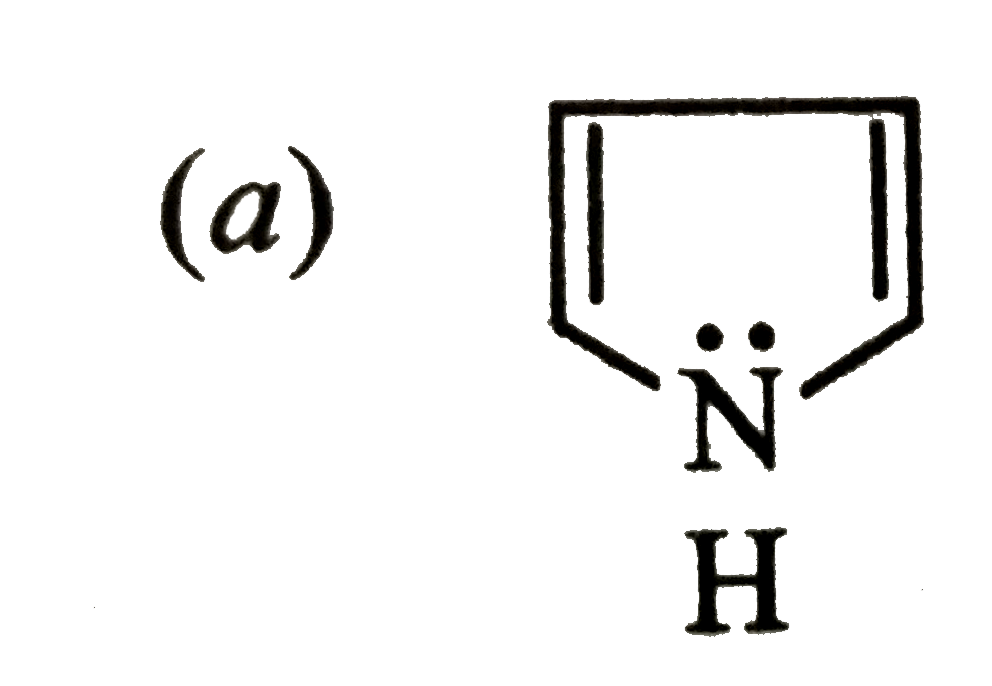Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which one of the following is used as an insecticide and as a soil sterilising agent? |
|
Answer» CHLOROFORM |
|
| 2. |
Thermodynamics is not concerned about .............. |
|
Answer» ENERGY changes involved in a CHEMICAL REACTION. |
|
| 3. |
What is the mass percent of oxygen in ethanol ? |
|
Answer» 0.5214 MOLAR mass of ethenol of `2xx12.01+6xx1.008+16` =46.068g Mass percent of OXYGEN `=(16)/(46.068)xx100=34.73%` |
|
| 4. |
What are Fullurenes? How are they propared? |
|
Answer» SOLUTION :Carbon exhibits many allotropicforms, both crystallins as well as amorphous. Diamond and graphite are two well known crystalline forms of carbon, third form of corbon known as fullerences. Fullerences are makde by heating of graphite in an electric are in the presence of inert gases such as helium or argon. The sooty material formed by condensation of vapourised `C^(a)` small molecular consists of mainly C60 with smaller quantity of C70 and traces of fallerence CONSISTING of EVEN number of carbon atoms upto 350 or above. |
|
| 5. |
Which of the following alkali metal shows high melting point ? |
| Answer» Solution :DUE to the smallest size of Li. | |
| 6. |
Which of the following has larger dipole moment ? Explain. 1-Butyne or 1-Butene |
Answer» Solution :The DIRECTION of dipole moments of individual bonds in 1-butyne and 1-butene are shown below : 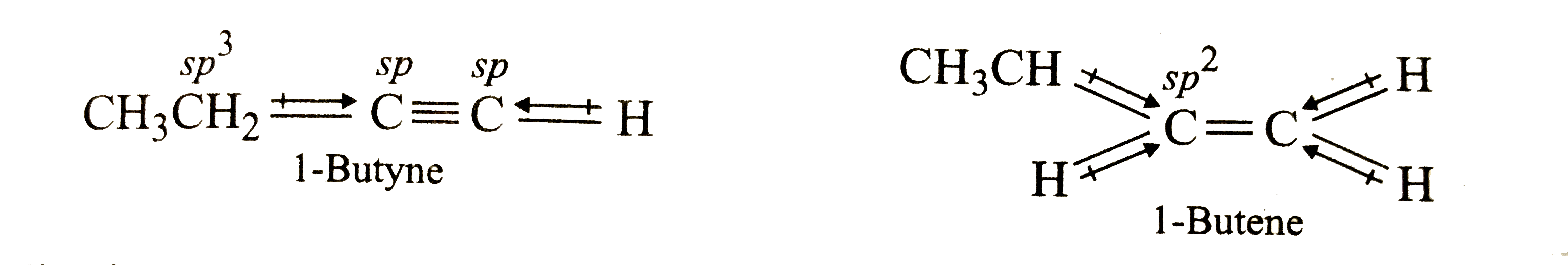 The resultant dipole moment of both 1-butyne and 1-butene is due to the dipole moment of `CH_3CH_2C` and C-H bonds which oppose each other . Since a SP CARBON is more electronegative than a `sp^2` carbon, therefore, the dipole moment of `CH_3CH_2(sp^3)`-C(sp) bond in 1-butyne is more than that of `CH_3CH_2(sp^3)-C(sp^2)` bond in 1-butene. As a result, the dipole moment of 1-butyne is more than that of 1-butene. |
|
| 7. |
