Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The reactivity of haloacids (HCI, HBr, HI) with alcohol is in the order…………………… . |
| Answer» SOLUTION :HI > HBR > HCI | |
| 2. |
The reactivity of carboxylic acids towards esterification is highest in |
|
Answer» ACETIC acid |
|
| 3. |
The reactivity of an element is very much related to its inoization enthalpy. Observe the following graph in wich the first ionozation enthalpies (delta_1H) of elements of the sercond period are ploptted agiains their atomic number (Z): |
| Answer» Solution :From the GRAPH it is clear that, Be has HIGHER inoization ENTHALPY than B. nitrogen has higher ionisation enthalpy than O. this is because Be has complitlely FILLED `(1s^2 2s^2)` 2sorbital. | |
| 4. |
The reactivity of alcohols with haloacid is ……………….. . |
|
Answer» `3^@ GT 2^@ gt 1^@` |
|
| 5. |
The reactivity of alcohols with halo acid is ……………….. . |
|
Answer» `3^@ GT 2^@ gt 1^@ ` |
|
| 6. |
The reactions of benzene with acetic anhydride in presence of anhydrous AlCl_3 is called _____ reaction and yields _____ |
|
Answer» |
|
| 8. |
The reaction which proceeds in the backward direction is |
|
Answer» `Fe_(3)O_(4)+6HCl=2FeCl_(3)+3H_(2)O` |
|
| 9. |
The reaction which proceeds in the forward direction is. |
|
Answer» `SnCl_4+Hg_2Cl_2rarrSnCl_2+2HgCl_2` (1) Stannous chloride is a powerful reducing agent. When an excess of stannous chloride solution is added to MERCURIC chloride solution, a white precipitate of mercurous chloride appears which slowly turns grey as further reduction to mercury occurs: `SnCl_2+2HgCl_2rarrHg_2Cl_2darr+SnCl_4` `Hg_2Cl_2+SnCl_2rarr2Hg_2darr+SnCl_4` (2) Cuprous iodie is precipitated as a BUFF solid when POTASSIUM iodide solution is added to the solution of cupric ions (e.g. copper sulphate solution): `2CuSO_4+2Klrarr2CuIdarr+I_2+2K_2SO_4` This reaction is used for estimating copper salts volumetrically by titrating the liberated iodine with standard SODIUM thiosulphate solution. (3) `NaOH` is a stronger base than `NH_3`, and thus, diplaces it from its salt: `NH_4Cl+NaOHrarrNaCl+underset(NH_3+H_2O)underset(darr)(NH_4OH)` |
|
| 10. |
The reaction which is in dynamic equilibrium, ensured us, that the reaction is reversible . But if that the reaction is in equilibrium. The reaction quotient predict either the reversiblereaction is in equilibrium or tries to achieve equilibrium. In those reactions which have not achieved equilibrium, we obtain reaction quotient Q_(c) in place of equilibrium constant (K_(c)) by substituting the concentration of reactant and product at the time, at whih we have to