Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Norbornene can be converted into exo-norborneol by |
|
Answer» hydroboration-oxidation reaction 
|
|
| 2. |
Non - zero dipole moment is shown by |
|
Answer» `CO_(2)` 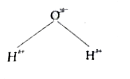
|
|
| 3. |
Non-stoichiometric cuprous oxidem,Cu_(2)O can bepreparedin thelaboratory. In this oxide, copper to oxygen ratio is slightly less than 2 :1 . Can you account for the fact that this substances is a a p -type semiconductor ? |
| Answer» Solution :The RATIO less than 2:1 in ` Cu_(2)O` shows that somecuprous `(Cu^(+))` ions have been replaced by cupric ` (Cu^(2+))` ions. To maintion electrical neutrality , EVERY two ` Cu^(+)` ionswill be DUE to presence of these POSITIVE HOLES, hence it is a p-type semiconductor. | |
| 4. |
Non-stoichiometric hydrides are formed by…........ |
|
Answer» PALLADIUM,vanadium |
|
| 5. |
Non-stoichiometric hydrides are formed by |
|
Answer» PALLADIUM, vanadium |
|
| 6. |
Non-stoichiometric cuprous oxide, Cu_2O can be prepared in the laboratory. In this oxide, copper to oxygen ratio is slightly less than 2:1. Can you account for the fact that this substance is a p-type semiconductor. |
| Answer» Solution :The ratio less than 2:1 in `Cu_2O` shows that some cuprous `(Cu^+)` ions have been replaced by cupric `(Cu^(2+))` ions To maintain electrical neutrality, every TWO `Cu^+` ions will be replaced by one `Cu^(2+)` ION thereby creating a hole As conduction will be due to PRESENCE of these positive holes, hence it isa p-type semiconductor | |
| 7. |
Non-stoichiometric Cu_(2)O in which coper to oxygen ratio is slightly less than 2:1 is |
|
Answer» an insulator |
|
| 8. |
Non polar compounds can also solidify because of |
|
Answer» Van DER Waals FORCES |
|
| 9. |
Non-Lanthanoid atom is |
|
Answer» La |
|
| 10. |
Non-ideal gases approach ideal behaviour at: |
|
Answer» HIGH TEMPERATURE and high pressure |
|
| 11. |
Nol. Of following reagents in which ppt A is soluble is AgNO_(3)+NaCl to A darr (i) aq. NH_(3) (ii) aq. KCN (iii) Na_(2)S_(2)O_(3) (iv) Conc. HCl (v) aq. NaoH (iv) aq. Kl |
|
Answer» `AgCl+KCNtoK^(+)[Ag(CN)_(2)]^(-)` `AgCl+Na_(2)S_(2)O_(3)("excess")toNa_(3)[Ag(S_(2)O_(3))_(2)]` `AgCl+conc.Hclto[AgCl_(2)]^(-)` `AgCl+aq.NaOHtoAg_(2)Odarr` |
|
| 12. |
Non-benzenoid aromatic compound(s) is/are |
|
Answer» Tropolone |
|
| 13. |
Nominationof electronis 5200 A.At whichvelocityof electronitswavelengthis equaltophoton. |
| Answer» SOLUTION :`1.4 xx 10^(3) ms^(1)` | |
| 14. |
Noble's oil is |
|
Answer» Fire extinguisher or Nitroglycerine is called as Noble OIL. |
|
| 15. |
Noble prize in green chemistry is not associated with |
|
Answer» RICHARD `R` schrock |
|
| 16. |
Noble gases have maximum ionisation energy. Justify. |
|
Answer» Solution :Noble gases have completely FILLED electronic COFIGURATION and are STABLE. (ii)It is difficult to remove the ELECTRONS from the valence shell. So the ionisation energy is MAXIMUM. |
|
| 17. |
Noble gases have almost zero electron affinity give reason. |
|
Answer» Solution :(i) Noble gases have completely filled ELECTRONIC CONFIGURATIONS. (ii)It has no tendency to attract the electrons TOWARDS itself. (iii)so the electron affinity of noble gases are almost ZERO. |
|
| 18. |
NO_2 gas is not obtained by heating which compound ? |
|
Answer» `AgNO_3` |
|
| 19. |
NO_2 is involved in the formation of smog and acid rain.A reaction that is important in the formation of NO_3 is O_3 (g) + NO(g) iff O_2 (g) + NO_2(g) K_C = 6.0 xx 10^(34) . If the airover a section of New Delhi contained1.0 xx 10^(6) M " of " O_3, 1.0 xx 10^(-5) Mof NO, 2.5 xx 10^(-4)M of NO_2 and 8.2 xx 10^(-3) Mof O_2 , what canwe conclude ? |
