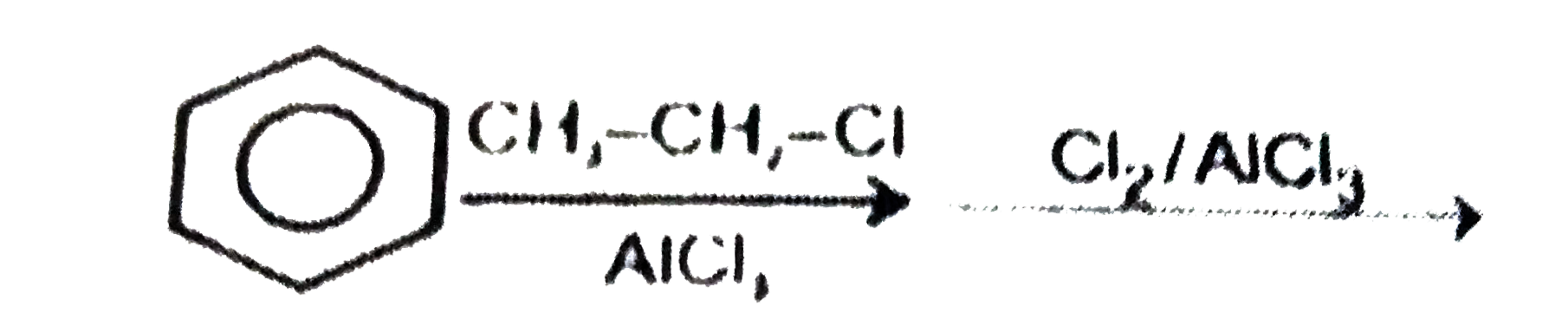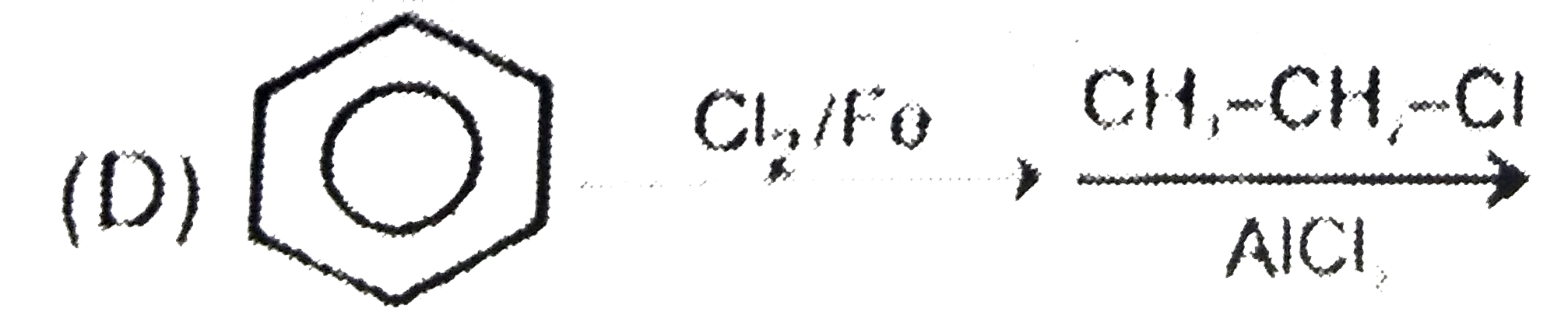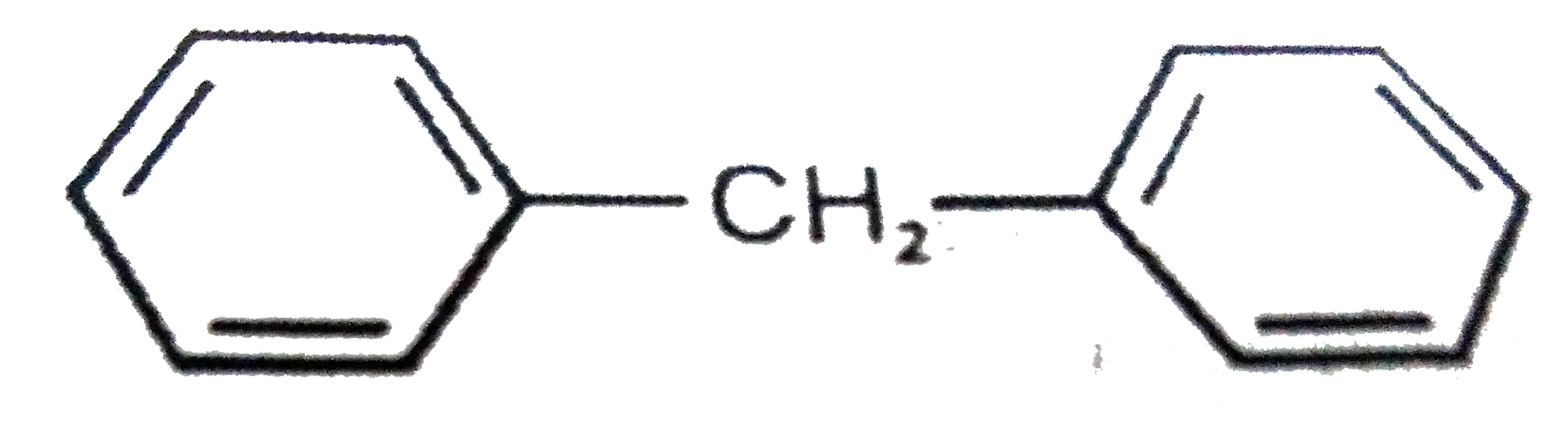Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Use the data given below to find the type of cubic lattice to which the crystal of iron belongs a/pm=286 , rho//g cm^(-3)=7.86 |
|
Answer» |
|
| 2. |
The test for unsaturations is confirmed by the decolourisation of which of the following. |
|
Answer» Iodine water |
|
| 3. |
What will be the spontaneous reaction between the following half cell reactions ? (i) Cr^(3+)(aq)+3e^(-) rarr Cr(s) ""E^@=-0.74V (ii) MnO_2(s)+4H^(+)+2e^(-)rarr Mn^(2+)(aq)+2H_2O(l)""E^@=1.28V Calculate E_("cell")^@ |
|
Answer» Solution : Since the reduction POTENTIAL REACTION (ii) is more than that of reaction (i), reaction (ii) will occur as reduction . Therefore , reaction (i) should be written as oxidation . To obtain the net reaction , we multiply by appropriate COEFFICIENTS so that electrons get