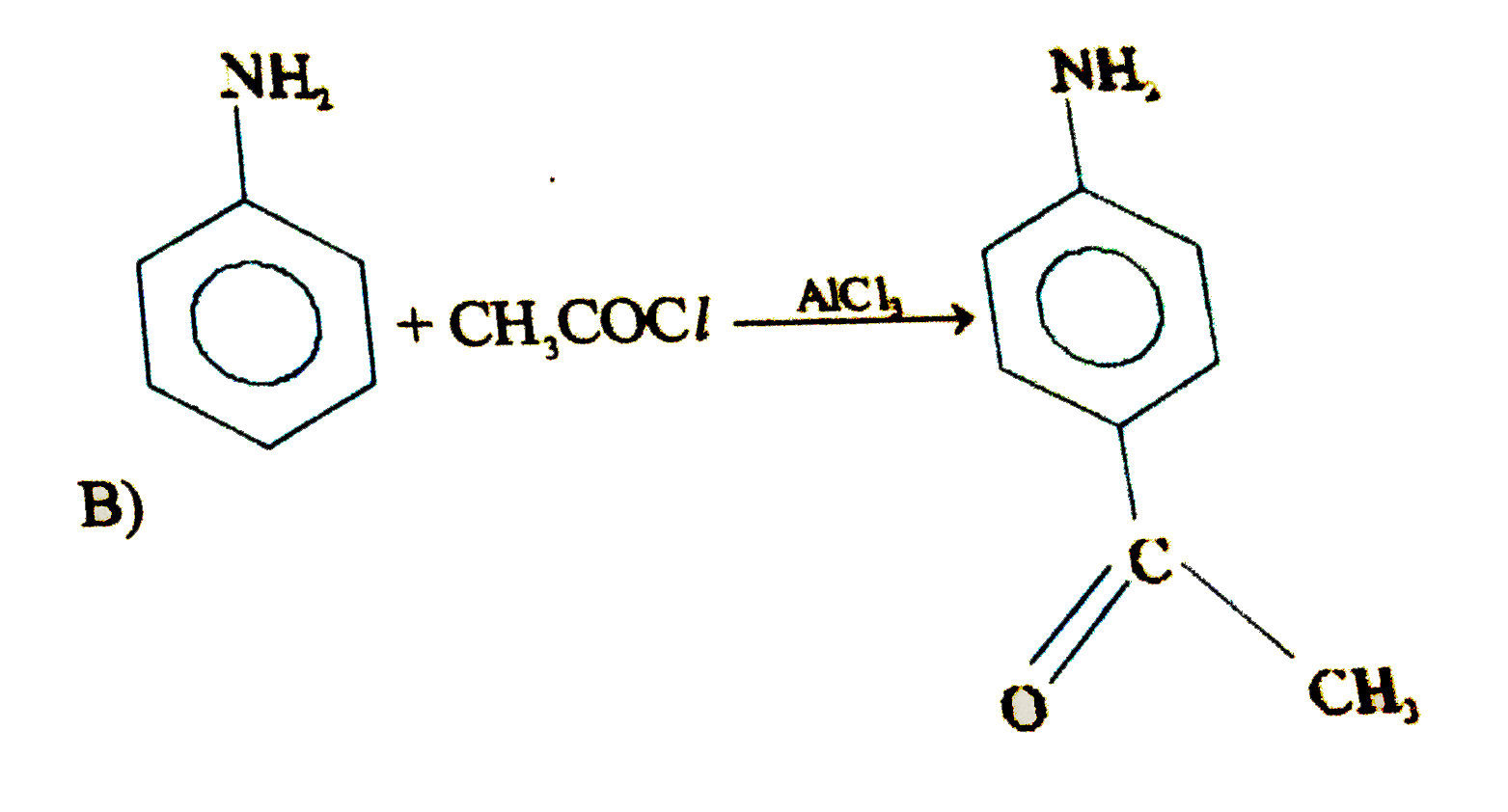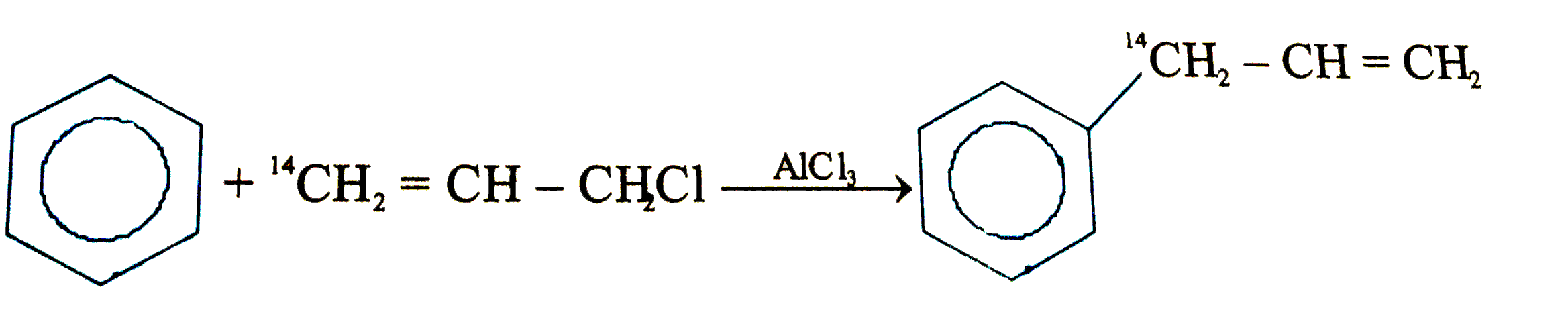Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following element do not form hydrated metal chloride ? |
| Answer» ANSWER :D | |
| 2. |
Which of the following statements regarding IV A group elements is/are correct |
|
Answer» They all FORM tetrahalides C) DUE to `SP^(3)` hybridisation D) `SiF_(4)+OH^(-)toSiO_(4)^(4-)+4F^(-)+4H_(2)O` |
|
| 3. |
The shape of CIF_(3) according to VSEPR model is |
|
Answer» PLANAR triangle |
|
| 4. |
Which of the following statements about amorphous solids is incorrect ? |
|
Answer» They melt over a range of temperature (a)are isotropic in nature. (B)do not have a sharp MELTING POINT. `(c)`do not have an orderly arrangement of ATOMS. All solids are rigid and incompressible. |
|
| 5. |
The value of K_c for the reaction 3O_(2(g)) hArr 2O_(3(g))is 2.0 xx10^(-50) at 25^@C. If the equilibrium concentration of O_2 in air at 25^@C is 1.6 xx 10^(-2), what is the concentration of O_3 ? |
|
Answer» Solution :`{:("Equilibrium reaction :",3O_(2(g)) hArr, 2O_(3(g))),("Constant of equilibrium :", 1.6xx10^(-2) , x):}` According to chemical equilibrium : `K_c=[O_3]^2/[O_2]^3` where, `K_c=2.0xx10^(-50)` `therefore 2.0xx10^(-50) = [O_3]^2/[1.6xx10^(-2)]^3` `[O_2]=1.6xx10^(-2)` `therefore [O_3]^2=2.0xx10^(-50) (1.6xx10^(-2))^3=8.192xx10^(-56)` `therefore [O_3]=(8.192xx10^(-56))^(1/2)` `=2.862xx10^(-28) "MOL L"^(-1)` |
|
| 6. |
What are the various factors due to which the ionization enthalpy of the main group elements tends to decrease down a group ? |
|
Answer» Solution : Within the main group elements, the ionization enthalpy decreases regularly as we move down the group. This is due to the following FACTORS : (i)Atomic size : On moving down the group, there is a gradual increases in atomic size due to an additional main energy shell (n). (ii) Shielding effect : There is increases in shielding effect on the outermost electron due to increase in the number of inner electrons. (III) NUCLEAR charge : In going from top to bottom in a group, the nuclear charge increases. The effect of increase in atomic size and the shielding effect is much more than the effect of increases in nuclear charge. As a result, the electron becomes less TIGHTLY held to the nucleus as we move down the group. Hence there is a gradual decrease in the IONISATION enthalpies in a group. |
