Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The kinetic energy of an electron is 5 xx 10^(5) eV (electron volts). Calculate the wavelength of the wave associated with the electron. The mass of the electron may be taken as 10^(-30) kg |
|
Answer» or `v^(2) = 16 xx 10^(6) or v = 4 xx 10^(3) m s^(-1)` Hence, `lamda = (h)/(mv) = (6.626 xx 10^(-34) kg m^(2) s^(-1))/(10^(-30) kg xx 4 xx 10^(3) ms^(-1)) = 1.66 xx 10^(-7) m` |
|
| 2. |
The kinetic energyof electronis 4.55 xx 10^(-25) Jandmas is 9.1 xx 10^(-31) kg thencalculatevelocity, momentum and wavelengthofelectron. |
| Answer» Solution :VELOCITY `=1XX 10^(3)ms^(-1)` MOMENTUM = `9.1 xx 10^(28) kg ms^(-1)lambda = 7.28 xx 10^(7)m` | |
| 3. |
The massof anelectronis 9.1 xx 10^(31) kgif its K.E.Is 3.0 xx 10^(25) J calculateitswavelength . |
| Answer» SOLUTION :`7.25 XX 10^(7) m` | |
| 4. |
The kinetic energy of an electron in the second Bohr orbit of a hydrogen atom is [a_(0) is Bohr radius] |
|
Answer» `(h^(2))/(4pi^(2) m a_(0)^(2))` `:. v = (nh)/(2pi mr)` `K.E. = (1)/(2) mv^(2) = (1)/(2) m (n^(2) h^(2))/(4pi^(2) m^(2) r^(2))`..(i) But `r = (a_(0) n^(2))/(Z)` for 2ND Bohor orbit of hydrogen, `n = 2, Z = 1` `:. r = (a_(0) xx 4)/(1) = 4a_(0)` Substituting in eqn (i) `K.E. = (1)/(2) (m(2)^(2) h^(2))/(4pi^(2) m^(2) (16a_(0)^(2))) = (h^(2))/(32pi^(2) ma_(0)^(2))` |
|
| 5. |
The kinetic energy of a sub-atomic particles is 5.85 xx 10^(-25)J. Calculate the frequency of the particle wave. (Planck's constant, h = 6.626 xx 10^(-34) kg m^(2) s^(-1)) |
|
Answer» SOLUTION :K.E. `= (1)/(2) mv^(2) = 5.85 xx 10^(-25) J` By de Broglie equation, `lamda = (h)/(mv)` But `lamda = (u)/(V) :. (u)/(v) = (h)/(mv) or v (mv^(2))/(h) = (2 xx 5.85 xx 10^(-25)J)/(6.626 xx 10^(-34) Js) = 1.77 xx 10^(9) s^(-1)` |
|
| 6. |
The Kinetic energy of a gas depends upon a) nature of the gas b) absolute temperature c) molecular weight of the gas d) number of moles of the gas |
|
Answer» B, d |
