Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
When Benzene is treated with chlorine under the influence of u.v. light, a solid material of molecular wt. 291 is formed. Quantitative analysis gives an empirical formula of CHCI. What statement(s) is/are correct regarding above information |
|
Answer» Product is aromatic 
|
|
| 2. |
Which of the following gases is used for welding |
|
Answer» Methane The combustion of acetylene is HIGHLY exothermic and the HEAT PRODUCED during the combustion can be used for welding PURPOSES in the form of oxy acetylene flame. |
|
| 3. |
The spin of the electron |
|
Answer» increases the angular momentum |
|
| 4. |
What type of semiconductor is obtained when silicon is doped with arsenic ? |
| Answer» SOLUTION :n-type SEMICONDUCTOR | |
| 5. |
What is the meaning, "Dipole moment is a vector quantity." How it is express ? |
|
Answer» Solution :Dipole moment is a VECTOR quantity, I.e., it has MAGNITUDE as well as direction. It is representedby an arrow with its TAIL at the positive CENTRE and BEAD pointing towards the negative end. `rarr` |
|
| 6. |
Which of the following metal carbonates is decomposed on heating? |
|
Answer» `MgCO_3` |
|
| 7. |
what is molal depression constant ? Does it depend on natureof the solute? |
|
Answer» SOLUTION :` K_f ` =molal freezing POINT depression constant or cryoscopic constant. ` DELTA T_f =K_f m,` Where ` Delta T_f ` =depression in freezing point m= molality of the solution ` K_f ` = cryoscopic constant ` 1_fm,=1 Delta T_f =K_f` i.e. cryoscopic constant depends on the MOLAR concentration of the solute particles ,` K_f` is directly PROPORTIONAL to the molar concentration of the solute