Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The ionisation energy of hydrogen is |
|
Answer» `1312 KJ "MOLE"^(-1)` |
|
| 2. |
The ionisation energy of He^(+) " is " 8.72 xx 10^(-18)J " atom"^(-1). Calculate the energy of the first stationary state of Li^(2+) |
|
Answer» Solution :`E_(n) = - (2pi^(2) mZ^(2) e^(4))/(n^(2) h^(2)) = - K (Z^(2))/(n^(2))` (K = constant) I.E. of `He^(+) = E_(oo) - E_(1) = 0 - (-K (2^(2))/(1^(2)))= 4K` Hence, `4K = 8.72 xx 10^(-18)J "atom"^(-1)` (GIVEN) or `K = 2.18 xx 10^(-18) J " atom"^(-1)` For `Li^(2+), Z =3` and for 1st stationary STATE, n = 1 `:. E_(1) = -K (Z^(2))/(n^(2)) = - 2.18 xx 10^(-18) xx (3^(2))/(1^(2)) = - 19.62 xx 10^(-18) J "atom"^(-1)` |
|
| 3. |
The ionisation energy of He^(+) is 19.6xx10^(-18)"J atom"^(-1) Calculate the energy of first stationary state of Li^(2+) ion. |
|
Answer» Solution :Ionisation energy of hydrogen LIKE species = Ionisation energy of `H XXZ^(2)` `"I.E of "He^(+)="I.E of H"xx4,"I.E of "H=(1.9xx10^(-18))/(4)"______(1)"` `"I.E of "LI^(2+)="I.E of "Hxx9," I.E of H"=("I.E of "Li^(2+))/(9)"____(2)"` `"Equating (1) and (2) "("I.E of "Li^(2+))/(9)=(19.6xx10^(-18))/(4)` `"I.E of "Li^(2+)=(19.6xx10^(-18)xx9)/(4)` The energy of first stationary state of `Li^(2+)` `=(-19.6xx10^(-18)xx9)/(4)=-4.41xx10^(-17)"J atom"^(-1)` |
|
| 4. |
The ionisation energies of Lithium and Sodium are 520kJ Mol^(-1) and 495kJ Mol^(-1) respectively. The energy required to convert all the atoms present in 7mg of Li vapour and 23 mg of sodium vapour to their respective gaseous cations respectively are |
|
Answer» 52 J , 49.5 J |
|
| 5. |