|
| 7. |
The kinetic energy (KR) of photoelectron emitted on irradiating a metal surface with frequency .vartheta. is related by KE = h vartheta - IE. The plots of K.E vs incident frequency is a straight line which shows |
|
Answer» SLOPE EQUAL to planck.s constant |
|
| 8. |
The kinetic energy of 1 mole of oxygen molecules in cal mol^(-1)at 27°C |
|
Answer» 300 |
|
| 9. |
The kinetic energy for 14 grams of nitrogen gas at 127^(@)C is nearly (mol. Mass of nitrogen=28 and gas constant=8.31J/mol/K.) |
|
Answer» 1.0J |
|
| 10. |
The kind of delocalization involving sigma bond and hybrid orbital's is called |
|
Answer» INDUCTIVE effect |
|
| 11. |
The kind of delocalization invloving sigma-bond orbitals is called............... . |
|
Answer» |
|
| 12. |
TheK_H for the solution of oxygen dissolved in water is4xx 10^(4) atm at a given temperature . If the partial pressure of oxygen in air is 0.4 atm the mole fraction of oxygen in solution is |
|
Answer» <P>`4.6 xx 10 ^(3)` `(P _(O_(2)))_(air) = K _(H) (x _(O_(2)))_("in solution ")` `0.4 =4 xx 10 ^(4) (x _(O_(2)))_("in solution ")` ` therefore (x _(O_(2))) _(" in solution ")= (0.4)/(4 xx 10 ^(4)) =1 xx 10 ^(-5)` |
|
| 13. |
The kind of delocalization involving sigma bond orbitals is called: |
|
Answer» INDUCTIVE EFFECT |
|
| 14. |
The K.E of N molecules of O_(2)is x Joules at -123°C. Another sample of O_(2) at 27°C has a KE of 2x Joules. The latter sample contains________N molecules of O_(2). |
| Answer» SOLUTION :`(K.E_1)/(K.E_2) = (n_1T_1)/(n_2T_2)` . | |
| 15. |
The Kc for the reaction I_(2 (g)) hArr 2I_((g)) is 4 xx 10^(-3) . If the equilibrium concentration of atomic iodine is 4 xx 10^(-2) M . What is the concentration of molecular iodine ? |
|
Answer» 0.8 M |
|
| 16. |
The K_(a) values of formic acid and acetic acid are respectively 1.77xx10^(-4) and 1.75 xx 10^(-5). The ratio of acid strengthof 0.1 N acids is |
|
Answer» 10 |
|
| 17. |
