Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is value of Z at 200 bar pressure for CO_(2), CH_(4) and O_(2) ? |
| Answer» Solution :This gas shows negative DEVIATION at 200 bat and `Z LT 1` but it shows positive deviation at 900 bat and `Z gt 1`. | |
| 2. |
Which one of the following is incorrect for ideal solutions? |
|
Answer» ` DELTA H_("MIX ") =0` |
|
| 3. |
What happened when chemical reaction occurs? |
| Answer» SOLUTION :(i) Bond FISSION and bond FORMATION. (ii) Displacement of electron. (iii) ENERGY change. (iv) Substract convert into product | |
| 4. |
Two gaseous samples were analysed. One contained 1.2 g of carbon and 3.2 g of oxygen. The other contained 27.3% carbon and 72.7% oxygen. The experiemental data is an accordance with |
|
Answer» LAW of CONSERVATION of mass |
|
| 5. |
Using Aufau priciple,write the ground state electronic configuration of following atoms.Boron(Z=5),(ii)Neon(Z=10),(iii)Aluminium(Z=14),(iv)Chlorine(Z=17),(v)calcium(Z=20),(vi)Rubidium(Z=37) |
|
Answer» Solution :(i) Boron(Z=5),`1s^(2)2s^(2)2p^(1)` (II) Neon(Z=10),`1s^(2)2s^(2)2p^(6)` (III)Almuminium(Z=13),`1s^(2)2s^(2)2p^(6)3S^(2)3p^(1)` (iv)Chlorine(Z=17),`1s^(2)2s^(2)2p^(6)3^(2)3p^(5)` (V)Cacium(Z=20),`1s^(2)2s^(2)2p^(6)3s^(2)3p^(6)4s^(2)` (vi)Rubidium(Z=37),`1s^(1)2s^(2)2p^(6)3p^(6)3d^(10)4s^(2)4p^(6)5s^(1)` |
|
| 6. |
Which is the number of Calcium in terms of elements are obtained from earth crust ? |
|
Answer» 4 |
|
| 7. |
What happens in real gas at high pressure and low temperature ? |
|
Answer» Solution :At high pressure, effective pressure change by `(an^(2))/(V^(2))` and deviation SHOWS between real gas and Ideal gas. At LOW pressure, SPEED of molecule decreases, distance, between molecule decrease, real VOLUME decreases to NB. It shows deviation between Ideal gas and real gas. |
|
| 8. |
Vinyl carbinol is |
|
Answer» `HO-CH_(2)CH=CH_(2)` 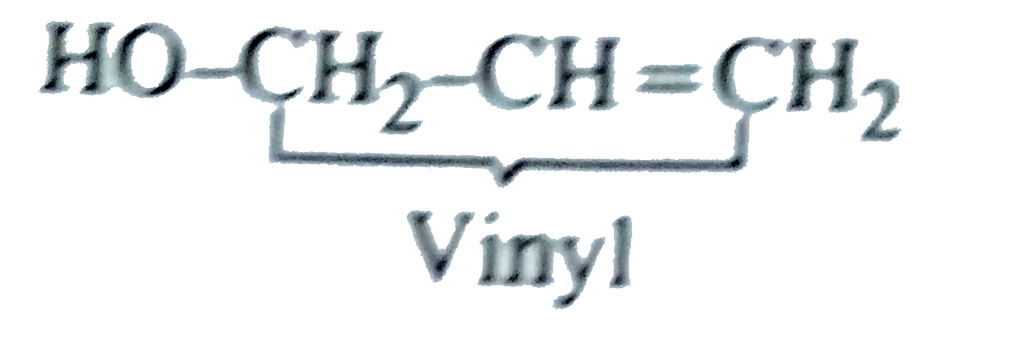 Methyl alcohol is called CARBINOL. The compound in QUESTION is called vinyl cabinol since it is a vinyl derivative of carbinol. |
|
| 9. |
Which of the following statement is incorrect ? |
|
Answer» `KO_(2)` and `KO_(3)` are paramagnetic species . |
|
| 10. |
What is the correct order for boiling point having same carbon in alkane, alkene and alkyne ? |
|
Answer» Alkane `gt` Alkene `gt` ALKYNE |
|
| 11. |
Which of the following sets of groups contains only electrophiles ? |
|
Answer» `NH_(2)^(-),NO_(2)^(+),H_(2)O,NH_(3)` |
|
| 13. |
When mercury is dropped over a glass surface the globules are spherical which is due to its |
|
Answer» VISCOSITY |
|
| 14. |
What are representative elements? |
|
Answer» |
|
| 15. |
The reagent NH_4Cl and aq. NH_3 will precipitate ions of group ….. |