calculate the value of Q_(c) . To determine the direction at which the net reaction will proceed to achieve equilibrium, we compare values of Q_(c) and K_(c). The three possible cases are shown as comparison of K_(c) and Q_(c) in the following figures. Change in Gibbs free energy, i.e., Delta G is the driving force of any reaction. For spontaneous reaction , Delta G =-ve For non-spontaneous reaction , Delta G=+ve For reaction at equilibrium , Delta G =0 Thermodynamically, we know that Delta G= Delta G^(@)+ RTln Q, where Q is reaction quotient and Delta G^(@)= change in Gibbs energy at standard condition. For equilibrium A(g) hArr B(g) (K_(eq) =1.732) If the pressure of the system [varied by introducing a stream of A (g) and B (g) is representedby the curve at constant temperature T. For equilibrium system N_(2)O_(4)(g) hArr2NO_(2) (g) N_(2)O_(4)(g) is in a cylinder which is fitted with movable piston. Assume that equilibriumpartial pressureof N_(2)O_(4) (g)and NO_(2)(g) are 10 and 14 atmospheres respectively. If the piston of the cylinder is pulledout in such way so that volume of the system than what will be the value of equilibrium partial pressure of the NO_(2) gas ? |
|
Answer» 10.7 atm |
|
| 11. |
The reaction which is in dynamic equilibrium, ensured us, that the reaction is reversible . But if that the reaction is in equilibrium. The reaction quotient predict either the reversiblereaction is in equilibrium or tries to achieve equilibrium. In those reactions which have not achieved equilibrium, we obtain reaction quotient Q_(c) in place of equilibrium constant (K_(c)) by substituting the concentration of reactant and product at the time, at whih we have to calculate the value of Q_(c) . To determine the direction at which the net reaction will proceed to achieve equilibrium, we compare values of Q_(c) and K_(c). The three possible cases are shown as comparison of K_(c) and Q_(c) in the following figures. Change in Gibbs free energy, i.e., Delta G is the driving force of any reaction. For spontaneous reaction , Delta G =-ve For non-spontaneous reaction , Delta G=+ve For reaction at equilibrium , Delta G =0 Thermodynamically, we know that Delta G= Delta G^(@)+ RTln Q, where Q is reaction quotient and Delta G^(@)= change in Gibbs energy at standard condition. For equilibrium A(g) hArr B(g) (K_(eq) =1.732) If the pressure of the system [varied by introducing a stream of A (g) and B (g) is representedby the curve at constant temperature T. If the gaseous substances A,B,C, be in equilibrium as under : {:(B(g),hArr,A(g),,),(10%,,60%,,):} {:(B(g),hArr,C(g),,),(10%,,30%,,):} then which of the following is the correct order of stability of A,B, and C in the equilibrium mixture ? |
