Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
When 22.4 litres of H_(2)(g) is mixied with 11.2 litres of Cl_(2)(g), each at S.T.P, the moles of HCl (g) formed is equal to : |
|
Answer» 0.5 mol of HCl (g) No. of moles in 11.2 L of `Cl_(2)(g)` at S.T.P = 0.5 mol `underset("1 mol")(H_(2)(g))+underset("1 mol")(Cl_(2)(g))rarrunderset("2 mol")(2HCl(g))` `Cl_(2)(g)` is the limiting REACTANT SINCE it has only 0.5 mole available for chemical reaction `:.` No. of moles of HCl (g) formed = 1mol. |
|
| 2. |
The strongest base among the following |
|
Answer» `C_(6)H_(5)NH_(2)` |
|
| 3. |
Which of the reagent is used in Kharash effect ? |
|
Answer» HI |
|
| 4. |
Weak Acid HA (K_a = 1.4 xx 10^(-5)) 0.1 M so in dissow in 2 lit. Find percentage of ionization and pH of solution. |
|
Answer» SOLUTION :`HA_((aq)) + H_2O_((l)) HARR H_3O_((aq))^(+) + A_((aq))^(-)` `K_a=1.4xx10^(-5)` initial concentration C=0.1 M `[H_3O^+]=SQRT(K_axxC)` `=(1.4xx10^(-5)xx0.1)^(1/2)` `=1.183xx10^(-3)` M `pH=-log [H_3O^+]` `=-log (1.183xx10^(-3))` =-(0.07306-3) =2.9269 `APPROX` 2.93 `K_a=(alpha^2C)/(1-alpha)` Now, `1-alpha approx 1` `K_a=alpha^2C` `alpha=sqrt(K_a/C) = sqrt((1.4xx10^(-5))/0.1)` `alpha=1.183xx10^(-2)` % of dissociation = `alpha xx 100` `=1.183xx10^(-2) xx100` =1.183 % |
|
| 5. |
Which of the following undergo electrophilic substitution reactions faster than benzene ? |
|
Answer» Phenol |
|
| 6. |
Which of the following does not have any effect with water? |
| Answer» ANSWER :d | |
| 7. |
What happens when ethylene is passed through cold dilutealkaline potassium permanganate |
|
Answer» Solution :Ethylenereactswithcolddilutealkaline`KMnO_(4)` SOLUTIONTO giveethyleneglycol `CH_(2) = CH_(2)+ H_(2) Ounderset((O)273 K) OVERSET( KMnO_(2)OH^(@))(to)CH_(2) - UNDERSET(OH ) underset(|) (C H_(2))` |
|
| 9. |
Write formulas for the following compounds : Thallium (I) sulphate |
| Answer» SOLUTION :`Th_(2)SO_(4)` | |
| 10. |
Which one is remedy to protect depletion of ozone layer ? |
|
Answer» Prevent the use of ODS |
|
| 11. |
Which of the following is not tetrahedral? |
| Answer» Answer :C | |
| 12. |
What is the nameof Thomsonmodel ? |
| Answer» SOLUTION :Thomsonmodelof ATOMIS KNOWNAS plumpuddingrisinpuddingor WATERMELON. | |
| 13. |
Write the disproportionation reactions of the following species : (i) ClO^(-) (ii) ClO_3^(-) (iii) Cl^(-) (iv) ClO_3^(-) (v) ClO_4^(-) (vi) Tl^+ |
|
Answer» Solution :(i) `OVERSET(+1)(3Cl)O^(-)rarroverset(-1)(2C)l^(-)+overset(+5)(ClO_3^(-))` (ii) `overset(+5)(4ClO_3^(-))rarroverset(-1)(Cl^-)+overset(+5)(2ClO_3^(-))` (iii) `Cl^-` does not DISPROPORTIONATE. (iv) `overset(+3)(ClO_2^-)rarroverset(-1)(Cl)+overset(+5)(2ClO_3^(-))` (v) `ClO_4^(-)` does not disproportionate because in this oxoanion , chlorine is present in its highest oxidation state i.e., +7. (iv) `3Tl^(+)hArroverset(0)(2Tl)+Tl^(3+)` |
|
| 14. |
Which of the following is free radical? |
|
Answer» `NO_2` `Cl^-`: NUCLEOPHILE, `Cl^+` Electrophile |
|
| 15. |
Which of the following reagent will convert Propanoic acidto Propan-1-ol? |
|
Answer» `KMnO_(4)` |
|
| 16. |
Which of the following set of quantum number is vaild / invalid . In case of valid set write the symbol of the orbital it respresent [may more than one answers] &in case it is invalid mention the resason.{:("n",1,m),(3,"not known (but less than 3)",0),(-2,""-1,+1),(4,""2,-1):} |