particles ` Delta T_f= (K_f xx W_B xx 1000 )/( M_B xx W_A ) ` ` W_B= ` mass of the solute `W_A `=mass of solvent `M_B ` =molecular mass of the solute. |
|
| 8. |
What happens when HI reacts with tert. Butyl alcohol? |
| Answer» SOLUTION :`UNDERSET("Tert butyl alcohol")(CH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-OH+HI) underset(H_(3)PO_(4))overset(KI)to underset("Tert butyl IODIDE")(CH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-I+H_(2)O)`. | |
| 9. |
Which type of elements form interstitial hydrides? |
|
Answer» s-BLOCK and p-block |
|
| 10. |
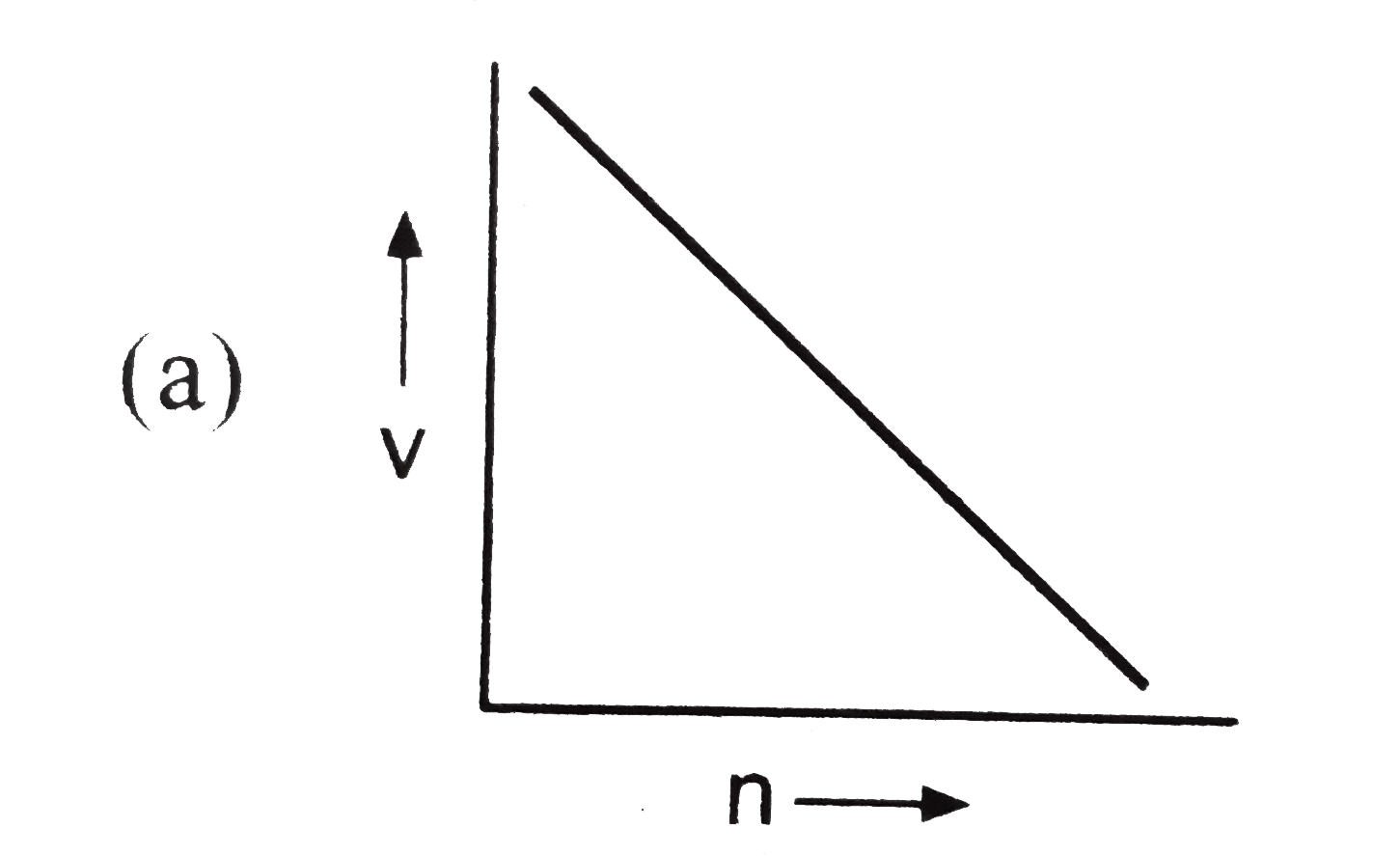
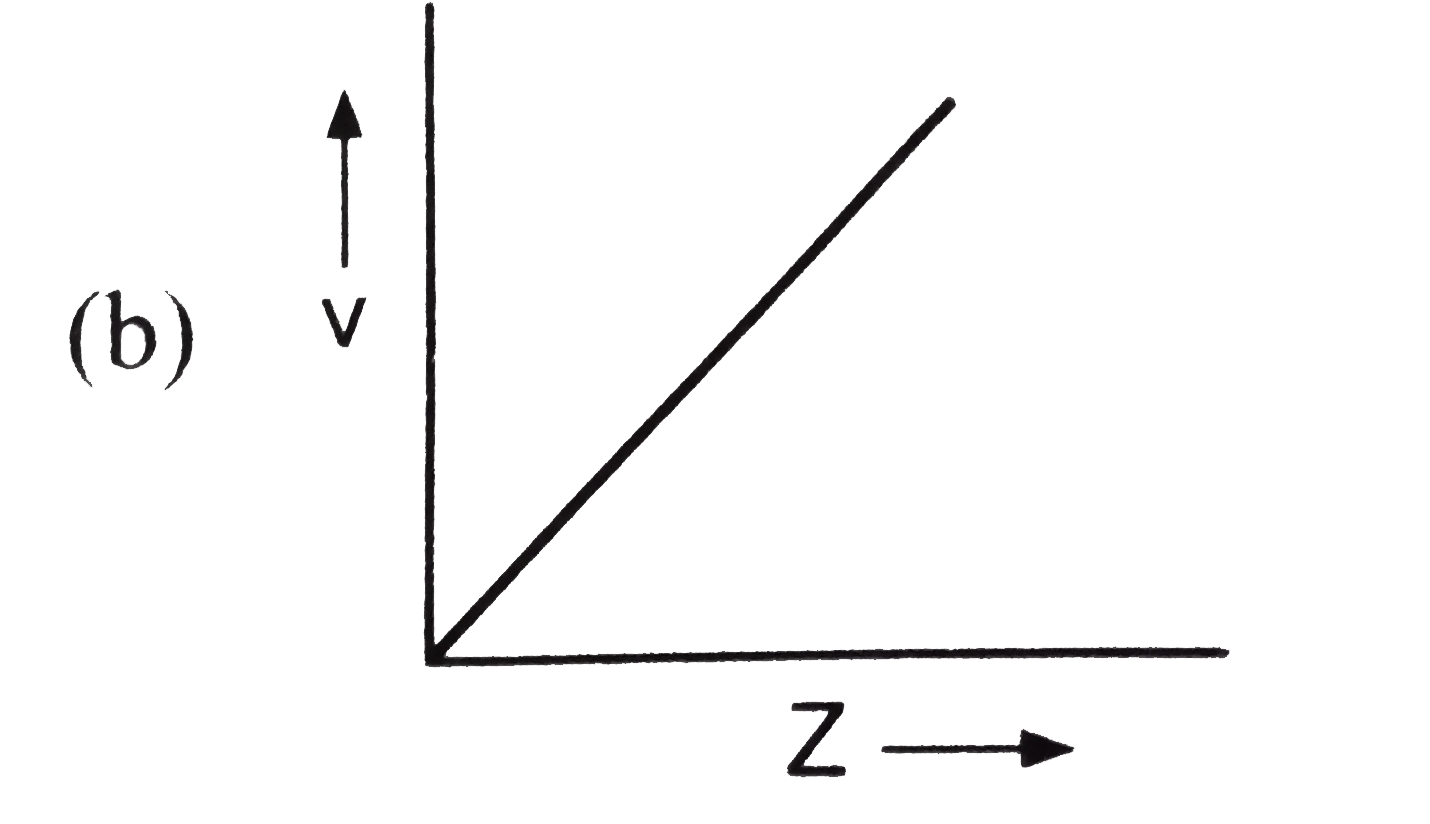
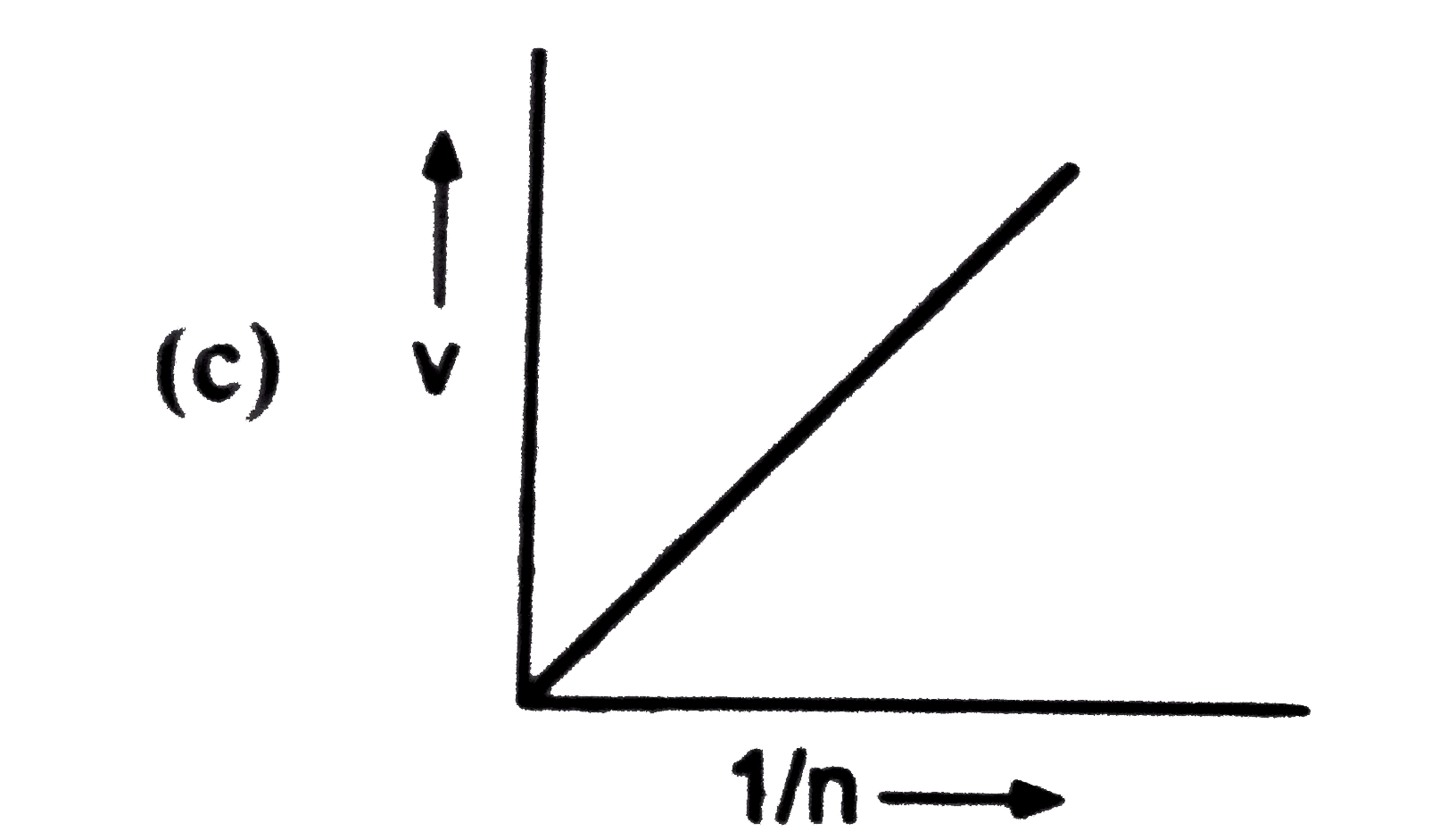
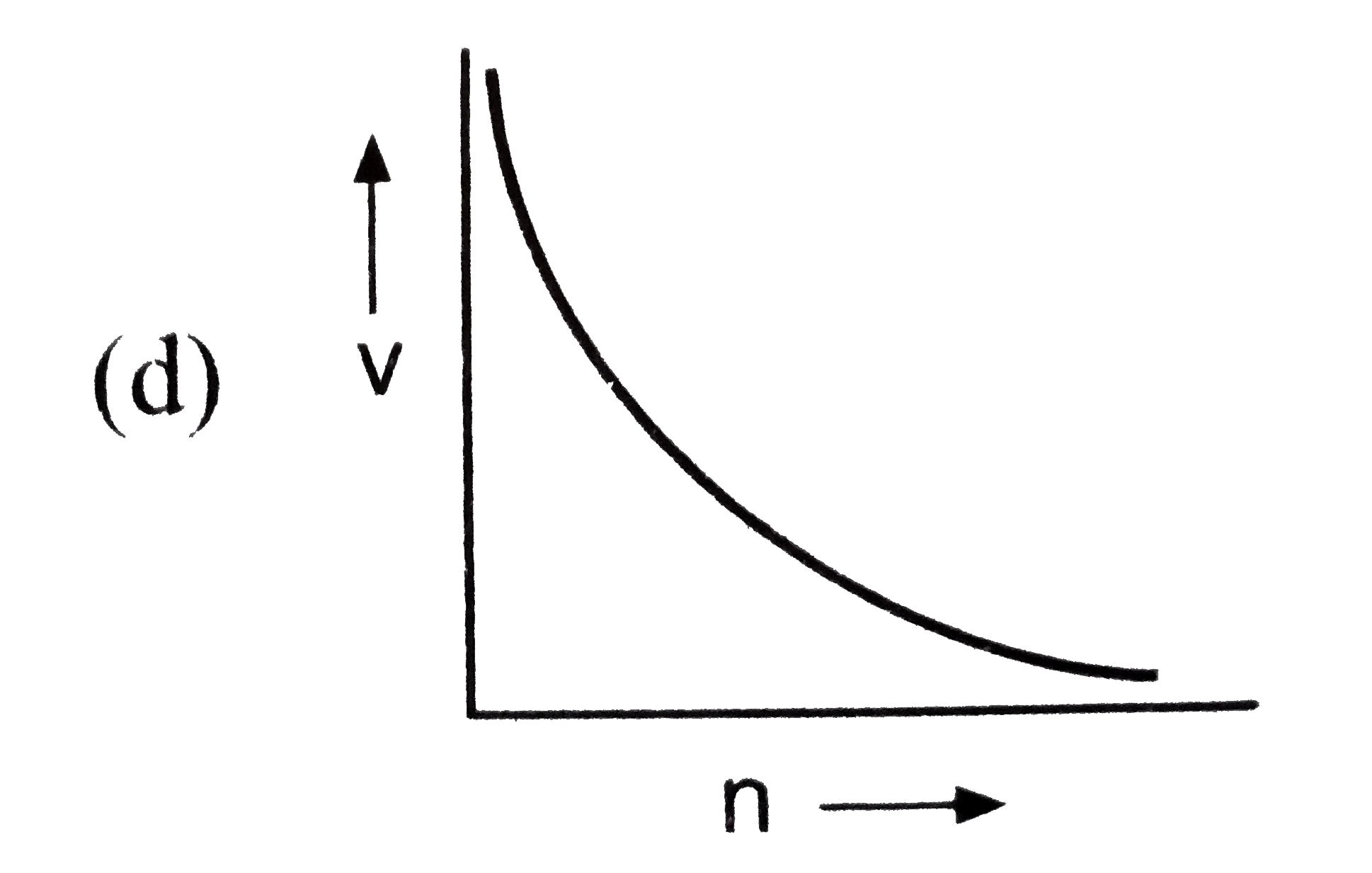
Which of the following is not correct for the velocity of electron? |
|
Answer»
|
|
| 11. |
