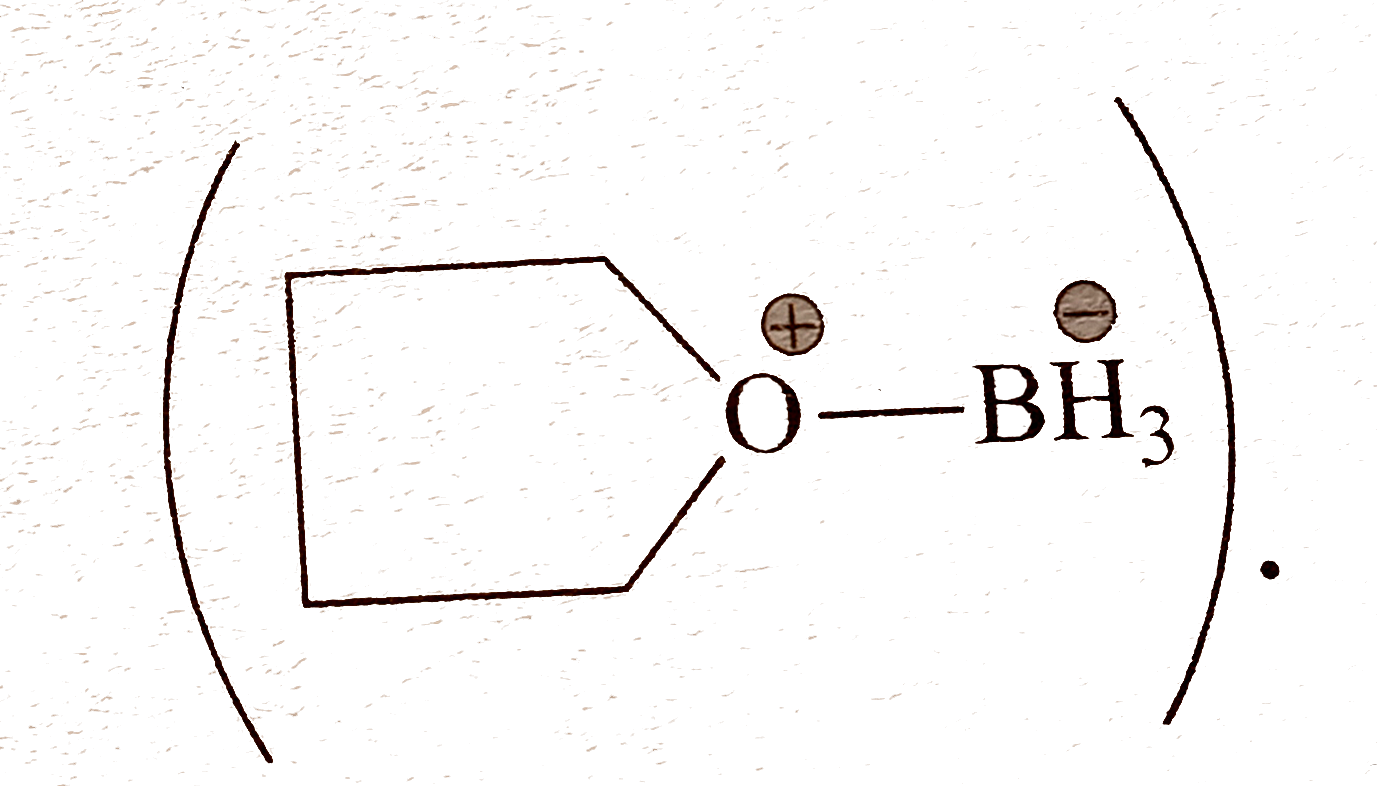Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
When H_2O_2 is added to ice cold solution of acidified potassium dichromate in ether and the contents are shaken and allowed to stand |
|
Answer» A blue colour is OBTAINED in ether due to FORMATION of `Cr_2(SO_4)_3` `Cr_2 O_7^(-2) + 2H^(+) + 4H_2O_2 to 2CrO_5 + 5H_2O` blue colour in ether |
|
| 2. |
Which of the following factors will not distrub the equilibrium state of the reaction N_(2) (g) + O_(2) hArr 2NO(g) ? |
|
Answer» CHANGE in pressure |
|
| 3. |
What is electron negativity ? How does it change in a period as well as in a group ? |
| Answer» SOLUTION :The ability of an atom to attract the shared electron pair (of a COVALENT bond) in a molecule towards itself is called electro NEGATIVITY. In a period from LEFT to right the electro negativity increases. Down a group electro negativity value decreases. | |
| 4. |
Which one of the following reagent react with methyl magnesium iodide followed by acid hydrolysis to give ethyl acetate? |
|
Answer» Chlorodimethyl ETHER |
|
| 5. |
Which of the following statements is/are not correct? |
|
Answer» Metamerism BELONGS to the category of structural isomerism |
|
| 6. |
What is the angular momentum of an electron in (i) 2 s orbital (ii) 4f orbital ? |
|