|
Answer» Solution :(i)valid ,`3s//3p_(x)//3p_(y)//3p_(z)//3d_(x^(2)-y^(2))//3d_(z^2)//3d_(XY)//3d_(XZ)//3d_(YZ)` (ii)Invaild 'n' cannot be NEGATIVE (III) Vaild `4d_(x^(2)-_y^(2))//4d_(z^(2))//4d_(xy)//4d_(xz)//4d_(yz)` |
|
| 17. |
Which of the following is wrong . |
|
Answer» The maximum COVALENCY of Boron is 4 where as that of Al is 6 . |
|
| 18. |
Traces of which metal is present in the sulphide ore of Zn ? |
|
Answer» Indium |
|
| 19. |
Which of the following compounds can show optical isomerism as well as geometrical isomerism? |
|
Answer»
|
|
| 20. |
Which of anions in the column-I showns one or more observations from the column-II? |
|
Answer» |
|
| 21. |
Which of the following properties show similar trends down the group among the elements of group 1 and 17? |
|
Answer» METALLIC character |
|
| 22. |
What is indicated by Psi for atomic and molecular orbitals ? How the molecular orbital is obtained by Schrodinger wave equation ? OR What is LCAO. |
|
Answer» SOLUTION :Atomic orbitals and `Psi` : According to wave mechanics, the atomic orbitals can be EXPRESSED by wave functions `(Psi)` which represent the amplitude of the electron WAVES. These are obtained form the solution of schrodinger wave equation. MOLECULAR orbital and `Psi` and LCAO : Schrodinger wave equation can not be solved fore any system containing more than one electron, molecular orbitals which are one electron wave functions for molecules are difficult to obtain directly from the solution of Schrodinger wave equation. To overcome this problem, an approximate method known as linear combination of atomic orbitals (LCAO) has been adopted. LCAO means method for solution of molecular orbitals having more than one electron by Schrodinger wave equation. |
|
| 24. |
Which one isthe correct unit for entropy? |
|
Answer» KJ MOL |
|
| 25. |
Write down the resonance structures of CO_3^(2-) and HCO_3^(-) |
|
Answer» (##ANE_PKE_CHE_0XI_C11_E01_008_S02##) |
|
| 26. |
What volume of dioxygen is required for complete combustion of 2 volume of acetylene gas at NTP ? |
|
Answer» 2 volumes 1 vol. of `C_2H_2` REQUIRES = `5/2 " vol. of " O_2` 2 vol. of `C_2H_2 " requires " = 5/2xx2=5 vol. " of " O_2` |
|
| 27. |
Which of the following sets of compounds cannot turn clear orange solution of CrO_(3)//aq. H_(2)SO_(4) of greenish opaque solution |
|
Answer» I, IV |
|
| 28. |
What iscatenation ? |
| Answer» Solution :PROPERTY of self-linkingof atoms to form chains or ringsof various sizesis calledcatenation. Carbonhas THEMAXIMUM tendencyfor catenation due to highbond STRENGTH of `C-C` BONDS. | |
| 29. |
Which of the following is a cyclic oxo acid ? |
|
Answer» <P>`H_(4)P_(2)O_(7)` `(HPO_(3))_(5)=H_(5)P_(5)O_(15)` cannot be regarded as a cyclic oxo acid since ten-membered rings are difficult of form. Therefore, it has a CHAIN STRUCTURE. |
|
| 30. |
What is alkane substitution reactions ? Give different types of substitution reactoins name and possibilities. |
|
