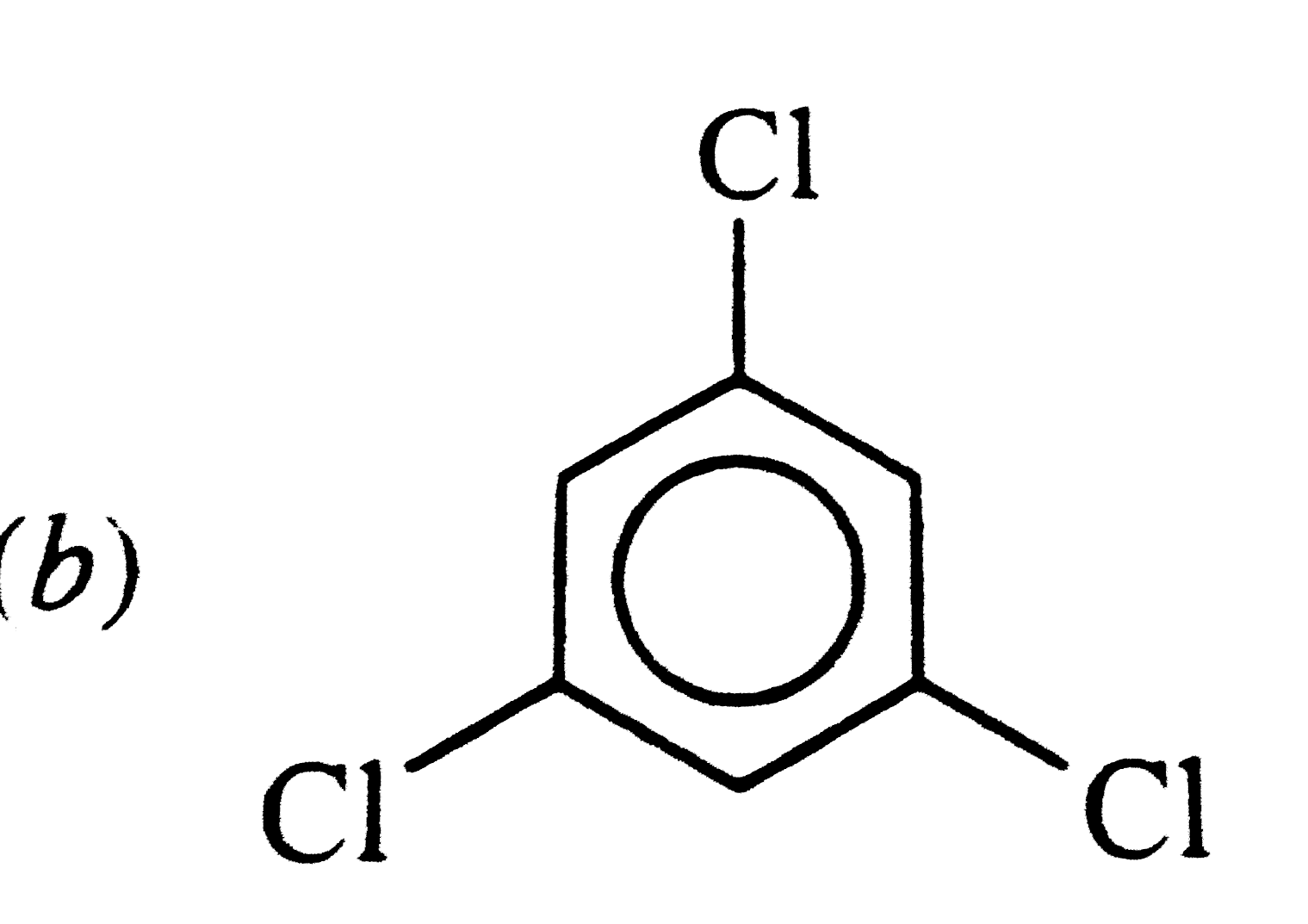
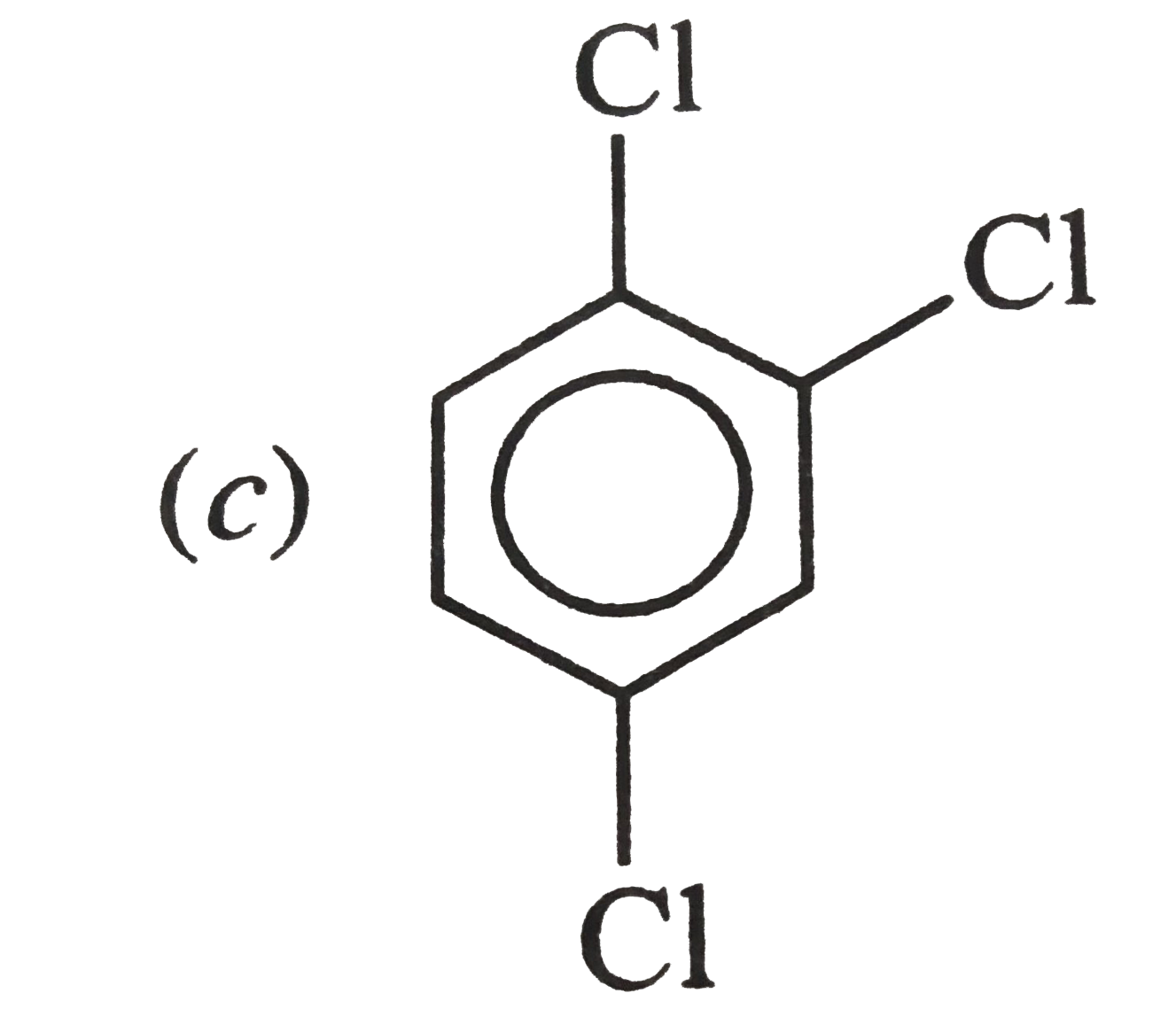
Which of the following has maximum dipole moment ? |
|
Answer»
 , RESULTANT bond MOMENT of , RESULTANT bond MOMENT of`C_(1) - Cl and C_(3) - Cl` bondsadds up to the bond moment of `C_(2) -Cl` . HENCE ,it has maximum value . |
|
| 3. |
Which anion forms the smallest number of insoluble salts? |
| Answer» Answer :B | |
| 4. |
Which of the following is useful for making pure water from a solution of salt in water? |
|
Answer» Filtration |
|
| 5. |
Two elements A (at. wt. = 75) and B (at. wt. = 16) combine to give a compound having 75-8% of A. What is the formula of the compound? |
|
Answer» SOLUTION :100 g of the COMPOUND should contain 75.8 g of A and 24.2 g of B. Moles of `A= ("WEIGHT of A")/( "atomic wt.of A") =(75.8)/(75) =1 ` Moles of B = `("Weight of B")/("atomic wt.of B) = (24.2)/(16)=1.5 ` Thus, molar ratio of A and B in WHOLE numbers is 2:3, and the formula is, therefore, `A_(2),B_(3)`. |
|
| 6. |
Which is//are true statements? |
|
Answer» The layer of ice on the surface of river in the winter acts as a thermal insulator between the water below and the air above |
|
| 7. |
The standard substances used in the enthalpy of combustion of a substance in bomb calorimeter is ____ |
| Answer» SOLUTION :BENZOIC ACID | |
| 8. |
Which of the following allotropic forms of carbon is isomorphous with crystallines silicon ? |
|
Answer» Graphite |
|
| 9. |
Which one of these is not compatible with arenes ? |
|
Answer» Electrophilic |
