Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
When a beam of photons of a suitable frequency strikes the metal surface, electrons are emitted from metal. This process is called photoelectric effect. The effect occurs when energy of incident beam is greater than that of the work function The graph between photo electron current (c) and intensity of photon (I) |
|
Answer»

|
|
| 2. |
When a beam of photons of a suitable frequency strikes the metal surface, electrons are emitted from metal. This process is called photoelectric effect. The effect occurs when energy of incident beam is greater than that of the work function. Which of the following graphs represents photoelectric effect ? |
|
Answer»

|
|
| 3. |
When a beam of photons of a suitable frequency strikes the metal surface, electrons are emitted from metal. This process is called photoelectric effect. The effect occurs when energy of incident beam is greater than that of the work function. Kinetic energy of photoelectron varies with |
|
Answer» Nature of metal |
|
| 4. |
Which are the particulate pollutants ? |
|
Answer» Solution :Particulate pollutant are of two types : (a) Viable particulate pollutant : Bacteria, Fungi, Moulds, Algae ETC. (B) Non-viable particulate pollutants : Smoke, DUST, Mist, FUMES, Smog. |
|
| 5. |
Which of the following has the ,highest dipole moment |
|
Answer» `AsH_(3)` |
|
| 6. |
What is the importance of heavy water ? |
|
Answer» Solution :(i) Heavy water is extensively used as a moderator in nuclear reactors. (ii) It is widely used as a TRACER COMPOUND for STUDYING the mechanism of many reasons. (III) Heavy water is used in preparation of other deuterium compounds like `CD_2 , D_2SO_4` , etc. |
|
| 7. |
Which of the following elements were unknown at that time of Mendeleev? |
|
Answer» NA, Mg |
|
| 8. |
Which of the following is not among short comings of Bohr's model? |
|
Answer» Bohr theory could not ACCOUNT for the fine lines in the atomic spectrum |
|
| 9. |
Which one of the following is used as a standard for atomic mass? |
| Answer» Answer :A | |
| 10. |
Which one of the following molecular hydrides acts as a Lewis acid ? |
|
Answer» `CH_(4)`  This is because BORON do not have SUFFICIENT valence electrons to from conventional two electron bond with all the H-atoms. |
|
| 11. |
The structure of benzene can also be explained by |
|
Answer» Valence BOND theory |
|
| 12. |
Which of the following is planar in all its conformation |
|
Answer» `CH_(2)=CH-CH=CH_(2)` |
|
| 13. |
Which S_(N)1 reaction would you expect to take place more rapidly ? (a) (i) (CH_(3))_(3)C Cl+H_(2)O to (CH_(3))_(3)COH+HCl Or (ii) (CH_(3))_(3)C Br+H_(2)O to (CH_(3))_(3)COH+HBr (b) (i) (CH_(3))_(3)C Cl+H_(2)O to (CH_(3))_(3)COH + HCl Or (ii)C_(5)H_(5)Cl+H_(2)O to C_(5)H_(5)OH + HCl (c) (i) (CH_(3))_(3)C Cl+ H_(2)O to (CH_(3))_(3)COH + HCl Or (ii) (CH_(3))_(3)C Cl+ CH_(3)OH to (CH_(3))_(3)COCH_(3)+ HCl (d) (i)(CH_(3))_(3)C Cl(1.0M)+CH_(3)CH_(2)O^(-)(1.0M) overset(C_(2)H_(2)OH)to (CH_(3))_(3)COCH_(2)CH_(3)+Cl^(-) Or (ii) (CH_(3))_(3)C Cl(1.0M)+CH_(3)CH_(2)O^(-)(2.0M) overset(C_(2)H_(2)OH)to (CH_(3))_(3)COCH_(2)CH_(3)+Cl^(-) |