|
Answer» `AGTBGTC` |
|
| 12. |
The reaction which is in dynamic equilibrium, ensured us, that the reaction is reversible . But if that the reaction is in equilibrium. The reaction quotient predict either the reversiblereaction is in equilibrium or tries to achieve equilibrium. In those reactions which have not achieved equilibrium, we obtain reaction quotient Q_(c) in place of equilibrium constant (K_(c)) by substituting the concentration of reactant and product at the time, at whih we have to calculate the value of Q_(c) . To determine the direction at which the net reaction will proceed to achieve equilibrium, we compare values of Q_(c) and K_(c). The three possible cases are shown as comparison of K_(c) and Q_(c) in the following figures. Change in Gibbs free energy, i.e., Delta G is the driving force of any reaction. For spontaneous reaction , Delta G =-ve For non-spontaneous reaction , Delta G=+ve For reaction at equilibrium , Delta G =0 Thermodynamically, we know that Delta G= Delta G^(@)+ RTln Q, where Q is reaction quotient and Delta G^(@)= change in Gibbs energy at standard condition. For equilibrium A(g) hArr B(g) (K_(eq) =1.732) If the pressure of the system [varied by introducing a stream of A (g) and B (g) is representedby the curve at constant temperature T. For the equilibrium n Butane (g) hArrIsobutane (g),(K_(eq)=1.732)If the pressure of the system (varied by introducing a stream of n butane and isobutane ) is represented by the curve at constanttemperature T. At a particular point Q, which of the following statements holds good ? |
|
Answer» The REACTION moves in the backward direction. |
|
| 13. |
The reaction which is in dynamic equilibrium, ensured us, that the reaction is reversible . But if that the reaction is in equilibrium. The reaction quotient predict either the reversiblereaction is in equilibrium or tries to achieve equilibrium. In those reactions which have not achieved equilibrium, we obtain reaction quotient Q_(c) in place of equilibrium constant (K_(c)) by substituting the concentration of reactant and product at the time, at whih we have to calculate the value of Q_(c) . To determine the direction at which the net reaction will proceed to achieve equilibrium, we compare values of Q_(c) and K_(c). The three possible cases are shown as comparison of K_(c) and Q_(c) in the following figures. Change in Gibbs free energy, i.e., Delta G is the driving force of any reaction. For spontaneous reaction , Delta G =-ve