|
| 11. |
Which of the following salts are responsible for temporary hardness of water ? |
|
Answer» `CA(HCO_3)_2` |
|
| 12. |
Write expression for the work done by 1 moleof the gas in each of the following cases : (i) For irreversible expansion of the gas from volume V_(1) to V_(2). (ii) For reversible isothermal expansion of the gas from volume V_(1) toV_(2). (iii)For expansion of the gas into an evaluated vessel. (iv) For reversibleisothermal compression of the gas from pressure P_(1) to P_(2) (v)For adiabatic expansion resulting into changeof temperature from T_(1) to T_(2). |
|
Answer» Solution :(i)IRREVERSIBLE EXPANSION takes place when external pressure `(P_(ext))` remains constant `w_("irrev")= - P_("ext") (V_(2)-V_(1))= -P_(ext) DeltaV` (ii)Reversible expansion takes place when internal pressure is infinitesimally greater than exterhanl pressure `(P_("int")~=P_(ext))` at EVERY stage. Thsu, externalpressurehas to be ADJUSTED throughout. `w_(rev)= -nRT LN. (V_(2))/(V_(1))` (iii) The expansion is irreversible. Further, as `P_(ext) =0`, therefore, `w= -P_(ext)DeltaV= 0 xx ( DeltaV) = 0` (iv) When gas is compressed, work is done on the gas. For isothermalreversible compression, `w= + nRT ln. (P_(2))/(P_(1))` (v) For adiabatic expansion, temperature falls, i.e.,`T_(2) ltT_(1))` `w=C_(v) (T_(2)-T_(1)),i.e., w` is -ve. |
|
| 13. |
The solubility product of aluminium sulphate is given by the expression |
|
Answer» `4S^(3)` |
|
| 14. |