cancelled . `MnO_2(s)+4H^(+)(AQ)_+2E^(-)rarrMn^(2+)(aq)+2H_2O(l)]xx3` `""Cr(s)rarrCr^(3+)(aq)+3e^(-)]xx2` `2Cr(s)+3MnO_2(s)+12H^(+)(aq)rarr2Cr^(3+)(aq)+3Mn^(2+)(aq)+6H_2O` `E_("cell")^@=E_("Substance reduced")-E_("Substance oxidised")` `=1.28-(-0.74)=2.02V` |
|
| 5. |
Which is soluble both in water and acetone? |
| Answer» Solution :N//A | |
| 6. |
The temporary hardness of water due to calcium bicarbonate can be removed by adding |
|
Answer» `CaCO_(3)` |
|
| 7. |
The weight of 350 ml of a diatomic gas is 1 gram at 0^@C and 2 atm. pressure. The weight of one atom in grams is |
|
Answer» `32/N` |
|
| 8. |
Which doesn't cause coagulation of colloidal solution ? |
|
Answer» Filtration |
|
| 9. |
Write Gibb's equation |
|
Answer» |
|
| 10. |
The triple point of CO_(2) occurs at 5.1 atm and -56^(@)C. Its critical temperature is 31^(@)C. Solid CO_(2) is more dense than liquid CO_(2). Under which combination of pressure and temperature is liquid CO_(2) stable at equilibrium? |