|
| 8. |
What is the density of N_2 gas at 227^@ C and 5.00atm. pressure? (R = 0.082 L atm K^(-1) mol^(-1)) |
|
Answer» `1.40g// ML` |
|
| 9. |
The study of different methods and techniques used for analysis of chemical compounds included in branch of ......... |
|
Answer» BIOCHEMISTRY |
|
| 10. |
Which of the following pairs has elements containing same number of electrons in the outermost orbit? |
| Answer» Answer :D | |
| 11. |
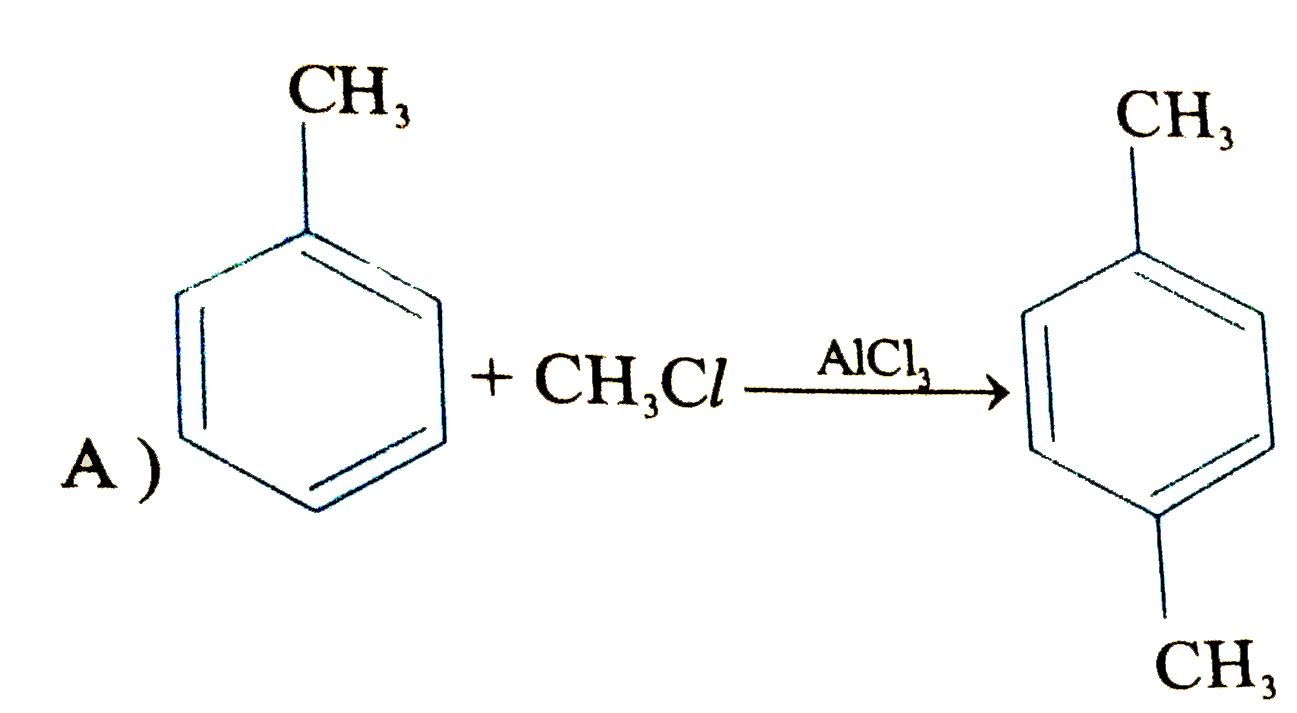
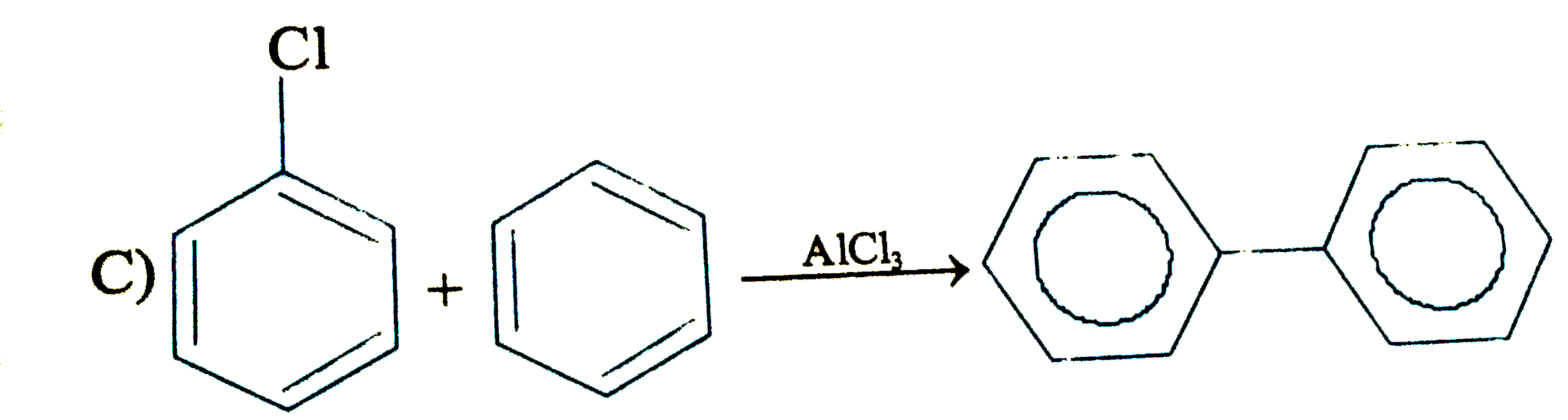
Which of the following will fal to product the product shown |
|
Answer»
|
|
| 12. |
Which of the following does not show the amomalous behaviour of lithium? |
|
Answer» LITHIUM reacts with nitrogen to FORM a nitride |
|
| 13. |
When heated in oxygen, lithium forms........, sodium forms......., while potassium forms......... |
| Answer» SOLUTION :`Li_2O, Na_2O_2, KO_2` | |
| 14. |
Write the Bronsted - Lowry principle for acids and bases. |
|
Answer» SOLUTION :The Danish chemist, Johannes Bronsted and the English chemist, Thomas M. Lowry gave a more general definition of acids and bases. DEFINATION "According to Bronsted-Lowry theory, acid is a substance that is capable of donating a hydrogen ion `(H^+)` and bases are substances capable of accepting a hydrogen ion, `(H^+)`." In short, acids are proton donors and bases are proton ACCEPTORS. e.g.-1 : Consider the example of dissolution of `NH_3` in `H_2O` represented by the following equation.  The basic solution is FORMED due to the presence of hydroxyl ions. In this reaction, water molecule acts as proton donor and ammonia molecule acts as proton acceptor and are thus, called Lowry-Bronsted acid-base, respectively. In the REVERSE reaction, `H^+` is transferred from `NH_4^+`to `OH^-`. In this case, `NH_4^+` acts as a Bronsted acid while `OH^-` acted as a Bronsted base. |
|
| 15. |
Which substance present in cotton cloth forms Hydrogen bond with Water ? |
|
Answer» NUCLEIC ACID |
|
| 16. |
