Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The intermediate formed in the acid catalysed dehydration of n-propyl alcohol is |
|
Answer» `CH_(3)-CH_(2)-CH_(2)^(+)` 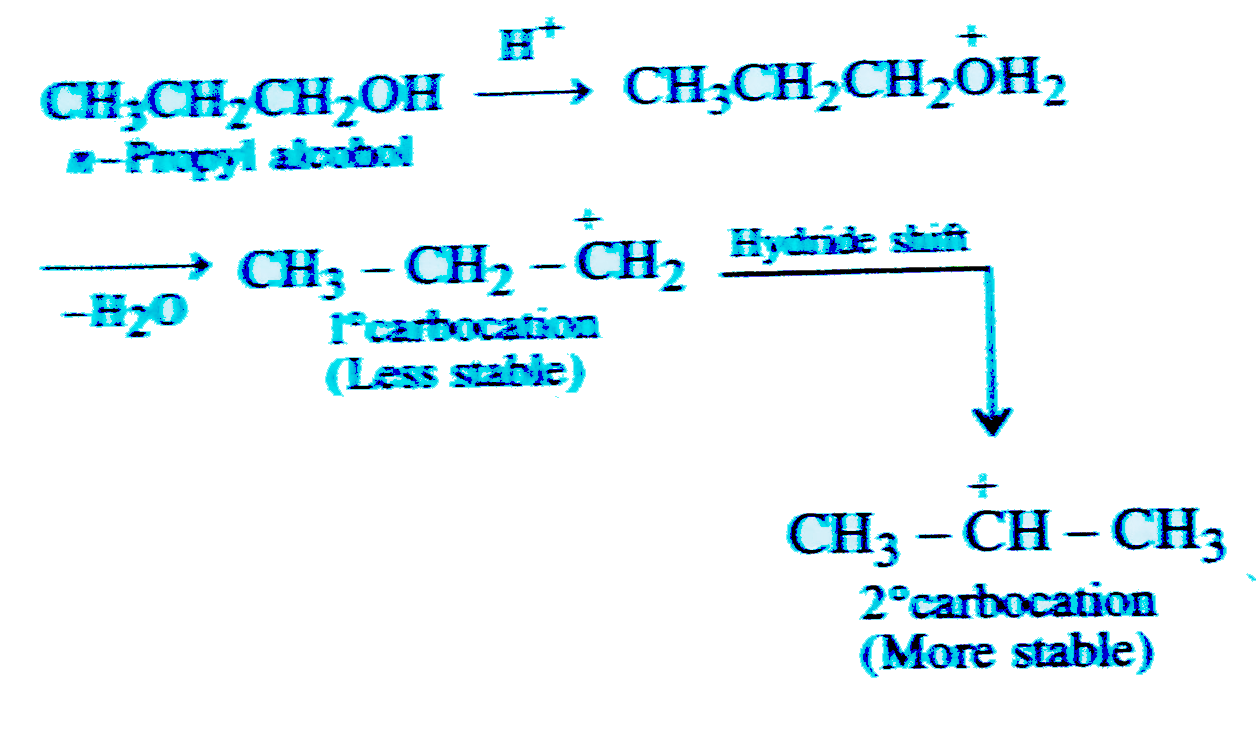
|
|
| 2. |
The intermediate during the addition of HCl to propene in the presence of peroxide is |
|
Answer» `CH_(3)overset(.)(C)HCH_(2)CL` |
|
| 3. |
The intermediate compound formed when acetylene is hydrated in presence of dil. H_(2)SO_(4) and HgSO_(4) is |
|
Answer» ACETALDEHYDE |
|
| 4. |
The intermediate carbocation formed in the reaction of HI, HBr and HCl with propene is the same and the bond energy of HCl, HBr and HI is 430.5 kJ mol^(-1), 363.7kJ mol^(-1) and 296.8 kJ mol^(-1) respectively. What will be the order of reactivity of these halogen acids ? |
|
Answer» Solution :Hydrogen halides (HCl, HBr, HI) ADD upto a alkanes to form alkyl halides. The order of reactivity of the hydrogen halides is `hI gt HBr gt HCl`. Reactivity of HX depends on bond dissociation enthalpy of HX. Bond enthalpy of HI `(296.kJ mol^(-1)) lt HBr (363.7 kJ mol^(-1)) lt HCl (430.5 kJ mol^(-1))` is in this order hence there activity is in reverse order : `HCl lt HBr lt HI` `CH_(3)-CH=CH_(2)+H^(+)` slow step `RARR CH_(3-) ""^(+)CH-CH_(3)+X^(-)` fast `rarr CH_(3)-CHX-CH_(3)` Slow step is RATE determining step and it depends on the availability of `H^(+)`. Availability of `H^(+)` depends on bond dissociation enthalpy of HX. |
|
| 5. |
The intermediate carbocation formed in the reactions of HI, HBr and HCl with propene is the same and the bond energy of HCl, HBr and HI is 430.5 "kJ mol"^(-1), 363.7 "kJ mol"^(-1) and 296.8 "kJ mol"^(-1) respectively. What will be the order of reactivity of these halogen acids ? |
Answer» Solution :In the reaction of propene with HCl , HBr or HI, first a `H^+` adds to give the same carbocation intermediate (I) which then undergoes nucleophilic attack by the HALIDE ion `(X^-)` to give the addition product. Here, first STEP is slow and hence is the rate-determining step of the reaction while the second step is first and hence does not affect the rate of the reaction. 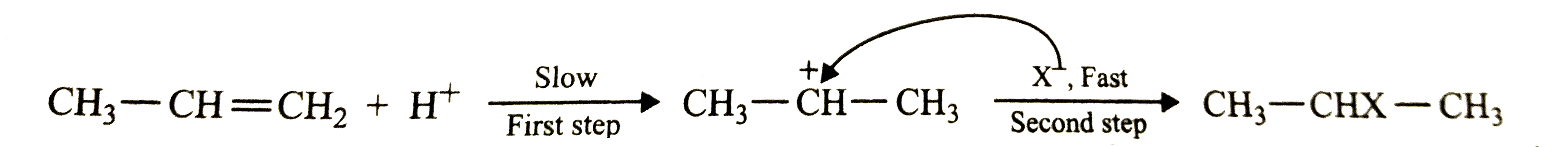 The rate of the first step depends upon how easily is the proton available. The availability of the proton, in turn, depends upon the bond dissociation energy of the H-X MOLECULE. Since the bond dissociation energy decreases in the order : HCl `(430.5 "kJ mol"^(-1)) GT HBr(36. "kJ mol"^(-1)) gt HI (296.8 "kJ mol"^(-1))`, therefore , the REACTIVITY of the halogen acids decreases in the reverse order : HI gt HBr gt HCl |
|
| 6. |
The intermediate as well as final products formed in the reaction . |
|
Answer»
|
|
| 7. |
The intermediate as well as final products feomed in the reaction . |
|
Answer»
|
|
| 8. |
The interionic distance for cesium chloride crystal will be |
|
Answer» a |
|
| 9. |
The interionic distance for cesium chloride crystals will be |
|
Answer» a |
|
| 10. |
The intercepts and slope of graph of logK_(p)^(@) "Vs" (1)/(T) are : |
|
Answer» `DeltaG^@, DELTAH^@` |
|
| 11. |