For non-spontaneous reaction , Delta G=+ve For reaction at equilibrium , Delta G =0 Thermodynamically, we know that Delta G= Delta G^(@)+ RTln Q, where Q is reaction quotient and Delta G^(@)= change in Gibbs energy at standard condition. For equilibrium A(g) hArr B(g) (K_(eq) =1.732) If the pressure of the system [varied by introducing a stream of A (g) and B (g) is representedby the curve at constant temperature T. Suppose the equilibrium system N_(2)O_(4)(g) hArr 2NO_(2)(g) N_(2)O_(4)(g)is in a cylinder fitted with a movable piston . Which of the following statements is correct ? |
|
Answer» If piston is pushed DOWNWARDS at constant temperature, `Q_(C) GT K_(c)` and the direction shifts in the LEFT direction. |
|
| 14. |
The reaction which is in dynamic equilibrium, ensured us, that the reaction is reversible . But if that the reaction is in equilibrium. The reaction quotient predict either the reversiblereaction is in equilibrium or tries to achieve equilibrium. In those reactions which have not achieved equilibrium, we obtain reaction quotient Q_(c) in place of equilibrium constant (K_(c)) by substituting the concentration of reactant and product at the time, at whih we have to calculate the value of Q_(c) . To determine the direction at which the net reaction will proceed to achieve equilibrium, we compare values of Q_(c) and K_(c). The three possible cases are shown as comparison of K_(c) and Q_(c) in the following figures. Change in Gibbs free energy, i.e., Delta G is the driving force of any reaction. For spontaneous reaction , Delta G =-ve For non-spontaneous reaction , Delta G=+ve For reaction at equilibrium , Delta G =0 Thermodynamically, we know that Delta G= Delta G^(@)+ RTln Q, where Q is reaction quotient and Delta G^(@)= change in Gibbs energy at standard condition. For equilibrium A(g) hArr B(g) (K_(eq) =1.732) If the pressure of the system [varied by introducing a stream of A (g) and B (g) is representedby the curve at constant temperature T. If A and B are enclosed in the cylinder and piston of the cylinder be moved downward so that volume of cylinder becomes half, then what will be the effect in K_(c) at constant temeprature ? |
|
Answer» <P>`K_(c)` will increase |
|
| 15. |
