Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Whichoneof thefollowingsamplesof metalscanproduducethe mosthydrogen byreacting withwater? |
|
Answer» 0.5MolOf Na |
|
| 2. |
Which will diffuse faster, ammonia or CO_(2)? What are their relative rates diffusion ? |
|
Answer» Solution :`M_(NH_(3))=17, M_(CO_(2))=44`. As rate `PROP sqrt((1)/(M))`, hence `NH_(3)` will DIFFUSE faster `(r_(NH_(3)))/(r_(CO_(2)))=sqrt((M_(CO_(2)))/(M_(NH_(3))))=sqrt((44)/(17))=1.609` |
|
| 3. |
What is the relationship between DeltaH and DeltaU for the reaction PCl_(5(g))hArrPCl_(3(g))+Cl_(2(g))? |
|
Answer» |
|
| 4. |
Which unit of concentration does not depends on temperature ? |
|
Answer» FORMALITY |
|
| 5. |
Which of the following statement sis inocrrect ? |
|
Answer» Sodium is the most abundant metla in earth's CRUST q |
|
| 6. |
The vapour density of completely dissociated NH_(4)Cl is |
|
Answer» double than that of `NH_(4)Cl` |
|
| 7. |
Using Stock notation, represent the following compounds : HAuCl_(4),Tl_(2)O,FeO,Fe_(2)O_(3),CuI,CuO,MnOandMnO_(2) |
|
Answer» Solution :By applying various rules of calculating the oxidation number of the desired element in a compound, the oxidation number of each metallic element in its compound is as follows: `HAuCl_(4)to` AU has 3 `Tl_(2)Oto` has 1 FeO `to` Fe has 2 `Fe_(2)O_(3)to` Fe has 3 CUI `to` Cu has 1 CuO `to` Cu has 2 MnO `to` Mn has 2 `MnO_(2)to` Mn has 4 THEREFORE, these compounds may be represented as : `HAu(III)Cl_(4),Tl_(2)(I)O,Fe(II)O,Fe_(2)(III)O_(3),Cu(I)I,Cu(II)O,Mn(II)O,Mn(IV)O_(2)` |
|
| 8. |
Using the bond dissciationenthalpies (BDE0 in the table , estimate DeltaH^(@) for the disproportionation of hydrazine described in the equation below: 3N_(2)H_(4)(g)to 4NH_(3)(g)+N_(2)(g) |
|
Answer» `+283KJ xxmol^(-1)` |
|
| 9. |
Which of the following has largest bond angle? |
|
Answer» `NO_(2)^(+)` |
|
| 10. |
When 20 ml of 2.5" M "H_(2)O_(2) aqueous solution is decomposed the S.T.P. volume of oxygen obtained is |
|
Answer» 56 ML |
|
| 11. |
What is Bond Angle ? Explain. |
|
Answer» Solution :It is defined as the angle between the orbitals containing bonding ELECTRON pairs around the central ATOM in a molecule/complex ion. Bond angle is expressed in degree which can be experimentally determined by spectroscopic methods. Uses : It gives some idea regarding the DISTRIBUTION of orbitals around the central atom in a molecule/complex ion and HENCE it HELPS us in determining its shape. For example H - O - H bond angle in water can be represented as under : 
|
|
| 13. |
Which of the following patterns has voids fraction of 0.26? |
|
Answer» AAAA…….. |
|
| 14. |
Which one is strong form sigma & pi bond ? Why ? |
|