What are the atomic numbers of elements whose outermost electrons are represented by (i) 3S^(1) (ii) 3P^(5)? |
|
Answer» |
|
| 12. |
Which bond angle theta would result in the maximum dipolemoment for the traitomic molecule XY_(2) |
|
Answer» `THETA =90^@` |
|
| 13. |
Which of the following is/are corret statements ? |
|
Answer» `Mg` burns in air releasing dazzling light rich in UV RAYS Eg : CHOROPHYLL pigment |
|
| 14. |
Which of the following statement (s)are wrong. |
|
Answer» one MOL of photons having ebnergy 400 KJ/mol will always breaks 4 male bonds of a molecule `A^(2)` (b)`(n_(A))/(n_(B))=(40xx2000)/(30xx3000)=(8)/(9)` (D) Time PERIOD`n^(3)` `(T_(1))/(T_(3))=(1)/(9)` |
|
| 15. |
Which of the following species has lowest first ionization potential ? |
|
Answer» O while in `O_(2) , O_(2)^(+) and O_(2)^(-)` , electron is to be removed from`pi^(**) (2p_(x)) or pi^(**) (2p_(y))` which has higher ENERGY and hence less stable . Further , `O_(2)^(-)` is least stable DUE to GREATER number of ELECTRONS in the antibonding molecularorbitals . |
|
| 16. |
We have 1.6 g CH_(4),1.7gNH_(3)and1.8gH_(2)O select correct the alternate(s). |
|
Answer» There are equal number of moles of each reactant atoms = `0.1N_(0)xx5,0.1N_(0)xx4,0.1N_(0)xx3` H-atoms = `0.1N_(0)xx4,0.1N_(0)xx3,0.1N_(0)xx2` no. of `C=N=O` |
|
| 17. |
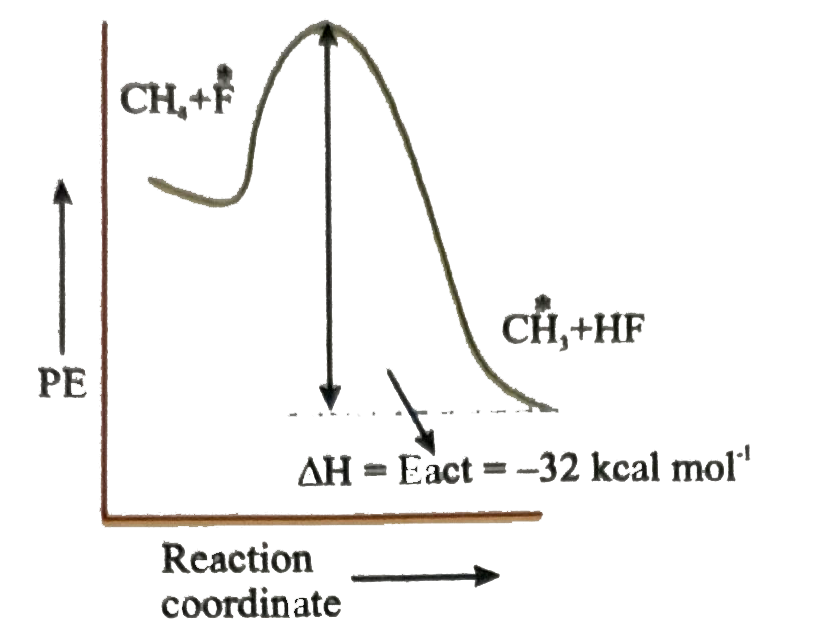
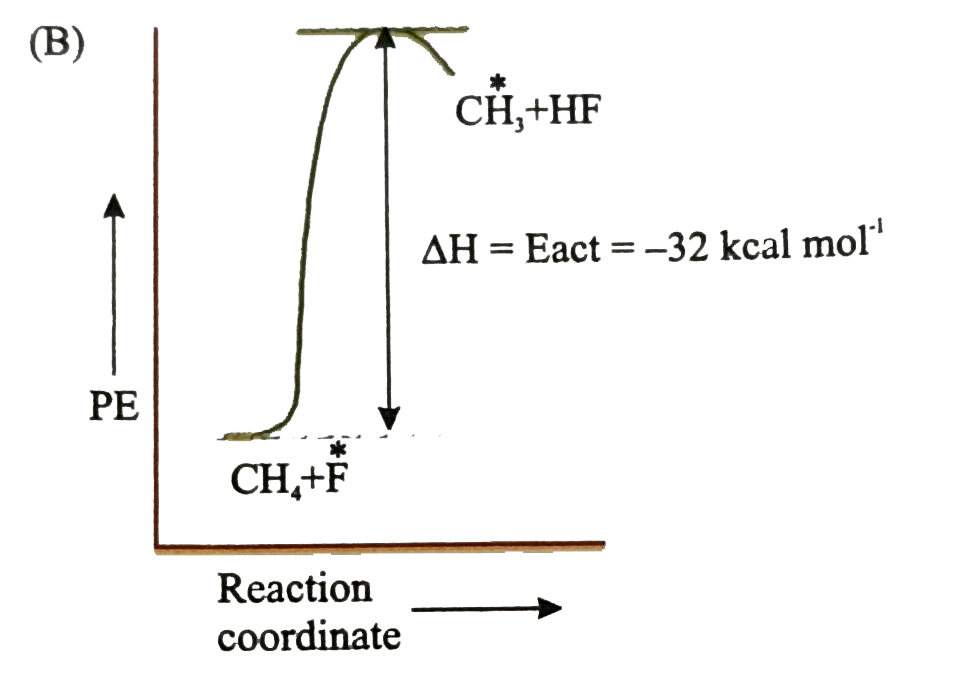
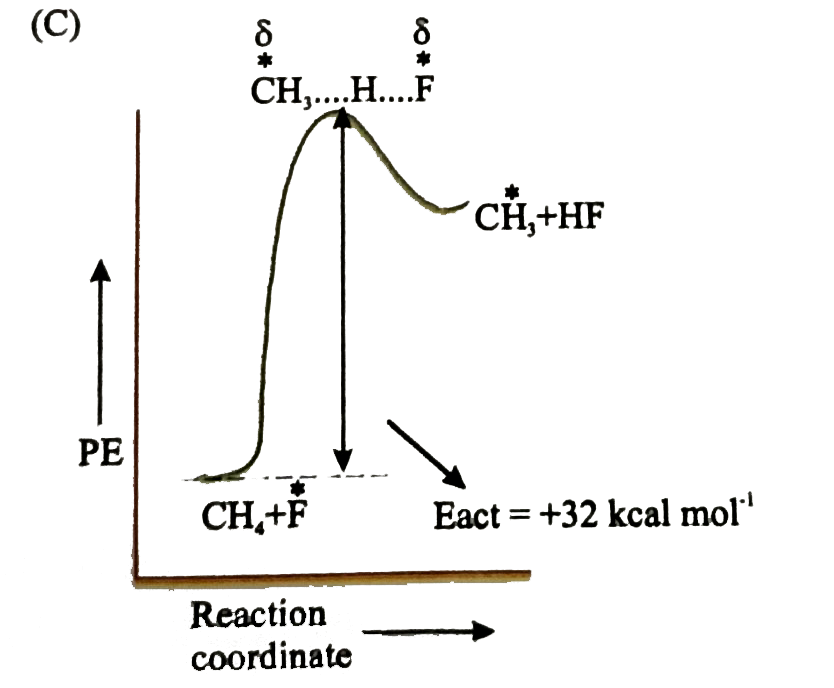
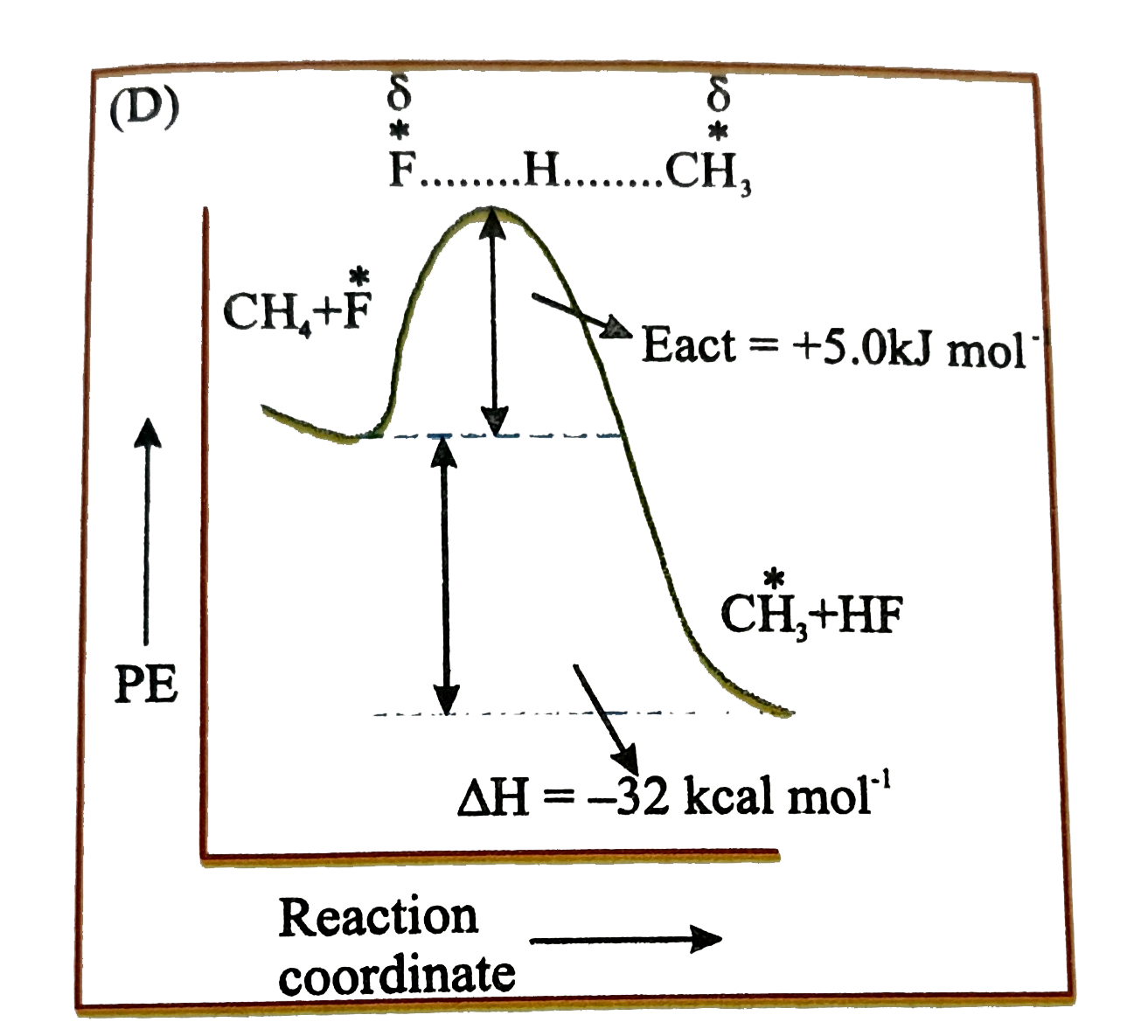
Which of the following is orrect potential energy diagram for the given chain propagating step. underset(B.E.=435kJ//mol)(CH_(3)-H+F)tounderset(B.E.569kJ//mol)(CH_(3)+H-F) DeltaH^(@)=-32kcal//mol |
|
Answer»
|
|
| 18. |
Which one of the following is the correct quadraticform of Ostwald's dilution law equation ? |
|
Answer» `alpha^(2)C+alphaK-K=0` `K=(C alpha^(2))/(1-alpha)` or `K-K alpha = C alpha^(2)` or, `C alpha ^(2) + K alpha-K=0` or, `alpha^(2)C + alpha K - K=0` |
|
| 19. |
Write thermo chemical equation for vaporization of ethanol |
| Answer» Solution :`C_(2)H_(5)OH(1)toC_(2)H_(5)OH(G),DeltaH=+QkJ` | |
| 20. |
Which of the following compound(s) gives brownish-black precipitate, on boiling with Na_(2)CO_(3) to their respective aqueous salt solution ? |
|
Answer» `PB(OH)_(2).2PbCO_(3)` |
|
| 21. |
Which of the following is shown the Lewis formation of N_(2) ? |
|
Answer»
Therefore in lewis structure there are THREE bonding electron pair of triple bond present between two N and one-one electron pair present on both N. 