|
Answer» Solution :(a) Reaction (II) because `Br^(-)` ION is a better leaving group than `Cl^(-)` ion, (b) Reaction (i) because the reactant is a tertiary halide. MOREOVER, phenyl HALIDES are not reactive in `S_(N)1` reactions. (c) Reaction (i) because water is more polar solvent than methanol and `S_(N) 1` reaction takes place faster in more polar solvent. (d) Both reactions occur at the same rate because `S_(N)1` reactions are independent of the concentration of the NUCLEOPHILE. |
|
| 14. |
Which of the following process do not involveabsorption of energy ? |
|
Answer» `O^(-)(G) +e^(-) to O^(2-) (g)` |
|
| 15. |
Water drops stick to a glass surface due to |
|
Answer» Cohesion |
|
| 16. |
Which of the following reaction is most suitable for the preparation of n-propylbenzene? |
|
Answer» Friedel-Crafts alkylation |
|
| 17. |
Which of the following is least thermally stable |
|
Answer» `MgCO_(3)` |
|
| 18. |
Which of the following is not a general characteristic of equilibrium involving physical process |
|
Answer» Equilibrium is possible only in a closed system at a given temperature. |
|
| 19. |
What are the bonds present in ammonium chloride? |
|
Answer» Only IONIC and dative |
|
| 20. |
Which among the following is the strongest base ? |
|
Answer» `C_2H_5NH_2 "" K_b=5.6xx10^(-4)` |
|
| 21. |
Which of the above mentioned reactions would yield white precipitate which on standing converts into black precipitate? |
|
Answer» 2 `PbS_(2)O_(3) rarr PbS darr`(Black) 4 `Bi_(2)(S_(2)O_(3))_(3) rarr Bi_(2)S_(3)darr` (Black) 5 `Cu_(2)(S_(2)O_(3))rarr Cu_(2)Sdarr` (Black) |
|
| 22. |
The wave function of a molecular orbital formed by reinforce of wave functions of Psi_(A)and Psi_(B) of atomic orbital A and B is represented as |
|
Answer» `psi_(A)+psi_(B)` |
|
| 23. |
When the vapour distillation is applied? |
| Answer» SOLUTION :STEAM distillation is applied to separate SUBSTANCES which are stem volatile and are immiscible with WATER | |
| 24. |
When radioactive minerals like clevelte, monozite and pitch blende are heated to 1273 K in vacuo the noble gas obtained is |
|
Answer» Rn |
|
| 25. |
Which of the following increasing order is not correct as per the property indicated against it? |
|
Answer» `CsCl lt RbCl lt KCl lt NaCl lt LiCl` (lattice ENERGY) `Cs^(+) lt Rb^(+) lt K^(+) lt Na^(+) lt Li^(+)` |
|
| 26. |
What is unuranium element ? |
| Answer» SOLUTION :After ELEMENTS of URANIUM are KNOWN as unurenium elements. | |
| 27. |
Which one of the following has maximum acid strength ? |
|
Answer» <P>`o-`Nitrobenzoic ACID 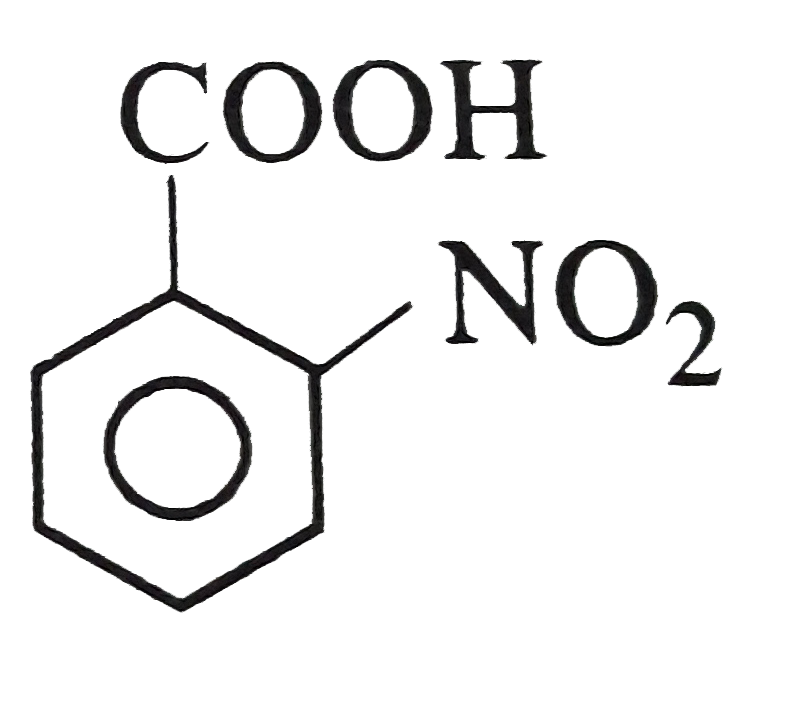
|
|
| 28. |
Which of the following magnetic moment values will correspond to the highest ionisation energy for the element which is placed in 4th period and 7th group of long form of periodic table? |
|
Answer» `2sqrt(2)` |
|
| 29. |
Which of the following can be prepared in good yield by Wurtz reaction? |
|
Answer»
|
|
| 31. |
Which of the following statements about photochemical smog is incorrect? |
|
Answer» it has high concentration of oxidizing AGENTS |
|
| 32. |