The ionisation energy and electron affinity of an element are 13.0ev and 3.8ev respectively. Its electronegativity is |
|
Answer» 2.8 |
|
| 6. |
The ionisation energies from Ga to TI do not decrease due to |
|
Answer» SHIELDING EFFECT |
|
| 7. |
The ionisation constant of NH_3 is 1.77 xx 10^(-5). What is the 'a' and 'pH' of 0.05M ammonia solution? |
| Answer» SOLUTION :`alpha=0.018, pH=10.97` | |
| 8. |
The ionisation constant of chloroacetic acid is 1.35xx10^(-3) .What will be the pH of 0.1 M acid and its 0.1 M sodium salt solution ? |
|
Answer» Solution :PH of 0.1 M chloroacetic ACID `(ClCH_2COOH)` : This is a weak acid and suppose x M of 0.1 M is ionised and equilibrium is established. So At equilibrium `x=[H_3O^+] = [ClCH_2COO^-]` `{:(,ClCH_2COOH_((aq)) +H_2O_((l)) hArr , ClCH_2COO_((aq))^(-) + , H_3O_((aq))^(+)),("At equilibrium" , 0.1 M, 0.0 , 0.0),(,(0.1 -x)M,x M, xM),(,approx 0.1, ,):}` `K_a=([ClCH_2COO^(-)][H_3O^+])/([ClCH_2COOH])` `therefore 1.35 xx 10^(-3) =((x)(x))/0.1` `therefore x^2=1.35xx10^(-4)` `therefore x=1.162xx10^(-2)M=[H_3O^+]` pH=log `[H^+]` `=-log (1.162xx10^(-2))` =-(0.0652-2) =1.9348 `approx` 1.93 `therefore` pH of this weak base is 1.93 Acid `ClCH_2COOH` and SALT `ClCH_2COONa` present in solution so solution is buffer. The pH of this buffer is calculate by following equation. `pH=pK_a + "log"([A^-])/([HA])` `[A^-]=[ClH_2COO^-]=0.1` [HA]=`[ClCH_2COOH]`=0.1 where `pK_a=-log (K_a)` =-log `(1.35xx10^(-3))` =2.8690 pH=2.869 +log `(0.1/0.1)`=2.869 |
|
| 9. |