Which one of the following is isoelectronic with Ne? |
| Answer» Answer :D | |
| 8. |
What is Delta U for the process described by figure. Heat supplied during the process q= 100kJ |
|
Answer» `+50kJ` W = area under graph`= (1)/(2) xx 1 xx (1 +2) = (3)/(20 xx 10^(5)N - m^(2)` `Delta U = +100 - (3)/(2) xx (10^(5))/(1000) = 100-150= -50J` |
|
| 10. |
Which of the following does not result in an increase in the entropy ? |
|
Answer» CRYSTALLISATION of SUCROSE from solution |
|
| 11. |
The units of constant b used in van der Waals' equation are ………….. . |
| Answer» SOLUTION :`L MOL^(-1)` | |
| 12. |
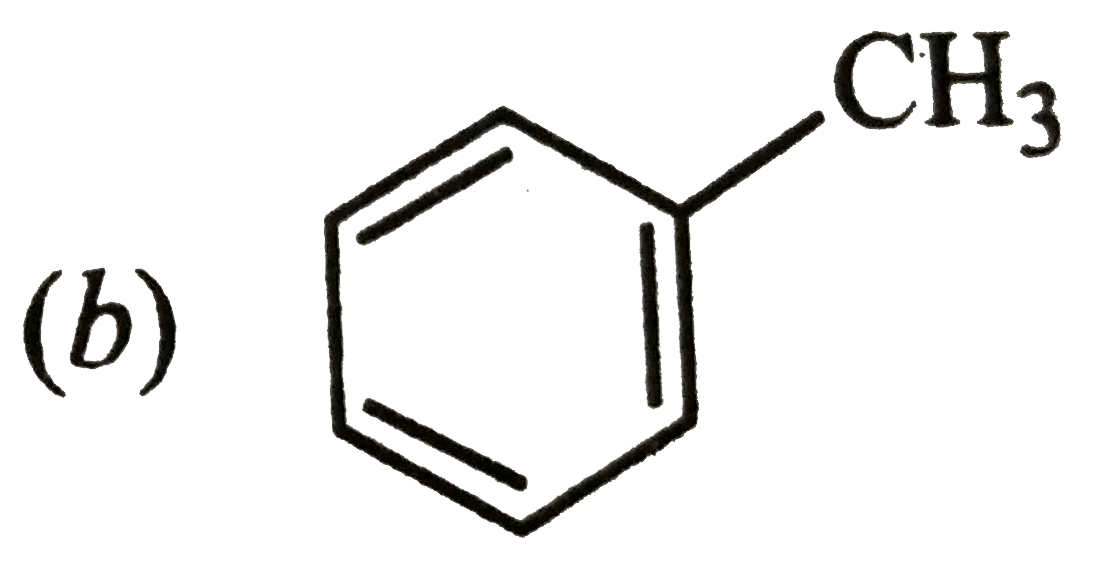
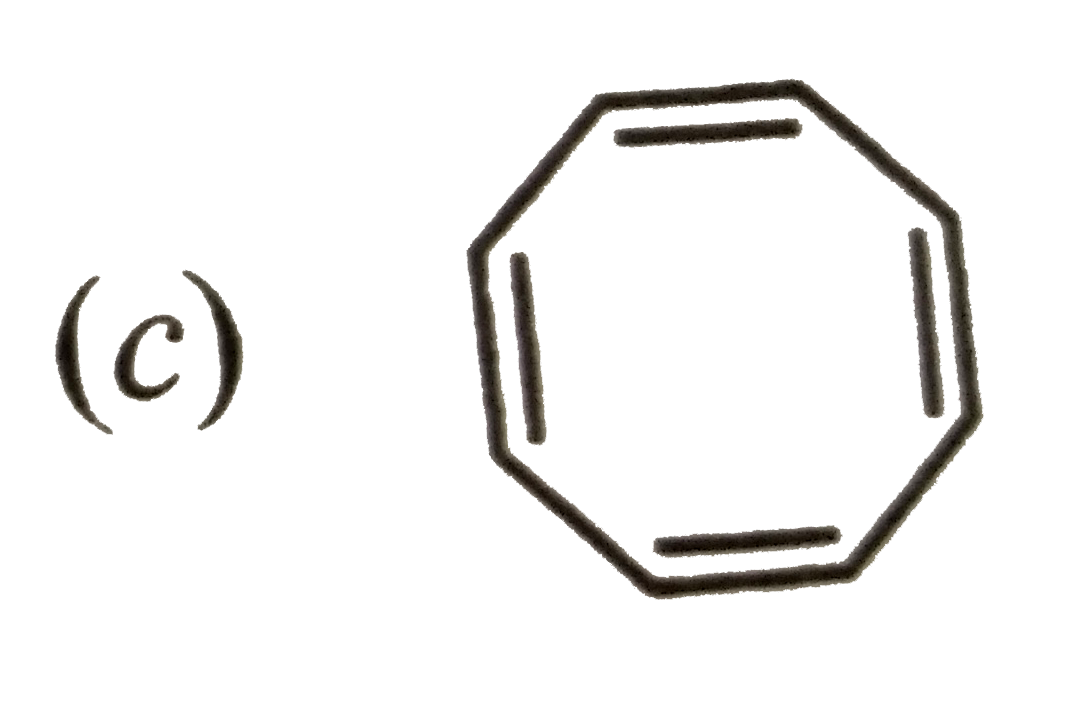
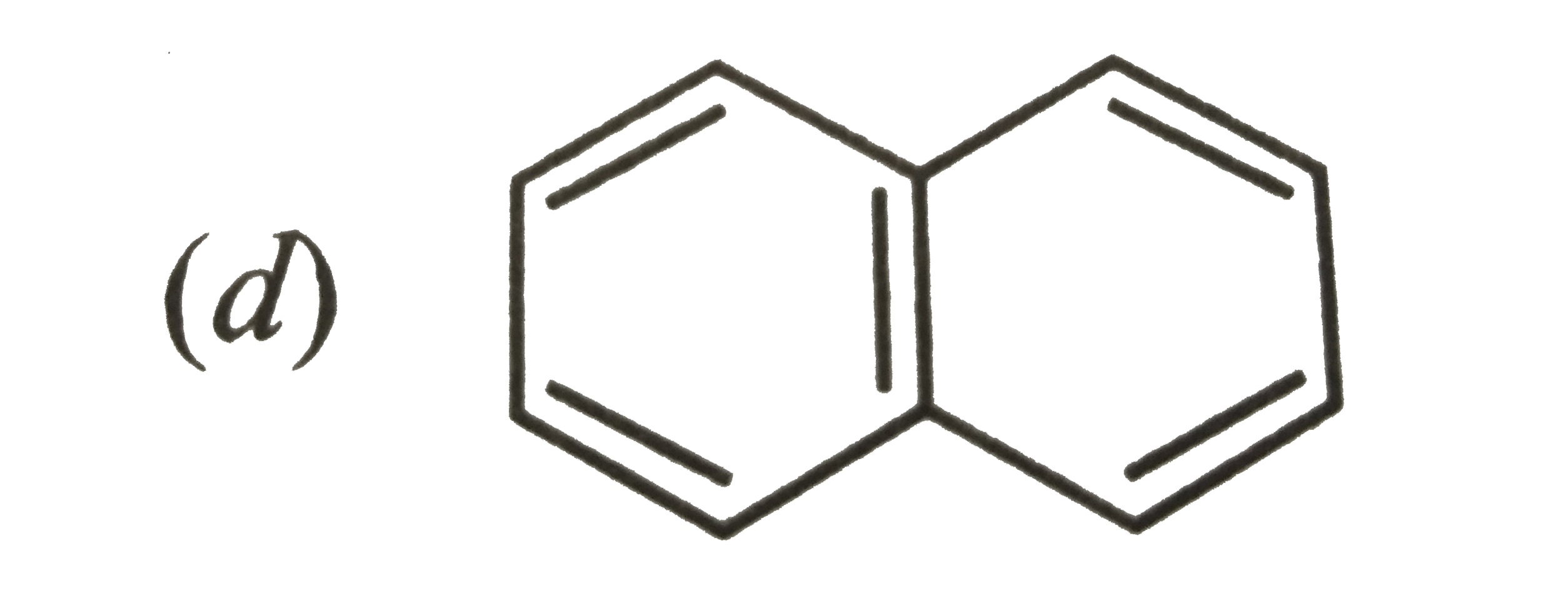
Which is a non-aromatic compound ? |
|
Answer»
|
|
| 13. |
Which of the following statement is wrong- |
|
Answer» nitrogen cannot form `dpi-p PI` bond |
|
| 14. |
What will be the temperature when the rms velocity is four times of that at 300 K ? |
|
Answer» 300 K HENCE, `upropsqrttrArr(u_(1))/(u_(2))=sqrt((T_(1))/(T_(2)))` From Eq. (i), `(1)/(4)=sqrt((300)/(T_(2)))` `(1)/(16)=(300)/(T_(2))` `T^(2)=16xx300` 4800 K |
|
| 15. |
Write Lewis representation of following molecules / Ions. (i) H_(2)(ii) O_(2) (iv) NF_(3) (iv) CO_(3)^(2-) (vi) HNO_(3) |
Answer» SOLUTION :LEWIS REPRESENTATION of molecule/ions are SHOWN in FOLLOWING table : 
|
|
| 16. |
Which of the following is ferromagnetic? |
|
Answer» CALCIUM METAL |
|
| 17. |
Which of the following species is an electrophile ? |
|
Answer» `H_2O` |
|
| 18. |
Threshold frequency, v_(0) is the minimum frequency which a photon must possess to eject an electron from a metal. It is different for different metals. When a photon of frequency 1.0 xx 10^(15) s^(-1) was allowed to hit a metal surface, an electron having 1.988 xx 10^(-19)J of kinetic energy was emitted. Calculate the threshold frequency of this metal. Show that an electron will not be emitted if a photon with a wavelength equal to 600 nm hits the metal surface. |