There are as many notes as number of oxygen atoms in 24.8 gm Na_(2)S_(2)O_(3).5H_(2)O (Mw = 248). A note counting machine counts 48 milion notes per day. Number of days taken to count these notes is |
|
Answer» `10^(17)` `48xx10^(6)rarr1" day"` `0.8xx6xx10^(23)rarr?=10^(16)` |
|
| 15. |
Which of the following bases are strong or weak than water ? ClO_4^(-) , Cl^(-) , Br^(-), I^(-) , NO_3^(-) , HSO_4^(-) |
| Answer» Solution :These all are MUCH weak BASE then water because they are conjugate base of strong `HClO_4, HCL, HBr, HI, HNO_3 , H_2SO_4`. | |
| 16. |
Which of the following is a by product of alcohol industry |
| Answer» Answer :D | |
| 17. |
What is the maximum numbers of electrons that can be associated with the following set of quantum numbers ? |
|
Answer» <P>4 either`3 p_(X)` or ` 3p_(y)` |
|
| 18. |
When 1 mole of super cooled water freezes, its temperature suddenly rises. The source of heat for the process is |
|
Answer» ethalpy of fussion |
|
| 19. |
The solubility of BaSO_(4) in water is 2.33 g 100 mL^(-1). Calculate the percentage loss in weight when 0.2g of BaSo_(4) is washed with a. 1L of water b. 1L of 0.01 NH_(2)SO_(4).[Mw_(BaSO_(4)) = 233 g mol^(-1)] |
|
Answer» Solution :a. Solubility is in general expressed in `gL^(-1)`, so soubility of `BaSo_(4) = 2.33 XX 10^(-3) gL^(-1)` Loss in weight of `BaSO_(4) =` Amount of `BaSO_(4)` soluble. `rArr %`loss `= (2.33 xx 10^(-3))/(0.2) xx 100 = 1.16%` b. `0.01 NH_(2)SO_(4) -= 0.01 N SO_(4)^(2-) ions` `-= 0.0055 M SO_(4)^(2-) ION (n` factor for `SO_(4)^(2-) = 2)` Now presence of `SO_(4)^(2-)` prior