The interactions that are results of temporary dipoles induced in the ordinarily non polar molecules are |
|
Answer» Dispersion FORCES |
|
| 12. |
The interaction energy of London force is inversely proportional to sixth power of the distance between two interacting particles but their magnitude depends upon |
|
Answer» charge of interacting particles. More complex or large are the molecules, higher is the magnitude of London forces. This is due to the fact that the large ELECTRON clouds are easily polarised. HENCE, higher the polarisability of interacting particles, higher is the magnitude of the interaction energy. |
|
| 13. |
The interaction energy of London force is inversely proportional to sixth of the distance between two interacting particles but their magnitude depends upon |
|
Answer» charge of intercting PARTICLES |
|
| 14. |
The inter molecular forces present in inert gases are |
|
Answer» Ion-ion |
|
| 17. |
The intensive property among these quantities is |
|
Answer» Weight |
|
| 18. |
Theintensityof linesin spectrumdependson whichfactor ? |
| Answer» SOLUTION :Emitteda ABSORB NUMBEROF PHOTON. | |
| 19. |
The instrument measuring heat changes at constant pressure .......... |
|
Answer» PH meter |
|
| 20. |
The inorganic compound obtained by the auto oxidation of 2-alkylanthraquinol is |
|
Answer» `H_(2)O` |
|
| 21. |
The inner shell to which the electrons jump from the outer shells in the formation of Pfund series in the hydrogen spectrum is |
|
Answer» |
|
| 22. |
The initial temperature of a gas 1s 27 to temperature 80 cm^(3) of the gas should be heated to increases the volume by 25%. |
|
Answer» Solution :`v_(1)80 cm^(3), v_(2)+80+(25)/(100)xx80=100 cm^(3)` `T_(1)=27+273=300 K, T_(2)=?` `(v_(1))/(T_(1))=(v_(2))/(T_(2)) orT_(2)=(T_(1)V_(2))/(V_(1))=(300xx100)/(80)` `:. T_(2) =375 K=375-273=102^(@)C` `:. T_(2)=102^(@)C` `:. T_(2)=102^(@)C` |
|
| 23. |
The initial concentrations of A and B are same for A +2B harr 3C and when equilibrium concentrations of B and C are same then K_(c) is |
|
Answer» 0.25  [B]=[C] `x=a // 5` `K_(C)=([C]^(3))/([A][B]^(2))=(((3A)/(5))^(3))/((4a // 5)(3a // 5)^(2))=(3)/(4)` |
|
| 24. |
The inflow of nutrients from fertilizers into lakes resulting in the increase of phosphate in lake water and depletion of dissolved oxygen causing the fish to die is called _____ |
|
Answer» |
|
| 28. |
The indicator used in iodometric titrations is : |
|
Answer» phenolphthalein |
|
| 29. |
Theincreasingorder orreactivity amonggroup 1 element is Li lt Na lt K lt Rb lt Cs whereasthe of group17isF gt C1 gt Br gt I. Explain. |
|
Answer» Solution :Theelements of group1 haveonly oneelectronin theirrespectivevalenceshellsand thusa strong tendencyto losethiselectron. Thetendencyto loseelectrons in turn DEPENDS upon theionizationenthalpy. Sincethe ionizationenthalpydecreasesdownthe groupthereforethe reactivityof group1 elements increasesin the sameorder : `LI lt Na lt K lt Rb ltCs.