Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which one of the following gives red colour in flame test? |
|
Answer» LITHIUM |
|
| 2. |
Which of them will not show geometrical isomerism? |
|
Answer»
|
|
| 3. |
What will be effect on bond length as bond multiplicity increases ? |
| Answer» Solution :As the BOND order between TWO CARBON INCREASES, bond LENGTH decreases. | |
| 4. |
Two similar reactions are given below : H_(2)(g) + (1)/(2)O_(2)(g) rarr H_(2)O(g), DeltaH = = DeltaH_(1) H_(2)(g)+(1)/(2) O_(2)(g) rarr H_(2)O(l), DeltaH= DeltaH_(2) In terms of magnitude of DeltaH, |
|
Answer» `DeltaH_(1) gt DeltaH_(2)` |
|
| 5. |
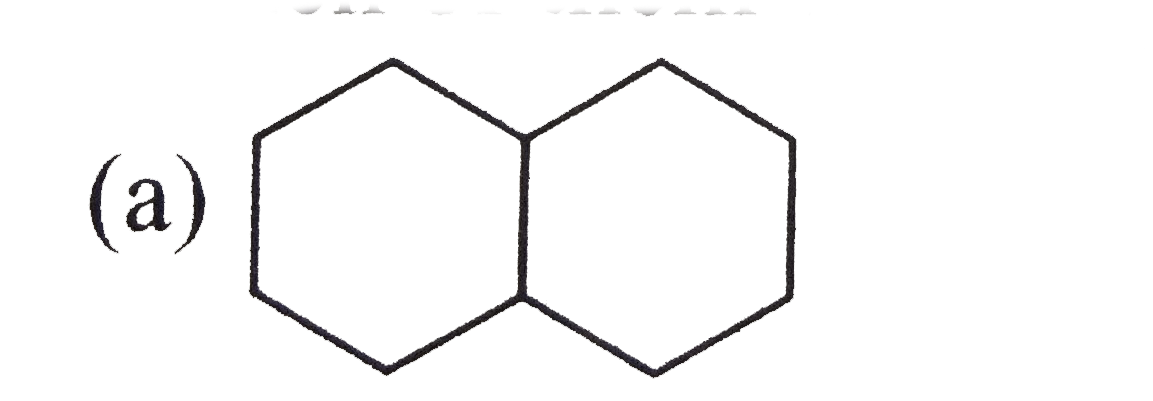
Which of the following compounds will react with Na to form 4,5-diethyloctane? |
|
Answer» `CH_(3)CH_(2)CH_(2)CH_(2)Br` 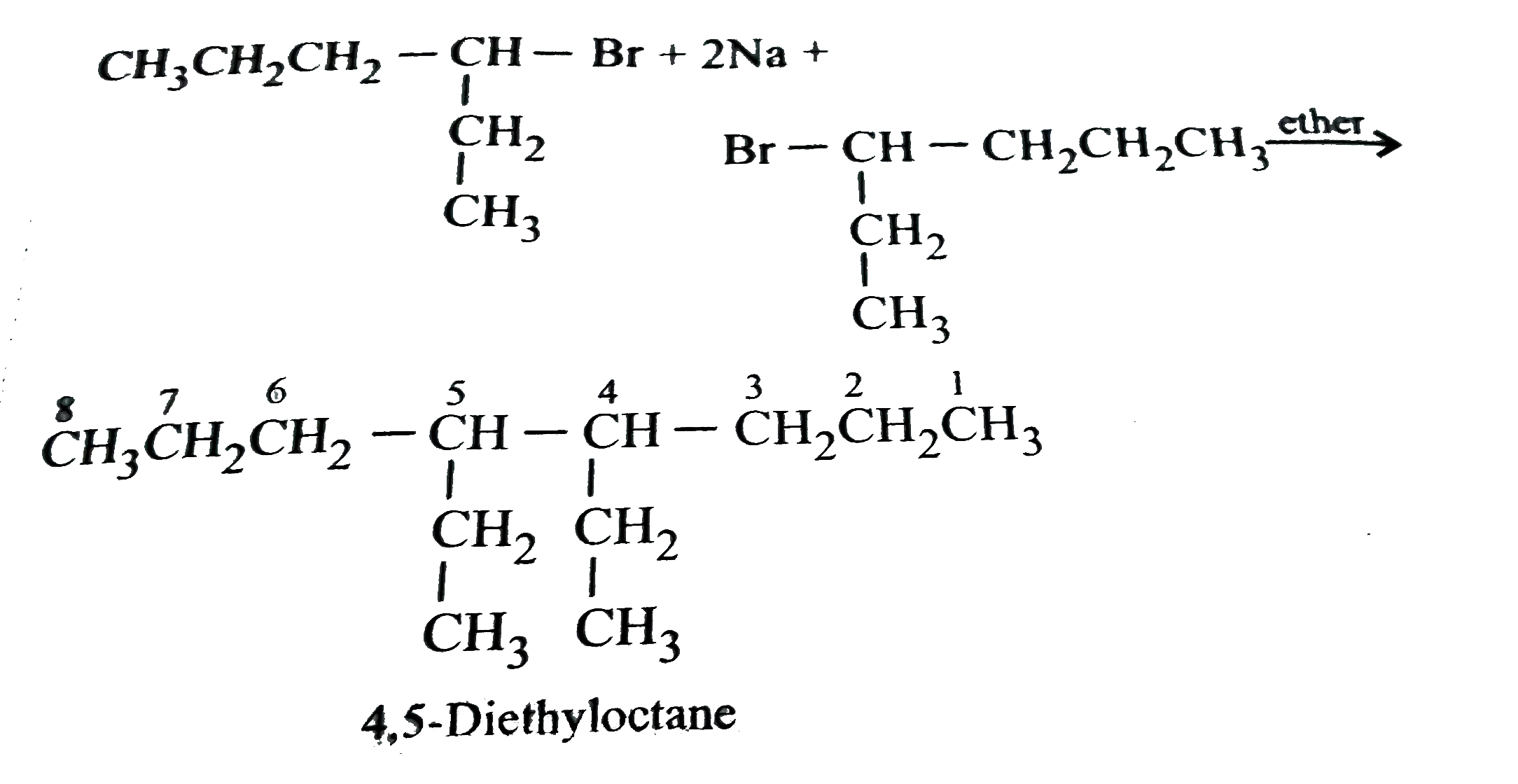
|
|
| 6. |
Write formula for the following compound: Thallium (I) sulphate |
| Answer» SOLUTION :`Tl_2` (I) `SO_4` | |
| 7. |
When acidified dichromate is treated with H_2O_2, a blue coloured compound is formed. It is stabilised by adding |
| Answer» Answer :A | |
| 9. |
whichof thefollowingelementformscompounds that arepartlyionicandpartlycovalent? |
|
Answer» CA |
|
| 10. |
Which of the following has directional character? |
|
Answer» IONIC bond |
|
| 12. |
Two different gases 'A' and 'B' are filled in separate containers of equal capacity under the same conditions of temperature and pressure. On increasing the pressure slightly the gas 'A' liquefies but gas B does not liquefy even on applying high pressure until it is cooled. Explain this phenomenon. |
| Answer» SOLUTION :GAS A is below its critical TEMPERATURE WHEREAS gas B is above its critical temperature. | |
| 13. |
Write note about The International system of units (SI). |
|
Answer» The CGPM is an intergovernmental treaty organizationcreated by a DIPLOMATIC treaty known as Metre Convention which was signed in Paris in 1875. The SI system has seven base units and they are listed in Table. These units pertain to the seven fundamental scientific quantities. The other physical quantities such as speed, volume, density etc. can be DERIVED from these quantities. The definitions of the SI base units are given in Table. The SI system allows the use of prefixes to indicate the multiples for submultiples of a unit.   
