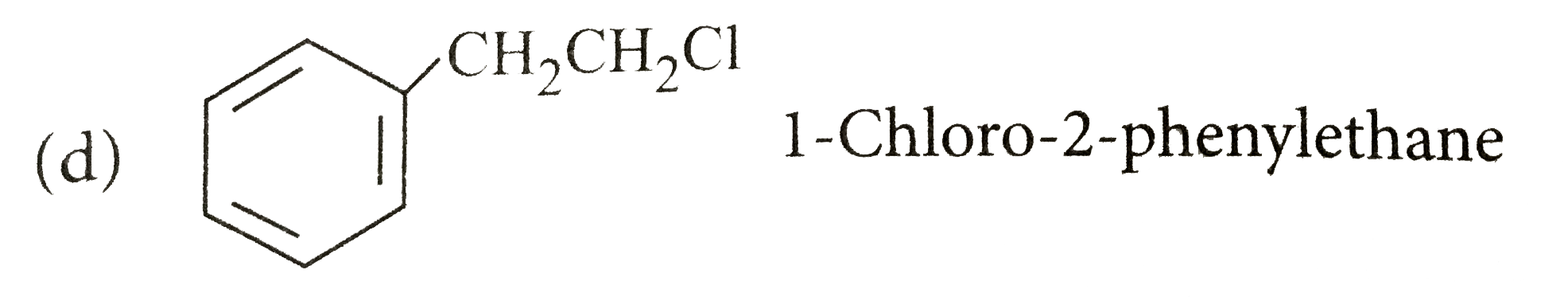Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following is decomposition reaction |
|
Answer» `2HgO to 2Hg +O_(2)` |
|
| 2. |
Write the IUPAC names of the following compounds. |
Answer» SOLUTION :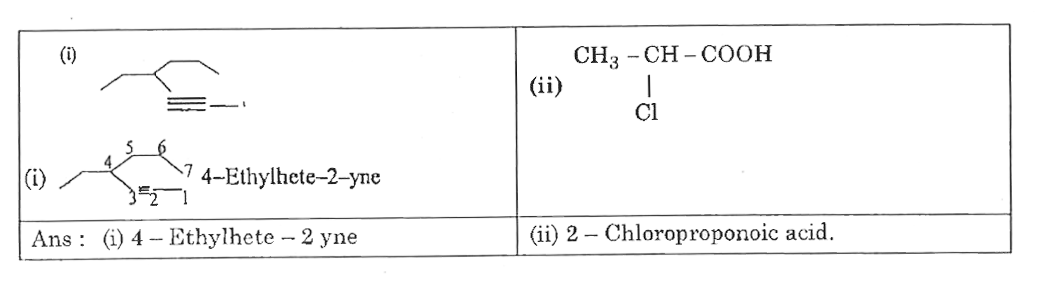
|
|
| 3. |
Which element containing Uuh series? |
|
Answer» 104 Atomic number = 116 |
|
| 4. |
Write the structures and IUPAV names of the functional isomers having the molecular formula C_(2)H_(5)O_(2)N. |
Answer» SOLUTION :THREE functional isomers and their IUPAC name are : 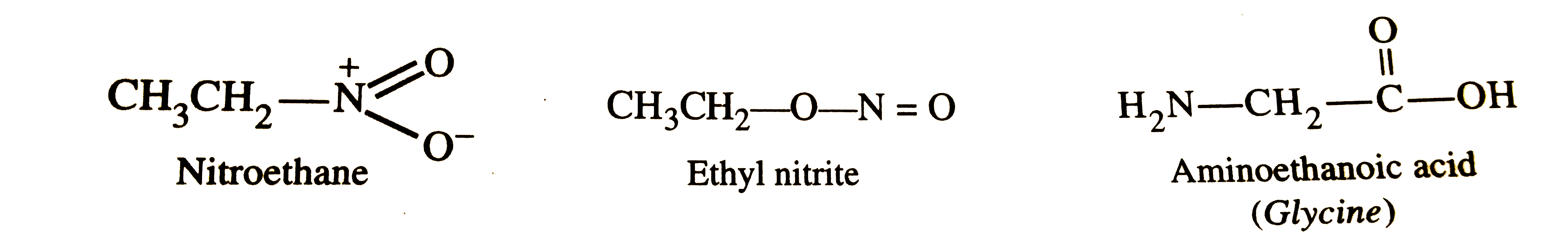
|
|
| 5. |
which of the following processes are accompanied by an increase of entropy . (i) Dissolution of iodine in solvent (ii) HCl added to AgNO_(3) solution and precipitate of AgCl is obtained (iii) A partition is removed to allow two gases to mix. |
|
Answer» (i) & (II) |
|
| 6. |
Which statement is/are correct. |
|
Answer» No primary KINETIC isotope effect is observed during nitration of BENZENE |
|
| 7. |
Which of the following statements are wrong? |
|
Answer» pH of NEUTRAL water is always7.0 (b) is wrong because at the end point, pH depends upon the NATURE of acid and base being titrated (c) is wrong because higher the pH than 7, more basic is the solution (d) is correct because AgCl DISSOLVES in `NH_(3) ` to form a soluble complex. |
|
| 8. |
Which is the correct sequence of solubility of carbonates of alkaline earth metals ? |
|
Answer» `BaCO_(3) GT SrCO_(3) gt CaCO_(3)gt MgCO_(3)` |
|
| 10. |
What should be the minimum concentration ofNH_(4)Cl that must be present to prevent precipitation, when 0.01 M NH_(4)OH is added to 0.01 (M)M^(2+) solution? (K_(b) of NH_(4)OH = 1.75' 10^(-5) and K_(sp) of M(OH)_(2) = 44.1' 10^(-13)) |
|
Answer» `0.025 M` `K_(b) = 1.75 xx 10^(-5) = ((b+x)x)/(0.01)` (`x = 2.1' 10^(-6)` from `K_(sp)` of `M(OH)_(2)`) `(b xx 2.1 xx 10^(-6))/(0.01) = 1.8 xx 10^(-5)` `rArr (1.75)/(2.1) xx (10^(-7))/(10^(-6)) = (1.75)/(10) = 0.083 M` |
|
| 11. |
What are hybridisation states of each carbon atom in the following compounds ? CH_(2) = C = O, CH_(3)CH= CH_(2), ""(CH_(3))_(2) CO,"" CH_(2) = CHCN"and "HCONH_(2) |
Answer» SOLUTION :
|
|
| 12. |
What is soda ash ? How it obtained ? |
|
Answer» Solution :Sodium carbonate DECAHYDRATE commonly known as WASHING soda `Na_(2)Co_(3).10H_(2)O`. Upon heating, it looses the water of crystallization to FORM monohydrate Above 373K, the monohydrate becomes COMPLETELY ANHYDROUS and changes to a white powder called soda ash `Na_(2)CO_(3).10H_(2)O overset(/_\^(@))to Na_(2) CO_(3).H_(2)O + 9H_(2)O` `Na_(2) CO_(3).H_(2) overset(/_\^(@))to underset(Soda ash)(Na_(2)CO_(3)) + H_(2)O` |
|
| 13. |
Which of the pairs of gases diffuse at a slower rate than CO_(2)? |