Answer» SOLUTION :Angular momentum of ELECTRON in any orbital `= SQRT(l(l + 1)) = (h)/(2pi)` `:.` For 2 s orbital, `l = 0, " " :.` angular momentum `= sqrt(0(0 + 1)) (h)/(2pi) = 0` For 4f orbital, `l = 3, " " :.` angular momentum `= sqrt(3(3 +1)) (h)/(2pi) = 2 SQRT3 (h)/(2pi) = sqrt3 (h)/(pi)` |
|
| 7. |
Which of the following solutions will have pH close to 1.0? |
|
Answer» 100 ML of M/10 HCl+100 mL of M/10 NaOH |
|
| 8. |
The value of Delta G^(@) for the phosphorylation of glucose in glucolysis is13*8kj//mol. Find the value of K_(c) at 298 K. |
|
Answer» Solution :`DELTAG^(@) = 13*8 " Kjmol"^(-1)= 13*8 xx 10^(3)" J mol"^(-1)` `Delta G^(@) = - 2*303" RT "log K_(c)` `13*8 xx 10^(3) "J mol"^(-1) = -2*303 xx ( 8*314 " JK"^(-1)" mol"^(-1))(298 K) " log "K_(c)or log K_(c) = -2*4186 = bar(3)*5814` `K_(c) = " ANTILOG " (bar(3)*5814) = 3*815 xx 10^(-3)` |
|
| 9. |
Which of the hydrrides araea ionic ? |
|
Answer» `CaH_(2)` |
|
| 10. |
Write the electronic configuration of Na and K. |
|
Answer» Solution :`NA (11) : 1 s^(2) 2s^(2) 2P^(6) 3s^(1)` `K(19) : 1s^(2) 2s^(2) 2p^(6) 3s^(2) 3p^(6) 4s^(1) K` |
|
| 11. |
What material is used preparebullet proof vests ? |
|
Answer» Solution :When boron oxide is REDUCED with carbon in an ELECTRIC furnance, boron carbide `(B_(4)C)`is formed `2B_(2)O_(3)(s) + 7 C (s) underset(1873 K)overset(" Electric furnace")rarrB_(4)C(s) + 6CO(G)` Boron carbide is EVEN hardar than diamond . Therefore , it is used or making bullet-proof vests. It is also used as a shield or ratioactive radiations end also as an abrasive. |