|
|
| 14. |
Which acts as autocatalyst during titration of KMnO_(4) and oxalic acid in presence of H_(2)SO_(4)? |
| Answer» Answer :D | |
| 15. |
What happened if we mix oil in H_(2)O ? Why ? |
| Answer» SOLUTION :SPREADING oil on the WATER and form UPPER layer as surface TENSION of `H_(2)O` is more than oil. | |
| 16. |
Zn_((s)) + Cu_((aq))^(2+) to Zn_((aq))^(2+) + Cu_((s)) . In this reaction, which gets oxidised ? |
|
Answer» `Cu^(2+)` (LOSS of electron = oxidation) Zn gets oxidised |
|
| 17. |
The temperature at which real gases obey the ideal gas laws over a wide range of low pressure is called: |
|
Answer» CRITICAL temperature At low pressure `Z=1+(b-(a)/(RT))(1)/(V_(m))` At `T_(B)=a//Rb, " "Z=1` because `TgtT_(i)` and if gas expanded adiabatically then `Tuarr` |
|
| 18. |
Which of the following on heating with aq KOH produces butanaldehyde |
|
Answer» `CH_(3)CH_(2)CH_(2)CH_(2)Cl` |
|
| 19. |
The r.m.s. velocity of molecules of a gas of density 4 kg m^(-3) and pressure 1.2xx10^(5)Nm^(-2) is |
| Answer» Answer :A::C::D | |
| 20. |
Which of the following is not a characteristic of a crystalline solid ? |
|
Answer» Definite and CHARACTERISTIC HEAT of fusion |
|
| 21. |
Which of the following will be most reactive in the addition reaction with HBr? |
|
Answer» `CH_3 -C -=C - CH_3` |
|
| 22. |
Which one of the following statements is not correct? |
|
Answer» The kinetic energy of a gas is inversely proportional to temperature (ink). |