The K_(c) for given reaction will be A_(2(g))+2B_((g))hArrC_((g))+2D_((g)) |
|
Answer» `K_(C)=([C][D]^(2))/([A_(2)][B]^(2))` |
|
| 18. |
The K_(a) value of formic acid and acetic acid are respectively, 1.77 xx 10^(-4) " and 1.75 xx 10^(-5). The ratio of the acid strength of 0.1 N acids is |
|
Answer» 10 LET `C MOL L^(-1)` be the conc. and `alpha` its DEGREE of dissociation `:. K_(a) = (C alpha xx C alpha)/(C (1 - alpha)) = C alpha^(2)` (where `alpha` is very small) `:. alpha = sqrt((K_(a))/(C))` where C is constant, `[H^(+)] prop alpha` `:. (alpha_(1))/(alpha_(2)) = sqrt((K_(a_(1)))/(K_(a_(2)))) = sqrt((1.77 xx 10^(-4))/(1.75 xx 10^(-5)))` `= sqrt(10) = 3.178` |
|
| 19. |
The K_a value for HCN is 10^(-9). What is the pH of decimolar HCN solution ? |
|
Answer» SOLUTION :Dissociation constant, `K_a = 10^(-9)`, CONCENTRATION, `C=0.1 = 1 XX 10^(-3) mol L^(-1)` `[H^+]= sqrt(K_aC) = sqrt(10^(-9) xx10^(-1) = 10^(-5)) mol L^(-1)` pHof thesolution`pH=-log 10^(-5) = 5` |
|
| 20. |
The K, of an indicator HIn is 9 x 10^(4), The percentage of the basic form of indicator is 10x in a solution of pH = 4. What is x ? |
|
Answer» ` 9= ( In^(-))/( HI_n) RARR % " of " In^(-)=90 %` |
|
| 21. |
The K for the reaction A + B hArr C + D is at 25^@C temperature is 2 xx 10^(-23) and At 50^@C temperature 2 xx10^(-12) this reaction will be endothermic or exothermic ? |
| Answer» Solution :To INCREASE the temperature K will be increase i.e. FORWARD reaction occurs and increase PRODUCTS. Therefore reaction is ENDOTHERMIC. | |
| 22. |
The K, L and M shells of an atom are full. Its atomic number is ________. |
|
Answer» 18 |
|
| 23. |
The Joule -Thomson coefficient for a gas is zero at . There are no intermolecular attractive forces in an ideal gas . |
| Answer» SOLUTION :EXPLANATION is CORRECT REASON for STATEMENT . | |
| 24. |
The IVA element with highest and lowest first ionisation potential values |
| Answer» Solution :1st I.E. order is `C gt Si gt GE gt Pb gt Sn` | |
