Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is the correct order of ionisation energy |
|
Answer» `K LT CU lt Cu^(+)lt K^(+)` |
|
| 2. |
Which fungicide decomposed in soil and their products are harmful ? |
| Answer» Solution :Mercuric compounds | |
| 3. |
Whichtypeofdimensionrequireforpositionof electron ? |
| Answer» SOLUTION :Thedimensionof measurementmustbe LESSTHAN thedimensionelectron. | |
| 4. |
What is a redox reaction give one example |
| Answer» SOLUTION :OXIDATION REDUCTION rections are called redox reaction `Mg+F_(2)rarrMg^(2+)(F)_(2)` here Mg is oxidised to `Mg^(+)` while `F_(2)` is reduced to `F^(-)` IONS | |
| 5. |
Among the following what is necessary condition for spontanity of a process? |
| Answer» Answer :D | |
| 6. |
Which of the following is a correct statement ? |
|
Answer» HF is a STRONG acid than HCl in aqueous medium. |
|
| 7. |
Which of the following is valid at absolute zero? |
|
Answer» KINETIC energy of the gas becomes ZERO but the MOLECULAR motion does not become zero |
|
| 8. |
Vapour phase diagram for a solution is given below if doted line represents deviation? (##SUR_CHE_XI_V02_C09_E02_044_Q01.png" width="80%"> Correct observation for this solution |
|
Answer» ` DELTA H_(MIX) : + ve` |
|
| 9. |
Which of the following is the weakest acid? |
|
Answer»

|
|
| 10. |
The unparied electrons in Al and Si are in 3p orbital. Which electrons will experience more effective nuclear charge from the nucleus? |
| Answer» Solution :Since the ATOMIC number of Si is more than that of Al, the unpaired ELECTRONS of Si will EXPERIENCE more EFFECTIVE nuclear charge. | |
| 11. |
Which of the followings point defects are shown by AgBr(s) crystals ? (A) schottky defect (B) Metal excess defect © Metal deficiency defect (d)Metal deficiency defect |
|
Answer» (A) and (B) |
|
| 12. |
Which is the correct order of ionic sizes (At, No : Ce=58,Sn=50,Yb=70 and Lu=71)? |
|
Answer» `CegtSngtYbgtLu` |
|
| 14. |
Which of the following orbitals will have zero probability of finding the electron in the yz plane |
|
Answer» `P_x` |
|
| 15. |
Whichof the followingis optically active |
|
Answer» 2-methylpentane |
|
| 16. |
White phosphours reacts with chlorine and the product hydrolyses in the presence of water. Calculate the mass of HClobtained by the hydrolysis of the product formed by the reaction of 62 g of white phosphorus with chlorine in the presence of water. |
|
Answer» Solution :The CHEMICAL EQUATIONS for the reactions in involved are : `{:(,P_(4)+6Cl_(2) to 4PCl_(3)),(,underline(PCl_(3) + 3H_(2)O to H_(3) PO_(3)+3HCl]xx4),(,underset(4xx31=124 G)(P_(4))+ 6Cl_(2)+12H_(2)O to 4H_(3)PO_(3)+underset (12 xx 36.5g)(12HCl)):}` Now, 124 g of white P give `HCl=12xx36.5g` `:.` 62 g of white P will give `HCl=(12xx36.5)/(124)xx62=219g` |
|
| 17. |
Using curved arrow notation show the formation of reaction intermediates when the following covalent bonds undergo heterolysis. (i) H_(3)C-CH_(2)-Cl""(ii) H_(3)C-C equiv N""(iii) CH_(3)ONa |