The vapour pressure of pure benzene (C_(6)H_(6)) at a given temperature is 640 mm Hg. 2.2g of non - volatile solute is added to 40 g benzene. The vapour pressure of the solution is 600 mm Hg. Calculate the molar mass of the solute ? |
|
Answer» Solution :`P_(C_(6)H_(6))^(0)=640` mm Hg, `W_(2)=2.2` G (no volatile solute), `W_(1)=40` g (benzene) `P_("solution")=600` mm Hg, `M_(2)=?` `(P^(0)-P)/(P^(0))=x_(2)` `(640-600)/(640)=(n_(2))/(n_(1)+n_(2))""[becausen_(1)gt gtn_(2),n_(1)+n_(2)~~n_(1)]` `(40)/(640)=(n_(2))/(n_(1))` `0.0625=(W_(2)xxM_(1))/(M_(2)xxW_(1))` `M_(2)=(2.2xx78)/(0.0625xx40)=68.64" g MOL"^(-1)` |
|
| 17. |
Which of the following compound(s) has/have percentage of carbon same as that in ethylene (C_(2)H_(4) )? |
|
Answer» Propene |
|
| 18. |
Weight of iodine required to oxidise 500 mL N Na_(2)S_(2)O_(3) solution, is : |
|
Answer» 6.35 g |
|
| 19. |
The vapour pressure of acetone at 20^(@)C is 185 torr. When 1.2 g of a non - volatile substance was dissolved in 100 g of acetone at 20^(@)C, its vapour pressure was 183 torr. The molar mass (g mol^(-1)) of the substance is : |
|
Answer» <P>32 Vapour pressure of solution, `P_(S)=183` torr Molar MASS of solvent, `M_(A)=58` g//mol as we know`(P_(A)^(@)-P_(S))/(P_(S))=(n_(B))/(n_(A))` `rArr (185-183)/(183)=(W_(B))/(M_(B))xx(M_(A))/(W_(A))` `rArr (2)/(183)=(1.2)/(M_(B))xx(58)/(100)` `rArr M_(B)=(1.2)/(2)xx(58)/(100)xx183=64` g/mol |
|
| 20. |
What are thermochemical equation? What are the conventions adopted in writing thermochemical equation? |
|
Answer» Solution :A thermochemical equation is a balanced stoichiometric CHEMICAL equation that includes the enthalpy change `(DeltaH)`. Conventions adopted in thermochemical equations: (i)The coefficients in a balanced thermochemical equation refer to number of MOLES of reactants and products involved in the reaction (ii)The enthalpy change of the reaction `DeltaH`has unit kJ (III)When the chemical reaction is reversed the value of All is reversed in sign with the same magnitude. (iv)Physical states (gas, liquid, aqueous and solid) of all species is important and MU. SPECIFIED in a thermochemical reaction since `DeltaH` depends on the phases of reactants products. (v)If the thermochemical equation is multiplied throughout by a number, the enthalpy change is also be multiplied by the same number value. (vi)The negative sign of `DeltaH_r^0`indicates the reaction to be an exothermic and the positive signof `DeltaH_r^` indicates an endothermic type of reaction. |