|
| 12. |
What will be the result if 100 mlL of 0.06 M Mg(NO_(2))_(2) is added to 50 ml. of 0.06 MNa_(2) C_(2) O_(4) ?[ Ksp of MgC_(2) O_(4) = 8.6 xx 10^(-5)] |
|
Answer» A precipitate will not be formed But after mixing ` M_1V_1 = M_2V_2 rArr0. 0 6 xx 100 = M_2 xx 150` ` [Mg^(+2)] =0.04 M rArr M_1 V_1 =M_2 V_2` ` 0.06 xx 50 = M_2 xx 150 rArr [C_2 O_4^(-2) ]= 0.02 M` ` {:(Mg^(+2)+, C_2O_4^(-2)hArr, MgC_2O_4),( 0.04 M , 0.02M , 0) ,( 0.02 , -,0.02M), (0.02M , , ):}` ` I.P =[Mg^(+2) ] [C_2O _4^(-2)]` ` =(4XX 10 ^(-2) ) (2xx 10 ^(-2)) = 8 xx 10 ^(_4) ` ` thereforeK_(sp)LT I. P` , ppt will from |
|
| 13. |
What is the lowest value of n that allows g orbitals to exist ? |
| Answer» SOLUTION :For g-subshell, `l=4`, As `l=0` to `n-1`, to have `l=4`, MINIMUM VALUE of `n=5`. | |
| 14. |
The second orbit in He^+ ion has radius as the first orbit in hydrogen atom. |
|
Answer» |
|
| 15. |
Which of the following does not possess zero magnetic momentum ? |
|
Answer» Transvinly chloride |
|
| 16. |
Use the periodic table to identify elements (a) With 5 electrons in the outer subshell (b) Would tend to lose two electrons (c) Would tend to gain two electrons |
|
Answer» |
|
| 17. |
Write the IUPAC names of : |