|
| 23. |
Which of the following reactions is an example of a redox reaction - |
|
Answer» `XeF_(6)+H_(2)OtoXeOF_(4)+2HF` `overset(6-1)(XeF_(6))+2H_(2)overset(-2)Otooverset(+6-2-1)(XeO_(2)F_(2))+4HF` `overset(+2-1)(XeF_(2))+overset(+5-1)(PF_(5))tooverset(+2-1)([XeF])overset(+5-1)([PF_(6)])` For the theses reactions ,there is no CHANGE in oxidation NUMBER of the respective elements .So these reactions are not reaction. `overset(+4-1)(XeF_(4))+overset(+4-1)(O_(2)F_(4))tooverset(+6-2)(XeF_(6))+overset(0)O_(2)`This is redox reaction because oxidation number of Xe increases and that of O decreases. |
|
| 24. |
The wave number of the firstline of Balmer series ofhydrogen is 15200 cm ^(-1) The wave number of the first Balmer line of Li^(2+) ion is |
|
Answer» `15200 CM^(-1)` |
|
| 25. |
Two samples of gases 'a' and 'b' are at the same temperature. The molecules of 'a' are travelling 4 times faster than molecules of 'b'. The ratio of M_a//M_b will be |
|
Answer» `1//4` |
|
| 28. |
Which among the following indicates change in the chemical composition due to dissociation |
|
Answer»
|
|
| 29. |
Which one is the correct order of the size of the iodine species? |
|
Answer» `I GT I^(+) gt I^(-)` |
|
| 30. |
Which of the following reactions requires high temperature and catalyst? |
|
Answer» `H_2 + F_2 to 2HF` |
|
| 31. |