|
Answer» 10 atm and `-25^(@)C` |
|
| 11. |
Which quantity out of Delta_(r)G andDelta_(r)G^(c-) will be zero at equilibrium ? |
| Answer» Solution :`Delta_(R)G= Delta_(r)G^(@) + RT LN K`. When reaction attains EQUILIBRIUM`Delta_(r) G=0` so that`Delta_(r)G^(@)=- RT` ln K . Thus `Delta_(r)G^(@) =0` only when `K+1` . For all other VALUES of `K, Delta_(r)G^(@) ` will be non-zero. | |
| 12. |
Tritium, ._(1)T^(3)(an isotope of H) combine wityh flurine to form a weakacid TF whch ionises to give T^(+) prepared dilute aquios solution of TF has a pt (euivalentof pH) fo 1.7 and frezze at - 0.372^(@)C. If 600mL of freshly prapared soltuion were allowed to stand for 24.8 years, calculate: (i() Ionisation constation of TE (ii) Charge carried by beta- particles emitted by tritium in faraday. Given: K_(f) for H_(2)O = 1.86, t_(1//2) (T) = 12.4yrs. |
|
Answer» |
|
| 13. |
Which of the following is not the configuration of lanthanoid |
|
Answer» `[Xe]4f^(10).6s^(2)` |
|
| 14. |
Which of the following compounds exhibits geometrical isomerism? underset((i))(CH_(2)=CHCH_(3))underset((ii))(CH_(3)CH=CHCH_(3)) underset((iii))(CH_(3)CH=C)=CHCH_(3)underset((iv))(CH_(3)CH=C=C=CHCH_(3)) |
|
Answer» (ii), (iv) |
|
| 15. |
What is electron gain enthalpy (Delta_(eg)H) ? When its value becomes positive and negative ? |
|
Answer» Solution :When an electron is added to a neutral gaseous atom (X) to convert it into a negative ion, the enthalpy change accompanying the process is defined as the .Electron Gain Enthalpy `(Delta_(eg) H)`.. Electron gain enthalpy provides a measure of the ease with which an atom adds an electron to form anion as REPRESENTED by EQUATION. `X_(Gg) + e^(-) to X_((g))^(-) : Delta_(eg) H ` Depending on the element, the process of adding an electron to the atom can be either endothermic or exothermic. For many elements energy is released when an electron is added to