|
| 21. |
What is the name of the Freon which have the formula C_(2)F_(3)Cl_(3) ? |
|
Answer» Freon-11 |
|
| 22. |
Write the balanced chemical equations for the combustion of the following hydrocarbons . (i) Butane (ii) Toulene |
|
Answer» Solution :(i)`C_(4)H_(10)+3/2O_(2)to5H_(20O+4CO_(2)` (ii) `C_(6)H_(5)CH_(3)+9O_(2)to7CO_(2)+4H_(2)O` |
|
| 23. |
What is electron gain enthalpy? |
|
Answer» Solution :When an electron is added to a NEUTRAL gaseous atom (X) to convert it into negative ion, the enthalpy CHANGE accompanying the process is DEFINED as the electron GAIN enthalpy. `X(g) + e^(-) to X^(-)(g) , Delta_(eg) H` `Delta_(eg) H` it will be positive or negative. |
|
| 24. |
What is standard state ? |
| Answer» Solution :1 ATMOSPHERE pressure and 298 K TEMPERATURE is KNOWN as standard state. | |
| 25. |
What is the major product of the reaction given below? |
|
Answer» `cis -2-`butene 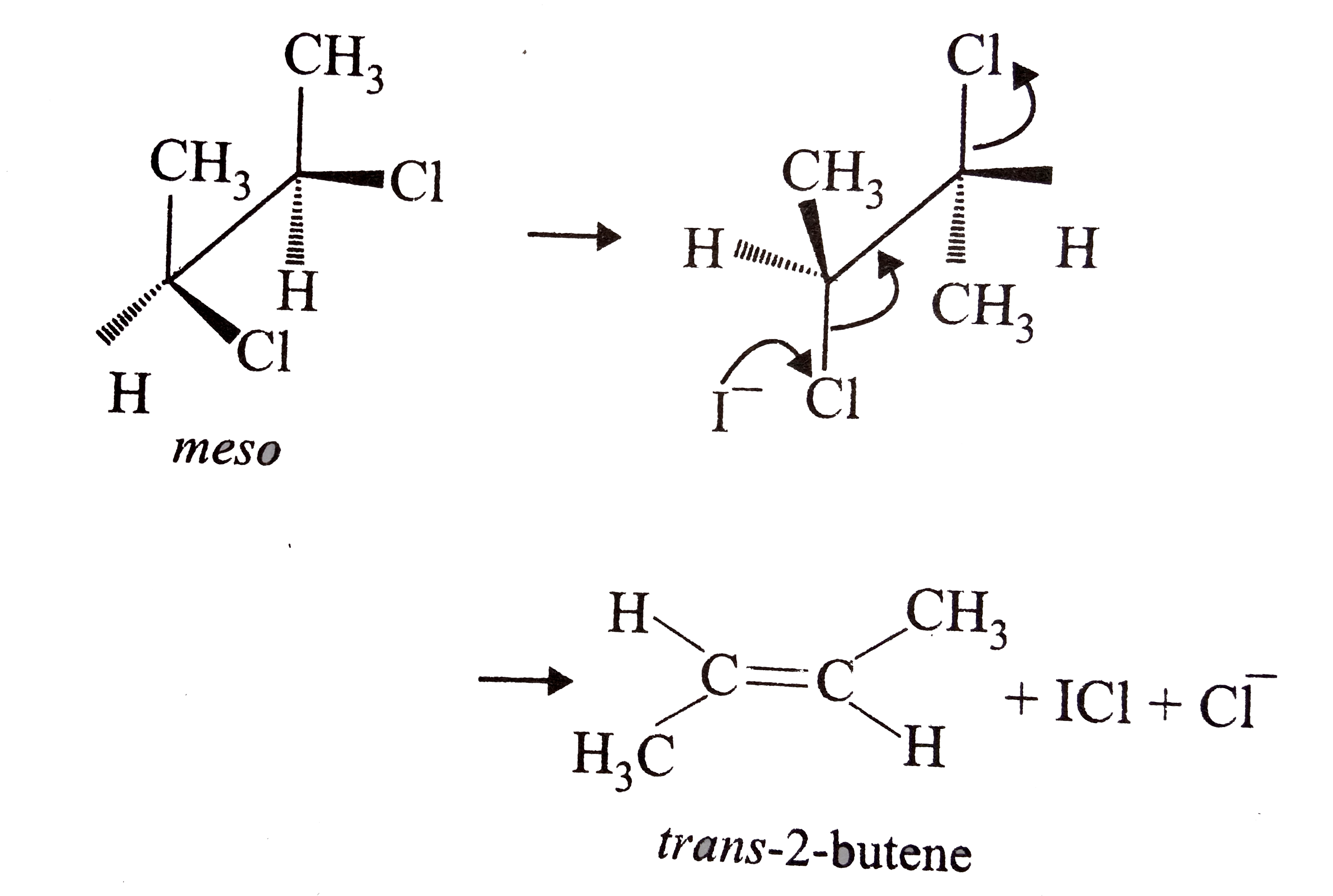
|
|
| 26. |
Write the names of hydrocarbons fuel used in daily life and write their uses ? |
|
Answer» Solution :In our daily LIFE hydrocarbon fuel is used as domesttic fuel. This gas and fuels are as follows : (i) LPG (Liquified Petroleum Gas) (ii) CNG (Compressed Natural Gas) (iii) LNG (Liquified Natural gas) (iv) Petrol diesel and kerosene oil are obtained by the FRACTIONAL distillation of petroleum. (v) Goal gas is obtained from destructive distillation of COAL. All these fuels contain MIXTURE of hdrocarbons which sources of energy. |
|
| 27. |
When metal carbides react with H_(2)O, the correct equations are |
|
Answer» `Al_(4)C_(3) +H_(2)O rarr CH -= CH` `CaC_(2) +2H_(2)O rarr Ca(OH)_(2) + C_(2)H_(2)` `Mg_(2)C_(3) + 4H_(2)O rarr 2Mg (OH)_(2) + CH_(3) - C -= CH` `Be_(2) C + 4H_(2) O rarr 2Be (OH)_(2) + CH_(4)` |
|
| 28. |
Which of the following fluoro-compounds is most likely to behave as a Lewis base ? |
|
Answer» <P>`CF_4` |
|
| 29. |