The reaction which is in dynamic equilibrium, ensured us, that the reaction is reversible . But if that the reaction is in equilibrium. The reaction quotient predict either the reversiblereaction is in equilibrium or tries to achieve equilibrium. In those reactions which have not achieved equilibrium, we obtain reaction quotient Q_(c) in place of equilibrium constant (K_(c)) by substituting the concentration of reactant and product at the time, at whih we have to calculate the value of Q_(c) . To determine the direction at which the net reaction will proceed to achieve equilibrium, we compare values of Q_(c) and K_(c). The three possible cases are shown as comparison of K_(c) and Q_(c) in the following figures. Change in Gibbs free energy, i.e., Delta G is the driving force of any reaction. For spontaneous reaction , Delta G =-ve For non-spontaneous reaction , Delta G=+ve For reaction at equilibrium , Delta G =0 Thermodynamically, we know that Delta G= Delta G^(@)+ RTln Q, where Q is reaction quotient and Delta G^(@)= change in Gibbs energy at standard condition. For equilibrium A(g) hArr B(g) (K_(eq) =1.732) If the pressure of the system [varied by introducing a stream of A (g) and B (g) is representedby the curve at constant temperature T. Suppose N_(2)O_(4)(g) is enclosed in a cylinder fitted with a movable piston which attains the following equilibrium N_(2)O_4)(g) hArr 2NO_(2)(g) Given that for the 10 atmosphere pressure of the equilibrium mixture,the content of NO_(2) is 8xx1^(5)ppm. if the piston of cylinder is moved upward in such a manner so that the volume of the gaseous mixture becomes double, then what will be new ppm of NO_(2)(g) in the cylinder ?( Assuming that the temperature of the cylinder remains constant ) |
|
Answer» `8.2 XX 10^(5) `PPM |
|
| 16. |
The reaction which is in dynamic equilibrium, ensured us, that the reaction is reversible . But if that the reaction is in equilibrium. The reaction quotient predict either the reversiblereaction is in equilibrium or tries to achieve equilibrium. In those reactions which have not achieved equilibrium, we obtain reaction quotient Q_(c) in place of equilibrium constant (K_(c)) by substituting the concentration of reactant and product at the time, at whih we have to calculate the value of Q_(c) . To determine the direction at which the net reaction will proceed to achieve equilibrium, we compare values of Q_(c) and K_(c). The three possible cases are shown as comparison of K_(c) and Q_(c) in the following figures. Change in Gibbs free energy, i.e., Delta G is the driving force of any reaction. For spontaneous reaction , Delta G =-ve For non-spontaneous reaction , Delta G=+ve For reaction at equilibrium , Delta G =0 Thermodynamically, we know that Delta G= Delta G^(@)+ RTln Q, where Q is reaction quotient and Delta G^(@)= change in Gibbs energy at standard condition. For equilibrium A(g) hArr B(g) (K_(eq) =1.732) If the pressure of the system [varied by introducing a stream of A (g) and B (g) is representedby the curve at constant temperature T. Which of the following is the correct statement at point R in the figure above ? |
