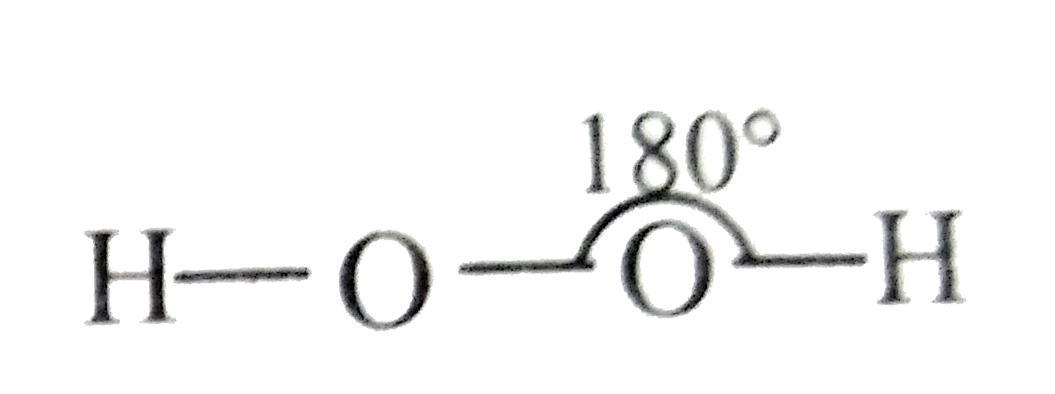Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Why are BeSO_(4) and MgSO_(4) readily soluble in water while CaSO_(4) , SrSO_(4) and BaSO_(4) are insoluble ? |
| Answer» SOLUTION :Because of big size of `SO_(4)^(2-)` ion as compared to `Be^(2+)` and `Mg^(2+)` ions , the lattice enthalpies of `BeSO_(4)` and `MgSO_(4)` are almost the same . But because of small size of `Be^(2+)` and `Mg^(2+)` ions their hydration enthalpies are much higher than their corresponding lattice enthalpies . As a RESULT , `BeSO_(4)` and `MgSO_(4)` are highly soluble in water . | |
| 2. |
Which statements are correct for the peroxide ion- |
|
Answer» it has five completely filled anti-bonding molecular orbitals `(sigma_(1s))^(2)(sigma_(1s)^(**))^(2)(sigma_(2s))^(2)(sigma_(2s)^(**))^(2)(sigma_(2p_(z)))^(2)` `(pi_(2px))^(2)(pi_(2py))^(2)(pi_(2p_(x))^(**))^(2)(pi_(2p_(y))^(**))^(2)` bond order`=(N_(b)-N_(a))/(2)=(10-8)/(2)=1` THUS, there is no unpared electron present for which it becomes diamagnetic. its total no. of electrons=18 (same as that of Ne). |
|
| 4. |
Which of the following is wrong for Bohr's model ? |
|
Answer» It establishes STABILITY of atom |
|
| 5. |
Which colour is shown by Calcium salts in flame test? |
|
Answer» LIGHT Green |
|
| 6. |
Which of the following alkanes may be synthesized from a single alkyl halide by a process involving coupling reaction? |
|
Answer» 2-Methylbutane 
|
|
| 7. |
Which one of the following alkali metals gives hydrated salts? |
|
Answer» Li |
|
| 8. |
X. and .Y. are two different solutions of hydrochloric acidwith concentrations 5M and 2M respectively. If one litre of 3M hydrochloric acid is to be prepared by mixing only X and Y calculate their volumes. |
|
Answer» Solution :Volume of solution .X. is V mL Volume of solution .Y. is (1000-V)mL MOLARITY desired to be obtained =3M Volume desired to be obtained =1000mL `M=(Vxx5(1000-V)2)/(1000)=3` SOLVING, V=333.3ml The volumes of Solutions .X. and .Y. are to be MIXED are RESPECTIVELY 333.3mL and 666.7mL. |
|
| 9. |
Which of the following halide is covalent |
|
Answer» `BeCI_(2)` |
|
| 10. |
Which of the following statements is // are correct |
|
Answer» Catenation is the property of self linking of identical atoms among themselves giving rise to CHAINS and rings |
|
| 11. |
Wrong reaction of the following |
|
Answer» `Li_(2)CO_(3)OVERSET(Delta)rarr Li_(2)O+CO_(2)` |
|
| 12. |
Which statements are correct for a process involving an ideal gas? |
|
Answer» `(Delta S)_("Isothermal, sys") = 2.303 N R "long" (V_(f))/(V_(i))` Isothermal `Delta S = nC_(V) "ln" (T_(2))/(T_(1)) + nR "ln" (V_(2))/(V_(1))` Adiabatic, rev `RARR q_("rev") = 0 rArr Delta S = 0` `rArr Delta S_("surr") = 0 rArr Delta S_("total") = 0` for irreversible, `Delta S_("total") gt 0` |
|
| 13. |
What is reaction quotient? |
|