|
|
| 22. |
When copper oxide is strongly heated with hydrogen it reduced to give metal. If the weight loss suffered by the oxide is 14.9g and weight of water formed is 16.78g, calculate the equivalent weight of oxygen. |
|
Answer» |
|
| 23. |
Valency of an element indicates |
|
Answer» COMBINING POWER with hydrogen |
|
| 24. |
Which of the following is most reactive towards electrophilic substitution reaction ? |
|
Answer» ANILINE |
|
| 25. |
Which is the correct statement for the given acids ? |
|
Answer» Phosphinic acid is a MONOPROTIC acid while phosphonic acid is a diprotic acid |
|
| 26. |
What is sigma bond ? How many sigma bonds are present in C_2H_2 ? |
| Answer» | |
| 27. |
underset((A))(CH_(3)-underset(Br)underset(|)overset(CH_(3))overset(|)(C)-CH_(3))+underset((B)(CH_(3)-overset(CH_(2)-Br)overset(|)(C)H-CH_(3)) CH_(3)-overset(CH_(3))overset(|)(C)H-CH_(3)overset(Br_(2))to the percentage yield of 'A' is (The relative reactivities of 1^(@),2^(@),3^(@)- hydrogens 1:3.8:5) |
|
Answer» 0.36 |
|
| 28. |
Which out of Cu^(2+), Fe^(2+) and Cr^(3+) has highest paramagnetism and why ? |
| Answer» Solution :`Cu^(2+)` has one, `FE^(2+)` has four and `Cr^(3+)` has three unpaired electrons. Hence, `Fe^(2+)` has HIGHEST PARAMAGNETISM | |
| 29. |
Which one of the following is the likely bond angles of sulphur tetrafluoride molecule ? |
| Answer» Answer :d | |
| 30. |
Which acts as a reducing agent ? |
|
Answer» `HNO_3` ELEMENT it its HIGHEST O.S acts as O.A `{:(COOH),(|),(COOH):},2x-8+2=0,x=+3` |
|
| 31. |
Which metals are used as an anode and a cathode in Denial cell ? |
| Answer» SOLUTION :Cu ACTS as cathode and ZN acts as an anode. | |
| 32. |
Which of the following gas has maximum ? |
|
Answer» Methane |
|
| 33. |
What happens when a ferromagnetic substance is heated to high temperature ? |
| Answer» SOLUTION :On heating to high TEMPERATURE, FERROMAGNETIC substances changes to paramagnetic. This is DUE to randomisation of domains (SPINS ) on heating | |
| 34. |
What is extensive property ? |
| Answer» Solution :A property WHOSE value depends on the QUANTITY or SIZE or value of MATTER is called extensive property. | |
| 35. |
Write the expression which gives the energy of electron in the nth shell of hydrogen like particles |
| Answer» SOLUTION :`E_(n) = - (2pi^(2) mZ^(2) e^(4))/(n^(2)h^(2))` where Z is the atomic number of the particles and m and e are the mass and CHARGE of the ELECTRON. | |
| 36. |
X' is a colourless salt giving following reactions : 'X' can be |
|
Answer» `AlCl_(3)` 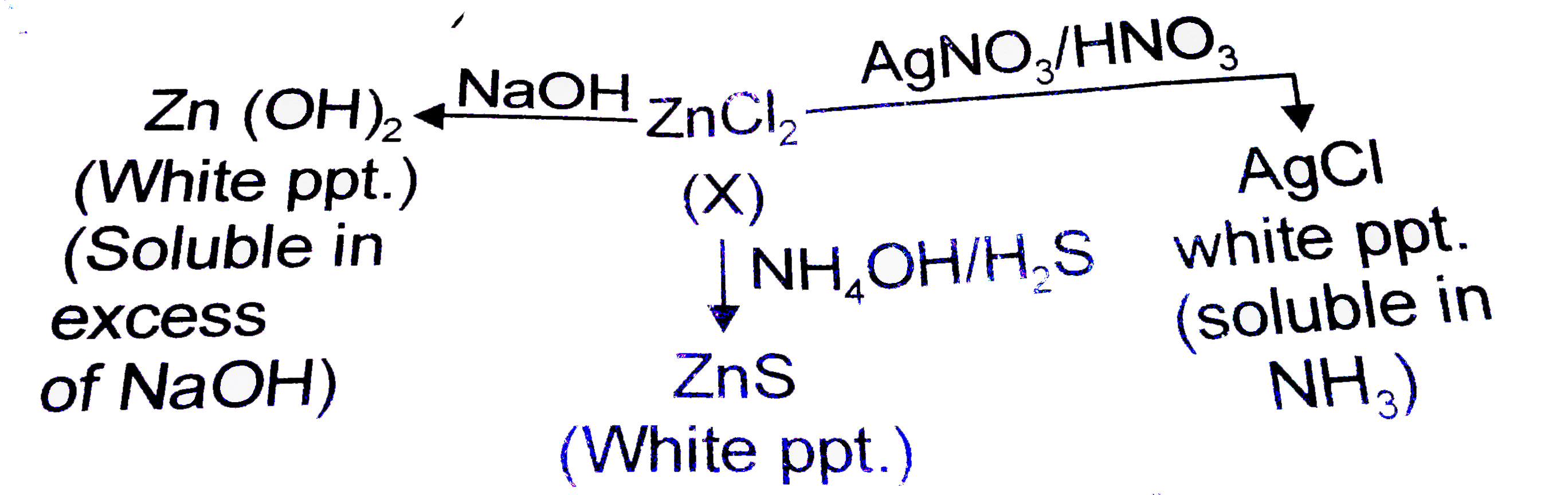
|
|
| 37. |
Which gas has greater contribution in global warming ? |
|
Answer» `CH_4` |
|
| 38. |
The volume of a concentrated H_(2)SO_(4), mixed with 0.5 N KOH to prepare 150 cm^(3) of 0.2 N KOH. Solution is : |
|
Answer» `50 cm^(3)` |
|
| 39. |
Which one of the following when dissolved in water, gives a solution with pH less than seven at 298 K ? |
|
Answer» `CH_(3)COCH_(3)` |
|
| 40. |
Which method is used to estimate sulphur? |
|
Answer» LASSAIGNE's test |
|
| 41. |
Which is not related to lanthanide contraction |
|
Answer» Atomic radii of 4d and 5d series elements are SIMILAR |
|
| 42. |
Which of the following outer electronic configurations corresponds to the d-block of the periodic table ? |
|
Answer» `3s^2 3p^3` |
|
| 43. |
What is the actual configuration of copper(Z=29)?Explain about its stability. |
|