| Answer» Answer :A | |
| 16. |
Whichof thefollowingstatements isINCORRECT ? |
|
Answer» Alkalimetalsformmonovalent`M^(+)` ion |
|
| 17. |
Which of the following statements about resonance is not correct? |
|
Answer» The differentresonance STRUCTURES of a molecule have fixed arrangement of atomic nuclei. |
|
| 18. |
Write the reaction of preparation of ethane from following reaction. (i) Ethane (ii) Ethyl chloride (iii) Bromo-methane (iv) Propanoic acid (v) Acetic acid (vi) Methane (vii) from acetate ion. |
|
Answer» Solution :(i) `underset("Ethene")(CH_(2)=CH_(2)) underset("Pt/Pd/Ni")overset(+H_(2), "Dihydrogenation")rarr CH_(3)-CH_(3)` (II) `underset("Ethyl chloride")(CH_(3)CH_(2)CL) underset(-HCL)overset(+H_(2), Zn, H^(+) " Reduction")rarr CH_(3)-CH_(3)` (iii) `underset("BROMO")(2CH_(3)Br) underset(-2NaBr)overset("2Na, dry ETHER Wurtz reaction")rarr CH_(3)-CH_(3)` (iv)  (v)  (vi) `2CH_(4) underset(-HCl, +Cl)overset(hv)rarr 2CH_(3) underset("combustion")overset("free radical")rarr CH_(3)-CH_(3)-` (vii) 
|
|
| 19. |
Which one has least energy in given molecular orbitals ? |
| Answer» Solution :`sigma_(1s)` | |
| 20. |
Total number of lone pair of electrons in I_3^-ion is : |
|
Answer» 3 Charge is -1 , so ACCEPTED `e^-` = 1  ` therefore `Total valence ELECTRONS = 22 = 9 Lone pair electrons |
|
| 21. |
When zeolite , which is hydrated sodium aluminium silicate is treated with hard water , the sodium ions are exchanged with which of the following ion(s) ? |
|
Answer» `H^(+)` ions |
|
| 22. |
What order or C-H bond lengths do you expect in C_(2) H_(6), C_(2) H_(4) and C_(2) H_(2) and why ? |
|
Answer» Solution :` C - H (C_(2) H_(6)) GTC - H (C_(2) H_(4)) gt C - H(C_(2) H_(2)) ` . This is because hybridorbrid ordbitals of carbon involved in overlapping with 1s ORBITAL of hydrogen are `SP^(3) gt sp^(2) gt sp` and ap respecitvely and their sizes are in the order ` sp^(3) gt sp^(2) gt sp ` . |
|
| 23. |
Weak acids and bases are not completely ionised when dissolved in polar medium like water H rarr H^(+) +A^(-) {:(t_(0),C,O,O),(t_(eq),C-Calpha,Calpha,Calpha):} K_(a)=(Calpha^(2))/(1-alpha)=Calpha^(2),alphasqrt((K_(a))/(C)) :. (alpha_(1))/(alpha_(2))=sqrt((Ka_(1))/(Ka_(2))),(alpha_(1))/(alpha_(2))=sqrt((C_(2))/(C_(1))) ( For two acids at same conc.) (for same acid at diff conc. ) 0.01M CH_(3),COOH is 4.24% ionised. What will be the pereentage ionisation of 0.1 M CH_(3),COOH. |
|
Answer» `1.33%` ` (18)/(alpha_2^(2))=10 , alpha_2^(2)= 1.8 rArr ALPHA =sqrt(1.8)=1.34%` |
|
| 24. |