Answer» SOLUTION :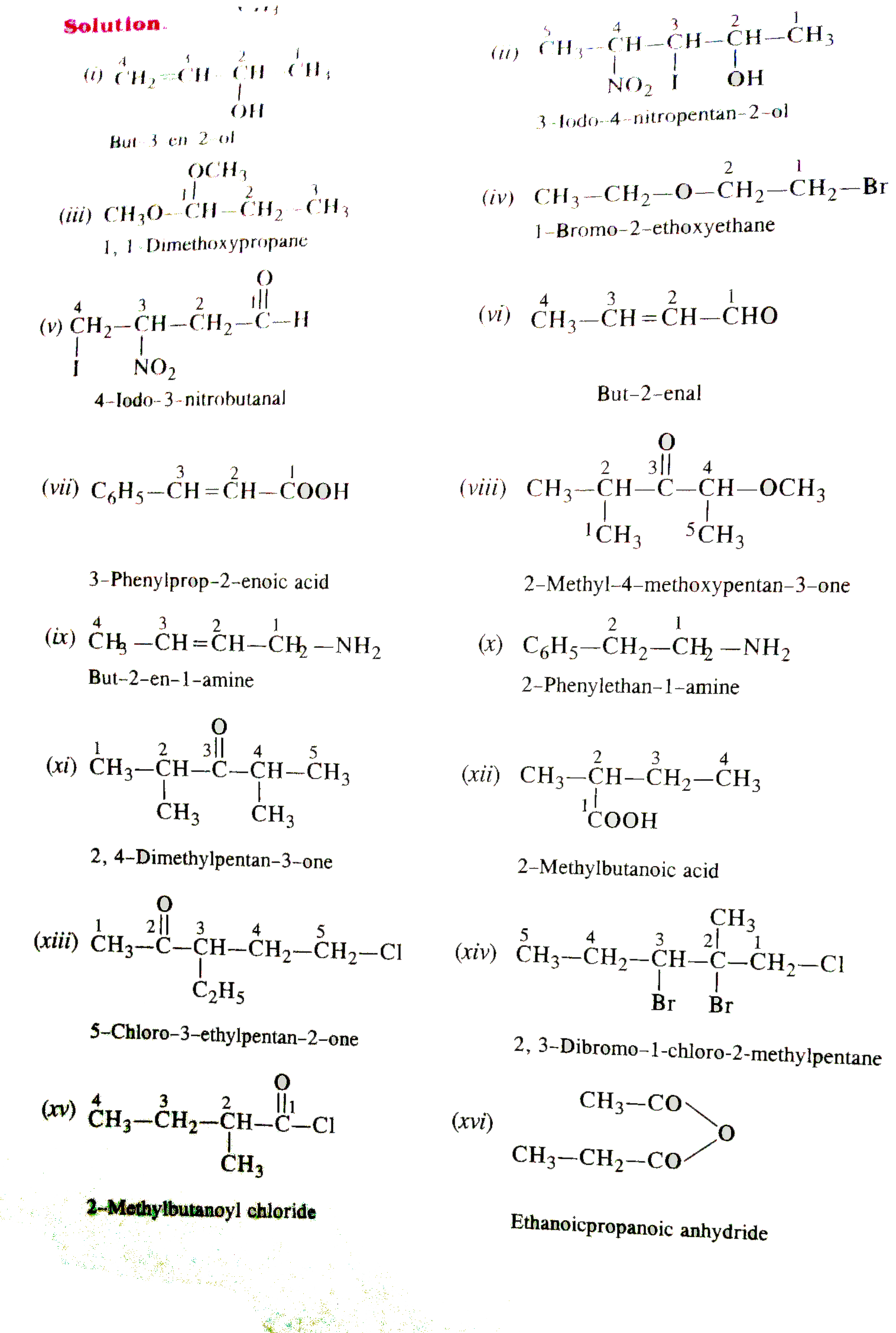
|
|
| 18. |
Which of the following is not an artificial sweetener ? |
|
Answer» Aspartame |
|
| 19. |
What is the valency possible to Arsenic with respect to oxygen and hydrogen? |
|
Answer» |
|
| 20. |
Which of the following is not a disadvantage of using hard water? |
|
Answer» In PRODUCTION of steam in boilers |
|
| 21. |
When CO_(2) is bubbled into an aqueous solution of Na CO_(3).......... is formed |
|
Answer» `H_(2)O` |
|
| 22. |
What happens when LiH reacts with Al_2C_(16)? |
|
Answer» |
|
| 23. |
Why alkali metals and alkaline earth metals are called s-block elements? |
| Answer» Solution :Alkali metals and alkaline earth metals (group 1 & 2) are those in which the last ELECTRON enters the OUTERMOST s-orbital. As the s-orbital can accommodate only TWO electrons, the two GROUPS belong to the s-block elements. | |
| 24. |
Which of the following reactions involve homogeneous equilibrium and which involve heterogeneous equilibrium ? Ag_(2)O_((s))+2HNO_(3(aq))hArr2AgNO_(3(aq))+H_(2)O_((l)) |
| Answer» SOLUTION :HETEROGENEOUS EQUILIBRIUM | |
| 25. |
Which of the following reactions involve homogeneous equilibrium and which involve heterogeneous equilibrium ? C_((s))+CO_(2(g))hArr2CO_((g)) |
| Answer» SOLUTION :HETEROGENEOUS EQUILIBRIUM | |
| 26. |