Which of the following is ketone compound? |
|
Answer» `H- underset(underset(O)(||))(C )-CH_(2) - underset(underset(OH)(|))(C H)-CH_(3)` |
|
| 30. |
The total wave function Psi may be symmetric (denoted by subscript S) or anti symmetric (denoted by the subscript A) depending on the symmtry of the function Psi and S. The function Psi = Psi_(S)S_(S) and Psi_(S) = Psi_(A)S_(A) are obviously symmetric whereas the function Psi_(A) = Psi_(A)S_(A) and Psi_(A) = Psi_(A)S_(S) are antisymmtric. All the functions Psi_(S) and Psi_(A) however do not corresponds to the state of an atom are governed by the Pauli principle. Pauli principle is regulated as one of the general principles underlying the modern quantum statistics. Now if two electron are supposed to have identical quantum numbers then transposition of these electrons will yield a state identical with the initial state and consequantly Psi_(12) = Psi_(21) (for symmetric) and Psi_(12) = - Psi_(21) (for antisymmetric). Hence the condition for satisfying only when the function is equal to zero. The number of radial nodes in 4d orbital is |
|
Answer» 3 |
|
| 31. |
The total wave function Psi may be symmetric (denoted by subscript S) or anti symmetric (denoted by the subscript A) depending on the symmtry of the function Psi and S. The function Psi = Psi_(S)S_(S) and Psi_(S) = Psi_(A)S_(A) are obviously symmetric whereas the function Psi_(A) = Psi_(A)S_(A) and Psi_(A) = Psi_(A)S_(S) are antisymmtric. All the functions Psi_(S) and Psi_(A) however do not corresponds to the state of an atom are governed by the Pauli principle. Pauli principle is regulated as one of the general principles underlying the modern quantum statistics. Now if two electron are supposed to have identical quantum numbers then transposition of these electrons will yield a state identical with the initial state and consequantly Psi_(12) = Psi_(21) (for symmetric) and Psi_(12) = - Psi_(21) (for antisymmetric). Hence the condition for satisfying only when the function is equal to zero. Which of the following set of quantum number is incorrect ? |
|
Answer» `n=5, n=0, L=0, s = +-(1)/(2)` |
|
| 32. |
Which bond is stronger sigma or pi ? Why ? |
|