Weak acids and bases are not completely ionised when dissolved in polar medium like water H rarr H^(+) +A^(-) {:(t_(0),C,O,O),(t_(eq),C-Calpha,Calpha,Calpha):} K_(a)=(Calpha^(2))/(1-aplha)=Calpha^(2),alphasqrt((K_(a))/(C)) :. (alpha_(1))/(alpha_(2))=sqrt((Ka_(1))/(Ka_(2))),(alpha_(1))/(alpha_(2))=sqrt((C_(2))/(C_(1))) ( For two acids at same conc.) (for same acid at diff conc. ) Relative strength of two weak monoprotic acids may be given as |
|
Answer» ` ([H^(+)])/([H^(+)]_2) ` |
|
| 25. |
Weak acids and bases are not completely ionised when dissolved in polar medium like water H rarr H^(+) +A^(-) {:(t_(0),C,O,O),(t_(eq),C-Calpha,Calpha,Calpha):} K_(a)=(C alpha^(2))/(1-alpha)=Calpha^(2),alphasqrt((K_(a))/(C)) :. (alpha_(1))/(alpha_(2))=sqrt((Ka_(1))/(Ka_(2))),(alpha_(1))/(alpha_(2))=sqrt((C_(2))/(C_(1))) ( For two acids at same conc.) (for same acid at diff conc. ) alphaand alpha are in the ratio 1:2 at same conc. Ka_(1), =2xx 10^(-4), what will be Ka_(2),? |
|
Answer» `8xx10 ^(-4)` |
|
| 26. |
Which of the following is arranged in order of increasing radius? |
|
Answer» `K_((AQ))^(+)ltNa_((aq))^(+)ltLi_((aq))^(+)` |
|
| 28. |
Write short notes on the following: (i) Vital force theory. (ii) General characteristics of organic compounds. (iii) Hybridization. (iv) Polar and non-polar bonds. (v) Hydrogen bonding. (vi) Resonance. (vii) Fractional crystallisation. (viii) Vacuum distillation. (ix) Mixed melting point. (x) Purity of an organic compound. |
| Answer» | |
| 29. |
What is the amount of work done when 0.5 mol of methane, CH_(4)(g) is subjected to combustion at 300K? (given, R = 8.314 JK^(-1)mol^(-1)): |
|
Answer» `-2494` J Work done,`W = -p DeltaV = -Deltan_(g) RT` `Deltan_(g)= 1 -3 = -2, T = 300K` `w = -(-2 xx 8.314 xx 300)` = 4988.4 J For 0.5 mol of `CH_(4)` ` w = (4988.4)/(2) = 2494.2` |
|
| 30. |
Wutrz reaction used in the preparationof ………………. |
| Answer» SOLUTION :higheralkanes | |
| 32. |
Which one of the following compound is a gas (at 0^(@)C) |
|
Answer» `BF_(3)` |
|
| 33. |
Write the descending order of electrons releasing tendensies of the Zn, Cu and Ag metals. Arrange the metals Zn, Cu and Ag in the descending order of their effective nuclear charge, |
| Answer» SOLUTION :Zinc, COPPER and SILVER the electro RELEASING thendency is the following order. | |
| 34. |
The volume strength of 1.5 N H_2O_2 solution is ….. |
|
Answer» 4.8 |
|
| 35. |
Which of the following is not aromatic ? |
|
Answer» Benzene |
|
| 36. |
When calamine is heated then a product (A) is formed then find the total number of correct options for compound (A): (a) Compound (A) is white in cold conditions. (b) Compound (A) is yellow in hot conditions. (c) Compound (A) is called philosopher's wool. (d) Compound (A) when combined with CoO, then compound (B) is formed and colour of new compound (B) is green (e) Compound (B) is called Rinmann's green |
|
Answer» |
|
| 37. |