|
Answer» there will betendency to FORM more `NO` and `O_ 2` `=8.2 xx 2.5 xx 10^(4) = 20.5 xx 10^(4)` As ` Q lt K_C`, the reaction will havea tendency to move forward . |
|
| 20. |
NO_2^(-) , F^(-) , H^(-) , CH_3COO^(-) like weak acid's strength of their conjugate base is more than water or less than water ? |
| Answer» Solution :All these are STRONGER BASE than water as these all are CONJUGATE base of WEAK acid. | |
| 21. |
NO_(2) and SO_(2) have some properties in common. Which property shown by one of these compounds, but not by the other? |
|
Answer» Forms acid |
|
| 22. |
NO_2 and O_3 react with unburnt hydrocarbons present in polluted air and produce |
|
Answer» FORMALDEHYDE |
|
| 23. |
No two electrons in an orbital can have parallel spin. This statement emerges from |
|
Answer» Hund.s rule |
|
| 24. |
No reaction occurs in which of the following equations? |
|
Answer» `I^(ө)-FE^(2+)rarr` So `F_(2)` can diplace `CI^(ө), Br^(ө)`, and `I^(ө)` but not VICE VERSA and `Br_(2)` can displace only `I^(ө)` but not vice versa. In (a), `Fe( "not" Fe^(2+))` is a better reducing AGENT than `I^(ө)`. |
|
| 25. |
No. of unit in SI system = ........... |
|
Answer» 5 |
|
| 26. |
No. of sigma - and pi - bonds in acrylonitrile |
|
Answer» `3PI` and `6sigma` |
|
| 27. |
No of revolutions made by the electron in one sec in 2^(nd) orbit of Be^(3+) |
|
Answer» `1.31 XX 10^(16)` |
|
| 28. |
No. of possible isomers C_(4)H_(10)O formula does have…… |
|
Answer» 7 `CH_(3)-CH_(2)-CH_(2)-CH_(2)-OH` `CH_(3)-underset(underset(OH)(|))(CH)-CH_(2)-CH_(3)` `CH_(3)-underset(underset(CH_(3))(|))(CH)-CH_(2)-OH` `CH_(3)-underset(underset(OH)(|))overset(overset(CH_(3))(|))(C )-CH_(3)` `CH_(3)-O-CH_(2)-CH_(2)-CH_(3)` `CH_(3)-CH_(2)-O-CH_(2)-CH_(3)` `CH_(3)-O-underset(underset(CH_(3))(|))(CH)-CH_(3)` |
|
| 29. |
No. of possible isomeric dioic acids for the molecular formulae C_(4)H_(4)O_(4) are |
|
Answer» |
|
| 30. |
No. of moles of H_(2) gas evolved when one mole of the following compound reacts with sodium. |
|
Answer» |
|
| 31. |
No. of moles in 100 mg of heptane is …………than those in 10 mg of propyne |
|
Answer» 4 times greater `= ((100xx10^(-3)g))/(("100g mol"^(-1)))=1xx10^(-3)` No. of moles in 10 mg of PROPYNE `(C_(3)H_(4))` `= ((10xx10^(-3)g))/(("40 g mol"^(-1)))=(1)/(4)xx10^(-3)` No. of moles of heptane are FOUR times the no. of moles of propyne. |
|
| 32. |
No. of moles of acetylene required for cyclic polymerization in a red hot copper tube to form2 moles of benzene is ________ |
|
Answer» |
|
| 33. |
No. of H_(2)O molecules in a drop of water weighing 0.05 g is : |
|
Answer» `1.15 xx 10^(23)` `=6.022 xx 10^(23)` 0.05 g of `H_(2)O` contain molecules `= ((0.05g))/((18.0g))xx6.022xx10^(23)` `=1.673xx10^(21)` molecules. |
|
| 34. |
No.of geometrical isomers possible for the compound CH_(3)-CH=CH-CH=CH-C_(2)H_(4) |
|
Answer» 2 |
|
| 35. |
No. of electrons in 1.8 mL of H_2O (l) is: |
|
Answer» `6.02 XX 10^(23)` |
|
| 36. |
No form of elemental silicon is comparable to graphite. |
|
Answer» Solution :In graphite, carbon is `sp^(2)`-hybridizedand eachcarbon is liked to three other carbon atoms by forming hexagonalrings.Eachcarbonis now LEFT with on unhybridized p-orbital which undergoes sidewaysoverlap to form three `ppi-ppi`double bonds.Thus,graphite has two dimensional sheet like (layered) structureconsistingof a number of benzenerings fusedtogether. Siliconon the other hand,does notforman anlogue ofcarbon becauseof the FOLLOWING REASON : DUE to bigger size kand smallerelectronegativityof Si than C, it does not undergo `sp^(2)`-hybridization and hence it does not form`ppi-ppi`double bondsneededfor graphite like structure. Instead, it PREFERS to undergoonly `sp^(3)`-hybridization and hencesiliconhas diamondlike three-dimensional network structure. |