|
Answer» `H_2 & He` |
|
| 14. |
Which of the following can act as sink for CO_2 and SO_2 |
| Answer» Solution :Sea water | |
| 15. |
Which atom of H is replaced by Br_(2) in presence of P from an acid : |
| Answer» Solution :SEE PROPERTIES of acids | |
| 16. |
Which of the following is not a protonic acid ? |
|
Answer» `SO_(2)(OH)_(2)` |
|
| 17. |
Which element do you think would have been named by Seaborg's group. |
| Answer» SOLUTION :SEABORGIUM (SG) or UNNILHEXIUM | |
| 18. |
The oxidation number of iron in brown ring complexes +x. What is value of x. |
|
Answer» Solution :The BROWN ring complex compound is GIVEN as: `[Fe(H_(2)O)_(5)NO] SO_(4)`. The complex ion is `[Fe(H_(2)O)_(5))NO]^(2+)` Oxidation number of `H_(2)O` is zero and NO is +1. Oxidation state of Fe is +1. Experimentally, the number of UNPAIRED ELECTRONS also support +1 state of Fe (But not +2 state). |
|
| 19. |
Which of the following set of quantum numbers belong to highest energy |
|
Answer» n=4,l=0,m=0,`s=+ 1/2` |
|
| 20. |
What is the IUPAC name of the insecticise DDT? Why is their use banned in most of the countries? |
|
Answer» SOLUTION :(i) The IUPAC name of the insecticide DDT is `P_(1)P^(1)`-dichloro-dipenyl trichloro ethane. (ii) Even DDT is an effective insecticide. Not a day it is banned because of its LONG term toxic affects. (iii) DDT is very PERSISTENT in the environment and it has a high affinity for fatty TISSUES. as a result, DDT gets accumulated in animals tissue fat, in particular that of birds of prey with SUBSEQUENT thinning of their eggs shells and impacting their of reproduction. that is why DDt. is banned in most of the countries. |
|
| 21. |
Which of the following reactions is/are feasible: |
|
Answer» `(i) & (II)` |
|
| 22. |
What isionizationenergyof an atom ? |
| Answer» SOLUTION :The energyrequireto moveelectronin `n_(PROP)`orbit is knownas ionization ENERGY | |
| 23. |
What is the integral values can take azimuthal quantum number ? |
|
Answer» ZERO to N |
|
| 24. |
What will be the minimum pressure required to compress 500dm^(3) of air at 1 bar to 200 dm^(3) at 30^(@)C ? |
|
Answer» <P> Solution :`P_(1)=1` bar,`V_(1)=500 dm^(3)``P_(2)=?,V_(2)=200 dm^(3)` As TEMPERATURE remains constant at `30^(@)C`, applying Boyle's law, `P_(1)V_(1)=P_(2)V_(2)` `1 barxx500 dm^(3)=P_(2)xx200 dm^(3)"or"P_(2)=(500)/(200)bar=2.5 bar` |
|
| 25. |
Which of the following names is not correctly written in front of the compound? |
|
Answer»
|
|
| 26. |
Which of the following statements is/are incorrect ? |
|
Answer» Nitrobenzene will give meta-nitrotoluene on reaction with `CH_3Cl//AlCl_3` (B)Chloro group is ortho-para director. (C )In Friedal Craft reaction the electrophile carbocation rearranges. (D)To AVOID polyalkylation aromatic substrate is taken in excess. |