Answer» SOLUTION :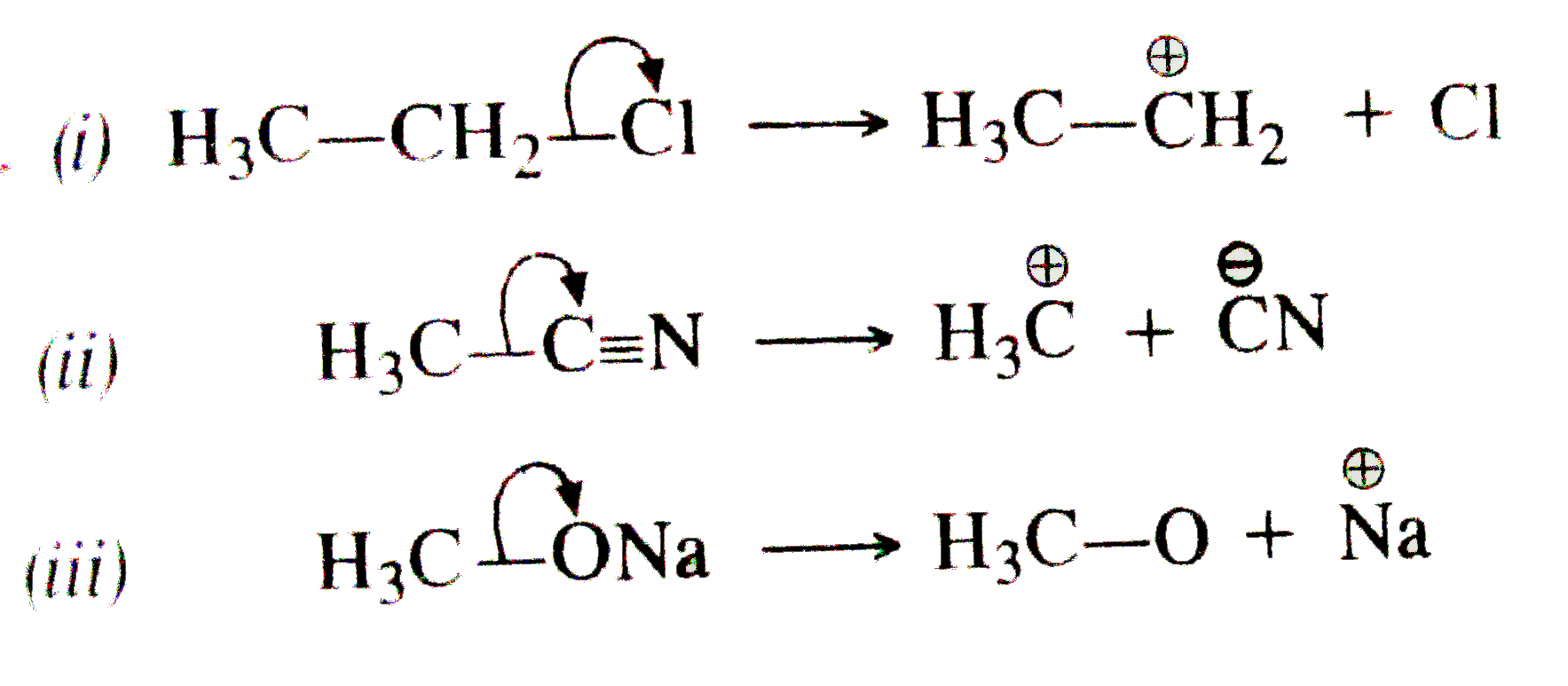
|
|
| 18. |
Which of the following incorrect statements for physiosorption? |
|
Answer» It is a reversible process |
|
| 19. |
What will be the minimum pressure required to compress 500 dm^(3) of air at 1 bar to 200 dm^(3) at 30^(@)C ? |
|
Answer» Solution :At CONSTANT temperature, VOLUME of GAS for fixed volume pressure will be calculated by Boyle.s formula. At `30^(@)C` Boyle.s equaltion, `p_(1)V_(1)=p_(2)V_(2)` where, `p_(1)=` Initial pressure = 1 bar `V_(1)=` Initial volume `= 500 dm^(3)` `V_(2)=` Final volume `= 200 dm^(3)` `p_(2)=` Final pressure = (?) `therefore p_(2) =(p_(1)V_(1))/(V_(2))` `= (("1 bar")xx(500 dm^(3)))/(200 dm^(3))=2.5` bar Minimum pressure to be KEPT is 2.5 bar. |
|
| 20. |
Which of the following indicates correct R (Gas constant) values in different units ? a) 1.987 "cals mole"^(-1)K^(-1) b) 8.314 xx 10^7 J "mole"^(-1)K^(-1) c) 0.0821 "lit-atm mole"^(-1) K^(-1) d) 82.1 "lit. mole"^(-1)K^(-1) |
| Answer» Answer :C | |
| 21. |
What is the oxidation state ofP in H_3PO_4 |
|
Answer» Solution :Oxidation NUMBER of P in `H_3PO_4` 3(+1)+x+4(-2)=0 +3+x-8=0 x=+5 Oxidation number of P in `H_3PO_4 =+5`. |
|
| 22. |
Which of the following will undergo reaction with Alcoholic Choloroform |
|
Answer»
|
|
| 23. |
Write notes about great London smog |
|
Answer» Solution :(i) The great smog of London (1952) was an instance of severe air pollution that atfected the London from 5TH December 9th of December, 1952 and then DISPERSED quickly when the whether changed. (ii) It causes major disruption by reducing the VISIBILITY and even penetrating indoor areas. (III) Government medical reports estimated that 4000 people had died as a direct result of smog and 100,000 were made ill by the smog effect on their respiratory tract. |
|
| 24. |
What is the atomic number of element whose outer most electrons are represented by 2p^3 |
| Answer» Solution :ELECTRONIC configuration is `1S^(2)2s^(2)2p^(3)therefore` Atomic NUMBER = 7 | |
| 25. |
Which of the following requires six times their volume of oxygen for complete combustion? |
|
Answer» n-Butane `(x+y)/(4)=6` `x=4implies(y)/(4)=2impliesy=8` |
|
| 26. |
The velocity of e^(-) in a certain Bohr orbitof the hydrogen atom bears the ratio 1:275 to the velocity of light. What is the quantum no. "n" of the orbit and the wave no. of the radiation emitted for the transition form the quantum state (n+1) to the ground state. |
|
Answer» `(2.18xx10^(6))/(n)=(1)/(275)` `(2.18xx10^(6))/(n)=(1)/(3XX10^(8))=(1)/(275)` `(2.18)/(n(300))=(1)/(275)""(1)/(n)=(300)/(599.5)` `n=(599.5)/(300)=(1)/(275)""(1)/(n)=(300)/(599.5)` `n=1.99~=2` |
|
| 27. |
The splitting of the spectral lines under the influence of magnetic field is called.... |
|
Answer» PHOTOELECTRIC EFFECT |
|
| 28. |