|
| 38. |
No crystal is found to be prefect at room temperature . The defects present in the crystals can be stoichiometric or non-stoichiometric. Due to non-stoichiometric defects, the formula of the ionic compound is different from the ideal formula. For example , the ideal formula of ferrous oxide should be FeO but actually in one sample , it was found to be Fe_0.93 O. This is because the crystal may have some ferric ions in place of ferrous ions. These defects change the properties of the crystals. In some cases , defects are introduced to havecrystals of desired properties as required in the field of electronics . Doping of elements of Group 14 with those of Group 13 or 15 is most common. In ionic compounds , usually impurities are introduced in which the cation has higher valency than the cation of the parent crystal , e.g. of SrCl_2 into NaCl NaCl was doped with 10^(-3) mol % SrCl_2. The concentration of cation vacancies is |
|
Answer» `6.02xx10^18 "MOL"^(-1)` |
|
| 39. |
No crystal is found to be prefect at room temperature . The defects present in the crystals can be stoichiometric or non-stoichiometric. Due to non-stoichiometric defects, the formula of the ionic compound is different from the ideal formula. For example , the ideal formula of ferrous oxide should be FeO but actually in one sample , it was found to be Fe_0.93 O. This is because the crystal may have some ferric ions in place of ferrous ions. These defects change the properties of the crystals. In some cases , defects are introduced to havecrystals of desired properties as required in the field of electronics . Doping of elements of Group 14 with those of Group 13 or 15 is most common. In ionic compounds , usually impurities are introduced in which the cation has higher valency than the cation of the parent crystal , e.g. of SrCl_2 into NaCl Which one of the following defects does not affect the density of the crystal ? |
|
Answer» SCHOTTKY DEFECTS |
|
| 40. |
No crystal is found to be prefect at room temperature . The defects present in the crystals can be stoichiometric or non-stoichiometric. Due to non-stoichiometric defects, the formula of the ionic compound is different from the ideal formula. For example , the ideal formula of ferrous oxide should be FeO but actually in one sample , it was found to be Fe_0.93 O. This is because the crystal may have some ferric ions in place of ferrous ions. These defects change the properties of the crystals. In some cases , defects are introduced to havecrystals of desired properties as required in the field of electronics . Doping of elements of Group 14 with those of Group 13 or 15 is most common. In ionic compounds , usually impurities are introduced in which the cation has higher valency than the cation of the parent crystal , e.g. of SrCl_2 into NaCl Which one of the following doping will produce p-type semiconductor ? |
|
Answer» Silicon doped with arsenic |
|
| 41. |
No crystal is found to be prefect at room temperature. The defects present in the crystals can be stoichimetric or non-stoichiometric. Due to non-stoichimetric defects, the formula of the ionic compound isdifferent from the ideal formula. For example , the ideal formula of ferrous oxide should be FeO but actually in one sample , it was found to beFe_(0.93)O.This is because the crystal may have some ferric ions in place of ferrous ions.These defects change the propeties of the crystals. In some cases, defects are introduced to have crystals of desired properties as required in the field of electronics. Dopingof elments of Group 14 with those of Group13 or 15 is most common. In ionic compounds, usually impurities are introduced in which the cation has higher valency than the cation of the parent crystal, e.g,SrCl_(2)into NaCl.NaCl was doped with10^(-3) " mol % " SrCl_(2) . The concentreation of cation vancancies is |
