Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following angle corresponds to sp^(2) hybridization ? |
|
Answer» `90^(@)` In `sp^(2)` hybridization, the geometry is triangular planar. So, bond angle is `120^(@)`.  Direction : The electronic CONFIGURATIONS of THREE elements, A, 8 and C are given below. Answer the questions from 14 to 17 on the basis of these configurations. A`1s^(2) 2s^(2) 2p^(6)` B ` 1s^(2)2s^(2) 2p^(6) 3s^(2) 3p^(3)` `C1 s^(2)2s^(2) 2 p^(6) 3s^(2) 3p^(5)` |
|
| 2. |
What is the difference between them? |
| Answer» SOLUTION :LOBES of `d_(x^2 - y^2)` lie along the x and y - AXES where those of `d_(xy)` lie between x and y axes. | |
| 3. |
Which of the following correctly represent the behaviour of an ideal gas |
|
Answer» `PV_m prop T` `P = CRT implies P prop CT, PM = DRT implies PM prop dT` . |
|
| 4. |
Tyrosine is a |
|
Answer» phenolic amino acid 
|
|
| 6. |
Which of the following has the highest dipolemoment ? |
|
Answer» `AsH_3` |
|
| 7. |
Which of the following can explain law of reciprocal proportions? |
|
Answer» `H_(2)O,CH_(4),CO_(2)` IMPLIES 3 ELEMENTS mutually combining. |
|
| 8. |
The table shown lists the bond dissociation energies (E_("diss")) for single covalent bonds formed between carbon and atoms of elements A,B,C, and D. Which element is the smallestatom? Bond between C and ""E_("diss") other atom""KJ"mol"^(-1) A. C-A""240 B. C-B""328 D. C-D""276 D. C-D""485 |
|
Answer» C |
|
| 9. |
Which is more acidic ? |
|
Answer» o-cresol 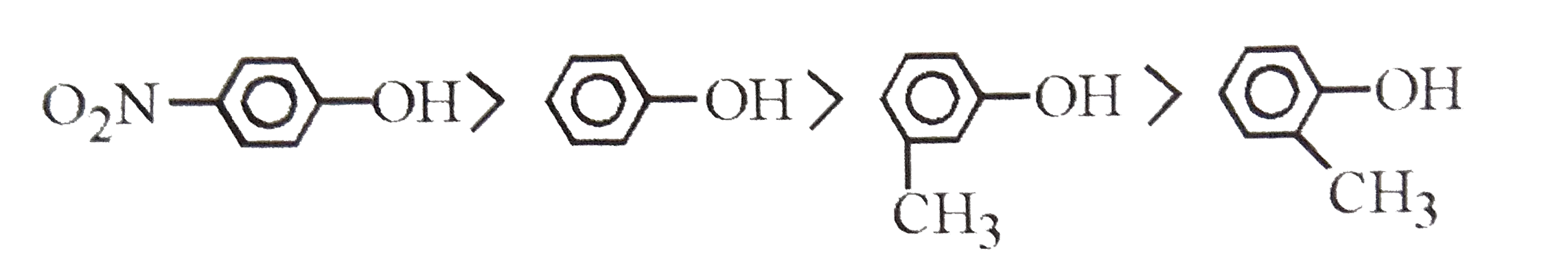
|
|
| 10. |
Which of the following has longest mean free path under identical conditions of temperature and pressure ? |
|
Answer» `H_(2)` where `sigma`=MOLECULAR diameter Under identical conditions of T and P,n is same, As `sigma` is LOWER for `H_(2)`, l will be LARGEST. |
|
| 11. |
The variation of lnKc vs. (l)/(T) gives straight line having an angle of 45^(@) . The value of heat of reaction inCal is _____ |
|
Answer» In `Kc=((-DeltaH)/(R))(1)/(T)` + In A for EXOTHERMIC `(DeltaH)/(R)=tan 45^(@)` `DeltaH = R=2cal` |
|
| 12. |
Which of the following is natural and human distrubance in ecology ? |
|
Answer» FOREST fire |
|
| 13. |
Which bicarbonate does not exist in solid state? |
|
Answer» `Ca(HCO_(3))_(2)` |
|
| 14. |
Write in brief for the preparation of alkene from vicinal dihalide. |
|
Answer» SOLUTION :Vicinal DIHALIDE MEANS carbon having dihalides in which two HALOGEN atoms are attached to two adjacent carbon atoms are known as vicinal dihalides. Dehalogenation : Vicinal dihalides on treatement with zinc METAL lose a molecule of `ZnX_(2)` to form an alkene. This reaction is known as dehalogenation. e.g., : `CH_(2)BrCHBrCH_(3)` Example : 
|
|
| 15. |
When temperature of any gas at constant pressure increase by 1^(@)C then what effect occur on volume ? |
|
Answer» Solution :Let INITIAL VOLUME `= V_(0)` After INCREASING on `1^(@)C`, Final volume = `V_(t)` `therefore V_(t)=V_(0)((273.15+t)/(273.15))` `= V_(0)(273.15+1)/(273.15)=V_(0)((274.15)/(273.15))` |
|
| 16. |
Which one of the following is produced as a result of incomplete combustion of coal? |
| Answer» SOLUTION :`CO_(2)` | |
| 17. |