The ionisation constant of an acid, K_(a) is the measure of strength of an acid. The K_(a) values of acetic acid, hypochlorous acid and formic acid are 1.74xx10^(-5), 3.0xx10^(-8) and 1.8xx10^(-4) respectively. Which of the following orders of pH of 0.1 mol dm^(-3) solutions of these acids is correct ? |
|
Answer» acetic acid `gt` hypochlorous acid `gt ` formic acid `{:(,HCO OH ,gt,CH_(3)CO OH,gt,HClO),(K_(a),1.8xx10^(_4),,1.74xx10^(-5),,3.0xx10^(-8).):}` |
|
| 10. |
The ionisation constant of an acid, K_a is the measure of strength of an acid. The K_a values of acetic acid, hypochlorous acid and formic acid are 1.74 xx 10^(-5), 3.0 xx 10^(-8) and 1.8 xx 10^(-4) respectively. Which of the following orders of pH of 0.1 mol dm^(-3) solutions of these acids is correct ? |
|
Answer» ACETIC acid `GT` hypochlorous acid `gt` formic acid `{:("Hypochlorous acid",lt,"Acetic acid",lt , "Formic acid"),((3.8xx10^(-8)),,(1.74xx10^(-5)),,(18xx10^(-4))):}` |
|
| 11. |
The ionisation constant of an acid base indicator is 3' 10^(-5). The acid from of the indictor is red and the basic form is blue. By how much must the p^(H) change in order to change the indicator from 75% red to 75% blue? |
|
Answer» `1.95` `K_(In)=K_(n)=([H^(+)][In^(-)])/(HIn),[H^(+)] = Ka(["acid"])/(["BASIC"])` for `75%` red, `[H^(+)] = ((3xx10^(-5))(75))/(25) = 9 xx 10^(-5), p^(H) = 4.05` The change in `p^(H) = 5 - 4.05 = 0.95` |
|
| 12. |
The ionic strength of solution containing 0.5 M MgSO_(4)0.1M AlCl_(3) and 0.2(NH_(4))_(2)SO_(4) is |
|
Answer» `0.75` `AlCl_(3)=0.1xx4=0.4` `(NH_(4))_(2)SO_(4)=0.2xx3=(0.6)/(2)` |
|
| 13. |
The ionic radius of H^+ ions and that of other ions is .......... pm and .......... pm respectively. |