|
Answer» ` 6.02 xx 10^(18) " mol"^(-1)` |
|
| 42. |
No crystal is found to be prefect at room temperature. The defects present in the crystals can be stoichimetric or non-stoichiometric. Due to non-stoichimetric defects, the formula of the ionic compound isdifferent from the ideal formula. For example , the ideal formula of ferrous oxide should be FeO but actually in one sample , it was found to beFe_(0.93)O.This is because the crystal may have some ferric ions in place of ferrous ions.These defects change the propeties of the crystals. In some cases, defects are introduced to have crystals of desired properties as required in the field of electronics. Dopingof elments of Group 14 with those of Group13 or 15 is most common. In ionic compounds, usually impurities are introduced in which the cation has higher valency than the cation of the parent crystal, e.g,SrCl_(2)into NaCl.which one of the following defects does not affect the density of the crystal ? |
|
Answer» SCHOTTKY DEFECT |
|
| 43. |
No crystal is found to be prefect at room temperature. The defects present in the crystals can be stoichimetric or non-stoichiometric. Due to non-stoichiometric defects, the formula of the ionic compound isdifferent from the ideal formula. For example, the ideal formula of ferrous oxide should be FeO but actually in one sample, it was found to beFe_(0.93)O.This is because the crystal may have some ferric ions in place of ferrous ions.These defects change the properties of the crystals. In some cases, defects are introduced to have crystals of desired properties as required in the field of electronics. Dopingof elements of Group 14 with those of Group13 or 15 is most common. In ionic compounds, usually impurities are introduced in which the cation has higher valency than the cation of the parent crystal, e.g,SrCl_(2)into NaCl.which one of the following doping will produces p-type semiconductor? |
|
Answer» Silicon doped with arsenic |
|
| 44. |
No crystal is found to beperfect at room temperature. The defects present in the crystals can be stoichiometric or non-stoichiometric. Due to non-stoichiometricdefects, the formula of the ionic compound is different from the ideal formula. For example, the ideal formula of ferrous oxide should be FeO but actually in one sample, it was found to be Fe_(0.93)O. This is because the crystal may have some ferric ions in place of ferrous ions. These defects change the properties of the crystals. In some cases, defects are introduced ot have crystals of desired properties as required in the field of electronics. Doping of elements of group 14 with those of group 13 or 15 is most common. In ionic compounds, usually the impurities are introduced in which the cation has higher valency than the cation of the parent crystal e.g.,SrCl_(2) into NaCl. Which one of the following defects does not affect the density of the crystal ? |
|
Answer» Schottky defect |
|
| 45. |