Two substance C and O_(2) are allowed to react completely to form CO and CO_(2) mixture , leaving none of the reactants . Its is known that when I mole of CO_(2) ,100 Kcal of energy is released and when 1 mole of carbon reacts with 0.5 mole of O_(2) to give of CO,25 Kcal is liberated . Using this information match column I and column II. |
|
Answer» <P> |
|
| 18. |
Which of the following tests can be used for differentiation among , 1^(@) , 2^(@)and 3^(@) alcohol? |
|
Answer» Lucas test |
|
| 19. |
The strongest acid amongst the following compound is |
|
Answer» `HCOOH` |
|
| 20. |
Which of the following methods does not remove hardness of water ? |
|
Answer» Clark.s method |
|
| 21. |
Which of the following is the least stable carbonium ion ? |
|
Answer» `overset(oplus)(C)F_(3)` |
|
| 22. |
Whal is an ionic bond ? With two suitable examples explain the difference between an ionic and a covaJent bond ? |
|
Answer» Solution :Ionic bond : The bond FORMED, as a result of theelectrostatic ATTRACTION between the positive and negative ions was TERMED as the electrovalent bond or ionic bond. e.g., the formation of NaCl from sodium and chlorine can be explained as similarly, the formation of `CaF_(2)` may be shown as Covalent bond : The bond formed between the two atoms by mutual sharing of electrons between them is called covalent bond. e.g., the formation of chlorine molecule can be explained as  Similarly, in the formation of HCL  Covalent bond : The bond formed between TEH two atoms by mutual sharing of electrons between them is called covalent bond e.g., the formation of chlorine molecule can be explained as 
|
|
| 23. |
Which quantity out of Delta_(r)G " and " Delta_(r) G^(Θ) will be zero at equilibrium ? |
|
Answer» Solution :Gibbs ENERGY for a REACTION in which all reactants and products are in standard STATE. `Delta_(r)G^(Θ)` is related to the equilibrium constant of the reaction as follows `Delta_(r)G = Delta_(r)G^(Θ) + RT LN K` At equilibrium, `0 = Delta_(r) G^(@) + RT ln K "" ( :. Delta_(r) G = 0)` or `Delta_(r) G^(Θ) = - RT ln K` `Delta_(r) G^(Θ) = 0` When `K = 1` For all other values of `K, Delta_(r)G^(Θ)` will be non-zero. |
|
| 24. |
Which of the following have same hybridisation but are not isostructural- |
|
Answer» `ClF_(3) and I_(3)^(-)` `BrF_(3)(sp^(3)d` T-shape), `NH_(3)(sp^(3)`, pyramidal) `CH_(4)(sp^(3),` tetrahedral), `NH_(4)^(+)(sp^(3)`, tetrahedral) `XeO_(3)(sp^(3)`, Pyramidal), `NH_(3)(sp^(3)`, pyramidal). |
|
| 25. |
The reduction half reaction represents the gain of electrons. |
|
Answer» |
|
| 26. |
Which of the following orbitals are not possible? 1p, 2s, 3f and 4d |
|
Answer» SOLUTION :(i) The first shell has only one sub-shell, i.e., 1s, which has only one ORBITAL, i.e., 1s orbital. Therefore, 1p orbital is not possible. (ii) The second sub-shell has two SUBSHELLS, i.e., 2s and 2p. Therefore, 2s orbitals are possible. (iii) The third subshell has three subshells, i.e., 3s, 3p and 3d. Therefore, 3f-orbitals are not possible (iv) The fourth shell has four subshells has four subshells, i.e., 4S, 4P 4d and 4f. Therefore, 4d-orbitals are possible |
|
| 27. |
Why are alkaline earth metal smaller than the Corresponding alkali metals. |