|
Answer» The reaction will move in backward direction in order to gain EQUILIBRIUM Higher the negative value of `Delta G^(@)`, higher will be the stability of the product. |
|
| 17. |
The reaction which is in dynamic equilibrium, ensured us, that the reaction is reversible . But if that the reaction is in equilibrium. The reaction quotient predict either the reversiblereaction is in equilibrium or tries to achieve equilibrium. In those reactions which have not achieved equilibrium, we obtain reaction quotient Q_(c) in place of equilibrium constant (K_(c)) by substituting the concentration of reactant and product at the time, at whih we have to calculate the value of Q_(c) . To determine the direction at which the net reaction will proceed to achieve equilibrium, we compare values of Q_(c) and K_(c). The three possible cases are shown as comparison of K_(c) and Q_(c) in the following figures. Change in Gibbs free energy, i.e., Delta G is the driving force of any reaction. For spontaneous reaction , Delta G =-ve For non-spontaneous reaction , Delta G=+ve For reaction at equilibrium , Delta G =0 Thermodynamically, we know that Delta G= Delta G^(@)+ RTln Q, where Q is reaction quotient and Delta G^(@)= change in Gibbs energy at standard condition. For equilibrium A(g) hArr B(g) (K_(eq) =1.732) If the pressure of the system [varied by introducing a stream of A (g) and B (g) is representedby the curve at constant temperature T. What will be the value of difference of standard Gibbs free energy to Gibbs free energy change at point Q in the figure above ? |
|
Answer» `-ve` `:. Delta G^(@)-Delta G=-RT ln Q` `=- ve (" Q GT 1.732 )` |
|
| 18. |
The reaction which does not liberate hydrogen is |
|
Answer» `Zn+NAOHTO` |
|
| 19. |
The reaction that is not involved in the ozone layer depletion mechanism in the stratosphere is ……………. |
|
Answer» `CF_2 Cl_(2(g)) OVERSET(uv)to Cl_((g)) + CF_2 Cl_((g))` |
|
| 20. |
The reaction that gives CO_(2) as one of the products is |
|
Answer» `Fe_(2)O_(3)+3C overset(250^(@)-400^(@)C)rarr` |
|
| 21. |
The reaction takes place at anode in H_2 - O_2fuel cell is |
|
Answer» `2H_2O + O_2 + 2E^(-) to 4OH^(-)` |
|
| 22. |
the reaction related to coal gassification |
|
Answer» `CO+H_(2)Ooverset(Fe_(2)O_(2)+Cr)toCO_(2)+H_(2)` |
|
| 23. |
The reaction related to coal gasification |
|
Answer» `CO+H_(2)O OVERSET(Fe_(2)O_(3)+Cr)(RARR)CO_(2)+H_(2)` |
|
| 24. |