Answer» Solution :(i) SIGMA bonds `sigma` are STRONGER than pi `(pi)` bonds. Because Sigma bonds are formed from bonding orbitals directly . Between the NUCLEI of thebonding ATOMS. Resulting in greater overlap and a strong sigma BOND ( axial overlapping). (ii) pi atrong results from overlap of atomic orbitals that are in contact through two are as of overlap ( lateral overlapping). Pi bonds are more diffused bonds than sigma bonds. |
|
| 33. |
Unit of vapour density is |
|
Answer» `mol KG^(-1)` |
|
| 34. |
Write the structures of followingalkanes (i) 2,3 - Dimethyl-6-(2- methylpropyl) decane (ii) 5-(2-Ethylbutyl)-3,3-dimethyldecane (iii)5-(1,2-Dimethylpropyl)-2-methylnonane |
Answer» SOLUTION :
|
|
| 35. |
Two elements P and Q combine to form a compound. If P has 2 and Q has 6 electrons in their outermosrt shell, what will be formula of the compound formed ? |
|
Answer» PQ `P - 2E^(-) to P^(2+)` `Q + 2e^(-) to Q^(2-)` SINCE both showa valency of 2, the formula for the compound will be PQ. |
|
| 36. |
Write the steps to be followed for writing empirical formula. |
|
Answer» Solution :Empirical formula shows the ratio of number of atoms of different elements in one molecule of the compound. Steps for finding the Empirical formula: The percentage ot the elements in the compound is determined by suitable methods and from the DATA collected, the empirical formula is determined by the following steps. (i) Divide the percentage of each element by its atomic mass. This will give the relative number of atoms of various elements present in the compound. (ii) Divide the atom value obtained in the above step by the smallest of them so as to GET a simple ratio of atoms of various elements. (iii) Multiply the FIGURES so obtained, by a suitable integer if NECESSARY in order to obtain whole number ratio. (iv) FINALLY write down the symbols of the various elements side by side and put the above numbers as the subscripts to the lower right hand of each symbol. This will represent the empirical formula of the compound. (v) Percentage of Oxygen =100- Sum of the percentage masses of all the given elements |
|
| 37. |
The structure of N(CH_(3)) is "………….." while that of N(SiH_(3))_(3) is "…………..". |