Write effect of concentration change on equilibrium and explain by suitable example. |
|
Answer» Solution :In general, when equilibrium is disturbed by the addition / removal of any reactant / product, Le-Chatelier.s principle predicts that: (i) The concentration stress of an added reactant/product is relieved by net reaction in the direction that consumes the added substance. (ii) The concentration stress of a removed reactant / product is relieved by net reaction in the direction that replenishes the removed substance. OR Le-Chatelier.s principle for concentration change: "When the concentration of any of the reactants or products in a reaction at equilibrium is changed, the composition of the equilibrium mixture changes so as to minimize the effect of concentration changes." To understand the effect of concentration changes the example of decomposition of HI: The synthesis of HI is due to the reaction between `H_2` and `I_2` at the certain temperature and get equilibrium. `H_(2(g)) + I_(2(g)) hArr 2HI_((g))` (a) `H_2` is added in equilibrium : If `H_2` is added to the reaction mixture at equilibrium, then the equilibrium of the reaction is disturbed. In order to restore it, the reaction proceeds in a direction wherein `H_2` is consumed, i.e., more of `H_2` and `I_2` react to form HI and finally the equilibrium shifts in right (forward) direction.  (b) According to Le Chatelier.s principle : Which implies that in case of addition of a reactant/ product, a new equilibrium will be set up in which the concentration of the reactant/product should be less than what it was after the addition but more than what it was in the original mixture. (C) Explanation of reaction quotient `Q_c` : The same point can be explained in TERMS of the reaction quotient `Q_c` `Q_c=[HI]^2/([H_2][I_2])` and `K_c=[HI]^2/([H_2][I_2])` Addition of HYDROGEN at equilibrium results in value of `Q_c` being less than `K_c`. Thus, in order to attain equilibrium again reaction moves in the forward reaction. Removal of product : Similarly, the removal of product and increase the concentration of the products. Commercial application : The product is a gas or volatile substance so, the removal of products and increase the production. e.g.-1 : In vase of manufacture of ammonia, ammonia is liquefied and removed from the reaction mixture `(N_(2(g)) + H_(2(g)) = 2NH_(3(g)))` so that reaction keeps moving in forward direction. e.g.-2: In large scale production of Cao (used as important building material) from `CaCO_3`, constant removal of `CO_2` from the kiln drives the reaction completion `(CaCO_(3(s)) hArr CaO_((s)))` . It should be remembered that continuous removal of a product maintains `Q_c` at a value less than `K_c` and reaction continuous to move in the forward direction. |
|
| 29. |
The van der Waals radius in chlorine is 180 pm. What is the distance between two molecule of Cl_(2) ? |
| Answer» Solution :`180 XX 2 ` 360 PM | |