Which of the following reactions involve homogeneous equilibrium and which involve heterogeneous equilibrium ? CH_(3)COOC_(2(g))H_(5(aq))+H_(2)O_((l))hArrCH_(3)COOH_((aq))+C_(2)H_(5)OH_((aq)) |
| Answer» SOLUTION :HOMOGENEOUS EQUILIBRIUM | |
| 27. |
Which of the following reactions involve homogeneous equilibrium and which involve heterogeneous equilibrium ? 2SO_(2(g))+O_(2(g))hArr2SO_(3(g)) |
| Answer» SOLUTION :HOMOGENEOUS EQUILIBRIUM | |
| 28. |
Two metals (A) and (B) belongs to the same group of periodic table. Metal (A) forms an insoluble oxide but soluble sulphate. Metal (B) forms soluble oxide and insoluble sulphate. Then A and B are respectively |
|
Answer» `BA,Be` |
|
| 29. |
What is the coordination number of sodium in sodium oxide( Na_(2)O) ? |
|
Answer» 6 |
|
| 30. |
Using curved-arrow notation, show the formation of reactive intermediates when the following covalent bonds undergo heterolytic cleavage. (a) CH_(3) - SCH_(3), (b) CH_(3) - CH, (c ) CH_(3) - Cu |
Answer» Solution :(a) Bonding electron pair transfer on more negative S.  (b) The sp carbon of `C -=` is more negative then `H_(3)O (sp^(3))`.  (c ) In COMPARE to ELECTROPOSITIVE Cu metal, the negativecarbon atom ACCEPT bonding electron pair 
|
|
| 31. |
What is the relationshipbetweenq_(p) and q_(v) ? |
|
Answer» <P> Solution :`q_(p)= q_(v)+Deltan_(g)`RTwhere `Deltan_(g)=n_(p)-n_(r)` (GASEOUS) |
|
| 32. |