The van der Waals' coefficient of the inert gases He, Ar and Xe are given below : {:("Inert Gas",a ("atm "dm^(6) mol^(-2)),b (10^(-2) dm^(3) mol^(-1)),),(""He,""0.34,""2.38,),(""Ar,""1.337,""3.20,),(""Xe,""4.137,""5.16,):} Choose the appropriate pair to complete the following statement, ''The increase in the value of 'a' signifies the increasing importance of .... interaction while increase in the value of 'b' is due to....'' |
|
Answer» Ion-ion, increased atomic VOLUME |
|
| 32. |
Which of the following acid is important for digestion in human stomach ? |
|
Answer» `CH_3COOH` |
|
| 33. |
What are the products formed when Li_(2)CO_(3) undergoes decomposition? |
|
Answer» `Li_(2)O+CO` |
|
| 34. |
When electron jumps from 5^(th) energy level to 1^(st) energy level, to which series the spectral line belongs? |
|
Answer» Balmer |
|
| 35. |
What is the concentration of sugar (C_(12)H_(22)O_(11)) in mol L^(–1) if its 20 g are dissolved in enough water to make a final volume up to 2L? |
|
Answer» Solution :Molar mass of sugar `=(12 xx 12) + (1 xx 22) + (11 xx 16) = 342 g mol^(-1)` Number of moles in 20 g of sugar `=20/342= 0.0585` `THEREFORE` Molar concentration of sugar solution `=("Number of moles of sugar DISSOLVED")/("Volume of solution in litres") = 0.0585/2 = 0.02925 mol L^(-1)` |
|
| 36. |
Which of the following is correct order for ionization enthalpy of group 13 ? |
|
Answer» `B gt Al gt Ga gt In gt TL` |
|
| 37. |
Toluene reacts with halogen in presence of iron (III) chloride giving ortho- and para-halo-compounds. The reaction is |
