Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
When H_(2)O_(2) is added to blood, rapid evolution of gas occurs. Explain. |
|
Answer» SOLUTION :The enzyme present in the blood catalyst the OXIDATION of `H_(2)O_(2)` and hence rapid evolution of `O_(2)` takes place. `2H_(2)O_(2) underset("in blood")OVERSET("Enzyme present")to 2H_(2)O + O_(2) uarr` |
|
| 2. |
The temperature of an ideal gas is increased from 120K to 480K. If at 120K the root-mean-square velocity of the gas molecules is v, at 480K it becomes: |
| Answer» ANSWER :B | |
| 3. |
The total number of tetrahedral voids in the face centred unit cell is…… |
|
Answer» 6 |
|
| 4. |
What will be normality of the solution obtained by adding 200 mL of pure water to 100 mL of 0.2 N HCl solution ? |
|
Answer» `OVERSET(("conc."))(N_(1)V_(1))-=overset(("dilute"))(N_(2)V_(2))` `0.2xx100=N_(2)xx300orN_(2)=(0.2xx100)/(300)=0.067N`. |
|
| 5. |
Which is the correct statement? |
|
Answer» Hydrogen has the same IE as ALKALI METALS |
|
| 6. |
Which one of the following satements is//are correct? 1) H_(2)O_(2) soluytions are stored in plastic or wax coated glass vessels since contact with rough surfaces acceleartes its decomposition 2) Urea, sodium stannate, acetanilide, sodium pyrophosphate etc., act as stabilizers(inhibitors)(negative catalyst) for the decomposition of H_(2)O_(2) 3) Silica, MnO_(2),iron, manganese, alumina act as accelerters(positive catalysts) for teh decomposition of H_(2)O_(2) H_(2)O_(2) is a very powerful oxidising agent and a poor reducing agent |
|
Answer» All are correct |
|
| 7. |
Which of the following compound on acidic hydrolysis yields 1, 2-ethanediol ? |
|
Answer» Epoxyethane 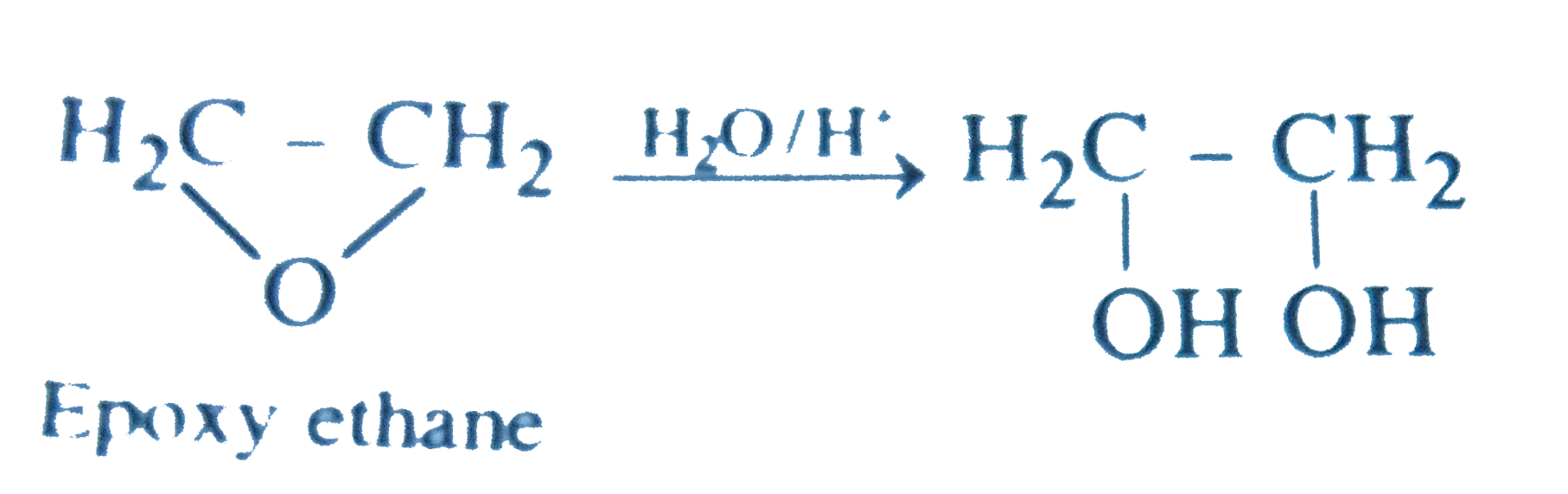
|
|
| 8. |
Which of the following is least ionic? |
|
Answer» `CaF_(2)` |
|
| 9. |
When sodium is dissolved in liquid ammonia, a solution of deep blue colour is obtained. The colour of the solution is due to |
| Answer» SOLUTION :DEEP BLUE, ammoniated ELECTRONS | |
| 10. |
Why axis bonds of PCl_(5)are longer than equatorial bonds ? |
| Answer» Solution : This is DUE to greater REPULSION on the AXIAL bond pairy by the equatorial bond parisof ELECTRONS . | |
| 11. |
Which contains the greatest number of moles of oxygen atoms? (i) 1 mol of ethanol (ii) 1 mol of formic acid (iii) 1 mol of H_(2)O |
|
Answer» Solution :(i) 1 mol of ETHANOL `C_(2)H_(5)OH` (ethanol) - Molar mass = 24 + 6 + 16 = 46 46 g of ethanol contains `1xx 6.023 xx 10^(23)` number of oxygen atoms. (ii) 1 mol of formic acid HCOOH (formic acid) - Molar mass = 2 + 12 +32 = 46 46 g of HCOOH contains `2xx 6.023 xx 10^(23)` number of oxygen atoms. (iii) 1 mol of `H_(2)O` `H_(2)O` (WATER) - Molar mass = 2+16=18 18 g of water contains `1 xx 6.023 x 10^(23)` number of oxygen atoms. `:.` 1 mole of formic acid contains the greatest number of oxygen atoms. |