Which fo the following peresent E from? |
|
Answer»
|
|
| 33. |
Which of the following is a more stable carbocation? |
|
Answer» Sec. PENTYL CARBOCATION |
|
| 34. |
What weight of HNO_3is needed to convert 5g of iodine into HIO_3according to the reaction I_(2)+HNO_(3) rarrHIO_(3)+NO_(2)+H_(2)O |
|
Answer» 12.4g `(w)/(63//1)=5/(254//10)IMPLIES w = 12.4 G` |
|
| 35. |
Total number of chiral carbon (Y) in following compounds. |
Answer» 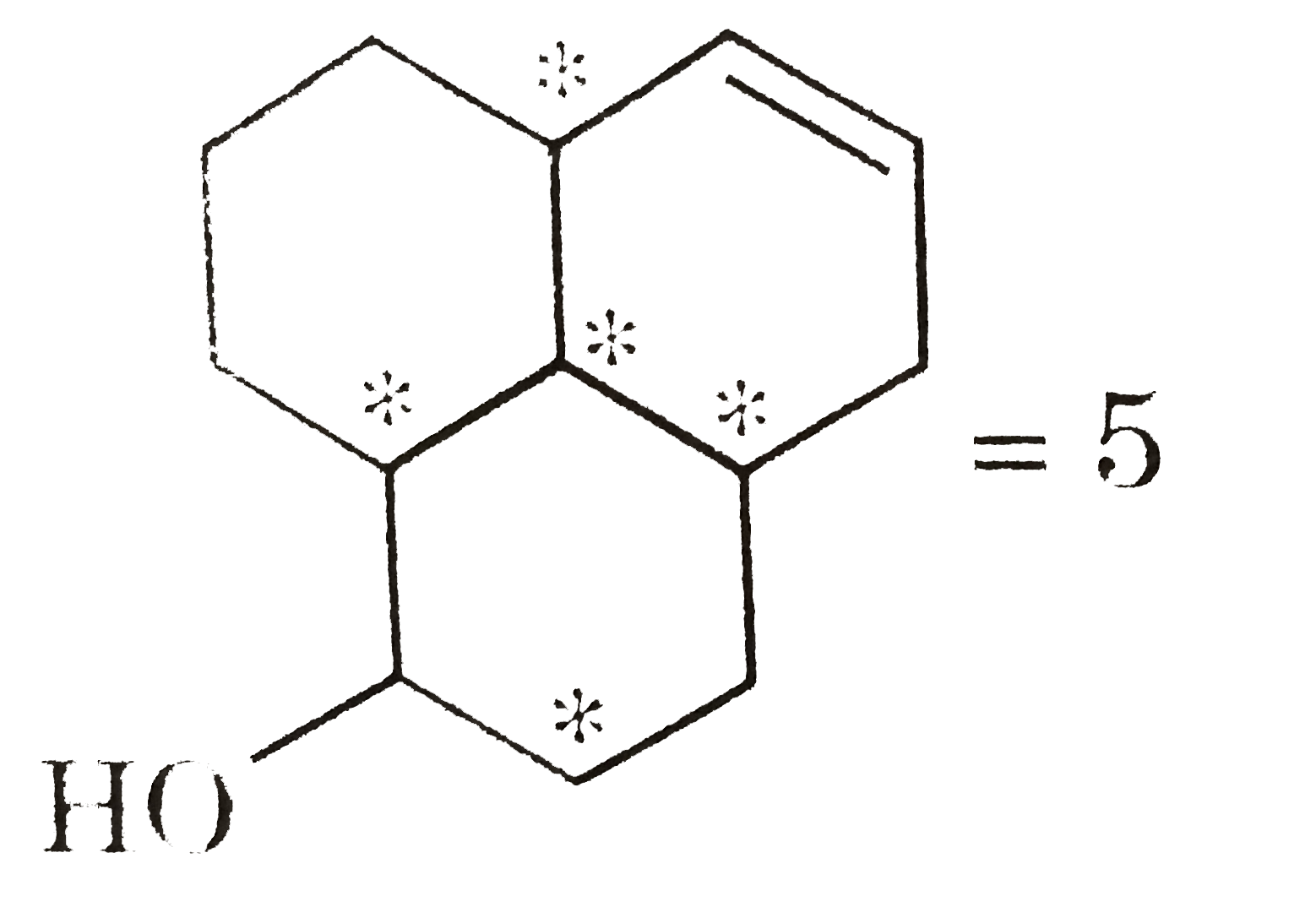
|
|
| 36. |
Whatismeant by entropy drivenreaction? How can the reaction with positivechange of enthalpy and entropydriven? |
|
Answer» Solution :The free energy change of a reaction is GIVEN by `DeltaG = DeltaH -TDELTAS` For a reaction to be SPONTANEOUS, `DeltaG` shouldbe `-ve`. If both `DeltaH`and `DeltaS` are positive, `DeltaG` can be`-ve` if `T Delta Sgt DeltaH`in magnitudee. Thus, entropy factor should dominate over enthalpyfactor. Such reactions are therefore, called entropy driven.This canhappen in either of the following two WAYS `:` (i) `DeltaS` shoul beso learge that even if T is low , `T DeltaS` should be GREATER than`DeltaH` (ii) If` DeltaS`is small, T should be so large that`T DeltaSgt DeltaH` |
|
| 37. |
Which of the following ions will not undergo disproportionation? |
|
Answer» `CL^(-)` `CI^(-)rarr(– 1)`It can undergo oxidation only. `CIO_(4)^(-)(+7)`It can undergo reduction only. THUS , `CI^(-)and CIO_(4)^(-)`ions cannot undergo disproportionation. |