|
Answer» <P>free RADICAL addition reaction |
|
| 38. |
write the structural formula for each of the following a. A 3^(@) amine with the formula C_(3) H_(9) N. b. Three ethers with the formula C_(4) H_(10) O. c. A 3^(@) alcohol with the formula C_(4) H_(8) O. d. Three 2^(@) alky1 halides with the formula C_(5) H_(11) C1. e. Three 1^(@) alcohols with the formula C_(4) H_(8) O. f. A 2^(@) wiht the foumula C_(3) H_(6) O. g. Two esters with the formula C_(3) H_(6) O. g. Two esters with the formula C_(3) H_(6) O_(2). h. Four 1^(@) alky1 halides with the formula C_(5) H_(11) C1. i. A 3^(@) alky1 halide with the formula C_(5) H_(11) CI. j. Three aldehydes with the formula C_(5) H_(10) O. k. Three ketones with the formula C_(5) H_(10) O. l. A 2^(@) amine with the formula C_(3) H_(9) N. m. Two amides with the formula C_(2) H_(5) NO. n. Two 1^(@) amines with the formula C_(3) H_(9) N. |
|
Answer» Solution :a. `(CH_(3))_(3) N` b. 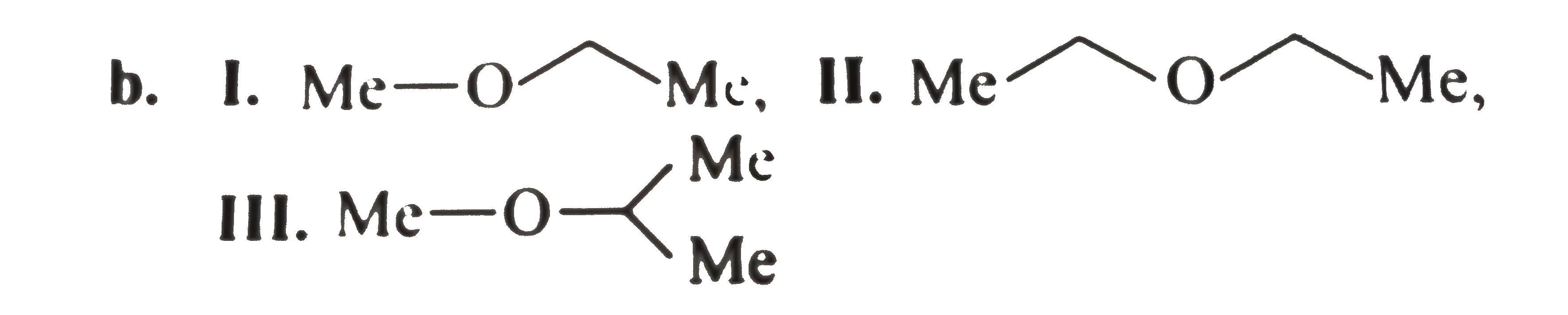 C. Degree unsaturation: `((2 n_(C ) + 2) - n_(H))/(2) = (10 - 8)/(2) = 1^(@)` `1^(@) D.U`. Suggests either unsturated alcohol or cyclic `3^(@)` alcohol four `C` atoms, `3^(@)` alcohol cannot be unsturated, so it is cyclic 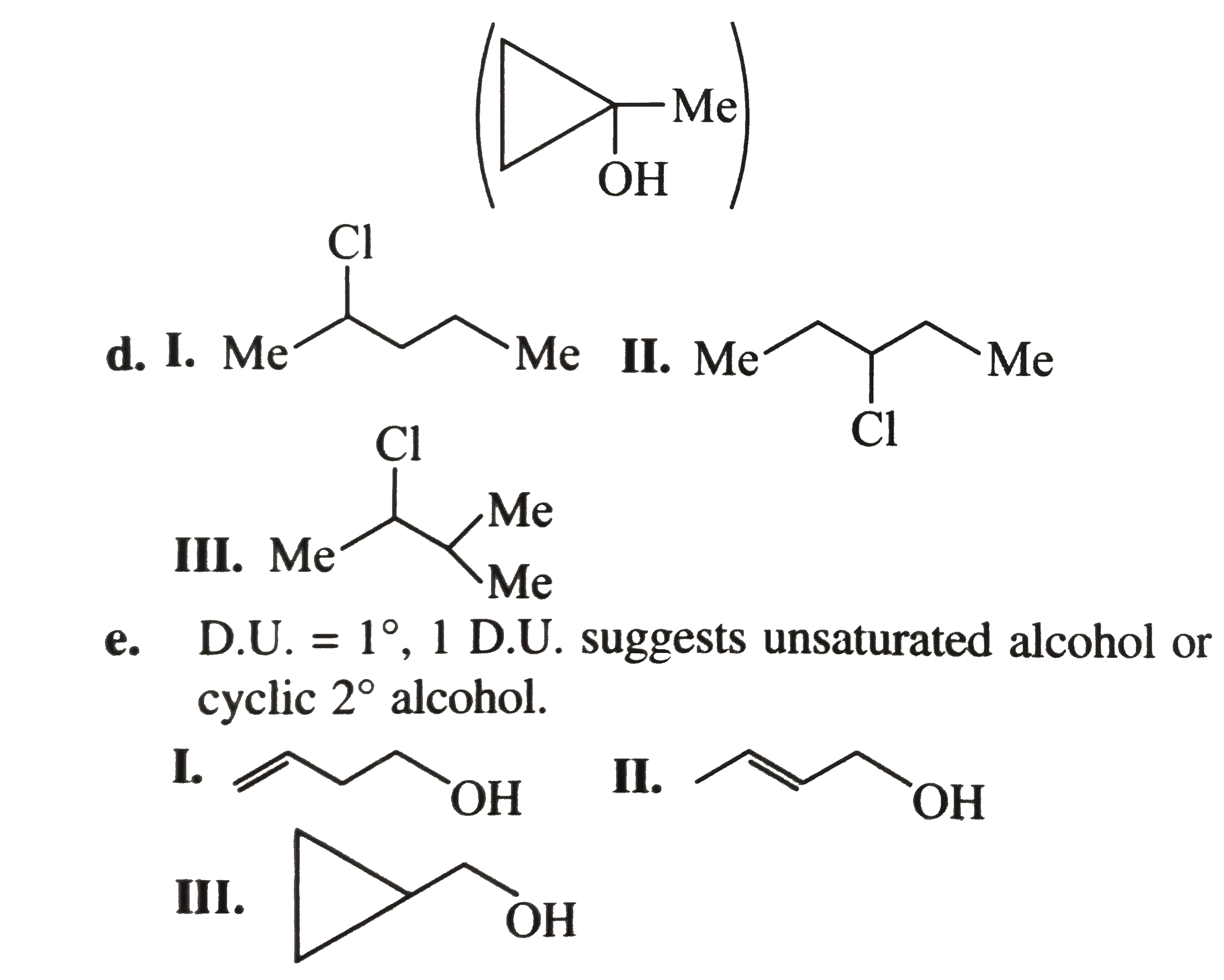 F. `D.U. = 1^(@). 1D.U.` suggests UNSATURATED alcohol or cyclic `2^(@)` alchohol THREE `C` , `2^(@)` alcohol cannot be unsaturated, so ITIS cyclic. 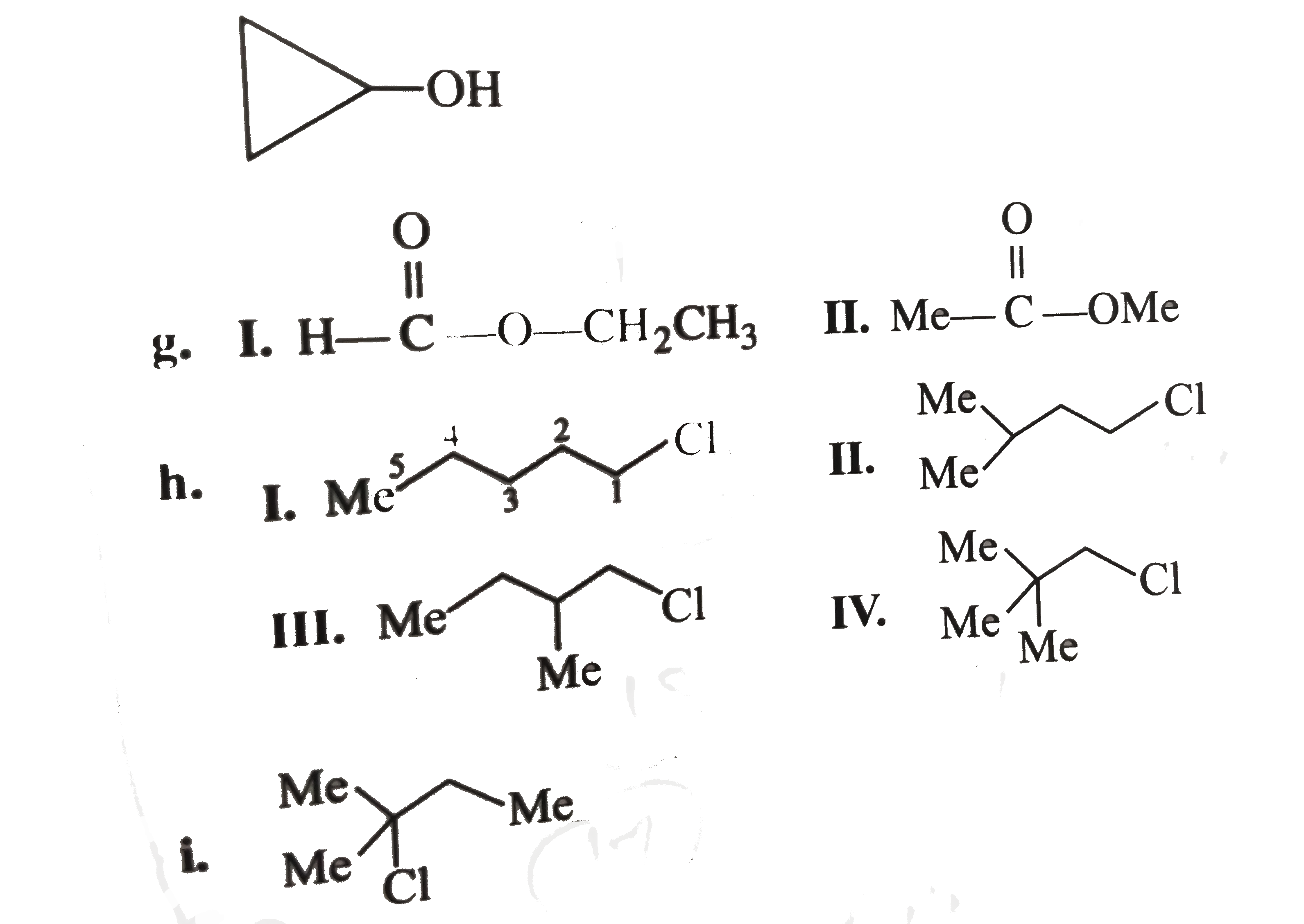 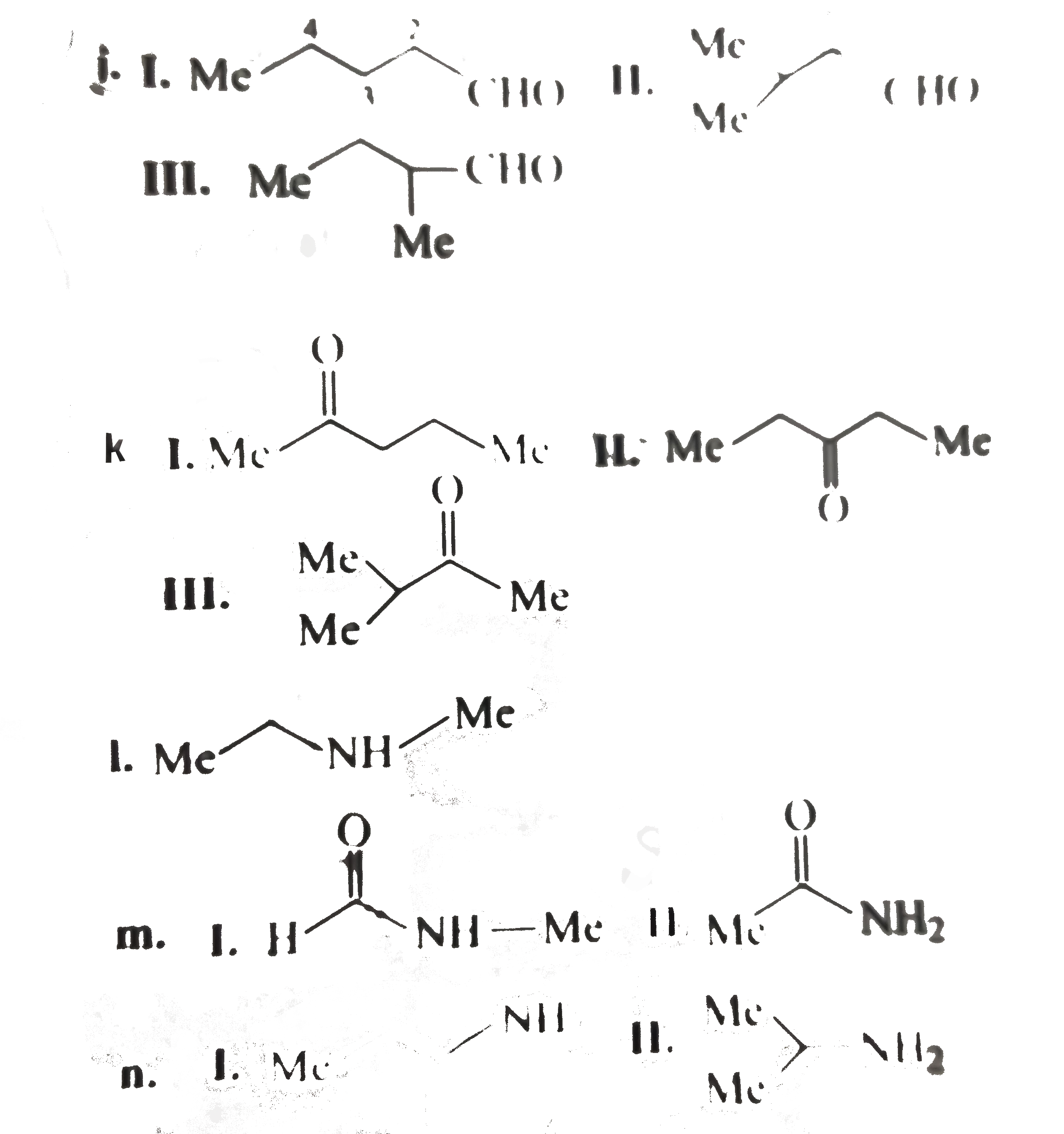