to washing `BaSO_(4)` will supress the solubility of `BaSO_(4)` (due to common ion effect. ) The supersion will be governed by `K_(sp)` value of `BaSO_(4)`. So, first calculate the `K_(sp)` of `BaSO_(4)`. Solubility of `BaSO_(4)` in fresh water `= 2.33 xx 10^(-3) gL^(-1)` `= (2.33 xx 10^(-3))/(33) ,mol L^(-1) = 10^(-5)M` `K_(sp)= [Ba^(2+)][SO_(4)^(2-)] = (10^(-5))^(2) = 10^(10)` Now let `x` be the solubility in `mol L^(-1) in H_(2)SO_(4)` `rArr [Ba^(2+)] `in solution `= x mol L^(-1)` and `[SO_(4)^(2-)]` in solution `= (x + 0.005) mol L^(-1)` Ionic product `= [Ba^(2+)] [SO_(4)^(2-)] = (x) (x + 0.005)` `K_(sp) =` Ionic product at equilibrium (SATURATION) `rArr 1.0 xx 10^(-10) = (x) (x + 0.005)` Assuming `x` to be a small number `(x + 0.005) ~~ 0.005` `rArr x = (10^(-10))/(0.005) = 2xx10^(-8)mol L^(-1)` `= 2 xx 10^(-8) xx 233 gL^(-1)` ` = 4.66 xx 10^(-6) gL^(-1)` `rArr 4.66 xx 10^(-6)g of BaSO_(4)` is washed away. `rArr % loss = (4.66 xx 10^(-6)xx100)/(0.2) = 2.33 xx 10^(-3) %` |
|
| 20. |
When 1-butanol is heated with an excess of conc. Sulphuric acid, the main product is |
|
Answer» 1-Butene |
|
| 22. |
Whose boiling point is less in cis and trans geometer ? Explain with example. |
|
Answer» SOLUTION :Trans has less boiling point than the cis isomer. e.g., : Cis-but-2-ene (277 K) has boiling point more than trans-but-2-ene (274 K). In trans GEOMETRY the SUBSTITUTION or molecule groups are opposite to each other, therefore there is less - inter NUCLEAR force and strong van der Waals force. 