` In contrastthe elements of group17 haveseven electrons in theirrespectivevalenceshellsand thushave astrong tendencyto accept one moreelectron Thetendency to accept electronsin turn dependsupontheirelectrode potentials.Sincetheelectrode potentialsof group17 elements decreasesin the order : `F (+ 2.87 V ) GT C1 ( + 1.36 V ) , Br ( 1.08 V )` and `I (+ 0.53 V)` thereforetheirreactivitiesalso decreases in the sameorder `: F gt C1 gt Br gt I` Alternativelytendency toaccept electrons can belinked to electrongain enthalpy. sinceelectrongainenthalpybecomesless and lessnegativeas we move fromC1 to I thereforereactivityincreasesfromto I, F ,howeveris themostreactive due toits lowbonddissociationenergy . |
|
| 30. |
Theincreasingorderof theionicradiiof the givenisoelectronicspecies is |
|
Answer» `S^(2-) , C1^(-),CA^(2+) ,K^(+)` |
|
| 31. |
The increasing order of the ionic radii of the given isoelectronic species is :- |
|
Answer» `K^(+),S^(2-),CA^(2+),CL^(-)` |
|
| 32. |
The increasing order of the ionic radii of the given isoelectronic species is : |
|
Answer» `CL^(-),Ca^(2+), K^(+), S^(2-)`  From `Ca^(2+) to K^(+)to Cl^(-) to S^(2-) ` nuclear chaege Z is DECREASING in them are EQUAL 18. THEREFORE the ionic radii of them is increasing. |
|
| 33. |
The increasing order of the density of alkali metals is |
|
Answer» `Li lt K lt NA lt Rb lt CS ` |
|
| 34. |
The increasing order of the first ionisation enthalpies of the element B, P, S and F is- |
|
Answer» <P>`F LT S lt P lt B` |
|
| 35. |
The increasingorder of thefirstionizationenthalpyfo the elementB, P, S andF (lowestfirst )is |
|
Answer» `B lt Plt S lt F` |
|
| 36. |
The increasing order of the acid strength is correct in |
|
Answer» PHENOL, ACETIC acid, Ethanol, Chloroacetic acid |
|
| 37. |
The increasing order of stability of the following free radicals is………. |
|
Answer» `(CH_(3))_(2) overset(.)(C )H lt (CH_(3))_(3) overset(.)( C) lt (C_(6)H_(5))_(2) overset(.)(C )H lt (C_(6)H_(5)) overset(.)(C )` `2^(@) lt 3^(@) lt "benzylic " 2^(@) lt "benzylic " 3^(@)` Stability of free readicals `ul(1^(@)lt 2^(@)lt 3^(@) 4^(@) lt "benzylic"1^(@))lt` benzylic `2^(@)` alkylic `lt` benzylic `3^(@)` |
|
| 38. |
The increasing order of stability of the following free radicals is |
|
Answer» `(CH_(3))_(2)overset(**)(C)H lt (CH_(3))_(3)overset(**)(C) lt (C_(6)H_(5))_(2)overset(**)(C)Hlt(C_(6)H_(5))_(3)overset(**)(C)` |
|
| 39. |
The increasing order of stability of the following free radicals is : |
|
Answer» `(CH_(3))_(2)overset(*)(CH) lt (CH_(3))_(3) overset(*) lt (C_(6)H_(5))_(2)overset(*)(CH) lt (C_(6)H_(5))_(3)overset(*)C` |
|
| 40. |
The increasing order of reduction of alkyl halides with zinc and dilute HCl is ….. |
|
Answer» `R-CL LT R-I lt R-Br` |
|
| 41. |
The increasing order of reduction of alkyl halides with zinc and dilute HCl is |
|
Answer» R-Cl lt R-I lt R-Br |
|
| 42. |
The increasing order of reactivity among group 1 elements isLi< Na< K< Rb < Cs whereas in group 17 F> Cl> Br> I Explain. |