|
Answer» `50-200,1.5xx10^(-3)` |
|
| 14. |
The ionic radius of H^+ ions and that of other ions is ........ pm and ........ pm respectively. |
| Answer» SOLUTION :`1.5xx10^(-3)` PM , 50-200 pm | |
| 15. |
The ionicradiusofCris minimum inwhichthe followingcompounds ? |
|
Answer» `K_(2) CrO_(4)` |
|
| 16. |
The ionic radius(in Å ) of N^(3-), O^(2-) and F^(-)are respectively . |
|
Answer» 1.71, 1.40 AND1.36 and `F^(-) (9+ 1=10) ` areisoelectronicions . Amongst isoelectronicions, greaterthe negativechargeon theanion , largeris the ionicradius , Thusionicradiidecreae intheorder`N^(3-) (1.71 Å ) lt` `O^(2-)(1.40 Å) GT F^(-)(1.36 Å) . i.e., `option(a) ISCORRECT . |
|
| 17. |
The ionic radii of N^(3-) , O^(2-) , F^(-) " and " Na^(+) follow the order. |
|
Answer» `N^(3-) GT O^(2-) gt F^(-) gt Na^(+)` |
|
| 18. |
The ionic radii of A^(+) and B^(-) ions are0.98 xx 10^(-10) m and 1.81 xx 10^(-10) m.The coordination number of each ion in AB is |
|
Answer» 2 It liess in the range 0.414 - 0.732. Hence, coordination NUMBER of each ION will be 6, as in the case of NaCl. |
|
| 19. |
The ionic radii of A^+ and B^- ions are 0.92xx10^(-10) m and 1.81xx10^(-10) m . The coordination number of each ion in AB is |
|
Answer» 2 It lies in the RANGE 0.414-0.732 . HENCE, coordination NUMBER of each ION will be 6, as in the CASE of NaCl |
|
| 20. |
The ionic product of water is 10^(-14), The H^(+) ion concentration in 0.1 M NaOH soluiion is |
|
Answer» `10^(-11) M` ` [OH^(-) ] =10 ^(-1)=[H^(+) ] =10 ^(-13)M` |
|
| 21. |
The ionic product of water at 60^(@)C is 9.55 xx 10^(-14) "mole"^(2) lit^(-2) . The dissociation constant of water at the same temperature is |