When Benzoic acid is treated with LiAlH_(4) it for |
|
Answer» Benzaldehyde |
|
| 38. |
What is gram equivalent weight of hydrogen peroxide as reductant? |
|
Answer» Solution :As reductant hydrogen perioxide oxidises to OXYGEN. `H_(2)O_(2) to O_(2)` The CHANGE in oxidation number is 2 The molar mass =34G GRAM equivalent weight of `H_(2)O_(2)=(34)/(2)=17g` Equivalent weight =152 |
|
| 39. |
What makes lithium to show properties uncommon to the rest of the alkali metals ? |
| Answer» Solution :The unusual PROPERTIESOF lithium as compared to other ALKALI metals is due to (i) the exceptionally small size of its atom and ion, (ii) HIGH polarising power (charge/size ratio) , (ii) high ionization ENTHALPY and (iv) absence of d-orbitals . | |
| 40. |
Which one of the following is an endothermic process ? |
|
Answer» `CH_4 + 2O_2 to CO_2 + 2H_2 O` |
|
| 41. |
The root mean square velocity of a gas is double when temperature is |
|
Answer» increased four times `(V_(rms))/(V'_(rms))=sqrt((T)/(T'))rArr(1)/(2)=sqrt((T)/(T'))rArrT'=4T` |
|
| 42. |
What is not correct for SO_(2(g)) ? |
|
Answer» It is ANGULAR in shape |
|
| 43. |
Which one has highest boiling point ? |
|
Answer» Butan-2-ol |
|
| 44. |
What is reversible process? Give an example. |
| Answer» Solution :The PROCESS in which the system and SURROUNDINGS cannot be RESTORED to the initial STATE from the final state is called a REVERSIBLE process eg. All the processes occurring in nature are irreversible processes. | |
| 45. |
Which will have a higher value of electronegativity for fluorine, the pauling scale or mulliken scale? |
|
Answer» |
|
| 46. |
The volume of a given mass of gas is 0.6dm^3. If its pressure is increased by 3 times and absolute temperature is increased by 5 times, what would be its new volume? |
| Answer» SOLUTION : 1 `DM^(3)` | |
| 47. |
Which of the main functional group in the following ? (a) CH_(2)OH -CH_(2)-CHF-CH_(2)COOH (b) (CH_(3))_(2) CHCOCOOH (c ) CH_(3)CH_(2) underset(underset(NH_(2))(|))(C )HOH (d) CH_(3)COCH_(2)CH_(2)OH (e ) CH_(2)CH(NH_(2))COOH (f) CHOCH_(2)COCH_(2)CH_(3) |
| Answer» Solution :(a) `-COOH` (B) `-COOH` (c ) `-NH_(2)` (d) `-CO-` (E ) `-COOH` (f) `-CHO` | |
| 48. |
Which of the following decarboxylates readily in the temperature range 100 - 150^(@)C ? |
|
Answer» `CH_(2) = CH.CH_(2)COOH` |
|
| 49. |
Which of the following decolourises bromine water and does not give white precipitate with AgNO_(3)? |
|
Answer» `C_(6)H_(6)` |
|
| 50. |
Which bond is more polar in the following pairs of molecules: (i) H_(3)C- H, H_(3)C- Cl (ii) H_(3)C- OH, H_(3)C-NH_(2) (iii) H_(3)C- SH, H_(3)C-OH |
| Answer» SOLUTION :(i) `C-Cl` since Cl is more ELECTRONEGATIVE than H (ii) `C-O` (iii) `C-O` | |