| Answer» Solution :The ELEMENTS of group I have only one electron in their respective VALENCE shells and thus have a strong tendency to lose this electron. The tendency to lose electrons in turn, depends upon the ionization ENTHALPY. Since the ionization enthalpy decreases down the group therefore, the reactivity of group 1 elements increases in the same ORDER LiltNaltKltRbltCs. In contrast. the elements of group 17 have seven electrons in their respective valence shells and thus have strong tendency to ACCEPT one more electron to make stable configuration. It is linked with electron gain enthalpy and electronegativity. Since both of them decreases down the group, the reactivity also decreases. | |
| 43. |
The increasing order of pK_(a) values of the following compounds is : |
|
Answer» `C LT B LTA lt D` |
|
| 44. |
The increasing order of reactivity among group-1 elements is Li lt Na lt K lt Rb lt Cs whereas that among group 17 elements is F gt CI gt Br gt I. |
|
Answer» SOLUTION :The elements of GROUP-1 have only one electron in their outermost shells and therefore, have strong tendency to lose this electron. The tendency of these elements to lose the valence electron depends upon the ionization enthalpy. Since ionization enthalpy decreases down the group, therefore the reactivity of group-1 elements increases in the same order `Li lt Na lt K lt KB lt Cs`. On the other hand, the elements of group-17 have seven ELECTRONS in their respective valence shells and therefore, they, have strong tendency to accept one more electron . The tendency to accept ADDITIONAL electrons depends upon the electrode potentials of group17 elements. The electrode potential of group 17 elements decreases fromF to I `[F = +2.86V, Cl = + 1.36V, " Br " = + 1.08 V " and " I = +0.53V]` and therefore , their reactivities also decrease in the same order as `F gt Cl gt Br gt I`. |
|
| 45. |
The increasing order of ionic character of CsF, LiI, NaBr and KCl is |
|
Answer» `NaBr lt KCL lt LII lt CsF` |
|
| 46. |
The increasing order of first ionization enthalpies of the elements B, P, S and F (lowest first) is ......... |
|
Answer» `P lt S lt B lt F` ACCORDINGLY first ionisation enthalpy of `B lt F` (both of second period) and of `P lt S` (both of third period) The `I^(ST)` ionisation enthalpy decreases from top to botton in a GROUP . ` :. I^(st)`ionisation potential order should be: `B lt F " and " P lt S` `P [Ne] 3s^(2) 3px^(1) 3py^(1) 3p_(2)^(1)` and `S [Ne] 3s^(2) 3px^(2) 3py^(1) 3p_(z)^(1)` P has stable half filled (p) Electronic configuration, hence`Delta_(i)H(P) gt Delta_(i)H(S)` ` :. ` The correct order is `B lt F " and " S lt P ` F is of smallest size among these all. `:. F`has highest `Delta_(i)H` ` :. `Orderis `B lt S lt P lt F` |
|
| 47. |
The increasing order of hyperconjugative effect ofCH_(3)-,C_(2)H_(5)-,(CH_3)_(2)CH-" and "(CH_3)_(3)C- when one of these is attached to benzene ring is |
|
Answer» `CH_(3),C_(2)H_(5),(CH_3)_(2)CH" and "(CH_3)_(3)C` |
|
| 48. |
The increasing order of electron donating inductive effect of alkyl groups is |
|
Answer» `-H lt -CH_(3) lt -C_(2)H_(5) lt -C_(3)H_(7)` |
|
| 49. |
The increasing order of effusion among the gases H_2O_2, NH_3 and CO_2 is |
|
Answer» `H_2 , CO_2 ,NH_3 ,O_2` |
|