|
Answer» `1.09 XX 10^(-15)` |
|
| 22. |
The ionic product of water at 310 K is 2.7xx10^(-14). What is the pH of neutral water at this temperature ? |
|
Answer» SOLUTION :`[H^(+)]=SQRT(K_(w))=sqrt(2.7xx10^(-14))=1.643xx10^(-7)M` `pH= - LOG [H^(+)]=-log (1.643xx10^(-7))=7-0.2156=6.78` |
|
| 23. |
The ionic product of water at 310 K is 2.7 xx10^-14. What is the pH of neutral water at this temperature. |
|
Answer» SOLUTION :`K_w=[H^+][OH^-]=2.7xx10^-14` `THEREFORE[H^+]=SQRT(2.7xx10^-14)=1.643xx10^-7` `therefore pH=-log(1.643xx10^-7)=7-0.2156=6.7844=6.78` |
|
| 24. |
The ionic mobility of Li^(+) is less than that of the Na+ ion in solution because |
|
Answer» Li+ ION has a high MELTING point |
|
| 25. |
The ionic mobility of alkali metal ions in aqueous solution is maximum for |
|
Answer» `K^(+)` |
|
| 26. |
The ionic compounds of alkali metals colourless. Why? |
| Answer» Solution :ALKALI metals FORM unipositive ions which have STABLE CONFIGURATION of the nearest inert gas. Alkali metal salts are diamagnetic and colourless because they do not have unpaired ELECTRONS. | |
| 27. |
The ionic chloride is |
|
Answer» `PbCl_(2)` |
|
| 29. |
The ion that has pseudo-octet configuration |
|
Answer» `Mn^(2+)` |
|
| 30. |
The ion exchange resin which removes metal ions from hard water consists of giant organic molecule having |
|
Answer» `-CL` group |
|
| 31. |
The ion having highest mobility in aqueous solution is |
|
Answer» `Be^(+2)` |
|
| 32. |
The ion acting as an oxidising agent in the reaction, Cr_(2)O_(7)^(2-)+6Fe^(2+)+14H^(+)to2Cr^(3+)+6Fe^(3+)+7H_(2)O is |