The reaction, RCOOH + N_(3)H underset("conc.")overset(H_(2)SO_(4))rarr RNH_(2) + CO_(2) + N_(2) is called : |
|
Answer» HVZ reaction |
|
| 25. |
The reaction, RCOOAg + Br overset(C Cl_(4))rarr RBr + AgBr + CO_(2) is called : |
|
Answer» HVZ reaction |
|
| 26. |
The reaction RCH_(2)CH_(2)COOH underset(Br_(2))overset("Red P")rarr R - CH_(2) - underset(Br)underset(|)(CH) - COOH is called |
|
Answer» Reimer-Tiemann REACTION |
|
| 27. |
The reaction, RC-=CR underset("Lindlar's catalyst")overset(H_(2))toGives the main product as |
|
Answer» cis-alkene |
|
| 28. |
The reaction, R_(2)CO+4[H] underset("Conc. HCl")overset("Zn-Hg")rarr R_(2)CH_(2)+H_(2)O is well known as : |
|
Answer» WURTZ reaction |
|
| 29. |
The reaction quotient (Q) for the reaction N_(2 (g)) + 3 H_(2 (g)) hArr 2 NH_(3 (g)) is given by Q = ([NH_(3)]^(2))/([N_(2) ] [H_(2)]^(3)) . The reaction will proceed from right to left if |
| Answer» Answer :D | |
| 30. |
The reaction proceeds through _____. |
|
Answer» Electrophilic addition |
|
| 31. |
The reaction, Ph-CH_(2)-CH=CH-overset(OH)overset(|)(CH)-CH_(3) underset("ketone(excess)"//Delta)overset(Al(BuO)_(3))rarrPhCH_(3)CH=CH-overset(O)overset(||)(C)-CH_(3) is known as : |
|
Answer» Wolff-kishner reduction |
|
| 32. |
The reaction, PCI_(5)hArrPCI_(3)+CI_(2) is started in a five litre container by taking one mole of PCI_(5). If 0.3 mol PCI_(5) is there at equilibrium, concentration of PCI_(3) "and" K_(c) will respectively be: |
|
Answer» `0.14,(49)/(150)` Total mole of `PCI_(3)=0.7` Concentration`=0.14` `K_(c)=(X^(2))/((1-x)V)=(0.7xx0.7)/(0.3xx5)=(49)/(150)` |
|
| 33. |
The reaction P_(4)+3 NaOH+3 H_(2)O rarr 3 NaH_(2)PO_(2)+PH_(3) is an example of |
|
Answer» disproportination RECTION |
|
| 34. |
The reaction of zinc with dilute and concentrated nitric acid, respectively produces |
|
Answer» `N_(2)OandNO_(2)` `Zn+4HNO_(3)(CONC.)toZn(NO_(3))_(2)+2H_(2)O` |
|
| 35. |
The reaction of white phosphours with aqueous NaOH gives phosphine along with another compound. The reaction type , the oxidation states of phosphorus in phosphine and the other product are respectively |
|
Answer» <P>redox reaction , -3 and -5 It is a disproportionation reaction. In this reaction, oxidation state of P increases from 0 in `P_(4)" to "+1` in `NaH_(2)PO_(2)` and decreases from 0 in `P_(4)" to "-3" in "PH_(3)`. |
|
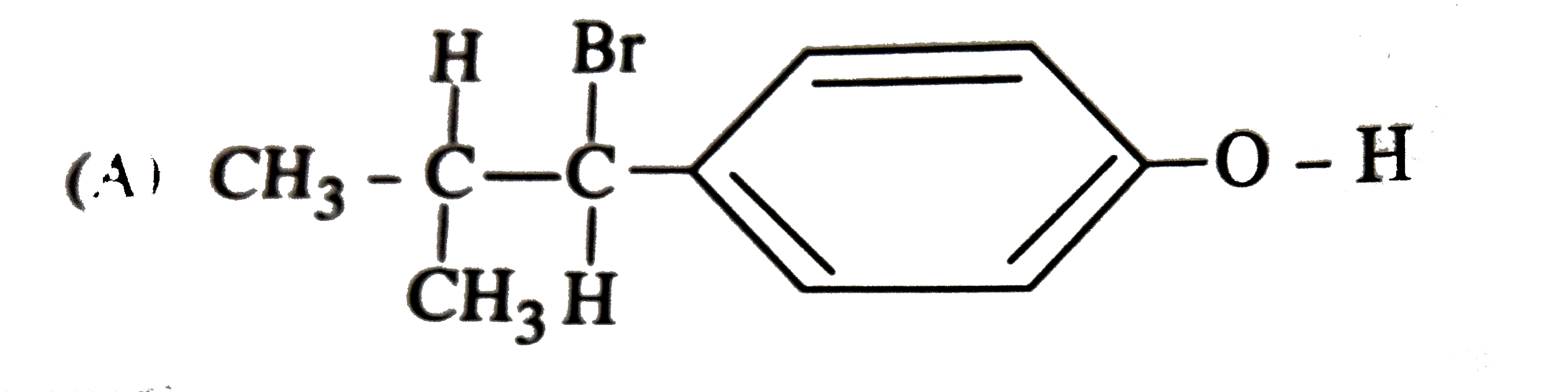
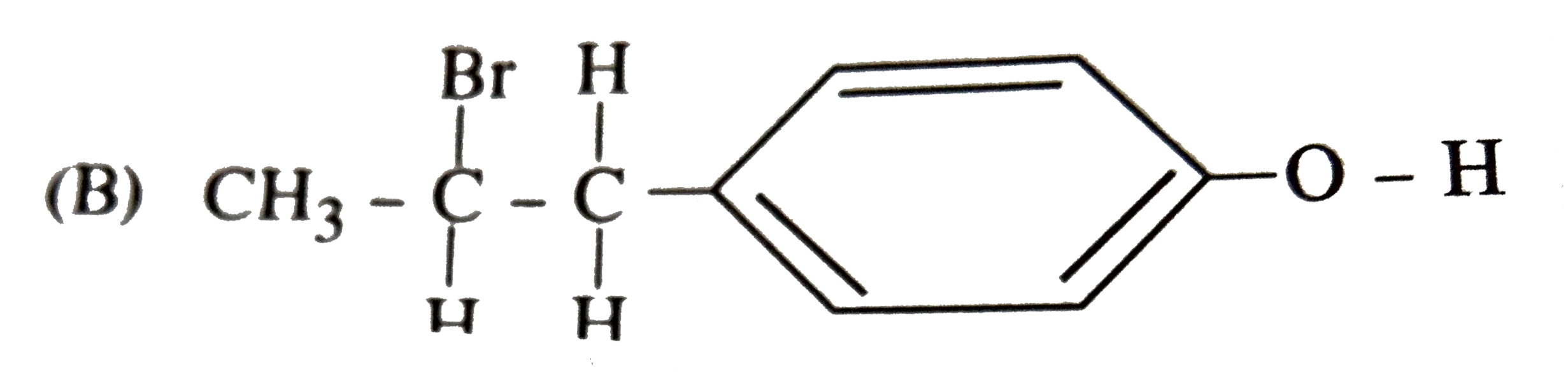
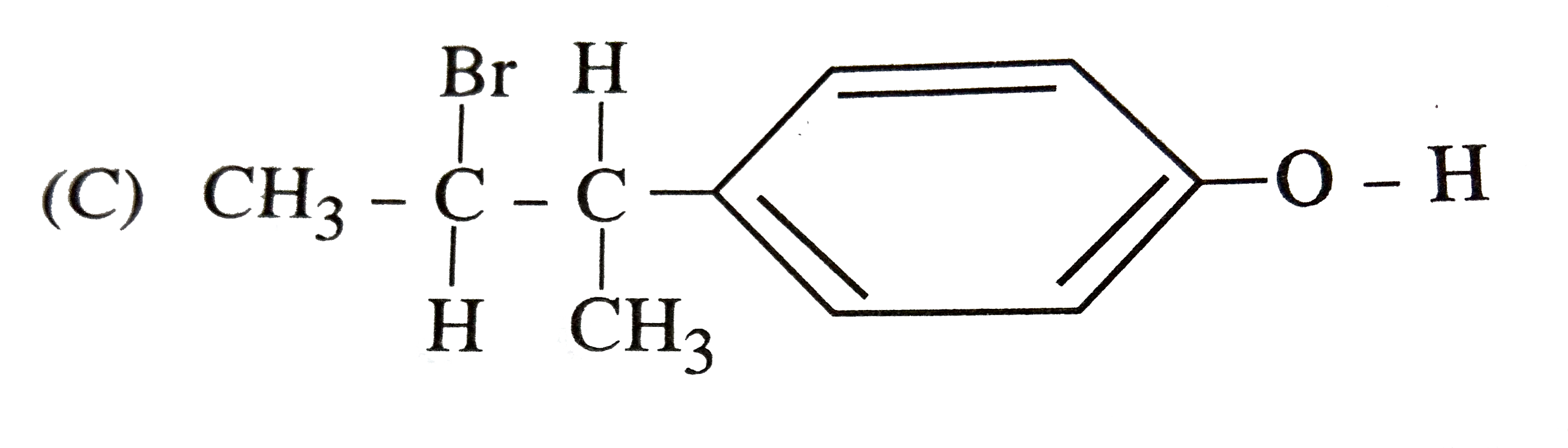
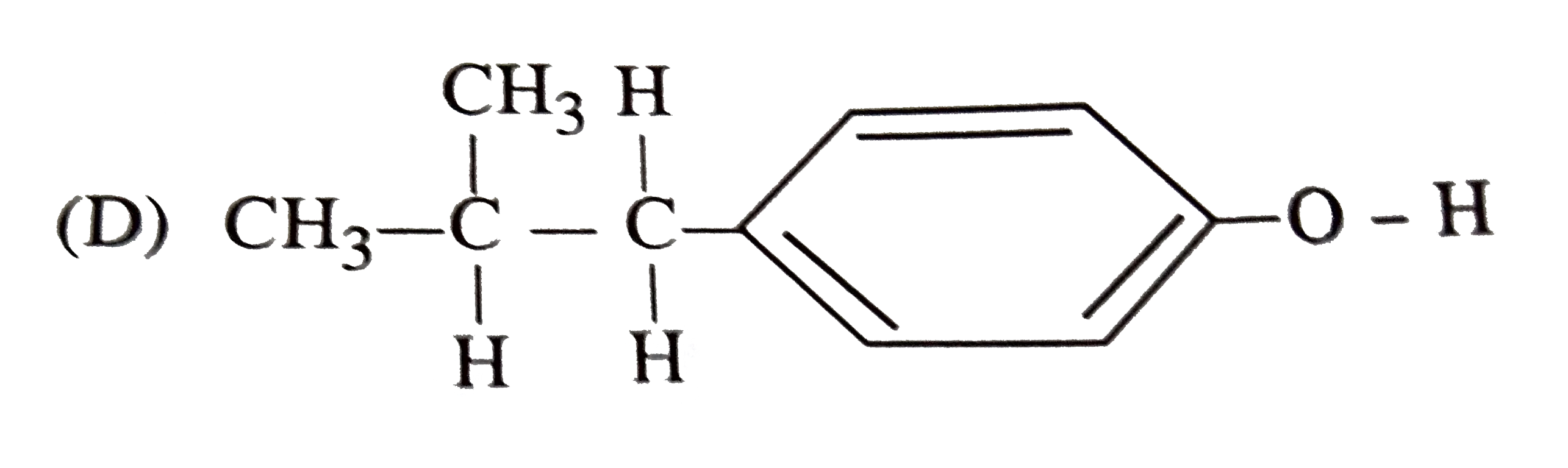
| 36. |
The reaction of with HBr gives predominantly |
|
Answer»
|
|
| 37. |