| 25. |
The IV A element with highest and lowest first ioniation potential values |
|
Answer» C, Pb |
|
| 26. |
The IUPAC of CH_(2)=CH-CH_(2)Cl is |
|
Answer» Ally] CHLORIDE |
|
| 27. |
Give IUPAC name of : CH_(3)-CO(CH_(2))_(2)-CH_(3) |
|
Answer» pentan-2-one |
|
| 28. |
The IUPAC name of the product formed when ethylene reacts with hypochlorous acid is |
|
Answer» ETHYLENE chlorohydrin |
|
| 29. |
The IUPAC name of the given structure (CH_3)_4C is |
|
Answer» DIMETHYL PROPANE |
|
| 30. |
The IUPAC name of the given compound CH_3-underset(Br)underset(|)(CH)-overset(CHO)overset(|)(CH)-overset(CONH_2)overset(|)(CH)-underset(COCl)underset(|)(CH)-COOH |
|
Answer» 2-BROMO-4-carbamoyl-5-chloroformyl-3 - formyl hexanoic acid |
|
| 31. |
The IUPAC name of the given structure (CH_3)_2CH-CH(CH_3)_2 is |
|
Answer» 2,2-Dimethyl BUTANE |
|
| 32. |
The IUPAC name of the given compound CH_3-CH=CH-COOC_2H_5 is : |
|
Answer» ETHYL 1-butenoate |
|
| 33. |
The IUPAC name of the given compound |
|
Answer» 2-ethylhex-4-en-2-ol |
|
| 34. |
The IUPAC name of the following compound is : H_(3)C-overset(CH_(3))overset("|")"CH"-overset(OH)overset("|")"CH"-CH_(2)-COOH |
|
Answer» 4-Methyl-3-hydroxypentanoic acid Carboxylic group has HIGH priority than hydroxyl group so NUMBERING starts from carbon of carboxylic group. 3-hydroxy-4-methylpentanoic acid |
|
| 35. |
The IUPAC name of the following compound is CH_(3)-underset(CH_(3))underset(|)CH-underset(CH_(2))underset(|)underset(CH_(2))underset(|)CH-CH_(2)-CH_(3) |
| Answer» SOLUTION :3-Ethyl 2- METHYLPENTANE | |
| 36. |
The IUPAC name of the following compound is CH_(3)-underset(CH_(3))underset("|")("C")H-CH_(2)-CH=CH_(2) |
|
Answer» 2-Methylpent-4-ene |
|
| 37. |
The IUPAC name of the following compound is CH_3 - undersetoverset(|)(CH_3)(C )H - CH_2 -CH = CH_2 |
|
Answer» 2- Methylpent -4-ene |
|
| 38. |
The IUPAC name of the following compound is |
|