|
Answer» |
|
| 38. |
Which of the following compound is metallic acid ferromagnetic ? |
|
Answer» `CrO_2` |
|
| 39. |
What is the oxidation state ofS in H_2 SO_4, |
|
Answer» SOLUTION :`H_2SO_3:` 2(+1)+x+3 (-2)=0, x=4 Oxidation STATE of S in `H_2 SO_3`=+4. |
|
| 40. |
What characteristics do you expect from an electron deficient hydride with respect to its structure and chemical reaction? |
|
Answer» Solution :It is EXPECTED to be a Lewis acid. For example, DIBORANE `B_(2)H_(6) (DIMER of BH_(3))` forms a complex with LIH which gives `H^(-)` ion (Lewis base) `underset("LITHIUM hydride")(2LiH)+`underset("Diborane")(B_(2)H_(6)) to `underset("Lithium borohydride")(2Li^(+)[BH_(4)]^(-))` Similarly, it also combines with trimethuylamine (organinc compound) and carbon monoxide which act as Lewis base to form addition compound. `B_(2)H_(6)+underset("Trimethylamine")((CH_(3))_(3)) ddot(N) to underset("Addition compound")(2H_(3)B) leftarrow N(CH_(3))_(3)` `B_(2)H_(6)+CO to underset("Addition compound")((2H_(3)B) leftarrow CO` |
|
| 41. |
Which instrument can be use to measurement of pressure and temperature ? |
|
Answer» SOLUTION :(i) PRESSURE of atmosphere : Barometer Pressure of gas : MANOMETER (ii) Temperature : THERMOMETER `(""^(@)C, ""^(@)F, K)` |
|
| 42. |
When 1.82 g a of mixture of Al and an unkown metal. Arranged in the series of the standard reduction electrode potential below hydrogen, was dissolved in HCl, 0.672 L of H_2, measured at STP was liberated. To oxidise this mixture, 0.56 L of O_2 measured at STP was needed which unknown metal was taken? Determine the mass percentage of metals in the mixture. |
|
Answer» SOLUTION :(a). Only Al gives `H_2`, not the other METAL below `H_2`. `2Al+6"HCl"to2AlCl_3+3H_2` Ew of `H_2=" EQ of "Al (xg)` `(0.672)/(11.2)=((x)/(27))/(3)` `(1 " mol of "H_2=2 " Eq of "H_2=2.4L)` `(1 " Eq of "H_2=11.2L)` `thereforex=0.54 of Al` The weight of unknown metal `(M)=1.82-0.54=1.28g` (b). `4Al+3O_2to2Al_2O_3` `{:(4 mol Al, 3 " mol of "O_2),(4xx27g,3xx32g O_2):}` `4xx27g of Al implies3xx32g O_2` `0.54 of "Al "implies(3xx32)/(4xx27)xx0.54 g of O_2` `(1 " mol of "O_2=32g=22.4L)` Volume of `O_2=(22.4)/(32)xx(3)/(4)xx(32)/(27)xx0.54` `=0.336 