Answer» Solution :In alkane, one or more hydrogen is substituted can be substituted with halogen, nitro group and sulphuric acid group. These reactions in which hydrogen atoms of alkanes are substituted are known as substitution reactions. (a) Nitration : Hydrogen of alkane is substituted with `_NO_(2)` group, Known as nitration. (b) Sulphonation : Hydrogen of alkane is substituted with `-SO_(3)H` group, known as sulphonation. (c) Halogenation : Hydrogen of alkane is replaced with halogen (F, Cl, Br, I) Known as halogenation. Rate of reaction of alkanes are : `F_(2) gt Cl_(2) gt Br_(2) gt I_(2)` (d) Fluorination : Fluorination is too violent to be controlled. `(CH_(4)+F_(2)(N_(2) " or " Ar) rarr CH_(3)F + HF)` (e) Iodination : Iodination is vert slow and a reversible reaction. It can be carried out in the PRESENCE of oxidizing aganets like `HIO_(3)` or `HNO_(3)`. `CH_(4) + I_(2) hArr CH_(3)I + HI` `HIO_(3) + 5HI rarr 3I_(2) + 3H_(2)O` (f) Chlorination and bromination : They are easy reactions. If chlorine is in required amount then all the hydrogens are substituted with -Cl ATOM. `CH_(4) underset(-HCl)overset(hv, Cl_(2))rarr CH_(3)Cl underset(-HCl)overset(hv, Cl_(2))rarrCH_(2)Cl_(2)` `underset(-HCl)overset(hv, Cl_(2))rarrCHCl_(3) underset(-HCl)overset(hv, Cl_(2))RARRC Cl_(4)` ....(i) `underset("Ethane")(CH_(3))-CH_(3)+Cl_(2) overset(hv) rarr underset("Chloroethane")(CH_(3))-CH_(2)Cl+HCl` ...(ii) Chlorination and bromination takes place either at temperature 573 to 773 K or in presence of diffused sunlight or UV light. In alkane, series of substitution of H is `3^(@) gt 2^(@) gt 1^(@)`. e.g., `CH_(3)CH_(2)CH_(3) underset(-HCl)overset(hv, Cl_(2))rarrunderset("1-Chloropropane")underset(45%)(CH_(3)H_(2)CH_(2)Cl)+underset("1-Chloropropane")underset(55%)(CH_(3)CHCLCH_(3))` Substitution of `2^(@)` - hydrogen `gt` Substitution of `1^(@)` hydrogen rate (g) Bromination : Reaction is slow than that of chlorine and mainly monobromo is formed. |
|
| 31. |
Two flasks of equal volumes have been joined by a narrow tube of negligible volume. Initially both the flasks are 300 K and totally 9 mole of gas is present. One of the flasks is then placed in a thermostat at 600K. How many moles of gas is present in hot bulb. |
|
Answer» `(n_("hot"))/(n_("cold")) = ((PxxV)/(R XX 600))/(P xx V//R xx 300) = 1/2` `n_("hot") = 1/3 xx 9 = 3 `moles. |
|
| 32. |
Vapour pressure is very high for the following liquid at room temperature |
|
Answer» WATER |
|
| 33. |
The two equilibrium AB hArr A^(+) + B^(-) and AB+B^(-)hArrAB_(2)^(-) are simultaneously maintained in a solutio with equilibrium constant K_(1) and K_(2) respectively. The ratio of [A^(+)]to [AB_(2)^(-)] in the solution is |
|
Answer» directly proportional to the concentration of `B^(-)` |
|
| 34. |
Writethe mechanism of freidel craft acylation |
|
Answer» Solution :STEP :1Generationof `.^(@) CH_(3) COCI to`electrohile Step :2 Attackof theelectrophileon benzenering toformareniumion.  Step :3rearomatiosationof ARENIUMION. 
|
|
| 35. |
Write electron configuration, bond order and Magnetic property of Hydrogen (H_(2)) molecule. |
|