Answer» Solution :Consider a homogenous reversible reaction `xA+yBhArrlC+mD` For the above reaction under non-equilibrium conditions, reaction quotient Q is defined as the RATIO of the PRODUCT of active masses of reaction products RAISED to the respective stiochiometric coefficients in the balanced chemical EQUATION to that of the reactants. Under non equilibrium conditions, `Q=([C]^(l)*[D]^(m))/([A]^(x)*[B]^(V))` |
|
| 14. |
Which of the following MO s have one nodal plane ? . |
|
Answer» `SIGMA 1S` |
|
| 15. |
When hydrogen peroxide is added to acidified potassium dichromate, a blue colour is produced due to formation of : |
| Answer» Answer :C | |
| 16. |
Which oneis notcorrect ? |
|
Answer» `n=4,L =2 ,m=0` |
|
| 17. |
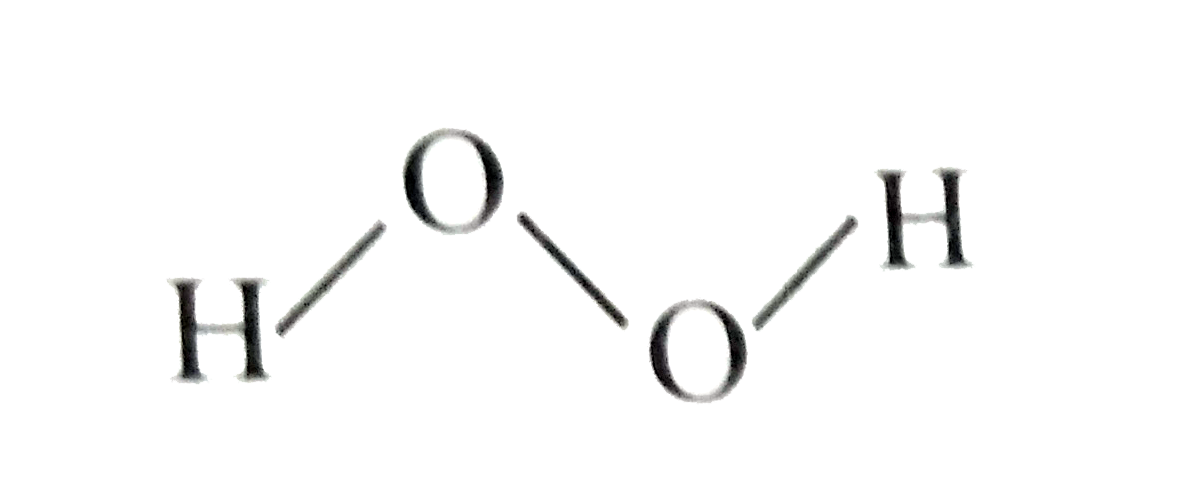
Which of the following is a true structure of H_(2)O_(2) in solid phase? |
|
Answer»
|
|
| 19. |
What is equivalent mass of (NH2_4)_2Cr_2O_7in the change (NH_(4))_(2)Cr_(2)O_(7) rarr N_(2) + Cr_(2)O_3 + 4H_(2)O? |
|
Answer» Solution :`(NH_4)_(2)Cr_(2)O_7` `2overset(-3)(NH_4^(+))rarroverset(0)(N_2)` `2x +8 =+2 , 2x =-6 , E =M/6` |
|
| 20. |
The volume of 10 moles of an ideal gas at 27^@C and latm pressure is 1 lit. What is the volume of 20 moles of same gas at same pressure and temperature? |
| Answer» ANSWER :A | |
| 21. |
Which of the following oxide behaves as acid as well as base |
|
Answer» `Cr_2O_3` |
|
| 22. |
What is meant by condensed structure? Explain with an example. |
|
Answer» Solution :The bond LINE structure can be further abbreviated by OMITTING all these dashes REPRESENTING covalent bonds and by indicating the NUMBER of identical groups attached to an atom by a subscript. The resulting expression of the compound is called a condensed structural formula. e.g., 1, 3-butadiene. `UNDERSET(underset("Condensed Structure")(H_(2)=CH-CH=CH_(2)))(H-overset(H)overset(|)C=overset(H)overset(|)C-overset(H)overset(|)C=overset(H)overset(|)C-H)` |
|
| 24. |
The vapour density of PCl_(5) at 473 is found to be 70*2. Find the degree of dissociation of PCl_(5) at this temperature. |
|
Answer» Solution :Theoretical vapour density of `PCl_(5)` , `D=(" Mol.mass of " PCl_(5))/2 = (31 + 5 xx 35*5)/2 = (208*5)/2 = 104 * 25` Observed vapour density , d= `70*2 ` `:. "Degree of dissociation " (ALPHA)=(104*25 - 70*2)/(70*2)=0*485` |
|
| 25. |
Which of the following represents a chemical reaction ? |
|
Answer» OXIDATION -REDUCTION reaction |
|
| 26. |
What is van't Hoff reaction isotherm ? |
| Answer» Solution :The reaction `DELTA G^(@)= - RT ` In K is CALLED van't HOFF reaction isotherm. | |
| 27. |
Which curve in the following graph represents the curve of ideal gas ? |
|
Answer» B only |
|
| 29. |
Which of the following shows geometrical isomerism. |
|
Answer» `BrCH=C=C=CHBr`<BR> |
|
| 30. |
xg of tartaric acid is dissolved in 30mL of 0.2M potash solution. The resultant mixture requires20ml of 0.1M HCl for titration. Report the numerical value of x. |
|
Answer» Solution :Tartaric ACID is a dibasic acid HOOC-CHOH-CHOH-COOH Gram molecular weight (GMW) = 150G , THEREFORE, GEW = 75g m.eq . Of NaOH =m.eq . Of HCl m.eq. of tartaric acid `=30xx0.2-20xx0.1=6-2=4` Weight of tartaric acid DISSOLVED = Number m.eq`xx(75)/(1000)=(4xx75)/(1000)=0.3` Numerical value of x=0.3 |