|
Answer» Solution :We KNOW thathv =`hv_(0)+ke` or `hv-ke =hv_(0)=-1.988xx10^(-19)` `hv_(0)=6.626 xx10^(-19)-1.988xx10^(-19)j` `hv_(0)=4.638xx10^(-19)j` `v_(0)=(4.638 xx10j)/(6.626xx10^(-34)=0.699xx10^(15)s^(-1)` when `lambda=600 nm =600 xx10^(-19) m` thus `v lt v_(0)`hence no electro will beemitted |
|
| 19. |
Which of the following expression is correct for a CsCl unit cell with lattice parameter,a? |
|
Answer» `r_(cs+)+r_(cl^(-))=2a` ` Rightarrow r_(Cs)+r_(Cl^(-))+(sqrt3)/(2)a` |
|
| 20. |
Thiosulphate reacts differently with iodine and bromine in the reactions given below 2S_(2)O_(3)^(2-) +I_(2)rarrS_(4)O_(6)^(2-)+2I^(-) S_(2)O_(3)^(2-)+2Br_(2)+5H_(2)O rarr 2SO_(4)^(2-) +2Br^(-) +10H^(+) Which of the following statements in correct ? |
|
Answer» a.Bromine is a STRONGER oxidant than iodine |
|
| 21. |
Which of the following properties of atom could be explained correctly by Thomson Model of atom |
|
Answer» Overall neutrality of ATOM. |
|
| 22. |
When copper is treated with a certain concentration of nitric acid, nitric oxide and nitrogen dioxide are liberated in equal volumes according to the following equation xCu + yHNO_(3)rarr Cu(NO_3)_2 + NO + NO_2 + H_(2)O . The coefficient of x is ______ |
|
Answer» `3Cu+8HNO_3Cu(NO_3)_(2)+2NO+4H_(2)O...(i)` `Cu+4HNO_(3) rarr Cu(NO_3)_(2)+2NO_(2)+2H_(2)O....(ii)` Here NO and `NO_2`are EVOLVED in equal volumes, therefore , adding Eqns. (i) and (ii) , have `4Cu + 12 HNO_3 rarr 4Cu(NO_3)_(2) +2NO_2 +2NO + 6H_(2)O " or " 2Cu+6HNO_3 rarr 2Cu(NO_3)_(2) +NO_2 +NO+3H_2O` Thus coefficients X and y of `Cu and HNO_3`respectively are 2 and 6. |
|
| 23. |
Which of the following reactions does not take place |
|
Answer»
|
|
| 24. |
Which of the following reactions shows reduction of water ? |
|
Answer» `2H_(2)O + 2Na to 2NAOH + H_(2)` |
|
| 25. |