Answer» Solution :`sigma`bond is strong form `sigma` & `pi` bond. The covalent bond in which extent of overlapping is more has more strength. The strength of any type of `sigma` or `pi`bond proportional to the magnitude of overlapping of atomic orbitals. In `sigma` bond formation of overlapping of atomic orbital is more and as a result `sigma` bond is strong. In `pi`bond formation the magnitude of over- LAPPING is LESS because of overlapping of atomic orbitals is SIDE WISE and as a result `pi`bond is weaker RELATIVE to `sigma` bond. |
|
| 15. |
Which of the following is not a reason and structure of CO_(2) ? |
|
Answer» O = C = O |
|
| 16. |
What is the common name given to the elements of group 2 ? |
| Answer» SOLUTION :ALKALINE EARTH METALS | |
| 17. |
What is meant by demineralised water? How is it obatined |
| Answer» Solution :Water free from cation `(Ca^(2+),MG^(2+)etc)` and ANIONS `(Cl^(-),SO_(4)^(2-),HCO_(3)^(-)etc).` responsible for hardness is known as de-ionised or demieralised water. It is formed by PASSING water SPECIALLY water repeatedly through cation and ANION exhanges. For more detail, consult section 9.10. | |
| 18. |
What is true but Ice |
|
Answer» Its DENSITY is more than water (d) ice is a good insulator |
|
| 19. |
What is the pHof 0.001 M anilinesolution ? The ionization constant of aniline is 4.27xx10^(-10). Calculate degree of ionization of aniline in the solution . Also calculate the ionization constant of the conjugate acid of aniline. |
|
Answer» SOLUTION :(i) `C_(6)H_(5)NH_(2)+H_(2)O hArr C_(6)H_(5)NH_(3)^(+)+OH^(-)` `K_(a)=([C_(6)H_(5)NH_(3)^(+)][OH^(-)])/([C_(6)H_(5)NH_(2)])=([OH^(-)]^(2))/([C_(6)H_(5)NH_(2)])` `[OH^(-)]=sqrt(K_(a)[C_(6)H_(5)NH_(2)])=sqrt((4.27xx10^(-10))(10^(-3)))=6.534xx10^(-7)M` `pOH=-log (6.534xx10^(7))=7-0.8152=6.18 :. PH = 14 - 6.18 = 7.82` `{:("(II)Also",C_(6)H_(5)NH_(2)+H_(2)O ,hArr,C_(6)H_(5)NH_(3)^(+),+,OH^(-),,),("Initial CONC.",C,,,,,,),("At. eqm.",C- C alpha,,C alpha,,C alpha ,,):}` `K_(b)=(C alpha . C alpha)/(C (1-alpha))=(C alpha^(2))/(1-alpha)=C alpha^(2) :. alpha=sqrt(K_(b)//C)=sqrt(4.27xx10^(-10)//10^(-3))=6.53xx10^(-4)` (iii) `pK_(a)+pK_(b)= 14` (for a pair of conjugate ACID and base) `pK_(b)=-log(4.27xx10^(-10))=10-0.62=9.38 :. pK_(a) = 14-9.38=4.62` i.e., `-log K_(a)=4.62 or log K_(a) = - 4.62 = bar(5).38 or K_(a) = ` Antilog `bar(5).38 = 2.399xx10^(-5)~=2.4xx10^(-5)` |
|
| 20. |
Velocity of 5 molecules of gas is 4 ms^(-1), 3 molecules is 10 ms^(-1) and 6 molecules is 6 ms^(-1), Then calculate u_(av), u_(rms) and u_(ms). |
|
Answer» Solution :`u_(av)=` AVERAGE of different VELOCITY `u_(av)= ("Sum of total different velocity")/("Total number of molecules")` `= (5xx4 ms^(-1))+(3xx10 ms^(-1))+(6+6 ms^(-1))/((5+3+6)"molecules")` `= ((20+30+36)ms^(-1))/(14)=6.143 ms^(-1)` `u_(rms)=` Average root mean square SPEED `= sqrt((u_(1)^(2)+u_(2)^(2)+u_(3)^(2))/(n))` `= sqrt(((5XX(4)^(2))+(3xx10^(2))+(6xx6^(2)))/(5+3+6))` `= sqrt((80+300+216)/(14))` `= sqrt(42.57)=6.527 ms^(-1)` Most probable speed `(u_(ms))=0.816xx u_(rms)` `= 0.816xx6.527` `= 5.326 ms^(-1)` |