The reduction of benzoyl chloride with H_(2)//Pd - BaSO_(4) produces |
|
Answer» Benzyl alcohol |
|
| 33. |
The weight percentage of carbon dioxide and carbon disulphide are respectively 27.27% and 15.79%. What is the composition of oxide of sulphur, if the weight percentage of oxygen is 50? |
|
Answer» Solution :WEIGHTOF OXYGEN that combines with 1 G of Carbon `=(72.73)/(27.27)=2.67g` Weight of Sulphur that combines with 1 g of Carbon `=(84.21)/(15.79)=5.33g` Ratio of weight of S and O that can combin with same weight of carbon is `5.33:2.67=2:1` The simple multiple ratio is `1:1` The oxide is sulphur DIOXIDE, `SO_(2)` |
|
| 34. |
Which of the following is relatively weak acid ? (pK_(a) values are given in brackets) |
|
Answer» HA (6,8) |
|
| 35. |
Yellow light emitted from a sodium lamp has a wavelength (lamda) of 580nm. Calculate the frequency (v) and the wave number (bar(v)) of the yellow light |
|
Answer» Solution :`LAMDA = 580 nm = 580 xx 10^(-9) m` Frequency, `V = (c)/(lamda) = (3.0 xx 10^(8) ms^(-1))/(580 xx 10^(-9) m) = 5.17 xx 10^(14) s^(-1)` Wave number, `bar(v) = (1)/(lamda) = (1)/(580 xx 10^(-9) m) =1.72 xx 10^(6) m^(-1)` |
|
| 36. |
Which the following statements about are true |
|
Answer» `H_(2)O_(2)` is used to clean oil paintings |
|
| 37. |
What is the correct expression for the representation of the solubility product constant of Ag_(2) CrO_(4) ? |
|
Answer» `[AG^(+)]^(2) [CrO_(4)^(2-)]` |
|
| 38. |
The shape of acetylene molecule is |
|
Answer» linear |
|
| 39. |
Write the structure of P and Q |
|
Answer» |
|
| 40. |
TheStandard enthalpy of formation of gaseous H_(2)O at 298K is -242 mol^(-1) . Calculate DeltaH^(@)"at"373K given the following values of the molar feat capacities at constant pressure . Molar heat capcity of H_(2)O(g)=33.5JK^(-1)mol^(-1) molar heat capacity of H_(2)(g)=28.8JK^(-1)mol^(-1) Molar heat capacity of O_(2)(g)=29.4 JK^(-1)mol^(-1) {Assume that the heat capacities are independent of temperature.} |
|
Answer» `508KJ mol^(-1)` |
|
| 41. |
Three different oxides of nitrogen each contain 7.00 g of hydrogen and weigh 15.0 g, 23.0g and 19.0g, respectively. What are their empirical formulas? |
|
Answer» `NO, NO_(2), N_(2)O_(3)` |
|
| 42. |
What is Polar Vortex ? What is its effect ? |
| Answer» Solution :A tight WHIRLPOOL of WIND FORMED in the stratosphere which surrounds Antarctica is called POLAR Vortex | |
| 43. |
The solubility of BaSO_4in water is 2.42 xx 10^(-3) gL^(-1)at 298 K. The value of its solubility product (Ksp) will be (Given molar mass of BaSO_4 = 233 g "mol"^(-1)) |
|
Answer» `1.08 xx 10^(-8) "mol"^2 L^(-2)` `K_(sp) " of " BaSO_4 = (s)^2` ` = ((2.42 xx 10^(-3))^2)/(233))` ` = 1.078 xx 10^(-10) "mol"^2 L^(-2)` `1.08 xx 10^(-10) "mol"^2 L` |
|
| 44. |
Which of the following is/are correct about the redox reation? MnO_(4)^(-) +S_(2)O_(3)^(2-) +H^(o+) rarr Mn^(2+) +S_(4)O_(6)^(2-) |
|