Write structures of different chain isomers of alkanes corresponding to the molecular formula C_(6)H_(14). Also write their IUPAC names. |
Answer» Solution :Hexane `(C_(6)H_(14))` has 5 ISOMERS which are as FOLLOWS : 
|
|
| 26. |
To find formula of a compound composed of iron and carbon monoxide Fe_(x)(CO)_(y),the compound is burned in pure oxygen and following reaction occurs: Fe_(x)(CO)_(Y) + O_(2) rightarrow Fe_(2)O_(3) + CO_(2) If 19.6gm of Fe_(x)(CO)_(y) is burnt and 8gm Fe_(2)O_(3) is formed, then empirical formula of Fe_(x)(CO)_(y) is: |
| Answer» Answer :D | |
| 27. |
Which is manufactured by electrolysis of fused sodium chloride? |
| Answer» SOLUTION :`Na`. | |
| 28. |
Two gases A and B having the same volume diffuse through a porous partition in 20 and 10 seconds respectively. The molecular mass of A is 49u. Molecular mass of B will be |
|
Answer» 50.00u |
|
| 29. |
What happens when warm aqueous Barium hydroxide solution is electrolysed? |
|
Answer» |
|
| 30. |
What are the harmful effects of photochemical smog and how can they be controlled ? |
|
Answer» Solution :The COMMON components of photochemical smog are ozone, nitric OXIDE, acrolein, formaldehyde and peroxyacetyl nitrate (PAN). Photochemical smog causes serious health problems. Both ozone and PAN act as powerful EYE irritants. Ozone and nitric oxide irritate the nose and throat and their high concentration causes headache, chest pain, dryness of the throat, cough and difficulty in breathing. Photochemical smog leads to cracking of RUBBER and extensive damage to plant life. It also causes corrosion of metals, stones, BUILDING materials, rubber and painted surfaces. |
|
| 32. |
which ofthe following represents correct order of conductivity in solids |
|
Answer» ` k_("metals")gtgt k_("insulators") lt k_("semiconductors")` |
|
| 33. |
Which one of the following equation is not correct ? |
|
Answer» `DELTA G^(@) = - RTlnK` |
|
| 34. |