The reaction of white phosphorus with aqueous NaO gives phosphine along with anotehr phosshporus containg compound the reaction type the oxidation states of phosphorus in phosphine and the other porduct are respectively |
|
Answer» <P>redox reaction ,-3 and -5 O.N of in `P_(4)` decreases FORM 0 to -3 in `PH_(3)` and INCREASES from 0 to +1 in `NaH_(2)PO_(2)` therefore it is a disproportionation reaction |
|
| 38. |
The reaction of white phosphorus iwht aqueous NaOH gives phosphine along with another phosphorus containing compund. The oxidation states of phosphorus in phosphine and the other product are respectively |
|
Answer» REDOX reaction,-3 and -5 white phosphorus reacts with NaOH in an inert atmosphere as FOLLOWS : `P_(4)+3NaOH+3H_(2)Orarr3NaH_(2)PO_(2) +PH_(3)` O.S of P in `NaH_(2)PO_(2),+1+2(+1)+x+2(-2)=0` or x+3-4=0, x==4-3=+1 O.S of P in `PH_(3),x+3(+1)=0,x=-3` The values donot match with any of CHOICES given in the various options.This MEANS that none of the options is correct. |
|
| 39. |
The reaction of toluene with Cl_(2) in the presence of FeCl_(3) gives X and the reaction in the presence of light gives Y. Thus, X and Y are : |
|
Answer» X = BENZYL CHLORIDE, Y = m-chlorotoluene 
|
|
| 40. |
The reaction of toluene with Cl_2 in presence of FeCl_3 gives predominantly |
|
Answer» BENZYL CHLORIDE |
|
| 41. |
The reaction of toluene with Cl_(2) in presence of FeCl_(3) gives predominantly |
|
Answer» m-chlorobenzene 
|
|
| 42. |
The reaction of solid calcium carbide with water produces ………….., a flammable gas. |
|
Answer» |
|
| 43. |
The reaction of propene with HOCl(Cl_(2)+H_(2)O) proceeds through the intermediate : |
|
Answer» `CH_(3)-CHCl-CH_(2)^(+)` 
|
|
| 44. |
The reaction of propene with HOCl proceeds via the addition of |
|
Answer» `H^+` in the FIRST step |
|
| 45. |
The reaction of propene with HBr in the presence of ROOR (peroxide) proceeds through which ofthe following intermediates? |
|
Answer» `CH_3 - overset(*)(CH)-CH_3` |
|
| 46. |
The reaction of propene with HBr in the presence of ROOR (peroxide) proceeds through which of the following intermediate? |
|
Answer» `CH_(3)-OVERSET(.)(C)H-CH_(3)`<BR>`CH_(3)-overset(.)(C)H-CH_(2)*Br` |
|
| 47. |
The reaction of propene with HBr in presence of peroxide proceeds through the intermediate |
|
Answer» `H_3C-dot(CH)-CH_3`<BR>`H_3C-dotCH-CH_2Br` 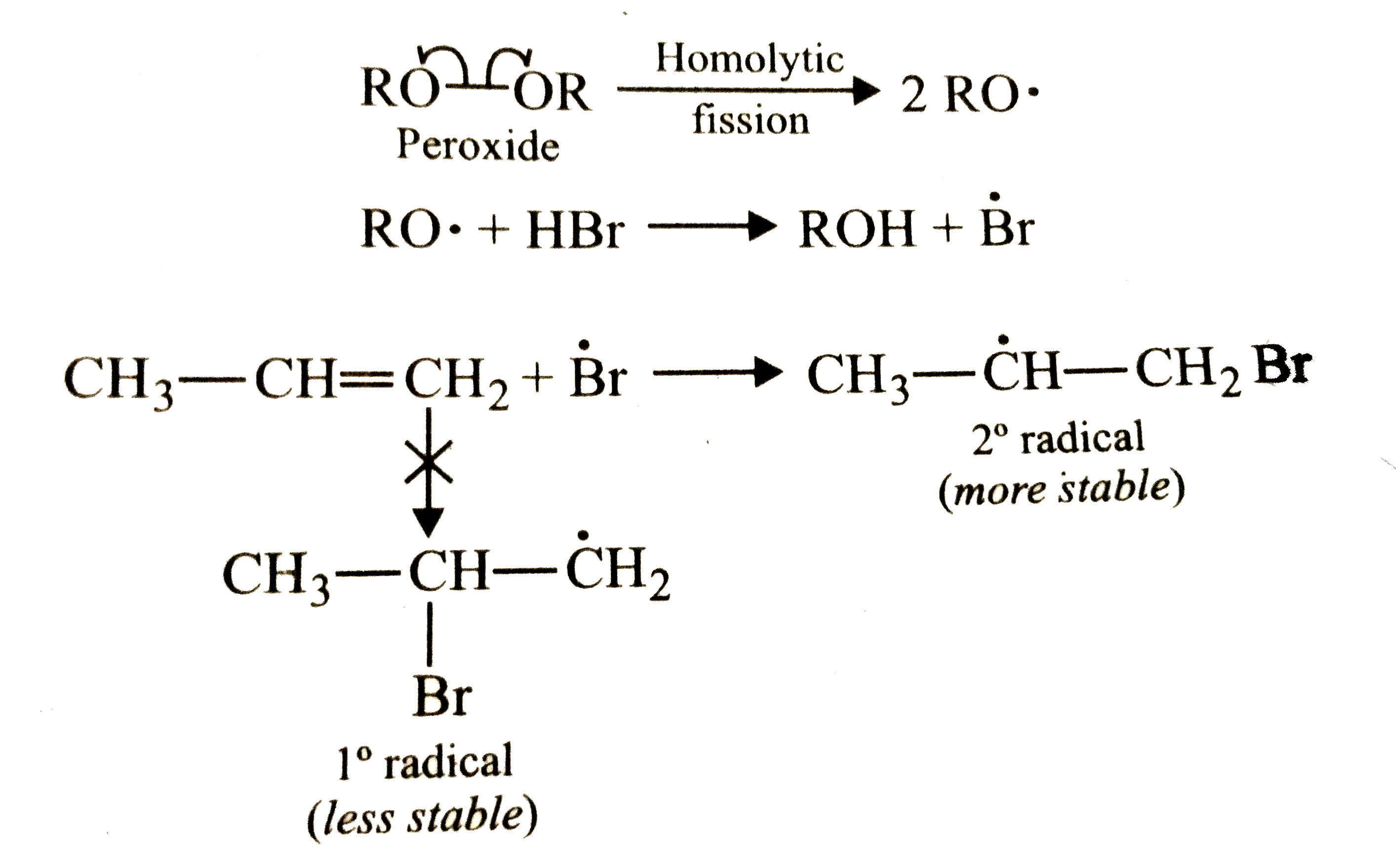
|
|
| 48. |
Thereactionof poweredBewithN_(2)produces______. |
|
Answer» `Be_(3) N_(2) ` |
|
| 49. |
The reaction of P_(4) with X leads selectively to P_(4)O_(6). The X is |
|
Answer» <P>Dry `O_(2)` `N_(2)` prevents further OXIDATION of `P_(4)O_(6)" to "P_(4)O_(10)`. |
|