| Answer» Solution :This is due to the fact the group 2 elements have HIGHER nuclear CHARGE that ALLOWS electrons to be attracted more strongly towards the nucleus thereby decreasing the ionic radii. THUS alkaline earth metals are smaller than the corresponding ALKALI metals. | |
| 28. |
Which of the following has no hindered rotation about carbon-carbon bond? |
|
Answer» ETHANE |
|
| 30. |
What is the two-dimensional coordination number of a molecule in square close packed layer ? |
| Answer» SOLUTION :In the two-dimensional square close packed layer, the ATOM touches 4 NEAREST neighbouring ATOMS. HENCE, its coordination number=4 | |
| 31. |
Thermal decomposition of gaseous X_(2) to gaseous X at 298 K takes place according to following equation X_(2) (g) hArr 2X(g) The standard reaction Gibbs energy Delta_(r)G^(@) of this reaction is positive . At the start of the reaction there is one mole of X_(2) " andno. " X. As the reaction proceeds the number of moles of X formed is given by beta. Thus beta_("equilibrium") is the the number of moles of X formed at equilibrium . The reaction is carried out a constant total pressure of 2 bar. Consider the gases to behave ideally. ("Given" : R =0.083 L " bar " K^(-1) " mol"^(-1)) The incorrect statement among the following for this reaction is |
|
Answer» Decrease in the total pressure will result in formation of more moles of GASEOUS X (a) If the pressure on the system is decreased , the equilibrium will shift in the direction in which pressure increases i.e.,increases in no. of moles takes place i.e., in forward idrection . (b) At the start of the reaction `Q lt K` thus the reaction will proceed in the forward direction i.e., reaction is spontaneous. (C) `"if "b_(eq) =0.7 " then " K_(p) =(8XX(0.7)^(2))/(4-(0.7)^(2)) gt 1` `DeltaG^(@) =- RT" In " K_(p) SO, DeltaG^(@) =- " ve but ginen " DeltaG^(@) = + " ve so, " K_(p) " should be LESS than " 1" Hence " beta_(eq) ne 0.7` (d)`K_(p) =K_(c) (RT)^(Dn).` `K_(c) lt K_(p) ne (":." RT gt 1)` `" If" K_(p)lt1 " then " K_(c) lt 1` |
|
| 32. |
When electrons are comtributed by one atom but one atom shared by both the atoms so as to complete their octets, the bond formed is called………………. . |
| Answer» SOLUTION :COORDINATE BOND or DATIVE bond | |
| 33. |
Which of the following sets of physical phenomena are shown by dual natuer of radiation ? |
|
Answer» INTERFERENCE and PHOTOELECTRIC effect |
|
| 34. |
When A reacts with Pb(NO_(3))_(2) then compound X is formed. Compound X is oxidized by atmospheric oxygen on boiling, then Y is formed whar is the colour of Y? |
|
Answer» Yellow |
|
| 35. |
What weight of Al will be completely oxidised by 44.8 lit of oxygen at STP? |
|
Answer» 18g |
|
| 36. |
Which oxide is not attacted by water? |
|
Answer» BeO |
|
| 37. |
Which of the following gives Deuter Acetylone with D_(2)O |
|
Answer» `CaC_2` (C ) `BaC_2 + 2D_2O to C_2 D_2+ BA(OD)_2` |
|
| 38. |
Write the expression for the comparison of the relative strengths of two weak acids in terms of their ionization constants. |
| Answer» Solution :`("STRENGTH of Acid"_(1))/("Strength of Acid"_(2))=sqrt(K_(a_(1))/K_(a_(2)))` | |
| 39. |
What happen if the saturated solution of CaCl_2 is heated ? The process is exothermic. |