What is the relationship between the members of following pairs of structure ? Are they structural or geometrical isomers or resonance contributors? (c ) H- overset(overset(""^(+)OH)(||))(C )-OH " " H - overset(overset(OH)(|))(C^(+))- OHs |
|
Answer» SOLUTION :(a) They are structural ISOMERS: `C_(5)H_(10)O` `CH_(3)-CH_(2)- OVERSET(overset(O)(||))(C )-CH_(2)CH_(3) and CH_(3)-CH_(2)-CH_(2)- overset(overset(O)(||))(C )-CH_(3)` In fact they are position isomer and metameres (b) They are trans and cis isomer and metameres (c ) They are RESONANCE structure because position of centre of atom are constant and the position of electrons are differ. |
|
| 35. |
What is the distance between two layers of carbon atom in graphite ? |
|
Answer» `1.42A^@` |
|
| 36. |
Which of the following compound have O-O bonding ? |
|
Answer» `H_2S_2O_6` |
|
| 37. |
What type of glass is obtained when borax is added that ? |
| Answer» Solution :PYREX GLASS it is a glass which is HEAT resistant . And can WITHSTAND a very high TEMPERATURE. | |
| 38. |
Which of the following has maximum bond cell ? |
|
Answer» `H_(2) O` |
|
| 39. |
Which scientist has obtained benzene first ? |
|
Answer» Kekule |
|
| 40. |
Which of the following is not formed when glycerol reacts with HI ? |
|
Answer» `CH_(2)=CH-CH_(2)I` |
|
| 41. |
Which one of the following is used as a bleach? |
| Answer» Answer :a | |
| 42. |
Which of the followingsets of quantumnumberis notpossible ? |
|
Answer» 1 and 3 |
|
| 43. |
The reason for small radius of Ga compared toAl is "……………….". |
|
Answer» POOR screening EFFECT of d and F orbitals |
|
| 44. |
What will be the concentration of iodine in solution? |
|
Answer» `1.1M` `n_(I_2)=(Mass_(I_2))/(Molar mass_(I_2))=(0.070g)/(254g mol^(-1))` `V_L=250 mLxx(IL)/(1000mL)` `:. M=(0.070g)/(254 g mol^(-1)xx250mL)xx((1000mL)/(1L))` `=0.0011 mol L^(-1)` |
|
| 45. |
Which of the following has the highest nucleophilicity? |
| Answer» Answer :C | |
| 46. |