|
|
| 23. |
What areideal gases ? |
|
Answer» <P> SOLUTION :An ideal GAS is defined as one is which all collisions between atoms or molecules are perfectly eleastic and in which there are no itermolecular attractive forces. An ideal gas is agas that obeys the ideal gas law, PV= nRT, where n is the number of moles of the gas, R is the ideal gas CONSTANT, pressure P, volume, V, and temperature T . |
|
| 24. |
There are two crystalline forms of PbO , one is yellow and the other isred. The standard enthalpies of formation of thesetwoforms are - 217.3 and -219.0 kJ per mole respectively. Calculate the enthalpy change for the solid - solid phase transition. PbO( yellow ) rarrPbO ( red) |
|
Answer» (II) `Pb(s) + (1)/(2) O_(2)(g) rarr PbO(` Red)`, Delta H = - 219 .0 kJ mol^(-1)` Eqn. (ii) - Eqn. (i) GIVES the requiredresult. |
|
| 25. |
Two moles of H_(2)SO_(4) will be neutralised by |
|
Answer» 1 mole KOH `2rarr4 2rarr2` |
|
| 26. |
Which of the following will produce the highest risein temperature? |
|
Answer» 67 mL of 1M NaOH `+`33 mL of 0.5M `H_(2)SO_(4)` |
|
| 27. |
The volume occupied by 0.32 g of a gas at S.T.R is 224 mL. Calculate the molecular mass of the gas. |
|
Answer» SOLUTION :`therefore` 224 mL of the gas at S.T.P WEIGH = 0.32 G `therefore 22400 mL` of the gas at S.T.P. will weigh `=0.32/224 xx 22400 = 32 g` HENCE, the molecular mass of the gas = 32 amu |
|
| 28. |
Which of the following is used to distinguish ethylene and acetylene. |
|
Answer» ALKALINE `KMnO_(4)` |
|
| 29. |
Which statement(s) is/are correct regarding alkali metals |
|
Answer» THERMAL stability of METAL nitrate increases on moving upward the group. `LiNO_(3) lt NaNO_(3) lt KNO_(3) lt RbNO_(3) lt CsNO_(3)` Metal carbonate solubility `Li_(2)CO_(3) lt Na_(2)CO_(34) lt K_(2)CO_(34) lt Rb_(2)CO_(3) lt Cs_(2)CO_(3)` Only `NaHCO_(3)` is insoluble in water due to association of `HCO_(3)^(-)` by intermolecular H-bonding 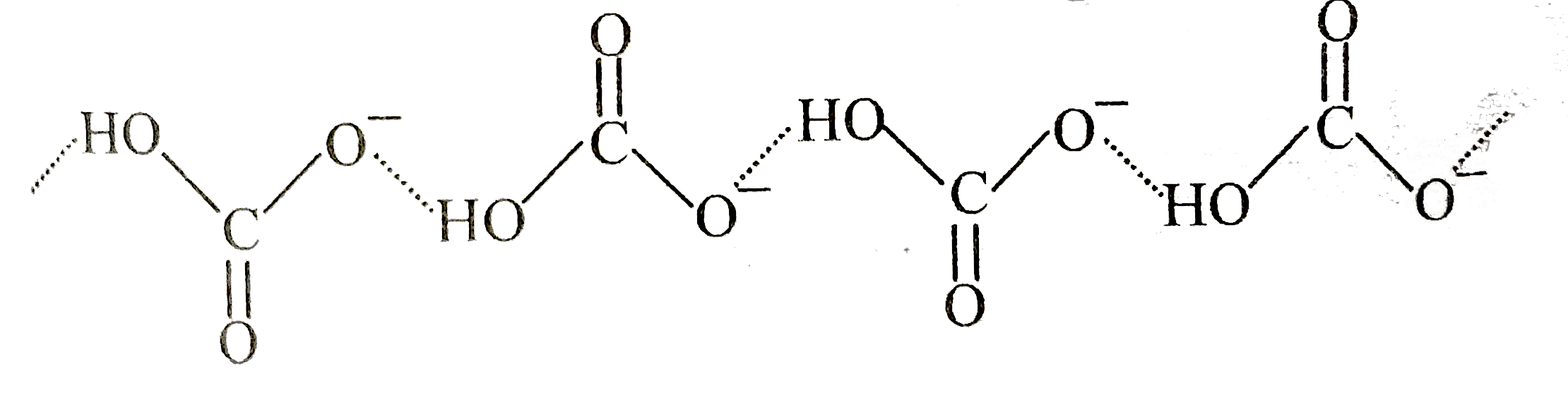