|
| 12. |
Which of the following is an unsaturated compound ? |
|
Answer» `C_(6)H_(14)` |
|
| 14. |
The rms velocity of an ideal gas at 300 K is 12240 ems^(-1), what is the most probable velocity at that temperature? |
|
Answer» `10000` |
|
| 15. |
Which of the following represents the correct order of increasing electron gain enthalpy with negative sign for the elements O, S, F and Cl ? |
|
Answer» `Cl ltF LT O lt S` |
|
| 16. |
Which is a good fuelCH_(4) orC_(2)H_(6) ? The standard enthalpy of formation of CH_(4), C_(2)H_(6), CO_(2) and H_(2)O are - 74.8, - 84.6 , - 393 . 5 and -286kJ mol^(-1) respectively. |
|
Answer» `CH_(4) + 2O_(2)RARR CO_(2)+ 2H_(2)O` and `C_(2)H_(6) + (7)/(2) O_(2) rarr 2CO_(2)+ 3H_(2)O` Then calculate `Delta H ` per gram |
|
| 18. |
What is true order of average speed mu_(av), maximum possible speed u_(mp) and root mean square speed u_(rms) for any gas ? |
|
Answer» `u_(MP)gt u_(av)gt u_(RMS)` |
|
| 19. |
What are the raw materials used in solvay's process? |
|
Answer» `NACL,NH_(3),CaCO_(3)` |
|
| 20. |
Value of universal gas constant (R ) is same for all gases. What is its physical significance ? |
| Answer» Solution :Unit of R depends upon those units in which p, V and T are measured as, `R = (pV)/(nT)`. If pressure is measured in Pascal, per mole volume is measured in `m^(3)` and TEMPERATURE is measured in KELVIN then. Units of .R. are `Pam^(3)K^(-1)mol^(-1)` or `J mol^(-1) K^(-1)`. Since, Joule defines the unit of work done so .R. is work done by the GAS per mole per kelvin. | |
| 21. |
What are spontaneous process ? |
|
Answer» SOLUTION :A reaction that occurs under the given set of conditions without any EXTERNAL DRIVING FORCE is called a spontaneous reaction. Spontaneous (irreversible) `DeltaHlt0` `DeltaSgt0` `DeltGlt0` |
|
| 22. |
The smog is essentially caused by presence of |
|
Answer» `O_2 and O_3` |
|
| 23. |
Which series of lines of the hydrogen spectrum lie in the visible region ? |
| Answer» SOLUTION :BALMER SERIES | |
| 24. |
Which of the following groups can neither donate a pair of electrons in resonance |
|
Answer» `-NO_(2)` |
|
| 25. |
What causes temporary and permanent hardness of water? |
| Answer» SOLUTION :For ANSWER, CONSULT SECTION 9.10 | |
| 26. |
Which one of the following statement is incorrect |
|
Answer» DIHYDROGEN is the most ABUNDANT ELEMENT in the universe. |
|
| 27. |
What do you understand by bond pairs and lone pairs of electrons? Illustrate with example. |
Answer» Solution :The shared pair of ELECTRONS which is RESPONSIBLE for the formation of bond is CALLED bond pairs of electrons.The pair of valence electrons not involved in BONDING are known as LONE pair of electrons.