Write the names of isotopes of hydrogen what is the mass ratio of these isotopes ? |
|
Answer» Solution :Hydrogen has three isotopes: protium, `(._1^1H)`, Deuterium, (`._1^2H` or D) and tritium, (`._1^3H` or T). These isotopes differ from one another in respect of the presence of NEUTRONS. Ordinary hydrogen, protium, has no neutrons, deuterium (also known as heavy hydrogen) has one and tritium has two neutrons in the nucleus. In the year 1934, an American scientist, Harold C. Urey, got Nobel Prize for SEPARATING hydrogen isotope of mass number 2 by physical methods. The predominant form is protium. TERRESTRIAL hydrogen contains 0.0156% of deuterium mostly in the form of HD. The tritium concentration is about one atom per `10^18` atoms of protium. Of these isotopes, only tritium is radioactive and emits low energy B- particles. Its half life period is (`t_(1/2)`=12.33 years) Since the isotopes have the same electronic configuration, they have almost the same chemical properties. The only difference is in their RATES of reactions, mainly due to their different enthalpy of bond DISSOCIATION. However, in physical properties these isotopes differ considerably due to their large mass differences. 
|
|
| 47. |
Which of the following is not true? |
|
Answer» Camphor |
|
| 48. |
What happens if a liquid is heated to the critical temperature of its vapours ? |
| Answer» Solution :The meinscus between the LIQUID and the VAPOUR disappears (SURFACE tension of the liquid becomes ZERO). | |
| 49. |
Whichelectronic configurationof an element hasabnormally highdifferencebetweensecondand thirdionizationenthalpy ? |
|
Answer» `1s^(2), 2s^(2)2p^(6) , 3S^(1)` |
|
| 50. |
Three lines are drawn from a single corner of an FCC unit cell to meet the other corner such that they are found to pass through exactly only 1 octahedrat void, no voids of any type and exactly 2 tetrahedral voids with 1 octahedral voids. Identify the line ? |
|
Answer» EDGE length 
|
|