L of O_2` `O_2` used for metal `M=(0.56-0.336)L=0.224L` Let M be monovalent. `1 " mol of "M= 1 " mol of "O_2` `implies(1.28)/("Atomic weight")=(0.224)/(22.4)" mol of "O_2` `therefore` Atomic weight`=(1.28)/(0.224)xx22.4` `=128` which is not possible So the metal is divelent `2 mol=1 " mol of "O_2` `therefore` Atomic weight `=(128)/(2)=64` i.e., metal is copper. |
|
| 43. |
Toluene reacts with halogen in presence of iron (III) chloride giving ortho and para-halo compounds. The reaction is : |
|
Answer» FREE redical addition REACTION |
|
| 44. |
Which of the following is not involved in the formation of photochemical smog ? |
|
Answer» NO |
|
| 45. |
Which of the following statements are correct ? (I) In ICl_(2)-CIF_(3) and TeCl_(4), the number of ions pairs (s) of electrons on central atoms are 3,2 and 1 respectively. (II) Amongst CO,CO_(2),CO_(3)^(2-),CH_(2)OH the correct order from the weakest to the strongest carbon-oxygen bond -=CH_(3)OH lt CO_(3)^(2-) lt CO_(2) lt CO. (III) The I^(-),b^(-)idisation of boron in BF_(2) is the same which nitrogen has in CINO molecule. |
|
Answer» Only `(I) & (III)` 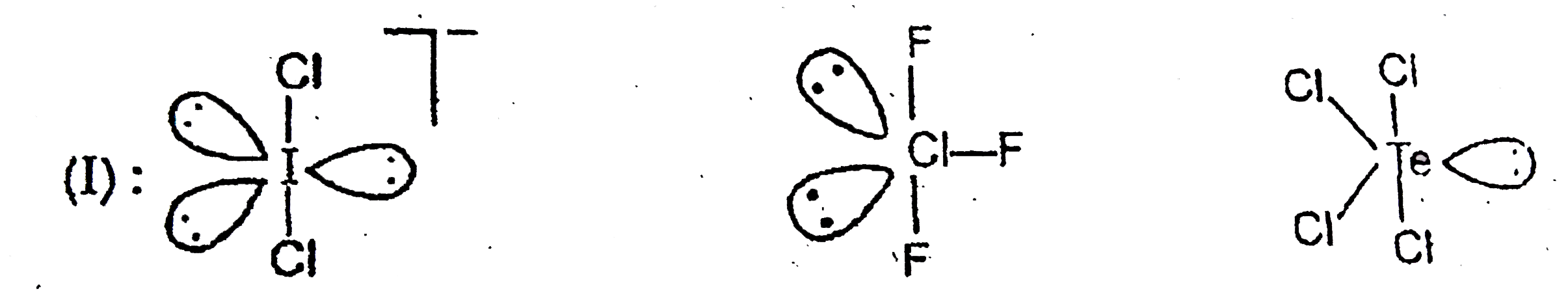 `(II)" ":C-O=1.43Å,C=O" "1.23Å,C=O" "1.09Å` `CO,Coverset(LARR)(-=)=O` partial triple bond character, borid LENGTH is intermediat of `C=O` and `C-=O` bonds `CO_(2),O=C=O,` double bond length 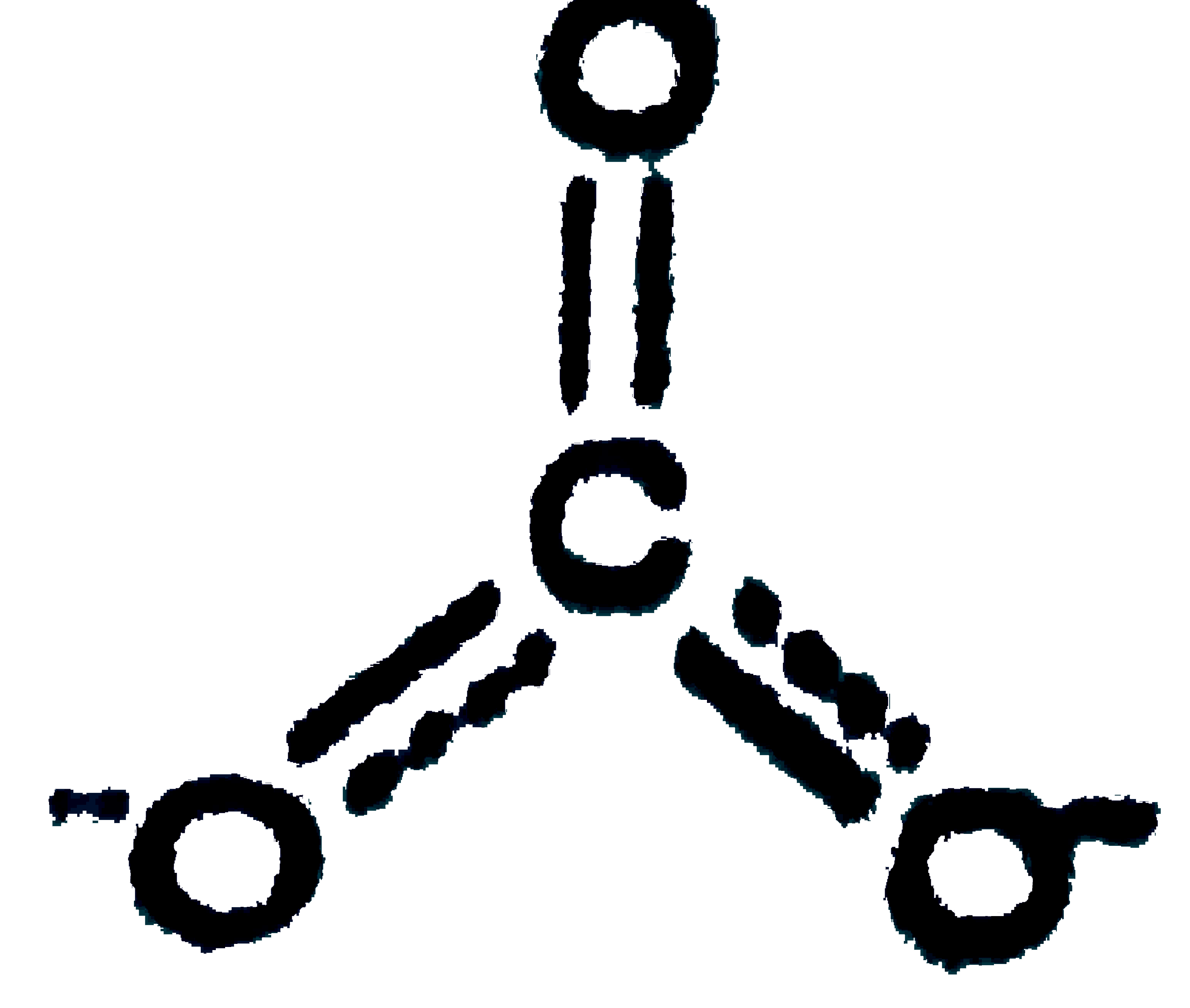 partial double bond character, intermediate of `C=O` and `C-O` partial double bond character, intermediate of `C=O` and `C-O`  single bond length. single bond length. 
|
|
| 46. |
Which type of bond is observed in O-Chlorophenol ? |
|
Answer» Metallic BOND |
|
| 47. |
What is the dihedral angle of H_2O_2 in gas and solid phase ? |
|
Answer» `111.5^(@)` and `90.2^(@)` |
|
| 48. |
Which negative ion is present in ortho-silicate ? |
|
Answer» `SiO_4^(-4)` |
|
| 49. |
What are thhe atomic numbers of elements whose outermost electrons are represented by (i) 3s^(1) (ii) 2p^(3) and (iii) 3d^(6) ? |
|
Answer» (ii) When last shell is `2p^(3)`, complete E.C. `=1s^(2) 2s^(2) 2p^(3)` (iii) When last shell is `3d^(6)`, complete E.C.`=1s^(2) 2s^(2) 2p^(6) 3s^(2) 3p^(6) 4S^(2) 3d^(6)`. |
|