|
| 31. |
The RMS velocity of nitrogen gas molecules is 'V cm/sec at a certain temperature. When the temperature is doubled, the molecules dissociated into individual atoms. Now the RMS velocity of nitrogen atoms is x V cm/sec. What is the value of 'x'? |
|
Answer» |
|
| 32. |
What are amphoteric substances? Give examples. |
|
Answer» SOLUTION :A SUBSTANCE or molecule which ACTS both as ACID as well as base, is called amphoteric. Examples: 
|
|
| 33. |
Why are potassium and cesium, rather than lithium used in photoelectric cells ? |
|
Answer» Solution : All the three, lithium, potassium, and cesium, are ALKALI metals. Still, K and Cs are USED in the photoelectric cell and not LI. This is because as compared to Cs and K, Li is smaller in size and therefore, requires high energy to lose an electron. While on the other HAND, K and Cs have low ionization energy. Hence, they can easily lose electrons. This property of K and Cs is utilized in photoelectric cells. |
|
| 34. |
When ethene gas is passed through an aqueous solution containing bromine and sodium chloride, three products are formed. Predict the products. |
Answer» SOLUTION :
|
|
| 37. |
What will be order of reactivity of following compounds towards addition of HBr ? |
|
Answer» |
|
| 38. |
Which of the following does not form volatile product with conc. H_(2)SO_(4)? |
|
Answer» `CO_(3)^(2-)` |
|
| 39. |
Which one of the following reactions does not form gaseous product? |
|
Answer» `PbO_(2)+H_(2)O_(2)to` |
|
| 40. |
The stable oxidation state of Thallium, a IIIA group element is |
| Answer» ANSWER :A | |
| 41. |
Which of the following acidic radicals produce colourless gas with dilute HCI which does not react with oxidising agent? |
| Answer» Answer :A | |
| 42. |
Which one of the following is a gem-dihalide? |
|
Answer» `CH_3CHCl_2` |
|
| 43. |
Which of the following chemical reactions depicts theoxidising behaviour of H_(2)SO_(4)? |
|
Answer» `2HI + H_(2) SO_(4) rarr I_(2) + SO_(2) + 2H_(2)O` |
|
| 44. |
Which mixture of gases is produced on heating Tin oxalate (SnC_2O_4) ? |
|
Answer» `CO+CO_2` |
|
| 45. |
The type of hybridisation of boron in diborane is (a) sp, (b) sp^(2) ,(c) sp^(3) ,(d) dsp^(2) |
|
Answer» SP |
|
| 46. |
Which scientist declared, "Properties of elements are periodic with their atomic mass"? |
|
Answer» NEWLAND |
|
| 47. |
Which of the following is the smallest in size? |
|
Answer» `NA^(-)` |
|
| 48. |
The rms speed of helium in ms^(-1) (atomic mass = 4.0g mol^(-1)) at 400K is |
|
Answer» 1580 |
|
| 49. |
What is similarity and difference between Lewis structure of H_(2)SO_(4) and SO_(4)^(2-) ? |
|
Answer» Solution :There are same NUMBERS of bonding and non bonding electrons in both atoms. `H_(2) SO_(4)` contain two H and neutral. In `SO_(4)^(2-), ` H is not PRESENT and CHANGE is - 2 because `H^(+) ` is remove but electron of H is remains in `SO_(4)`. |
|
| 50. |
Trans-2-Butene overset(HOBr)to P (product ) , 'P' is : |
|
Answer»
|
|