Answer» SOLUTION :Copper (Z=29) Expected configuration :`1s^(2)2s^(2)2p^(6)3s^(2)3p^(6)3d^(9)4S^(2)` Actual configuration:`1s^(2)2s^(2)2p^(6)3s^(2)3d^(10)4s^(1)` The reason is that electronic configuration[Ar]`3d^(10)4s^(1)` and not[Ar]`3d^(9)4s^(2)` due the symmetrical distribution andexchange ENERGIES of d electrons. Symmetry leads to stability.The FULL filled configuration have symmet distribution of electrons and hence they are more stable than unsymmetrical configuration. |
|
| 44. |
What happens when acethylene undergoes ozonolysis ? |
Answer» SOLUTION :When ACETYLENE unndergoes OZONOLYSIS, 
|
|
| 45. |
What is the mass in grams of : (i) 6.022 xx 10^(23) atoms of oxygen (ii) 1.0 xx 10^(23) molecules of H_(2)S (iii) 6.022 xx 10^(23) molecules of oxygen ? |
|
Answer» `6.022xx 10^(23)` MOLECULES of `H_(2)S` have mass = 34.0 g `1.0 xx 10^(23)` molecules of `H_(2)S` have mass `= (34.0xx10^(23))/(6.022xx10^(23))=5.645g` (iii) Mass of `6.022 xx 10^(23)` molecules of oxygen `(O_(2))=32g` |
|
| 46. |
Which of the following is/are chiral? |
|
Answer» Celing fan |
|
| 47. |
Write the postulate of Kossol for formation of Ionic bond. |
|
Answer» Solution :In the periodic Table, the highly electronegative halogens and the highly electropositive alkali metals are separated by the noble gases. The formation of a negative ion from a halogen atom and a positive ion from an alkali metal atom is associated with the gain and loss of an electron by the respective atoms. The negative and positive ions thus FORMED attain stable noble gas electronic configurations. The noble gases (with the EXCEPTION of helium which has a duplet of electrons) have a particularly stable outer shell configuration of EIGHT (octet) electrons `ns^(2) np^(6)`. The negative and positive ion are stabilized by electrostatic attraction. Kossel.s postulation provide the basis for modem concepts REGARDING ion-formation by electron transfer and the formation of ionic crystalline COMPOUNDS. |
|
| 48. |
Which of the following pair of reactants are involved the acid-catalyzed aldol reaction? |
|
Answer»
|
|
| 49. |
Why BBr_(3) is a strongerLewis acid as compared to BF_(3) throughflorine is more electronegative than bromine ? |
| Answer» Solution :The B atom in `BF_(3)`or `BBr_(3)`has only six electron inits valenceshell andhence can accepta pair ofelectronsto completeits octet.Therefore,both `BF_(3)` and `BBr_(3)` at as Lewisacids. But in `BF_(3)`,the sizes of empty `2p-`orbitalof B ANDTHE 2p-orbital ofF containing thelone pairof electronsare almostidenticaland henceeffective `ppi - ppi`boundingoccurs. As aresult,the lone pair of F is donate to Batom and hence the electrons deficiencyof borondecreases. Incontrast, in `BBr_(3)`.the size of 4p-orbitalov Br containingthe lone pair of electronsis much bigger than the empty 2p-orbitalof Band hencedonationof lone pairof electronsof Br toB does not occurto an significationextent. As a result, the electron deficiencyof B is much higherto `BBr_(3)`than in `BF_(3)`, and hence`BBr_(3)` i a stronger Lewisand than `BF_(3)`. | |
| 50. |
Which of the two, NO_(2)CH_(2)CH_(2)O^(oplus) or CH_(3)CH_(2)O^(oplus) is expected to be more stable and why? |
Answer» SOLUTION : (I) is more stable than (II) because`NO_(2)` group has `-I` effect and HENCE it tends to disperse the `-ve` charge on the O-atom. In CONTRAST, `CH_(3)CH_(2)` has `+I` effect.It THEREFORE, tends to intensify the `-ve` charge and hence destablizes it. |
|