| 30. |
Which of the following has the minimum bond length ? |
|
Answer» `O_(2)` |
|
| 31. |
Which one of the following statement regarding molecular orbital is incorrect ? |
|
Answer» The energy of the anti bonding M.O is more than the combining atomic orbital and it DESTABILIZES the molecule |
|
| 32. |
Which of the following is used as "Bioremedies" |
|
Answer» Nitrates & PHOSPHATES |
|
| 33. |
Write physical properties of Hydrogen. |
|
Answer» SOLUTION :(i) DIHYDROGEN is a colourless, odourless, TASTELESS, combustible gas. (ii) It is lighter than AIR and insoluble in WATER. |
|
| 34. |
Under the influence of electric field , which of the following statements is true about the movement of electrons and holes in a p-type semiconductor ? |
|
Answer» Electron will move towards are positively CHARGED plate through electron holes |
|
| 35. |
Which one of the following gives only one monochloro derivative? |
|
Answer» n-hexane |
|
| 36. |
X is: |
|
Answer» `NaBH_(4)//EtOH` |
|
| 37. |
Which of the following exists as covalent crystals in the solid state? |
|
Answer» Phosphorus |
|
| 38. |
Which metal cannot be obtained by electrolyte sis? |
|
Answer» Ca |
|
| 39. |
What is the characterstricsof electron microscopy ? It dependson whichprinciple ? |
|
Answer» Solution :An elecronmicroscopeis a powerful tool in MODERN scientific RESEARCH because it ACHIEVES amagnificationof about15 milliontimes. principle: it isbasedon wavenatureelectron. |
|
| 40. |
Which of the following wrong |
|
Answer» 4-methyl pentane does not exist in IUPAC nomenclature 
|
|
| 41. |
Which of the following equations depict the oxidising nature of H_(2)O_(2) ? |
|
Answer» `2MnO_(4)^(-) + 6H^(+) + 5H_(2)O_(2) to 2Mn^(2+) + 8H_(2)O+ 5O_(2)` |
|
| 42. |
What is the volume of ammonia obtained starting from 2 L of nitrogen, if the conversionis only 6% efficient in the given conditions? |
|
Answer» Solution :Conversion of nitrogen to ammonia is given as `N_(2)+3H_(2)rarr 2NH_(3)` 1 MOLE of `N_(2) = 2` moles of `NH_(3)` 1 VOLUME of `N_(2) = 2` volumes of `NH_(3)` Volume of ammonia with `100%` efficient conversion of 2 L nitrogen = 4 L Volume of ammonia with `6%` efficient conversion of the REACTION `=(6xx4)/(100)=0.24L` |
|
| 43. |
Which of the following type of hydrogen is not possible ? |
|
Answer» `3^(@)` |
|
| 44. |
What is dipole moment ? |
|
Answer» SOLUTION :The polarity of a covalent bond can be measured, in terms of dipole moment which is DEFINED as: `mu = q xx 2d,` where `mu` is the dipole moment, q is the charge. 2d is the distance between the TWO charges. (ii) The dipole moment is a vector quantity and the direction of the dipole moment points from the negative charge to positive charge. (iii) The unit of dipole moment is Coulomb METRE (C m). It is USUALLY expressed in Debye unit (D). (iv) 1 Debye `= 3.336 xx 10^(-30)` C m |
|
| 45. |
Which of the following method is not used in the preparation of Acetylene |