No crystal is found to beperfect at room temperature. The defects present in the crystals can be stoichiometric or non-stoichiometric. Due to non-stoichiometricdefects, the formula of the ionic compound is different from the ideal formula. For example, the ideal formula of ferrous oxide should be FeO but actually in one sample, it was found to be Fe_(0.93)O. This is because the crystal may have some ferric ions in place of ferrous ions. These defects change the properties of the crystals. In some cases, defects are introduced ot have crystals of desired properties as required in the field of electronics. Doping of elements of group 14 with those of group 13 or 15 is most common. In ionic compounds, usually the impurities are introduced in which the cation has higher valency than the cation of the parent crystal e.g.,SrCl_(2) into NaCl. The percentage of iron as Fe_(III)inFe_(0.93)O_(1.0)is |
|
Answer» 0.177 `"TOTAL -ve charge on "O^(2-)"IONS" = 2times100 = 200` `"LET no. of "Fe^(2+)"ions" = x` `therefore"""no. of "Fe^(3+)"ions" = 93-x` `therefore" Total +ve charge on "Fe^(2+)andFe^(3+)"ions"` =`2x+3(93-x)` For electrical neutrality of crystal, `2x+3(93-x)=200` `2x+279-3x = 200` x=79 `thereforeFe^(2+)=x=79`, `Fe^(3+)=93-x=93-79=14` `therefore"formula of oxide is ,"` `Fe_(79)^(2+)" " Fe_(14)^(3+) O_(100)orFe_(0.79)^(2+) "" Fe_(0.14)^(2+) O` `"Molar mass" = 0.93times56+16 = 68.08` `%" of Fe as "Fe^(3+) = (0.14times56)/(68.08)times100 = 11.5%` |
|
| 46. |
No crystal is found to beperfect at room temperature. The defects present in the crystals can be stoichiometric or non-stoichiometric. Due to non-stoichiometricdefects, the formula of the ionic compound is different from the ideal formula. For example, the ideal formula of ferrous oxide should be FeO but actually in one sample, it was found to be Fe_(0.93)O. This is because the crystal may have some ferric ions in place of ferrous ions. These defects change the properties of the crystals. In some cases, defects are introduced ot have crystals of desired properties as required in the field of electronics. Doping of elements of group 14 with those of group 13 or 15 is most common. In ionic compounds, usually the impurities are introduced in which the cation has higher valency than the cation of the parent crystal e.g.,SrCl_(2) into NaCl. NaCl was doped with 10^(-3) mol % SrCl_(2).The concentration of the cation vacancies is |
|
Answer» `6.02times10^(18)"mol"^(-1)` `therefore "1 mole of NaCl is doped with "SrCl_(2)` =`(10^(-3))/100"mole"= 10^(-5)"mole"` Since `Sr^(2+)`ion introduces one cation vacancy, therefore, concentration of cation vacancies `= 10^(-5)"mol//mol of NaCl"` =`10^(-5)times6.02times10^(23)"mol"^(-1)` =`6.02times10^(18)"mol"^(-1)`. |
|
| 47. |
No crystal is found to beperfect at room temperature. The defects present in the crystals can be stoichiometric or non-stoichiometric. Due to non-stoichiometricdefects, the formula of the ionic compound is different from the ideal formula. For example, the ideal formula of ferrous oxide should be FeO but actually in one sample, it was found to be Fe_(0.93)O. This is because the crystal may have some ferric ions in place of ferrous ions. These defects change the properties of the crystals. In some cases, defects are introduced ot have crystals of desired properties as required in the field of electronics. Doping of elements of group 14 with those of group 13 or 15 is most common. In ionic compounds, usually the impurities are introduced in which the cation has higher valency than the cation of the parent crystal e.g.,SrCl_(2) into NaCl. Which of the following dopings will produce p type semiconductor ? |
|
Answer» Silicon doped with ARSENIC |
|
| 48. |
NKg^(-1) is the unit of |
|
Answer» Momentum |
|
| 49. |
Nitromethane exists in the aci-form but nitrobenzene does not. Explain why ? |
Answer» Solution :Nitromethane has `alpha`-HYDROGEN and hence it exists in the aci-form. 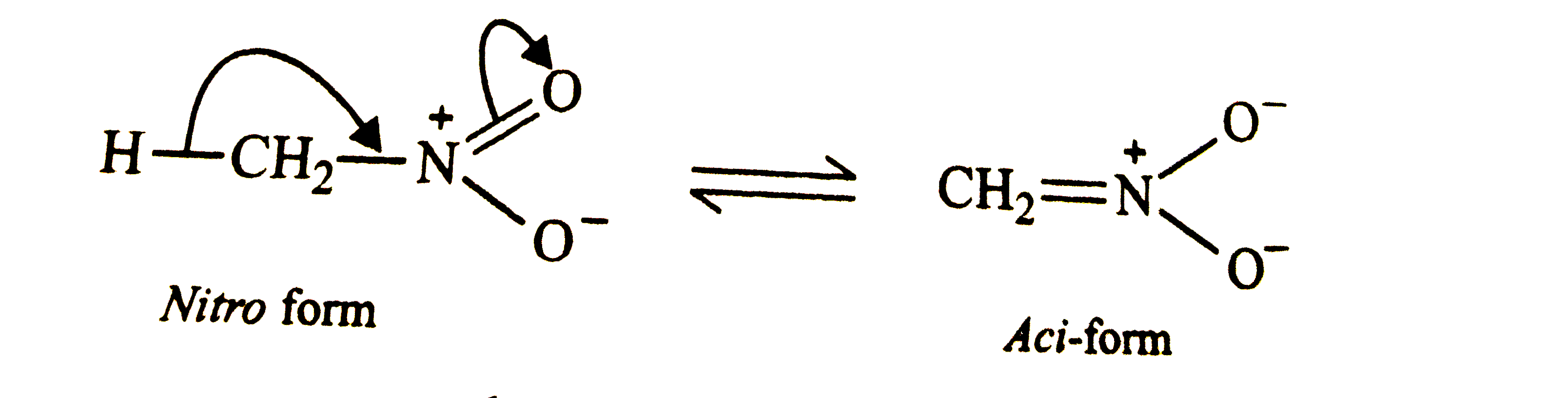 In contrast, NITROBENZENE does not have an `alpha`-hydrogen and hence it does EXIST in the aci-form. 
|
|