|
| 21. |
What is wrong with the following names ? Draw the structures they represent and give theircorrect name (i) 1, 1-Dimethylpentane(ii) 2-Methyl-2-propylhexane(iii) 3-Dimethylpentane (iv) 4, 4-Dimethyl-3-ethylpentane(v) 4-(2-Methylethyl)heptane |
Answer» SOLUTION :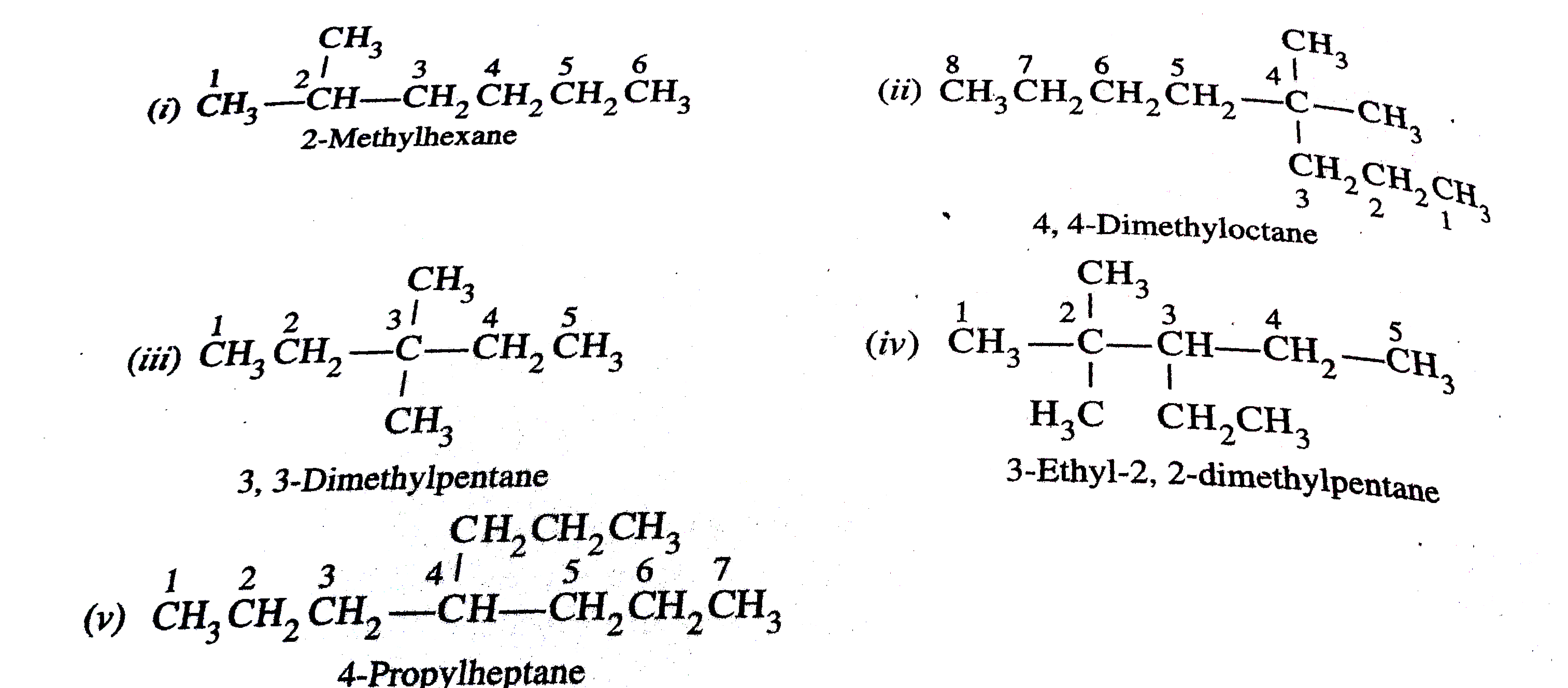
|
|
| 22. |
Two tetravalent elements A and B form dioxides.Both react with NaOH to form similar salts. lfloorOAO is 180^@ .lfloor ul(OBO)is 109^(@)28^(1). Both are acidic in nature. A and B are respectively |
| Answer» Answer :C | |
| 23. |
Write the reaction that takes place when BCl_3disolved in excess of heavy water. |
|
Answer» SOLUTION : In heavy water `BiCl_3`UNDERGO PARTIAL hydrolysis and FORMS BiOCI Bismathyl chloride. `BiCl_3 +D_2O to BiOCl + 2DCl` |
|
| 24. |
Write the electronic configuration and theblock to which an element with Z= 90 belongs |
| Answer» Solution :`[RN] 6S^(2) 7s^(2)` , f-block ELEMENT | |
| 25. |
The weight of a pure sample of KCIO_(3) to be decomposed in order to get 0.96g of O_(2) is |
|
Answer» 2.45g |
|
| 26. |
What is the correct order of bond angle of the following molecule |
|
Answer» `Coverset(alpha)F_(4),Coverset(BETA)(C)I_(4),Coverset(gamma)(B)r_(4), |__XMX` bond ANGLE `alpha gt beta gt gamma` (X= halgen atom) |
|
| 27. |
Which of the following is isobutyric acid ? |
|
Answer» `CH_(3)CH_(2)CH_(2)COOH` |
|
| 28. |
Zn_((s))+Fe_(aq)^(2+) Leftrightarrow Zn_(aq)^(2+) +Fe_((s). The vale of K_(c)for this reaction is 10^(23). Calculate the standard free energy change. |
|
Answer» Solution :Change in standard FREE energy, `Delta G ^(@) =- 2l303 RT log _(10) K _(c)` `Delta G ^(@) =- 2.303 xx 1.987 xx 298 xx log 10 ^(23)` `=- 13.570 CAL mol ^(-1)` Standard free energy change for the REACTION is `- 13.57 kcal mol^(-1)` |
|
| 29. |
Which of the following are the best method(s) for the preparation of cumene (A) ? |
|
Answer»

|
|
| 31. |
Which of the following will undergo nitration most easily? Why? Benzene, m-Dinitrobenzene, toluene. |
| Answer» SOLUTION :Toluene UNDERGOES nitration more easily. This is because`-CH_3` group in toluene is electron donating. It increases the electron DENSITY in BENZENE. So electrophilic substitution reactions such as nitration TAKE place more readily in toluene. | |
| 32. |
What are non-stoichiometric compounds ? |
| Answer» Solution :IF the actual ratio of the CATIONS and ANIONS is not same as REPRESENTED by the ideal chemical formula of the compound, it is called a non-stoichiometric compound. | |
| 33. |
Write formulas for thefollowing comounds (a) Mercury (II) chloride (b)Niclel (I) suphate (c ) Tin (iv) oxide (d) Thallium (I) suphate (e ) Iron (III) sulphate (f)chromium (III) oxide |