Answer» 1 mol of `S_(2)O_(3)^(2-)` is oxidised by 8 mol of `MnO_(4)^(-)` `2S_(2)O_(3)^(-) rarr S_(4)O_(6)^(2-) +2e^(-) (n = (2)/(2) = 1)` EQ of `MnO_(4)^(-) -=` Eq of `S_(2)O_(3)^(-)` `5xx `moles of `MnO_(4)^(-) -= 1 xx` moles of `S_(2)O_(3)^(2-)` `:. 1` mol of `S_(2)O_(3)^(2-) = 5` mol of `MnO_(4)^(-)` (B) pH changes from 4 to 10 (acidic to strongly basix) `e^(-) + MnO_(4)^(-) rarr MnO_(4)^(2-) (n = 1)` `S_(2)O_(3)^(2-) rarr 2SO_(4)^(2-) +8e^(-) (n = 8)` Eq of `MnO_(4)^(-) =` Eq of `S_(2)O_(3)^(-)` `:. 1` mol of `S_(2)O_(3)^(2-) = (1)/(8)` mol of `MnO_(4)^(-)` Hence with change of pH from 4 to 10, will change the stiochiometryof reaction and alo changes the product. (c) pH changes from 4 to 7 (acidic to neutral medium) `3E^(+) +MnO_(4)^(-) rarr MnO_(2) (n = 3)` `S_(2)O_(3)^(2-) rarr 2HSO_(4)^(-) +8e^(-) (n = 8)` Hence it will also effect the stoichiometry of reaction and nature of product. (d) At `pH = 7, S_(2)O_(3)^(2-)` is oxidised to `HSO_(4)^(-)` ion |
|
| 45. |
Which one of the following alkene will react fastest with H_2 under catalytic hydrogenationcondition ? |
|
Answer»
|
|
| 46. |
What do you mean by conformation and conformer (Rotamers) |
|
Answer» SOLUTION :Alkanes contain carbon-carbon sigma `(sigma)` bonds. ELECTRON distribution of the sigma molecular orbital is symmetrical around the internuclear axis of the C-C bond which is not disturbed due to rotation about its axis. This permits free rotation about C-C single bond. This rotation RESULTS into different SPATIAL arrangementts of atoms in space which can change into one another. Such spatial arrangements of atoms which can be converted into one another by rotation around a C-C single bond are CALLED conformations or conformers or rotamers. |
|
| 47. |
What mass of ice at 0.0^(@) C must be added to 100g H_(2)O at 25.0^(@)C to cool It to 0.0^(@)C? The heat of fusion of ice is 334Jg^(-1) |
|
Answer» `1.25g` |
|
| 48. |
Which of the above group has deactivator groups ? |
|
Answer» SOLUTION :Deactivator GROUPS : `-COCH_(3), -NO_(2), -Cl, -Br, -COOH` |
|
| 49. |
Write the folllowing redox rections using half equations (i) Zn(s) + PbCI_(2)(aq) rarr Pb(s)+ZnCI_(2)(aq) (ii)2Fe^(3+)(aq)+2I^(-)(aq)rarrI_(2)(s)+2 Fe^(2+)(aq) (iii)2Na(s)+CI_(2)(g)rarr2 NacI(s) (iv)Mg(s)+CI_(2)(g)rarrMgCI_(2)(s) (v)Zn(s)+2H^(+)(aq) rarr Zn^(2+)(aq)+H_(2)(g) In each of the reactions gien above mention (a) Which reactant is oxidized ?to what ?(b) which reactant is hte oxidiser ? (c )which reactant is reduced ? To what ?(d) which reactant is hte reducer? |
|
Answer» Solution :(i) `ZN(s) rarr Zn^(2+)+2e^(-) ("oxidation") , Pb^(2+)(aq)+2e^(-) rarrPb(s)("REDUCTION")` Zn is oxidised to `Zn^(2+), Pb^(2+) ("reduction"), Pb^(2+)` is the oxidiser and Zn is the REDUCER (ii) `2Fe^(3+)+2e^(-) rarr 2Fe^(2+) ("reduction"), 2I^(-)rarrI_(2)+2e^(-)("oxidation")` `Fe^(3+)` reduced to `Fe^(2+) I^(-)` is oxidiased to `I_(2), I^(-)` is the reducer and `Fe^(3+)` is the oxidiser (iii)`2Na rarr2Na^(+)+2e^(-)("oxidation"), CI_(2)+2e^(-)rarr2CI^(-)`(reduction) Na is oxidised to `Na^(+) "and" CI_(2)` is reduced to `CI^(-), CI_(2)+2e^(-) rarr 2CI^(-)` (reduction ) Mg is oxidised o `Zn^(2+)` while `H^(+)` is reduced to `H_(2)`, Zn is ht reducer and `H^(+)` is the oxidiser |
|
| 50. |
Which one is correct for ozone layer ? |
|
Answer» It prevent all rays of SUNLIGHT. |
|