|
|
| 29. |
Which scientist are the VSEPR theory ? |
|
Answer» SOLUTION :Sidgwick and POWELL GAVE this theory in 1940. This theory was improved by Nyholm and Gillespie. |
|
| 31. |
Which of thefollowing combination of aldehydes gives orange yellow ppt with 2,4- DNP |
|
Answer» CARBONYL COMPOUNDS |
|
| 32. |
When Zeolite, which is hydrated sodium aluminium silicate is treated with hard water, the sodium ions are exchanged with which of the following ion(s) ? |
|
Answer» `H^(+)` IONS |
|
| 34. |
Liquid drops attain spherical shape.Which property of liquids responsible for this? |
| Answer» SOLUTION :LIQUID tends to minimise their surface area. Spherical surface has small surface area. So liquid DROPS ASSUME spherical shape. | |
| 35. |
Which of the following is/are not known ? PCI_(3),AsCI_(3),SbCI_(3),NCI_(5),BiCI_(5). |
| Answer» Solution :(i) `NCI_(5)` (DUE to absence of d-orbitals) (ii) `BiCl_(5)(BI^(+5)` being an oxidising agent oxidises `Cl^(-)` ion to `Cl_(2)` and `Cl^(-)` ion being a reducing agent REDUCES `Bi^(+5)" to "Bi^(3+)`. | |
| 36. |
What is hybridization ? |
| Answer» Solution :It is a process where TWO or more ATOMIC orbital of comparable energy of valency SHELL of an atom MIX to form the same number of new orbital of same energy. | |
| 38. |
The shape of CIO_(3)^(-) is |
|
Answer» LINEAR |
|
| 40. |
Which of the following properties decreases from hydrogen to deuterium to tritium? |
|
Answer» Relative atomic mass `(g MOL^(-1))` |
|
| 41. |
The total number of anionic species which give coloured gas with conc. H_(2)SO_(4)are: CO_(3)^(2-),HCO_(3)^(-),SO_(3)^(2-),NO_(3)^(-),S_(2)O_(3)^(2-),CH_(3)COO^(-),SO_(4)^(2-). |
|
Answer» |
|
| 42. |
What is sublimation ? Give its uses and examples |
| Answer» Solution :Definition: On heating some solid substances change from solid to VAPOUR state without passing through LIQUID state. The purification technique based on the above principle is known as sublimation. Uses: It is used to separate sublimable compounds from NON sublimable impurities. Examples: Sublimation technique is used for purification of MIXTURE of calcium sulphate and camphor. Because camphor undergoes sublimation and separated and calcium sulphate settle at bottom without sublimation. | |
| 43. |
Which of the following IUPAC names (s) is/are correct? |
|
Answer» 2,2,3-Trimethyl HEXANE |
|
| 44. |
The temperature of the gas is raised from 27^(@)C to 927^(@)C, the root mean square velocity is |
|
Answer» `:. u_(2)=2u_(1)` |
|
| 45. |
Which of the following are used as catalyst in Friedel-Crafts reactions ? |
|
Answer» `AlCl_(3)` |
|
| 46. |
The rms velocity of CO gas molecules at 27^(@)C is approximately 1000 m//s . For N_(2) molecules at 600 K , the rms velocity is approximately "___________"' |
|
Answer» `2000 m//s` |
|
| 47. |
Which one of the following has highest bond order? N_(2), N_(2)^(+) or N_(2)^(-)? |
|
Answer» Solution :`N_(2)` (14 electrons) Bond ORDER = 3, B.O. = `(N_(b)-N_(a))/(2)=(10-4)/(2)=3` `N_(2)^(+)` (13 electrons) Bond order = 2.5, B.O. `=(N_(b)-N_(a))/(2)=(9 - 4)/(2)=2.5` `N_(2)^(-)` (15 electrons) Bond order `= 2.5, B.O. = (N_(b)-N_(a))/(2)=(10 -5)/(2)=2.5` So `N_(2)` has the HIGHEST bond order. |
|
| 48. |
What will be the enthalpy change of conversion of graphite into diamon |
|
Answer» `DELTAH=-393.12-(-391.25)=-1.87kJ` |
|
| 49. |
Vapour density of N_(2)O_(4) of x 60^(@) C is found to be 30.6 The degree of dissociation of N_(2) O_(4) is : |
|
Answer» 0.1 `ALPHA=(D-d)/(d(n-1))=(46-30.6)/(30.6)=0.5 IMPLIES 1-alpha=50%` |
|
| 50. |
The reversible expansion of an ideal gas under adiabatic and isothermal conditions is shown in the figure. Which of the following statements(s) is (are) correct? |
|
Answer» <P>`T_(1) = T_(2)` `T_(i) gt T_(a) ( :. T_(i) = T_(1) "&" T_(a) = T_(3) LT T_(1))` `P_(i) gt P_(a)` (from graph), `W_(i) gt W_(a)` (from graph) `Delta U_(i) gt Delta U_(a) ( :. Delta U_(i) = 0 "&" Delta U_(a) lt 0)` |
|