|
| 38. |
Which of the following elements is present as the impurity to the maximum extent in pig iron ? |
|
Answer» Phosphorus |
|
| 39. |
Which one of the following is used as a soil sterilizing agent? |
|
Answer» CHLOROFORM |
|
| 40. |
When salt solution 'A' is treated with dilute HCI it gives off gas 'B', which when passed through a filter paper, moistened with potassium iodate and starch solution, gives deep blue colouration. 'A' and 'B' respectively are: |
|
Answer» `A = Na_(2)SO_(3)` and `B = SO_(2)` |
|
| 41. |
Which of the following is a neutral salt ? |
|
Answer» `Na_3PO_4` |
|
| 42. |
Which of the following are true regarding hydroboration of alkene ?( More than one correct statements . ) |
|
Answer» Reaction proceeds through ionic intermediat and syn`-`addition take place. |
|
| 43. |
Which is not a use of plaster of pairs |
|
Answer» It isused in the manufacture of crucibles and models |
|
| 44. |
Which among the following factors decreases the activation energy of a chemical reaction ? |
|
Answer» Pressure |
|
| 45. |
The reactivity order towards hydrogenation of the following compounds is |
|
Answer» `I gt II gt III gt IV` |
|
| 46. |
Viscosity of a liuquid arises due to strong intermolecular forces existing between the molecules. Stronger the intermolecular forces, greater is the viscosity. Name the intermolecular forces existing in the following liquids and arrange them in the increasing order of their viscosities. Also given reason for the assigned order in one line.Water, hexane (CH_(3)CH_(2)CH_(2)CH_(2)CH_(2)CH_(3)),glycerine (CH_(2)OH CH (OH) CH_(2) OH) |
|
Answer» Solution :In `H_(2)O` and glycerine - Hydrogen bonding and dipole - dipole INTERACTIONS are present as polar molecules. Hexane - Dispersion forces / LONDON forces exist in non - polar nature. Order of intermolecular forces is, hexane `lt` water `lt` glycerine. Thus, hexane has WEAKEST intermolecular forces and glycerine the strongest intermolecular forces. Hence, hexane has minimum viscosity and glycerine has MAXIMUM viscosity. |
|
| 47. |
Which of the following element is not consider as alkaline earth metal ? |
| Answer» ANSWER :A | |
| 49. |
What is the pH of mix solution of ammonia and ammonium chloride ? Give equation. |
|
Answer» SOLUTION :This solution is basic buffer, So its pH is NEARER to pH `GT` 7 and 9.25. pH=`pK_b+"LOG"(["Conjugate Acid," NH_4^+])/(["BASE", NH_3])` |
|
| 50. |
The term not responsible for water pollution |
|
Answer» GREEN revaluation |
|
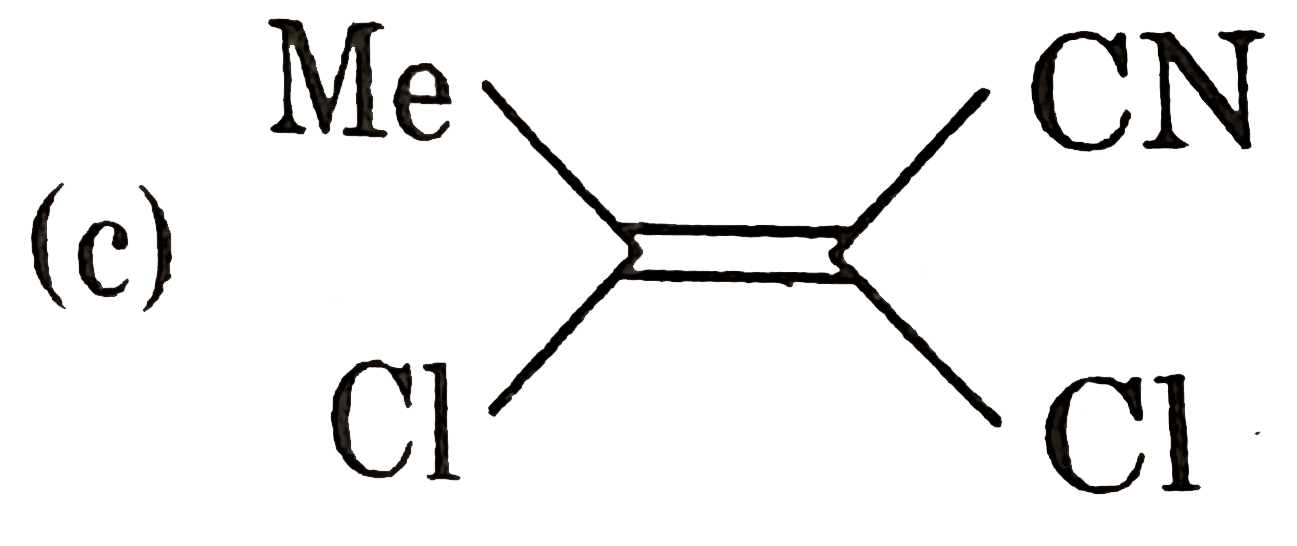
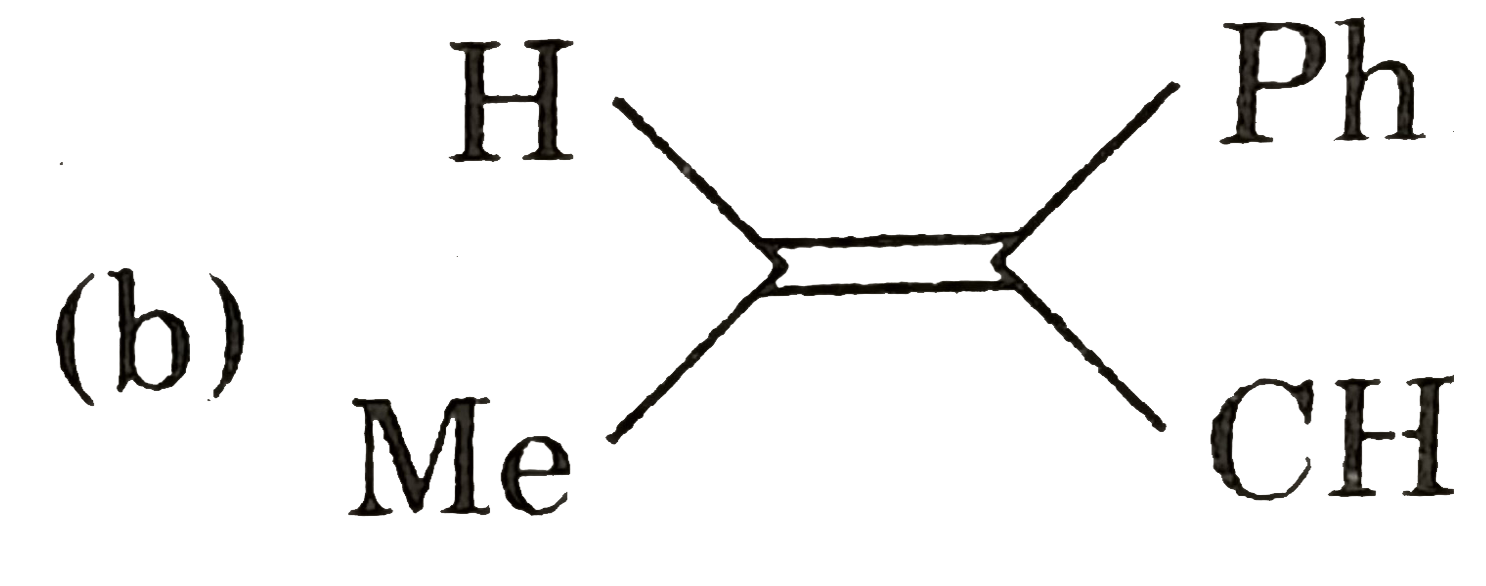
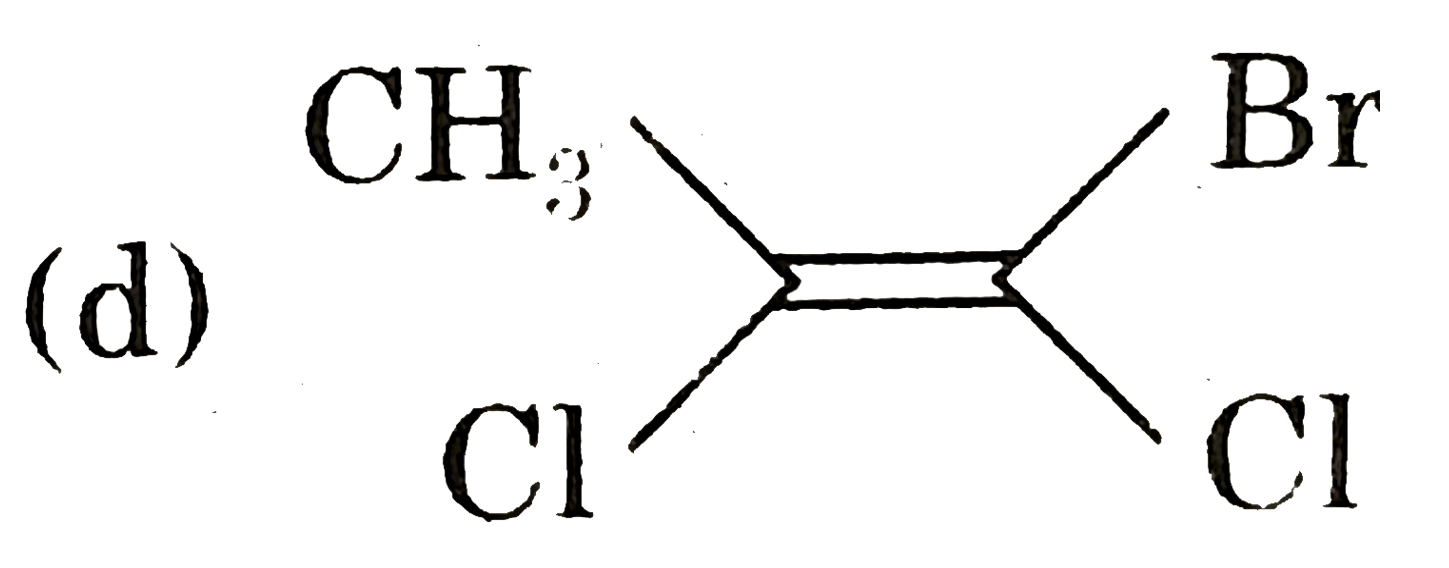
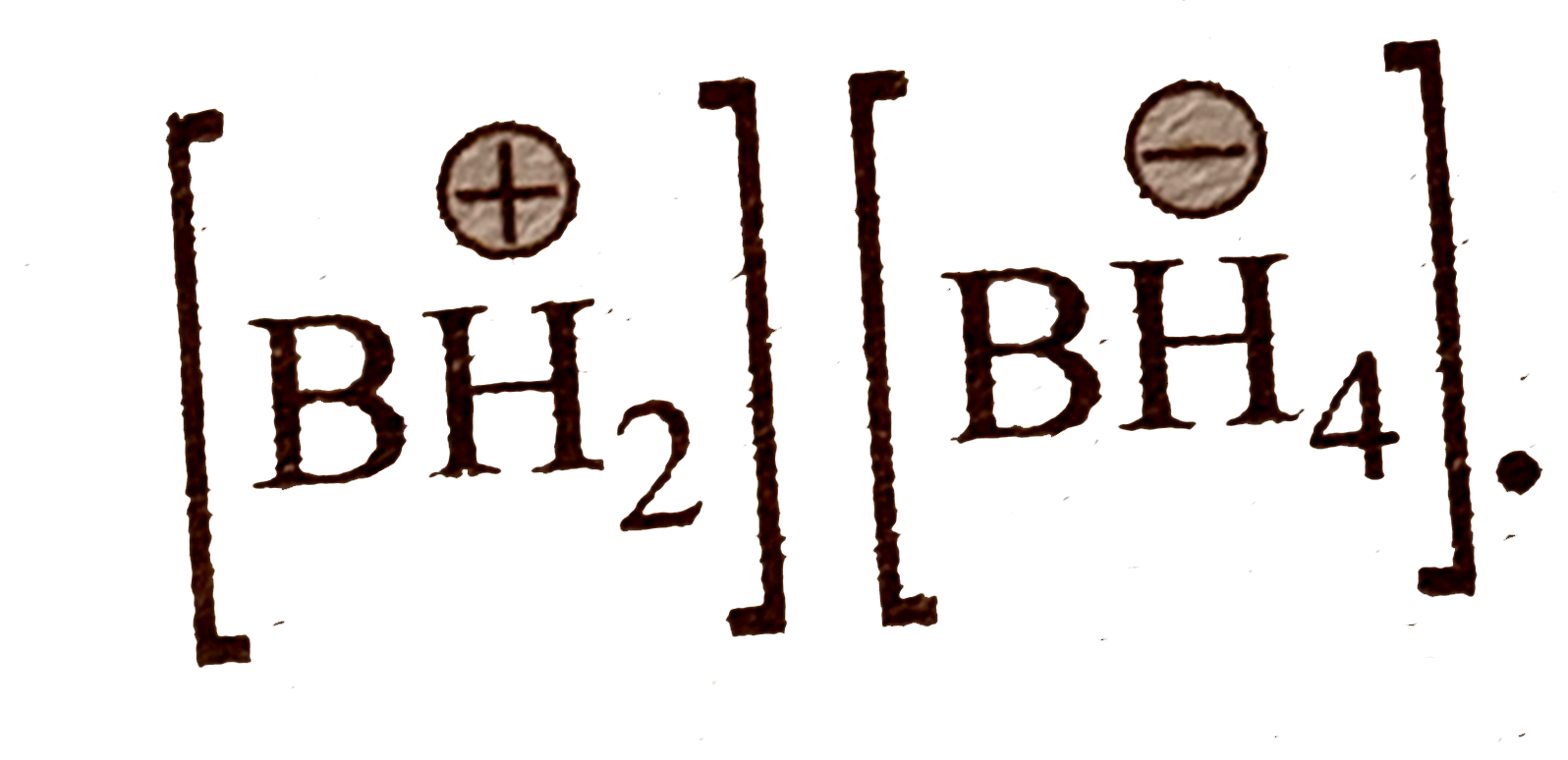 .
.