| Answer» Answer :A | |
| 33. |
The iodide content of a solution was determined by titration with sodium thiosulphate crystalline containing 11.2 % impurity. Calculate the normality of iodide ion solution in 250 mL of the iodide solution required 20 mL hypo (42 g hypo is dissolved in 1 litre). |
|
Answer» |
|
| 34. |
The iodine molecule dissociates into atoms after absorbing light of 4500Å. If one quantum of radiat is absorbed by each molecule, calculate the kinetic energy of iodine atoms (Bond energy I_(2) = 240 kJ mol^(-1)) |
|
Answer» Solution :Bond energy PER molecule of `I_(2) = (240 xx 1000J)/(6.022 xx 10^(23)) = 3.985 xx 10^(-19)J` Energy absorbed `= (HC)/(lamda) = ((6.626 xx 10^(-34) Js) (3 xx 10^(8) ms^(-1)))/(4500 xx 10^(-10)m) = 4.417 xx 10^(-19)J` The difference of energy absorbed and energy used for dissociation of bond in `I_(2)` gives the KINETIC energy one `I_(2)` molecule. `:.` K.E. of one `I_(2)` molecule `= (4.417 xx 10^(-19) - 3.985 xx 10^(-19)) J = 4.32 xx 10^(-20)J` `:.` K.E. of one iodine atom `= (4.32 xx 10^(-20))/(2) J= 2.16 xx 10^(-20)J` |
|
| 36. |
The inversion temperature for a gas is given by ________. |
|
Answer» `(2A)/(RB)` |
|
| 37. |
The introduction of neutron in nucleus of an atom would lead to the change i n ........ |
|
Answer» ATOMIC weight |
|
| 38. |
The intramolecular hydrogen bonding in molecules lead to ________ |
|
Answer» HIGH MELTING point |
|
| 39. |
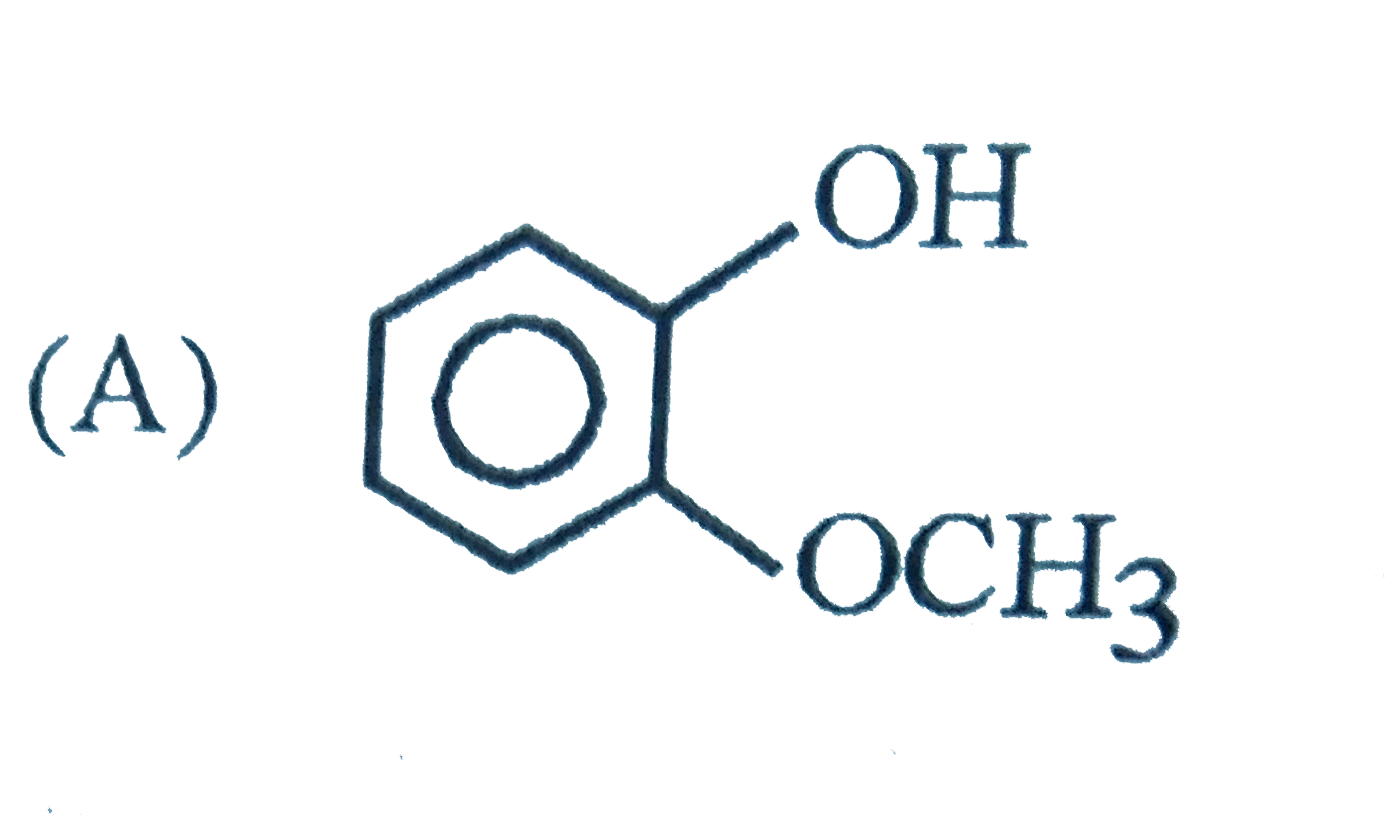
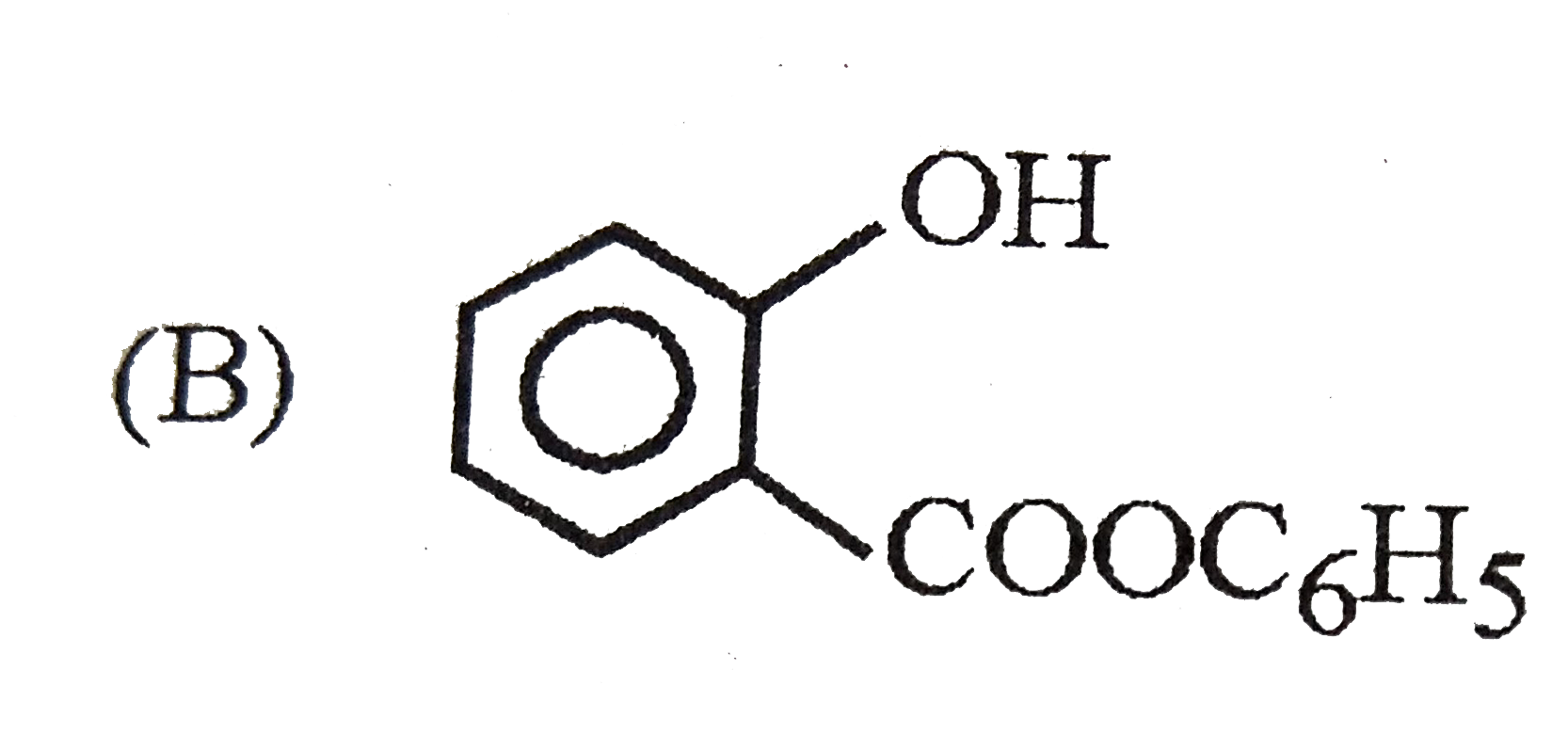
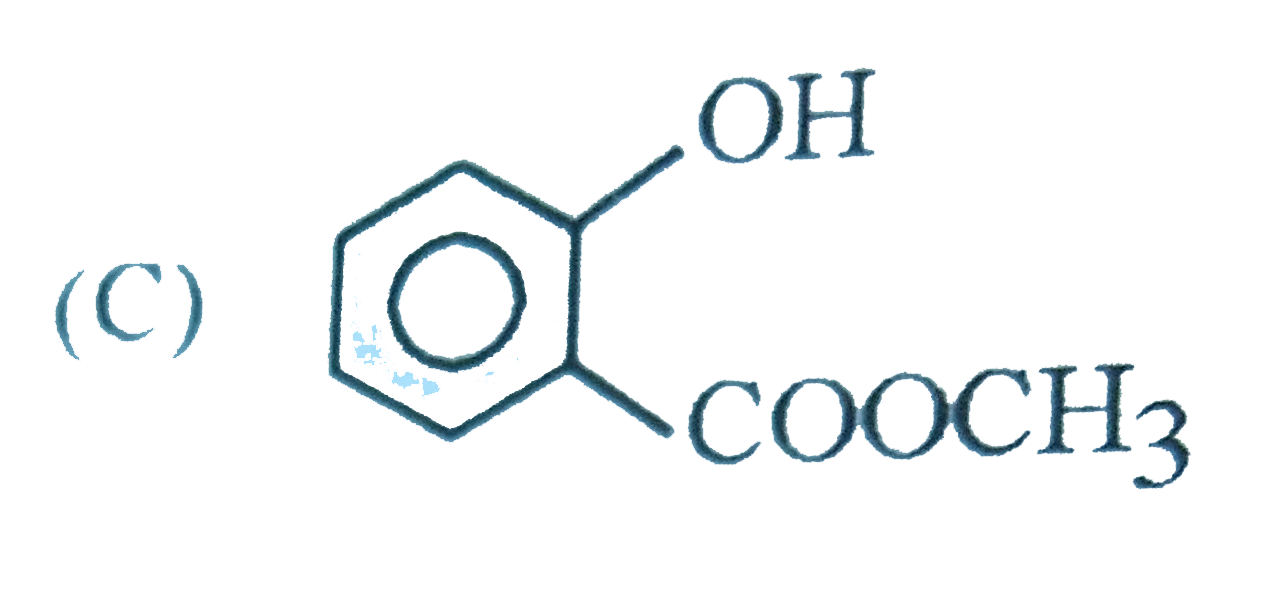
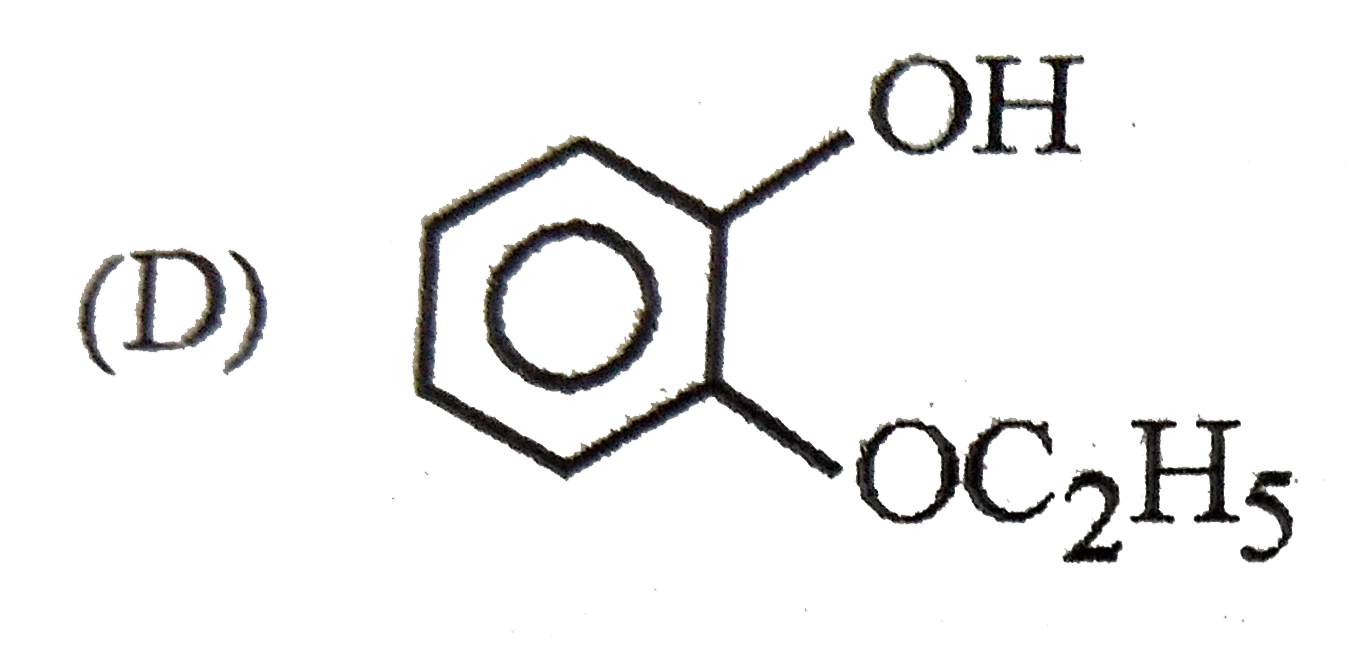