|
|
| 30. |
Which of the following samples of ideal gas will have maximum translation kinetic energy?? |
|
Answer» `(1)/(2)` mole of `CO(g)` at 400K |
|
| 31. |
Volume occupied by one molecule of water (density "1 g cm"^(-3)) is : |
|
Answer» `3.0 xx 10^(-23) cm^(3)` `= ((18g))/(6.022xx10^(23))=3xx10^(-23)g` Since density of water is 1g `:.` Volume occupied by one molecule of water `= 3 xx 10^(-23)` CC. |
|
| 32. |
When chloroform is treated with primary amine and KOH, we get ……………………. . |
| Answer» SOLUTION :OFFENSIVE ODOURS | |
| 33. |
What is buffer solution? Give one example of acidic buffer solution. |
| Answer» Solution :A solution which RESISTS any change in the pH by the addition of little acid or Base is called BUFFER solution. Eg: `CH_(3)COOH` and `CH_(3)COONA` (or) HCOOH and HCOONA | |
| 34. |
Total number of no bond resonance structures in tertiary pentyl cation is |
|
Answer» No. of ` ALPHA`- hydrogen = 8= no. of hyper conjugative strucure . |
|
| 35. |
Which are has highest ONO bond angle ? |
|
Answer» `NO_(3)^(-)` 
|
|
| 36. |
When the value of azimuthal quantum number, l=2, value of .n. will be _________ |
|
Answer» |
|
| 37. |
What is meant by Joule-Thomson effect? |
| Answer» Solution :The phenomenon of LOWERING of TEMPERATURE when a GAS is made to expand adiabatically froom a region of high pressure into a region of low pressure is known as Joule-Thomson EFFECT. | |
| 38. |
The sums of first and second ionization energies and those of third and fourth ionization energies ("in kJ mol"^(-1)) of nickel and platinum are: {:(,(IE_(1)),(IE)_(2)),(,Ni,2.49),(,Pt,2.66):} {:(,(IE_(1)),(IE)_(2)),(,Ni,8.80),(,Pt,6.70):} Justify using above data why Ni^(+2) and Pt^(+4) are more stable than their corresponding counter part ions ? |
|
Answer» Solution :Comparing `IE_1 + IE_2` and Ni and Pt, Ni has LOWER VALUE than Pt THEREFORE,`Ni^(+2)` will be more STABLE than `Pt^(+2)` where as on summating`IE_1, IE_2 , IE_3 and IE_4 . ` `Ni^(4+)=2.49+8.80=11.29` `Pt^(4+)=2.66+6.70=9.36` I.E. of Pt (IV) is LESS than Ni(V) Therefore `Pt^(4+)` is more stable |