|
Answer» Solution :`CaCl_(2(s)) + H_2O hArr CaCl_(2(aq))`+ HEAT So if the solution is heated the reaction TAKE PLACE in REVERSE direction so solid `CaCl_2` increases and solubility of `CaCl_2` decreases. |
|
| 40. |
Whatis reversible process in thermodynamics ? |
| Answer» Solution :A REVERSIBLE process in thermodynamics MEANS a process WHICHIS carried out infinitesimally slowly so that CHANGES occuring in the directly processcan be exactly reversed without disturbingthe equilibrium. | |
| 41. |
Which gas is useful in buffer system to control the pH of blood ? |
|
Answer» `CO_2` |
|
| 42. |
Which of the following is the most correct electron displacement for a nucleophilic reaction to take place? |
|
Answer»
|
|
| 43. |
Write the principles involved in the estimation of (i) Halogens (ii) Sulphur present in an organic compound. |
|
Answer» Solution :(i) Halongens: HALOGENS reacts with silver nitrate to for precipitate of silver halides. `X+Ag^(+)toAgX` X REPRESENTS a halogen -Cl-Br or -I (ii)) Sulphur `S^(+)[Fe(CN)_(5)NO]^(2)to[Fe(CN)_(5)NOS]^(4-)` Sulphur reacts with notropresside to from a violet COLOUR complex. |
|
| 44. |
Which type of element are likely to from anions ? Explain. |
|
Answer» |
|
| 45. |
Whatis theCORRECTnamefor CH_(3) CH_(2) CHO ? |
|
Answer» Ethana[ |
|
| 46. |
When total attractive forces between two atoms approaching each other is greater than total repulsive force then, potential energy of the system ________. |
|
Answer» increases |
|
| 47. |
Which of the following order is expected to be correct ? |
|
Answer» `pK_(a)(ClCH_(2)COOH) gt pK_(a)(CH_(3)COOH) lt pK_(a) (CH_(3)CH_(2)COOH)` |
|
| 48. |
What is polymerisation? Explain the two types of polymerisation reaction of acetylene. |
|
Answer» Solution :A polymer is a large molecule formed by the COMBINATION of large number of small MOLECULES (monomers). This process is known as polymerisation. Acetylene UNDERGOES two TYPES of polymerisation reactions, they are: (Linear polymerisation (1) Cyclic polymerisation (1) Linear polymerisation: Acetylene forms linear polymer, when passed into a solution of cuprous chloride and ammonium chloride. `underset("(Acetylene)")(2CH -=CH)overset(Cu_2Cl_2)underset(NH_4Cl)rarrCH_2=underset("(Vinyl acetylene)")underset(C-=CH)underset(|)"CH"` (ii) Cyclic polymerisation: Acetylene undergoes cyclic polymerisation on passing through red hot iron TUBE. Three molecules of acetylene polymerises to form benzene. 
|
|
| 49. |
What is the equilibrium constant K_(eq) for the following reaction at 400 K. 2NOCl_((g)) hArr 2NO_((g)) +Cl_(2(g)) , given that DeltaH^0=77.2 "kJ mol"^(-1) and DeltaS^0=122 "JK"^(-1) "mol"^(-1) . |
|
Answer» Solution :Given :T=400 K , `DeltaH^0=77.2 "kJ mol"^(-1) =77200 "J mol"^(-1) ,DeltaS^0 =122 "JK"^(-1) "mol"^(-1)` `DeltaG^0=-2.303 RT LOG K_(EQ)` `log K_(eq)=(DeltaG^0)/(2.303 RT)` `log K_(eq)=-((DeltaH^0-TDeltaS^0))/(2.303RT)` `log K_(eq)=-((77200-400xx122)/(2.303xx8.314xx400))` `log K_(eq)=-(28400/7659)` `log K_(eq)=-3.7080` `K_(eq)`=ANTILOG (-3.7080) `K_(eq)=1.95xx10^(-4)` |
|
| 50. |
Which of the following is not obtained by fractional distillation of coal tar |
| Answer» Solution :Vegetable oil is not obtained. | |