The intestinal antiseptic salol is correctly represented as |
|
Answer»
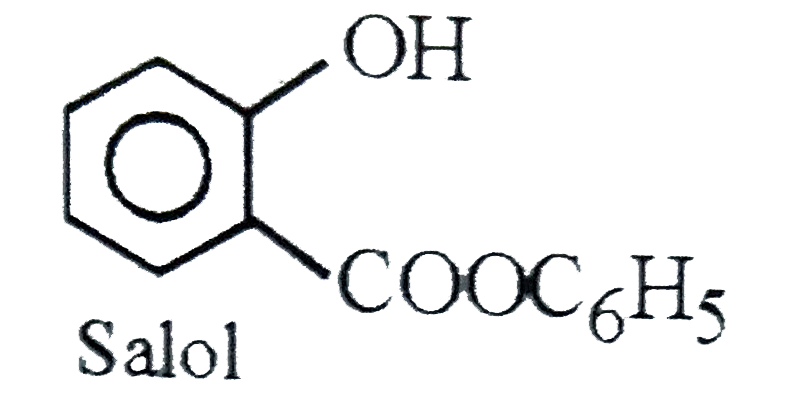
|
|
| 40. |
The internalenergy change ( Delta U )for the reaction CH_(4)(g) + 2O_(2)(g) rarr CO_(2)(g) +2H_(2)O(l)is - 885kJ mol^(-1) at298 K . What is DeltaH at298 K ? |
|
Answer» |
|
| 41. |
The internal resistance to flow in liquid is called |
|
Answer» Fluidity |
|
| 42. |
The internal pressure loss of one mole of vander Waal gas over an ideal gas is not equal to |
|
Answer» ZERO |
|
| 43. |
The internal energy of a system is ………….. Property and …………….function. |
|
Answer» INTENSIVE , SATE |
|
| 44. |
The internal energy change when a system goes from state A to B is 40 KJ/mole. If the system goes from A to B by a reversible path and returns to state A by an irreversible path what would be the net change in internal energy ? |
| Answer» ANSWER :D | |
| 45. |
The intermolecular interaction that is dependent on the inverse cube of distance between the molecules is : |
|
Answer» ion - ion interaction |
|
| 46. |
The intermolecular force of attraction present between NH_3 and C_6H_6 are |
|
Answer» DIPOLE - Dipole |
|
| 47. |
The intermolecular attraction that is dependent on the inverse cube of the distance between the molecules is |
|
Answer» Ion-ion interaction |
|
| 48. |
The intermetallic compound LiAg crystallizes in a cubic lattice in which Li and Ag atoms have C.N. of 8. To which crystal class the unit cell belongs |
|
Answer» NaCl |
|
| 49. |
The intermediate species involved in the acid catalysed dehydration of alcohol is |
|
Answer» Free radical |
|