|
| 39. |
Why ,benzene undergoes substitution reaction more readily than addition reaction ? |
|
Answer» SOLUTION :`(i)` Benzene CONTAINS delocalized `pi`-electrons which make the ring to ACT as an electro RICH centre. So electrophilic substiution reaction occurs in benzene. `(ii)` Benzene ring is stabilized by delocalized `pi` electrons. Though it is highly stable, it undergoes ADDITION and oxidation reaction under specic conditions. |
|
| 40. |
Which of the following is not a correct statement about enthalpy of solution? |
|
Answer» For most ionic compounds, `DeltaH_("soln.")^(@)` is positive and the dissociation PROCESS is endothermic. |
|
| 42. |
Which of the following resonance structures contributethe most to the resonance hybrid ? |
|
Answer»
|
|
| 43. |
Which one is correct statement of the following? |
|
Answer» `H_(2)^(+)` and `H_(2)^(-)` both are equal stable |
|
| 44. |
The weight of 1 molecule of the compoundC_(60)H_(122)is...(N_(A)=6.0xx10^(23)) |
|
Answer» `1.4xx10^(-21)g` `=720+122=842` `:.6xx10^(23)` molecule `C_(60)H_(122)` hasmass `= 842g` `:.1` molecule of `C_(60)H_(122)` has mass `= (842)/(6xx10^(23))` `=140.333xx10^(-23)g=1.4xx10^(-21)g` |
|
| 45. |
Which of the following alkyl halides will undergo S_(N)1 reaction most readily? |
|
Answer» `(CH_(3))_(3)C-F` |
|
| 47. |
Which is not the use of orthoboric acid? |
|
Answer» As an ANTISEPTIC and EYE wash. |
|
| 48. |
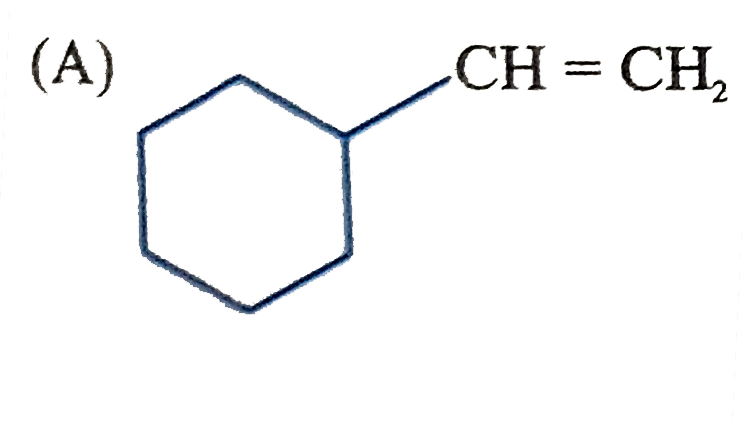
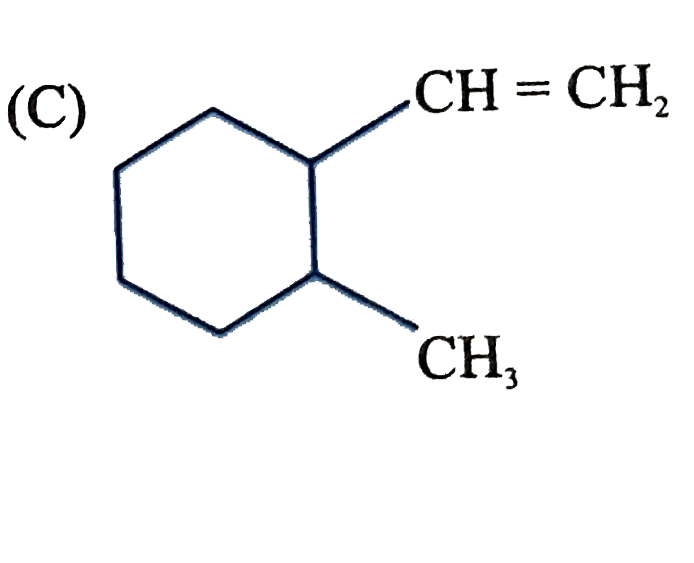
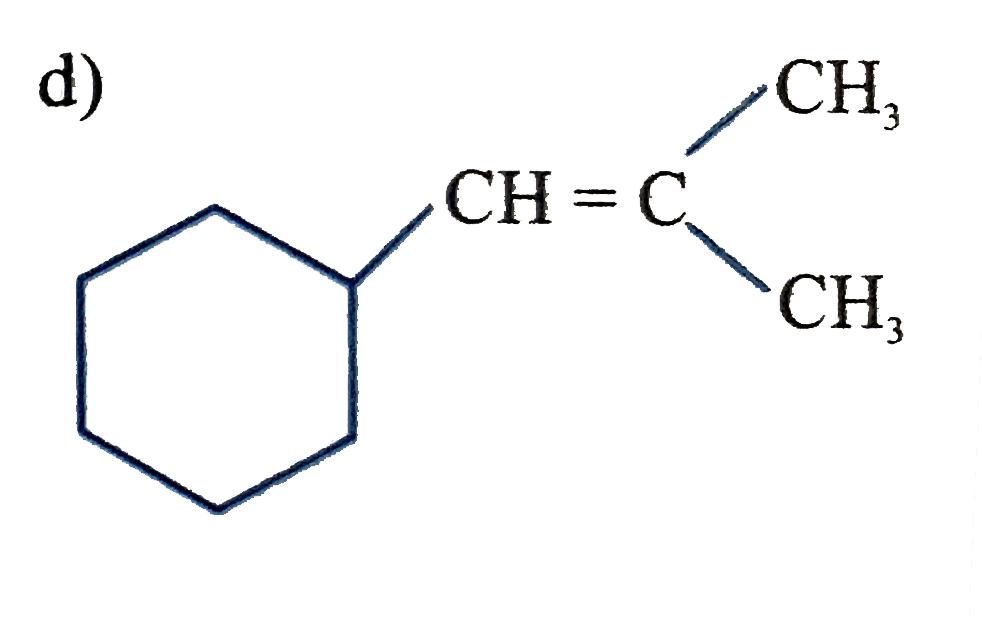
Which of the following compounds after complete hydrogenation will form three monochloro structural isomeric products ? |
|
Answer»
 Both STRUCTURE give three monochloro STRUCTURAL isomeric products. |
|
| 49. |
Which of the following will react fastest with Lucas reagent? |
|
Answer» Butan-1-ol |
|