|
|
| 39. |
Which of the following is not a transuranic element ? |
|
Answer» Pu |
|
| 40. |
What is redox couple ? |
| Answer» Solution :A redox couple is defined as having together the oxidised and reduced form of a substance taking PART in an oxidation or REDUCTION HALF reaction. | |
| 41. |
The weight of 350 mL of a diatomic gas at 0°C and 2 arm pressure is 1 g. The wt. of one atom is |
| Answer» Answer :A | |
| 42. |
Which free radicals are obtained on anode and cathode during electrolysis of RCOO^(-) Na^(+) in Kolbe's electrolysis ? |
| Answer» Solution :They PRODUCE FREE RADICALS LIKE `R∙` and `H∙` respectively. | |
| 43. |
There is a colllection of crystalline substances in a hexagonal close packing. If the density of matter is 2.6 g//cm^3, what would be the average density of matter in collection ? What fraction of the space is actually unoccupied ? |
|
Answer» SOLUTION :In HEXAGONAL CLOSE packing , packing efficiency is 74 % `therefore` Density of matter = Packing fraction x Total density =`74/100xx2.6=1.924 "g/cm"^3` % empty SPACE (space unoccupied ) =100-74=26% |
|
| 44. |
Water softening by Clarke's process used: |
|
Answer» `NaHCO_(3)` |
|
| 45. |
What is the state of hybridisation of carbon inCO_3^(2-) |
| Answer» SOLUTION :`CO_3^(2-)` : C in `CO_3^(2-)`is `sp^2` hybridised and is BONDED to three oxygen atoms. | |
| 46. |
Which of the following compound/s are carcinogenic? |
|
Answer» 1,2-Benzanthracene |