| Answer» Solution :(a) `Hg(II)CI_(2)(b) NI(II)SO_(4) (c ) Sn(iv)O_(2) (d)Th_(2)(I)SO_(4) (E )Fe_(2)(III)(SO_(4))_(3), (f) Cr_(2)(III)O_(3)` | |
| 35. |
What is maximum valency of the first order of elements in period ? |
|
Answer» Solution :Four. EXAMPLE : `[BF_(4)]^(-)` Because their valence ORBITALS are four. (one 2s and THREE 2P) |
|
| 36. |
Which changes observed with temperature to viscosity ? |
|
Answer» INCREASES |
|
| 37. |
Which of the following is the correct expression for the equation of state of van der Waals gas? |
|
Answer» <P>`(P+(a)/(N^(2)V^(2)))(V-nb)=NRT` |
|
| 38. |
The strenght of covalent bond will depend on the extent to which atomic orbitals____. |
|
Answer» |
|
| 39. |
The wavelength associated with a holf ball weighing 200 g and moving with a speed of 5km/h is of the order of : |
|
Answer» `10^(-10)m` |
|
| 40. |
The spontaneity means, having the potential to proceed without the assistance of external agency.The processes which occur spontaneously are |
|
Answer» flow of heat from COLDER to WARMER body |
|
| 41. |
Which of the following carbocation is more stable |
|
Answer» `OVERSET(+)(C)H_(3)` |
|
| 42. |
What is mother liquor? |
| Answer» Solution :For PURIFICATION of compound in crystalisation TECHNIQUE, the impure compound is soluble in suitable solvent and form HOT saturated solution. Then the filtration of solution and the REMAINING filtrate is known as MOTHER liquor. | |
| 43. |
Why are alkali metals not found in nature ? |
|
Answer» Solution :Alkali metals include lithium, SODIUM, potassium, rubidium, cesium, and FRANCIUM. These metals have only one electron in their valence shell, which they lose EASILY, OWING to their low ionization energies. Therefore, alkali metals are highly reactive andare not found in nature in their elemental state. |
|
| 44. |
Which step is chain propagation step in the following mechanism? (i) Cl_(2) overset(hv)to Cl^(**)+Cl^(**) (ii) Cl^(**)+CH_(4) to .^(**)CH_(3)+HCl (iii) Cl^(**)+Cl^(**) to Cl_(2) (iv) .^(**)CH_(3)+^(**)Cl to CH_(3)Cl |
| Answer» ANSWER :B | |
| 45. |
Which one of the following pairs of elements is called chemmical twins because of their very similar chemical properties |
|
Answer» MN and W |
|
| 46. |
Which one of the following is in solid state at room temperature? |
|
Answer» Bromine |
|
| 47. |
Whichoxide a carbon is an anhydrideof carbonic acid ? |
| Answer» Solution :SINCE CARBONIC acid decomposes to GIVE `CO_(2)` and `H_(2)O` , therefore, `CO_(2)` is regardedas an anhydride of carbonicacid: `H_(2)CO_(3) rarr H_(2)O + CO_(2)`. | |
| 48. |
The reactivity of the alkali metal sodium with water, is made use of |
|
Answer» in drying of alcohols |
|
| 49. |
The reactivity of IVA group element is highest with |
|
Answer» `F_(2)`<BR>`Cl_(2)` |
|