Answer» 5-cyano-3-formylcyclohex-3-en-1-carboxylic ACID |
|
| 39. |
The IUPAC name of the following compound is {:(CH_(2)-CH--CH_(2)),("|""|""|"),(CN""CN""CN):} |
|
Answer» 1, 2, 3-tricyanopropane |
|
| 40. |
The IUPAC name of the following compound is : |
|
Answer» 3-ethyl-2, 6-dimethyloct-2, 6-dien-1-ol 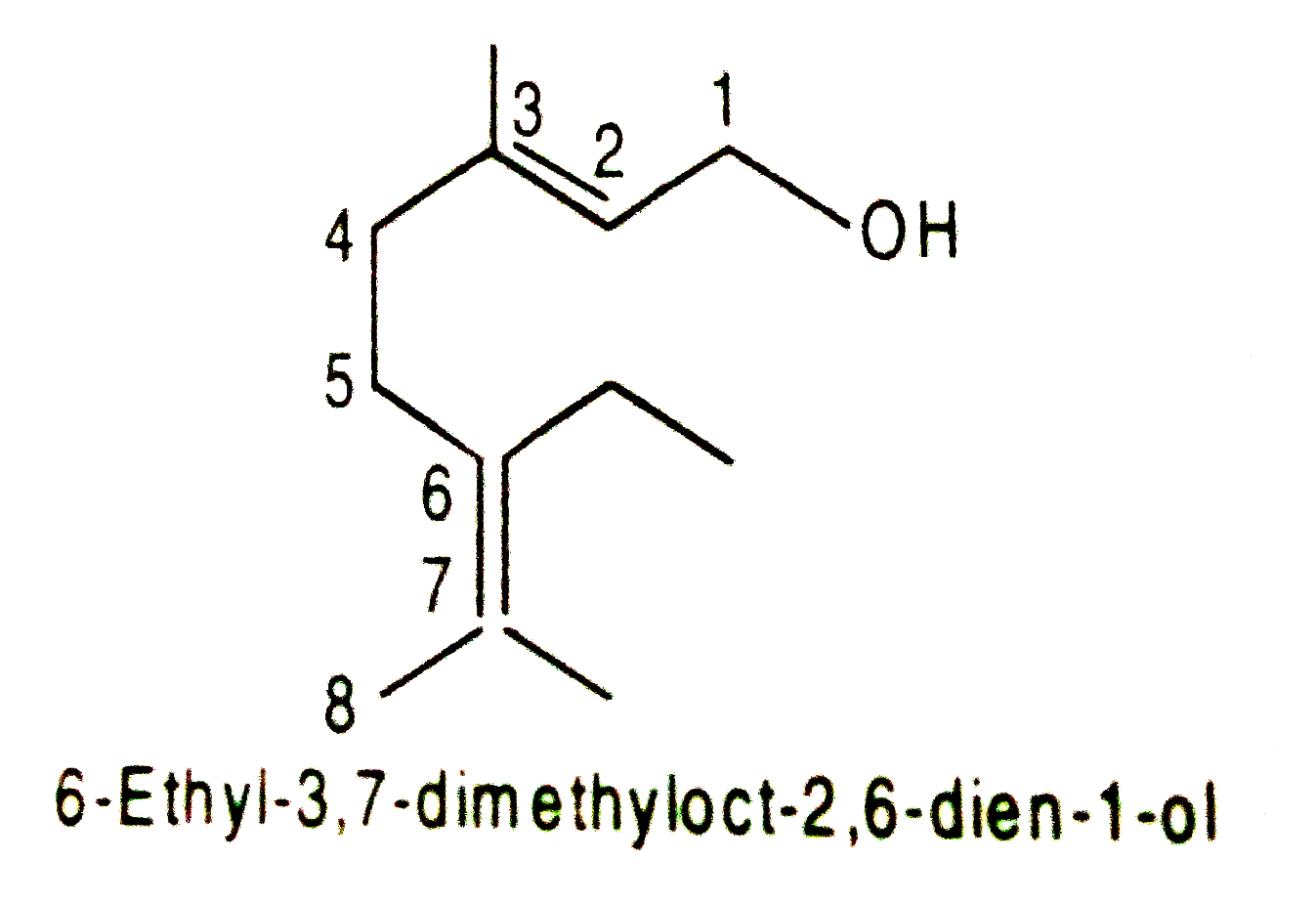
|
|
| 41. |
The IUPAC name of the element with atomic number 104 is.......... |
| Answer» SOLUTION :unnilquadrium. | |
| 42. |
The IUPAC name of the compound X in the following reaction (CH_(3))_(2)C = CHCH_(2) underset(H_(2)O_(2)//OH^(-))overset(B_(2)H_(6))rarrX is |
|
Answer» 2-Methylbutane Hydroboration oxidation of ALKENES TAKES place according to anti-Markownikov's rule. |
|
| 43. |
The IUPAC name of the compound, underset(OH)underset(|)(C)H_(2)-underset(NH_(2))underset(|)(C)H-COOH is |
|
Answer» 2-amino-3-hydroxypropanoic acid |
|
| 44. |
The IUPAC name of the compound shown below is: H-underset(H)underset(|)overset(H)overset(|)C-underset(H)underset(|)overset(H)overset(|)C-underset(H)underset(|)overset(Cl)overset(|)C-Cl |
| Answer» SOLUTION :1, 1-dicholoropropane | |
| 45. |
The IUPAC name of the compound shown below is …… |
|
Answer» 2-bromo-6-chlorocyclohex-1-ene 
|
|
| 46. |
The IUPAC name of the compound shown below is : |
|
Answer» 2-bromo-6-chlorocyclohex-1-ene  3-Bromo-1-chlorocyclohexene 3-Bromo-1-chlorocyclohexene
|
|
| 47. |
The IUPAC name of the compound is {:(HOOC - CH_(2) - CH - CH_(2) - CH_(2) - COOH),(""|),(""COOH):} |
|
Answer» 2-(Carboxymethyl)pentane-1, 5-dioic acid |
|
| 48. |
The IUPAC name of the compound OHC-CH_2-CH_2-underset(CHO)underset(|)(CH)-CH_3 is : |
|
Answer» 2-Formyl pentanal |
|
| 49. |
The IUPAC name of the compound is |
|
Answer» `1,1':3',1''-`Terycyclobutane 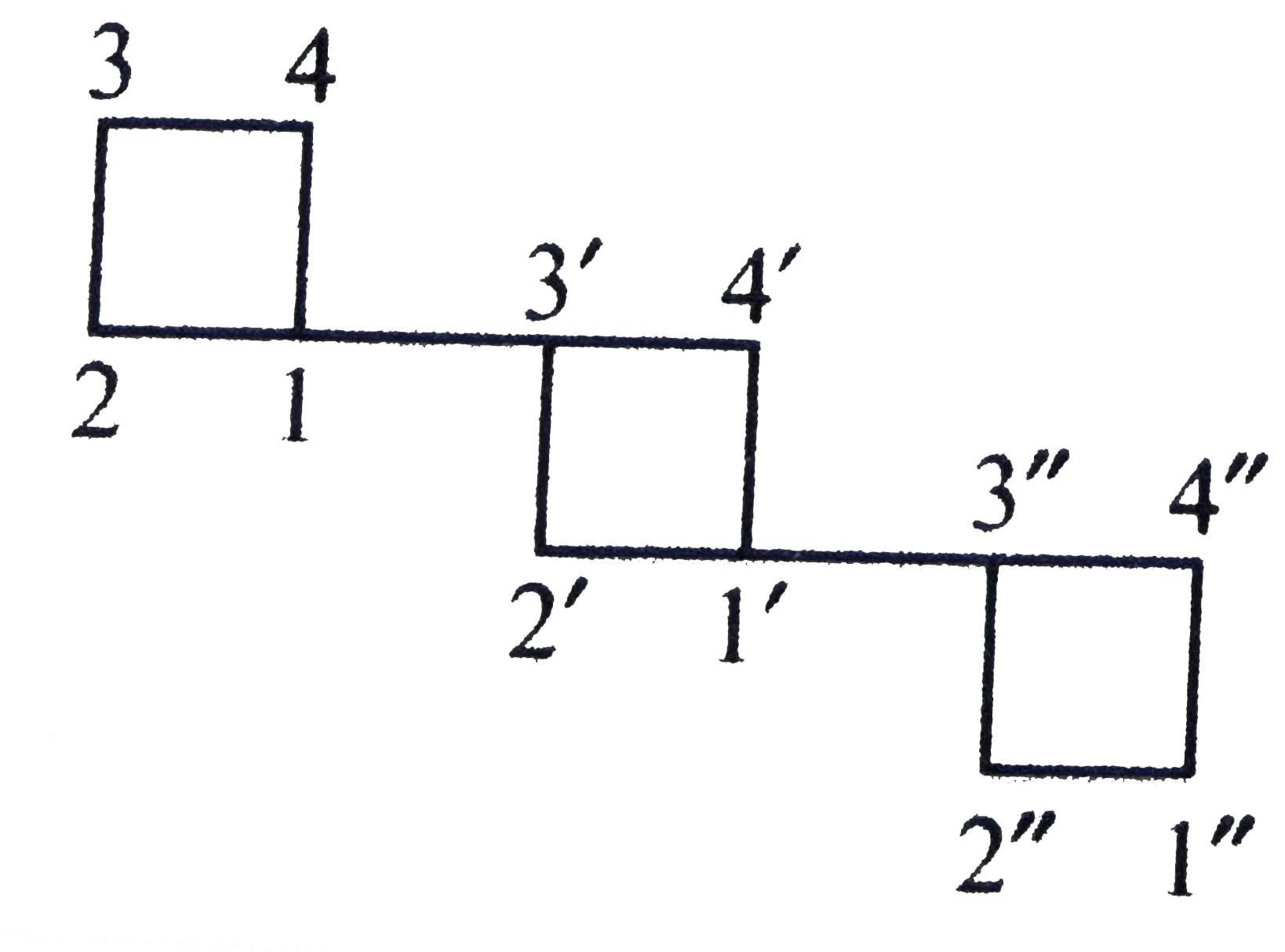 `1,1',3',1''-`Tercyclobutane |
|
| 50. |
The IUPAC name of the compound having the formula CH-=C-CH=CH_(2) is |
|
Answer» 1-butyn-3-ene |
|