|
Answer» DEHYDROHALOGENATION |
|
| 46. |
Which of the following hydrocarbons has the lowest dipole moment? |
|
Answer» cis-2-butene |
|
| 47. |
Which one of the following represents the correct order of increasing first ionization enthalpy of Ca, Ba, S, Se and Ar ? |
|
Answer» `Ca ltSlt Ba lt Ase lt Ar` `{:("Period"3^(rd),16^(S),18^(Ar)),("Period"4^(TH),20^(Ca),34^(Se)),("Period"6^(th),56^(Ba),):}` First ionization potentials increases in a period with increasing of atomic number `:.{:((i)Delta_(i)H(Ar)gt Delta_(i)H_(i)(S)),((ii)Delta_(i)H(Se)gt Delta_(i)H(Ca)):}}....(x) ` And in a group first ionization enthalpy DECREASES from top to BOTTOM `:.{:((i)Delta_(i)H(Ba)lt Delta_(i)H(Ca)),((ii)Delta_(i)H(Se)lt Delta_(i)H(S)):}}....(y) ` From (x)and (y) we can SAY that order of first ionization potential is as `Ba lt Ca lt Se lt S lt Ar` |
|
| 48. |
Which of the following species are expected to give the line spectrum similar to that of hydrogen atom? |
|
Answer» He(g) |
|
| 49. |
What do you mean by alkene ? Give its general formula and two examples. |
|
Answer» SOLUTION :ALKENES are unsaturated containing at least one double bond. General formula of ALKENE is `C_(n)H_(2N)`. In alkene, there is 1 double bond between carbon. Alkane has 2 hydrogen less than alkane. `{:("Name IUPAC","Structure","Molecular formula",),("Ethene (Ethelene)",CH_(2)=CH_(2),C_(2)H_(4),),("Propene (Propelene)",CH_(3)-CH=CH_(2),C_(3)H_(6),):}` |
|
| 50. |
Two mole ofPCl_(5)" were heated to" 327^(@)Cin a closed two litre vessel and when equilibrium was achieved, PCl_(5) was found to be 40% dissociated intoPCl_(3) and Cl_(2).Calculate the equilibrium constants K_(p) and K_(c) for this reaction. |
|
Answer» Solution :`PCl_(5) " dissociates as " :PCl_(5) hArr PCl_(3) + Cl_(2)` IntiaL amount of `PCl_(5) = 2" moles (Given)"` `% " age dissociation at equilibrium " = 40%` ` :. PCl_(5) " dissociated at equilibrium " = 40/100 xx 0.8 ` mole `:. " Amounts of " PCl_(5), Pcl_(3) and Cl_(2) " at equilibrium will be " ` `Pcl_(5) = 2- 0.8 = 1.2 ` mole `PCl _(3) = 0.8 ` `Cl_(2)-0.8 " " [:' 1 " mole of " PCl_(5) " on dissociation GIVES 1 mole of "PCl_(3) "and 1 mole of "Cl_(2) ]` Since the volume of the vessel is 2 litres is , therefore , the molar concentrations at equilibrium will be `[ PCl_(5)] = 1.2/2 = 0.6 " mol " L^(-1), [ PCl_(3)] = 0.8/2 = 0.4 " mol " L^(-1) and [ Cl_(2) ] = 0.8/2 = 0.4 "mol" L^(-1)` Applying the LAW of chemical equilibrium to the dissociation equilibrium, we get `K_(c)= ([PCl_(3)] [ Cl_(2)])/ ([PCl_(5)]) = (.4 " mol " L^(-1) xx 0.4" mol " L^(-1))/(0.6 " mol " L^(-1))= 0.267 "mol" L^(-1)` `K_(p) = K_(c) (RT)^(Delta N)` Here `Delta n = n_(p) - n_(r) = 2- 1 = 1 " mol " :. K_(p) = K_(c) (RT)` But `T= 327 + 273 = 600 K " (Given) "` ` R= 0.0821" L ATM"K^(-1) mol^(-1) :. K_(p) = 0.267 molL^(-1) xx 0.0821" L atm "K^(-1) mol^(-1) xx 600 K = 13.15 " atm"` |
|