|
| 47. |
The solubility product of a springly soluble metal hydroxide [M(OH)_2] is 5xx10^(-16)mol ^(3) L^(-3)at 298 K.Find the pH of its saturated aqucous solutions. |
|
Answer» ` 4S^(3)= 5 xx 10^(-16) rArrS = 5 xx 10 ^(-6)` ` [OH^(-) ] =2S =10 ^(-5), POH =5, PH = 9` |
|
| 48. |
The state of hybridisation of the central atom and the number of lone pairs over the central atom in POCl_3 are |
|
Answer» `SP,0` 
|
|
| 49. |
Write the structure and names of products obtained in there actions of sodium with a mixture of 1-iodo-2-methylpropane and 2-iodopropane. |
|
Answer» Solution :Products obtained are based on Wurtz reaction, n-alkali halide on treatment with Name talinpresence of ether gives alkane having double carbon atoms. `RX + 2NA + XR RARR R-R + 2NaX` `CH_(3) - CH(CH_(3))-CH_(2)I + 2Na + ICH_(2) - CH(CH_(3))-CH_(3) OVERSET("DRY ether")rarr` `UNDERSET("(2,5-dimethylhexane)")(2NaI + CH_(3) - CH(CH_(3)))-CH_(2)-CH_(2)-CH(CH_(3))-CH_(3)` `CH_(3) - CH(CH_(3))I + 2NA + I(CH_(3))CH-CH_(3) rarr underset("(2,4-dimethylbutane)")(2NaI + CH_(3)-CH(CH_(3)))-HC(CH_(3))-CH_(3)` `CH_(3)-CH(CH_(3))-CH2I+2Na+I-CH(CH_(3))-CH_(3) rarr underset("(2,4-dimethylpentane)")(2NaICH_(3)-CH(CH_(3)))-CH-CH_(3)-CH_(3)` |
|
| 50. |
The tatio of close packed atoms to tetrahedral holes in cubic close packingis |
|
Answer» `1:1` In CCP lattice atoms =`8 times1/8+6 times1/2=4 `therefore" Tetrahedral voids "= 4 times2=8` `" Ratio "4:8 i.e. 1:2`. |
|