|
| 27. |
The viscosity of four liquids P, Q, R and S are 85, 11.4, 18 and 12.3 respectively, then which flows slowly |
| Answer» ANSWER :A | |
| 28. |
Which of thefollowingrepresentsthe correctorderof increasingfirstionizationenthalpy for Ca, Ba , S, Se, and Ar ? |
|
Answer» `Ca lt Ba lt S lt Se lt Ar` `Ba lt Ca lt Se lt S lt Ar `,i.e., option (d) is correct. |
|
| 29. |
Which process is suitable for the purification of aniline ? |
|
Answer» SIMPLE DISTILLATION |
|
| 30. |
What is the percentage of water of crystallisation in a pure sample of washing soda (Na_(2)CO_(3).10 H_(2)O) ? |
|
Answer» PERCENTAGE of WATER of CRYSTALLISATION `= ((180u))/((286u))xx100=62.94%`. |
|
| 31. |
Which hybird orbitals are used by carbon atoms in the following molecules?CH_3-COOH |
Answer» SOLUTION :
|
|
| 32. |
Who had give information of covalent bond ? |
| Answer» SOLUTION :KOSSEL & LEWIS has given the octet rule for covalent bond. | |
| 33. |
Which one of the following is non-degradable pollutants ? |
|
Answer» DDT |
|
| 34. |
Which of the following species is not electrophile in nature ? |
|
Answer» `Cl^(+)` |
|
| 35. |
When alkyl halides are heated with dry Ag_(2)O they give, |
| Answer» Solution :diethyl ether | |
| 36. |
Which of the following electrodes will act as anodes, which connected to Standard Hydrogen Electrode ? |
|
Answer» `Al//Al^(+3)" "E^(Ө)=-1.66` |
|
| 37. |
Which one of the following is a state function? |
|
Answer» PRESSURE |
|
| 38. |
Which of the following will give acetic acid on acid hydrolysis ? |
|
Answer» ETHYL acetate |
|
| 39. |
When 5 litres of a gas mixture of methane and propane is perfectly combusted at 0^(@)C and 1 atmpsphere, 16 litres of oxygen at the same temperature and pressure is consumed. The amount of heat released from this combustion in kJ (Delta H _("comb.)) (CH _(4)) = 890 kJ mol ^(-1) , Delta H _("comb.) (C_(3) H _(8)) = 2220 kJ mol ^(-1)) |
|
Answer» 38 |
|
| 40. |
Use of hot air balloon in meteorological observation is based on which gas law ? Explain. |
| Answer» Solution :Charles law : According to Charles law, gases expand on heating. Since mass of gas in unchanged, INCREASE in volume DECREASES density. Thus hot air is less dense than COLD air. This causes hot air balloon to rise displacing the COOLER air of the ATMOSPHERE. | |
| 41. |
Why BaSO_(4) is insoluble whereas BeSO_(4) is soluble in water ? |
| Answer» Solution :The lattice enthalpy of `BaSO_(4)` is MUCH than its hydration enthalpy and hence it is INSOLUBLE in WATER . But on, the other hand , hydration enthalpy of `BeSO_(4)` is much higher than its lattice enthalpy because of small size of `Be^(2+)` ions . THEREFORE , it is HIGHLY soluble in water . | |