Answer» SOLUTION :Electron CONFIGURATION : It is formed by the combination of two hydrogen atoms (1s). Total number of electron in ` H_(2)` molecule = 2 In `H_(2)` molecule BMO : `sigma_(1s) and sigma_(1s)^(*)` `THEREFORE e^(-)` configuration of `H_(2)` molecule : `(sigma_(1s))^(2) (sigma_(1s)^(*))^(0)` bond order : BMO : `sigma`(1s) = 2 electron and ABMO : `(sigma_(1s)^(*)) = 0 ` electron `therefore ` Bond order = `(N_(b) - N_(a))/(2) = ((2 - 0))/(2) = 1 ` This MEANS two H atoms are bonded together by a single covalent bond. Magnetic property : In `H_(2)` molecule, no unpair~d electron is present and only electron paired present . `therefore H_(2)` molecule is diamagnetic . |
|
| 36. |
Which of the following has the highest positive electron gain enthalpy ? |
| Answer» Answer :B | |
| 37. |
The rms speed of hydrogen molecules at room temperature is 2400 ms^(-1). At room temperature the rms speed of oxygen molecules would be: |
| Answer» Solution :`(C_(O_(2)))/(C_(H_(2))) = sqrt((M_(H_(2)))/(M_(O_(2))))` | |
| 38. |
Which of the following compounds have finite dipole moments ? |
|
Answer» trans-2-Pentene |
|
| 39. |
Which is not evidence used to support the delocalisation model for benzene? |
|
Answer» Benzene has 6 equal length bonds, not alternating long/short bonds. |
|
| 40. |
What is the correct sequence of reagents for the following conversion : CH_3-undersetunderset(Br)(|)CH-CH_3 to CH_3-CH_2-CH_2-I |
|
Answer» `OVERSET(alc. KOH)to overset(HI)to` 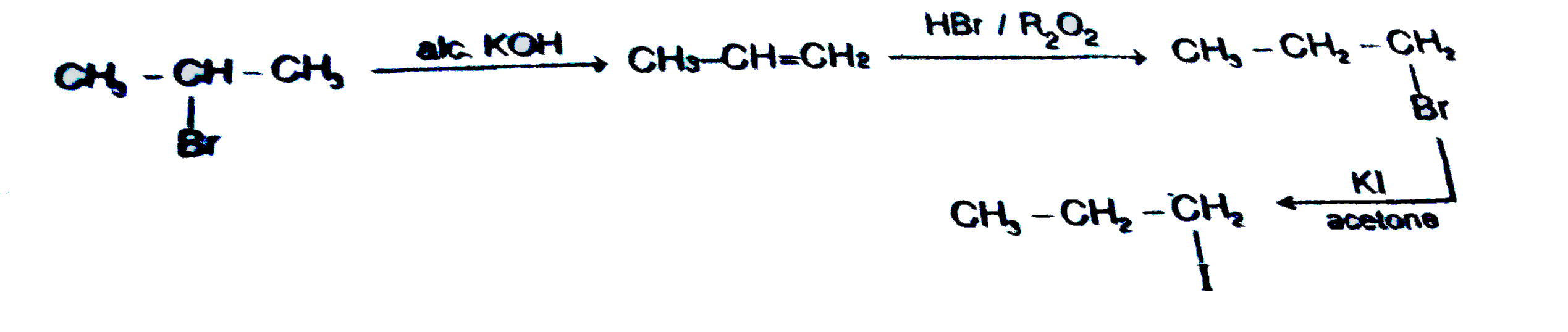
|
|
| 41. |
Which compound given below has sp^3, sp^2 and sp orbitals in the ratio of 6:3:2 |
|
Answer» `CH_3 - CH = CH - CH_2- C = C - CH_2` |
|
| 42. |
Which of the following statements are correct regarding hydrogen? i) The largest single use of dihydrogen is in the synthesis of NH_(3). Which is used in the manufacture of HNO_(3) and nitrogenous fertilizers. (ii) It is used to reduce heavy metal oxide (iii) It is used as rocket fuel. (iv) Atomic hydrogen and oxy hydrogen torches find use for cutting and welding purpose. |
| Answer» Answer :D | |
| 43. |
Titration curves for 0.10 M solution of three weak acids HX, HY and HZ with ionization constants H_(1), K_(2) and K_(3) respectively are plotted as shown in the figure below: |
|
Answer» `K_(2) = (K_(1) + K_(2))/(2)` |
|
| 44. |
The vapour pressures of benzene at 303k and 333k are respectively 1.53xx10^(4) and 5.2xx10^(4)Nm^(-2) Calculate the latent heat of evaporation of benzene over this temperature. |
|
Answer» SOLUTION :LOGE `P_(2)/P_(1)=(DeltaHv)/(2.30R)[(T_(2)-T_(1))/(T_(1)T_(2))]`,where `DeltaHv`is LATENT HEAT of evportation. `2.303"log" "(5.2xx10^(4))/(1.52xx10^(4))=(DeltaHv)/8.314[(333-303)/(333xx303)]` `DeltaH_(v)=34215J=34.215KJ` |
|
| 45. |
Which of the following pairs is isostructural? |
|
Answer» `BC l_(3) and BrCl_(3)`   . .
|
|
| 46. |
What happens when formic acid is heated with Cone. H_(2)SO_(4)? Give theequation. |
| Answer» Solution :Dehydration takes place or CO is FORMED `H-COOH UNDERSET(A)OVERSET(H_(2)SO_(4))(to)CO+H_(2)O` | |
| 47. |
Which element has smallest size? |
|
Answer» B |
|
| 48. |
What is the correct IUP AC name for CH_(3)-underset(CH_(3))underset(|)overset(H)overset(|)C-CH=CH-CH_(2)-overset(O)overset(||)C-OH ? |
|
Answer» 5-Methyl-3-hexenoic ACID |
|
| 49. |
Which changes of properties of periods shows reactivity ? |
|
Answer» SOLUTION :(i) Tendency of reduction and oxidation. (ii) Metallic and non-metallic property. (iii) Reaction of Halogen and oxygen. (IV) ACIDIC, basic, NEUTRAL and properties of oxide. |
|