| 43. |
Which of the following compounds does not form an ozonide? |
|
Answer» ETHANE |
|
| 44. |
What is shape of theorbital having I - I andm = 0 ? |
|
Answer» TRIANGULAR plana |
|
| 45. |
What mass of hydrogen peroxide will be present in 2 litres of a 5 molar solution ? Calculate the mass of oxygen will be liberated by the decomposition of 200 mL of this solution . |
|
Answer» Solution :Mol. Mass of `H_(2)O_(2)=34 g "mol"^(-1)` `therefore 1 L` of M solution of `H_(2)O_(2)` will contain `H_(2)O_(2)=34xx5` g `therefore 2 `L of 5 M solution will contain `H_(2)O_(2)=34xx5xx2=340 g` or 200 mL of 5 solution will contain `H_(2)O_(2)=(340)/(2000)xx200=34` g Now`underset(68 g)(2H_(2)O_(2))to 2H_(2)O + underset(32g)(O_(2))` Now 68 g of `H_(2)O_(2)` on DECOMPOSITION will give `O_(2)=32 g` `therefore 34 g ` of `H_(2)O_(2)` on decomposition will give `O_(2)=(32)/(68)xx34=16` g |
|
| 46. |
Writepostulatesof Bohr.smodelofhydrogenatom |
|
Answer» Solution :Nells Bohr (1913) was the firstto explainquqntitatively thegeneralfeaturesof hydrogenatomstructure andtisspectrum. Pitulatesof model: (i) Theelectron is the HYDROGENATOM can movearoundthenucleusin acircularpathcalledorbitsstationarystatesor allowedenergystates. Theseorbitsare arrangestconcentrically aroundthe nucleus. (ii) The energyof anelectronin the ORBIT doesnot changewith timeHowever the electron willmovefroma lowerstationarystateto ahigherstationarystatewhenrequiredor ENERGYIS emitted whenelectronmovesdoesnottakeplace in acontinuouschange (iii) The frequency of radiationabsorbedoremitted whentransitionoccursbetweentwostationarystatesthatdifferin energyby Where`E_(1)`and `E_(2)`are the energies of lowerand higherallowedenergystatesrespectivelythisexpressionis commonlyknown asbohrfrequencyrule Thusanelectroncan move only in thoseorbitsfor whichitsangularmomentumisintegralmultiple of `((h)/( 2pi))` that iswhyonlycertain fixedorbitsare ALLOWED. |
|
| 47. |
Write equilibrium constants of followingreactions.(1)2PCl_(3(g)) + O_(2(g)) hArr 2POCl_(3(g)) (2)2SO_(3(g)) hArr 2SO_(2(g)) + O_(2(g)) (3) H_2O_((g)) + Cl_2O_((g)) hArr 2HOCl_((g)) (4) CuSO_(4(s)) + 5H_2O_((s)) hArr CuSO_(4(s)) + 5H_2O_((g)) |
|
Answer» SOLUTION :(1)`K_c=[POCl_3]^2/([PCl_3]^2[O_2])` (2)`K_c=([SO_2]^2[O_2])/([SO_3]^2)` (3)`K_c=[HOCl]^2/([H_2O][Cl_2O])` (4)`K_c=[H_2O]^5` |
|
| 48. |
What is meant by octet structure? |
| Answer» Solution :The ATOMS having 8 electrons in the OUTER most orbit or having valence electronic CONFIGURATION. `ns^(2)p^(6)`. | |
| 49. |
Which of the following compounds will exhibit cis-trans isomerism? |
|
Answer» 2-Butene |
|
| 50. |
Which one of the following species is stable in aqueous solution? |
| Answer» Answer :A | |