the atom and the electron gain enthalpy is negative. For example, group 17 elements (the halogens) have very high negative electron gain enthalpies because they can attain stable noble gas electronic configurations by PICKING up an electron. On the other hand, noble GASES have LARGE positive electron gain enthalpies because the electron has to enter the next higher principal quantum level leading to a very unstable electronic configuration. It may be noted that electron gain enthalpies have large negative values toward the upper right of the periodic table preceding the noble gases. |
|
| 16. |
Which of the following has a regular geometry ? |
|
Answer» `CHCl_(3)` `XeF_(6)` tetrahedral octahedral `SF_(4)` = DISTORTED TRIGONAL bipyramidal . |
|
| 17. |
What do you understand by the term .auto protolysis. of water ? What is its significance ? |
|
Answer» Solution :The auto-protolysis (self-ionization) of WATER takes place as follows: `{:(H_2O_((l))+, H_2O_((l)) hArr , H_3O_((AQ))^(+)+, OH_((aq))^(-)),("acid-1", "base-2","acid-2","base-1"),("(acid)","(base)","(conjugate acid)","(conjugate base)"):}` (ii) REDOX Reactions Involving Water : Water can be easily reduced to dihydrogen by highly ELECTROPOSITIVE metals. `2H_2O_((l)) + 2Na_((S)) to 2NaOH_((aq))+ H_(2(g))` Thus, it is a great source of dihydrogen. Water is oxidised to `O_2` during photosynthesis. `6CO_(2(g)) + 12H_2O_((l)) to C_6H_12O_(6(aq)) + 6H_2O_((l)) + 6O_(2(g))` Withfluorine also it is oxidised to `O_2` `2F_(2(g)) + 2H_2O_((l)) to 4H_((aq))^(+) + 4F_((aq))^(-) + O_(2(g))` |
|
| 18. |
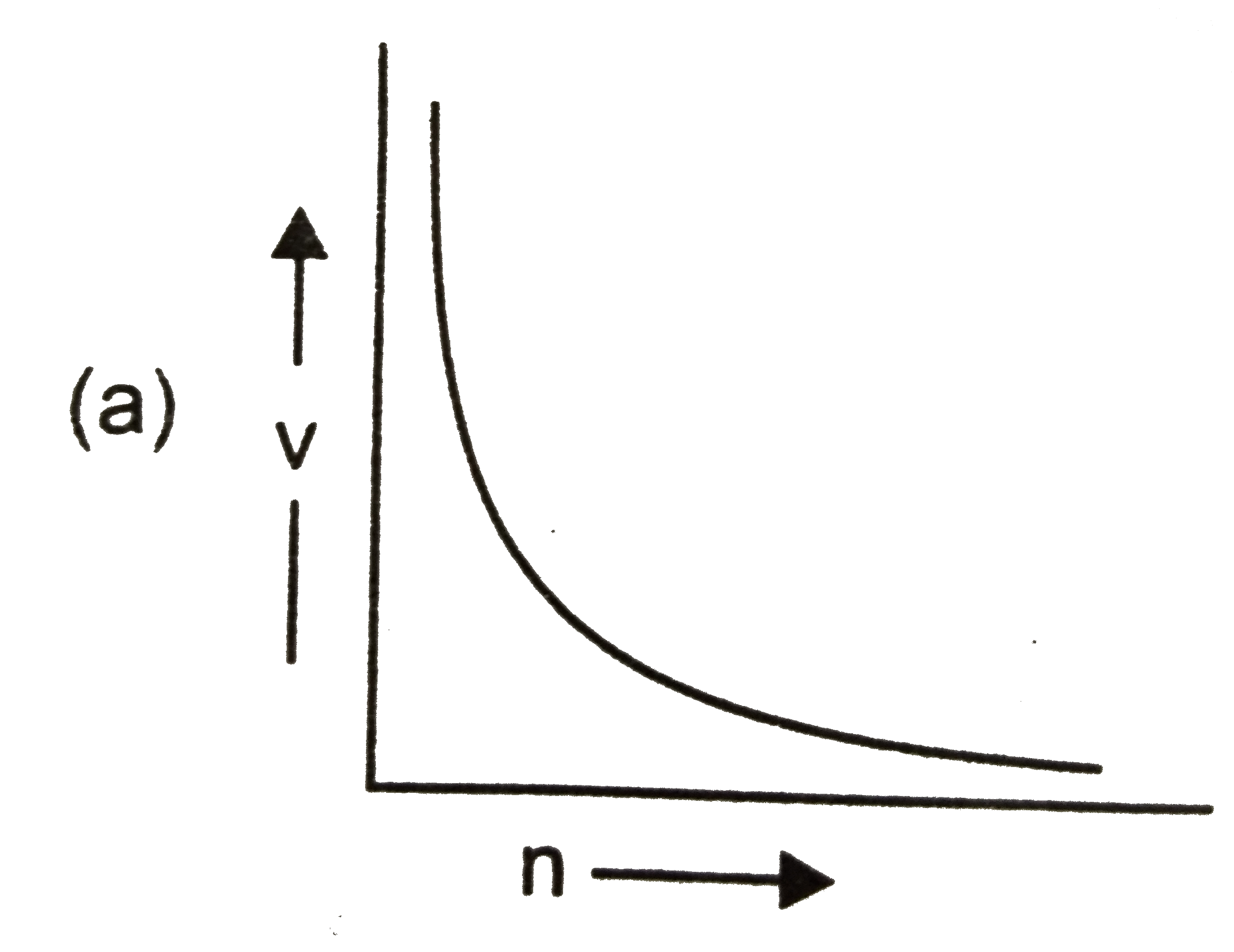
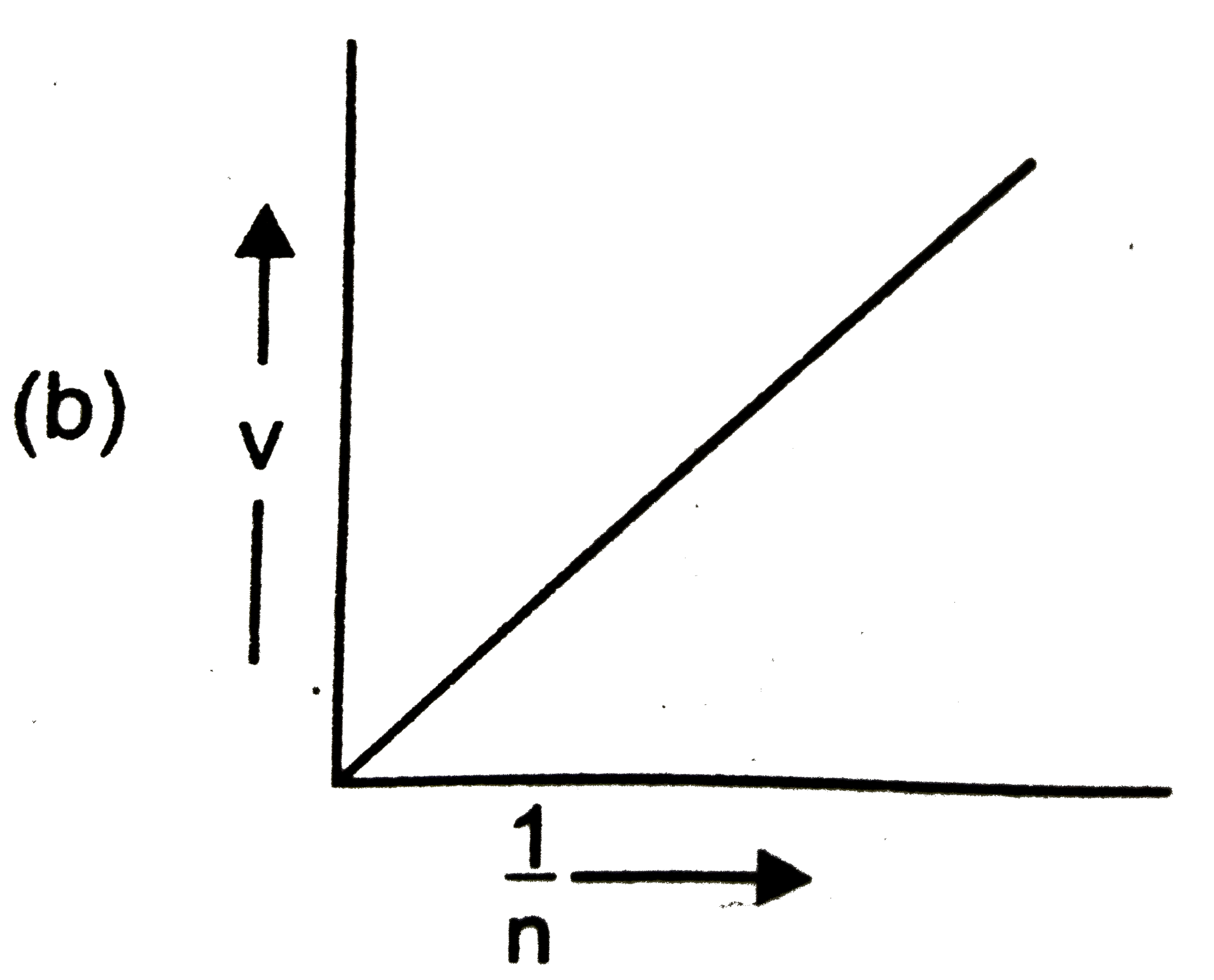
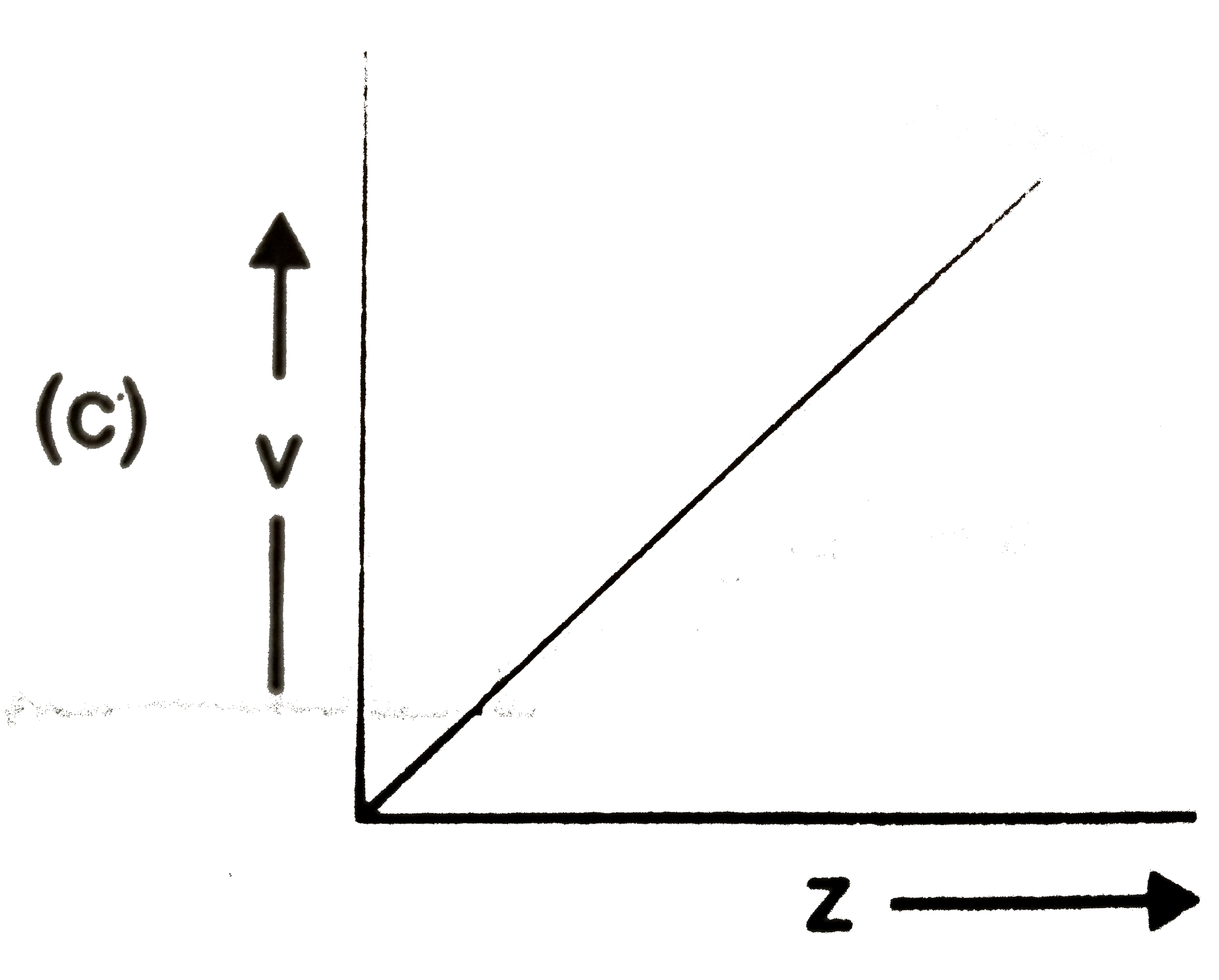
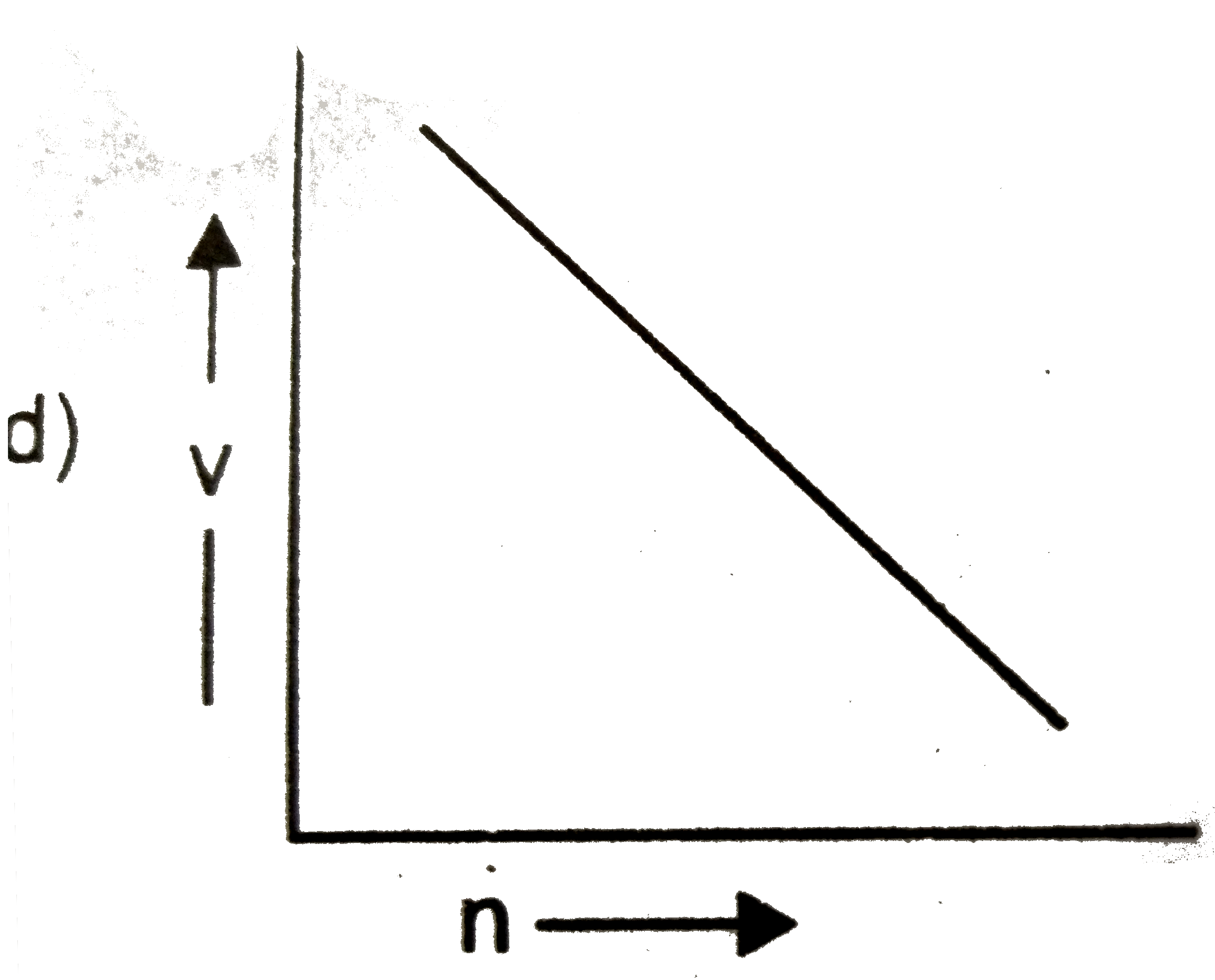
Which of the following graphs is incorrect? |
|
Answer»
|
|
| 19. |
Which point defects in crystals of a solid does not change the density of the solid ? |
| Answer» SOLUTION :FRENKEL DEFECT | |
| 20. |
What will be the change in valency down the group in the periodic table? |
|
Answer» INCREASES |
|
| 21. |
Which one is natural fertilizer ? |
|
Answer» Rhizobium |
|
| 22. |
What happen when nitrile undergoes acid hydrolysis ? |
|
Answer» Solution :When alkyl NITRILE undergoes acid HYDROLYSIS to GIVE amide,m which on further hydrolysis to give carboxylic acid. `underset("(NITRILA)")(R-CN)overset(H^(+)|H_(2)O)(to)R-underset("(Amide)")(CONH_(2))overset(H^(+)|H_(2)O)(to)underset("(carbosylic ackd)")(R-COOH)` |
|
| 23. |
The velocity of an electron of mass 9.1xx10^(-31) kg moving round the nucleus in the Bohr orbit (diameter of the orbit is 1.058Ã…) is 2.2xx10^(-6)m sec^(-1). If momentum can be measured within the accuracy of 1%, then calculate uncertainty in position (Deltax) of the electron |
|
Answer» |
|
| 24. |
What will be the nature of forces at critical conditions for a real gas? |
|
Answer» Attractive |
|
| 25. |
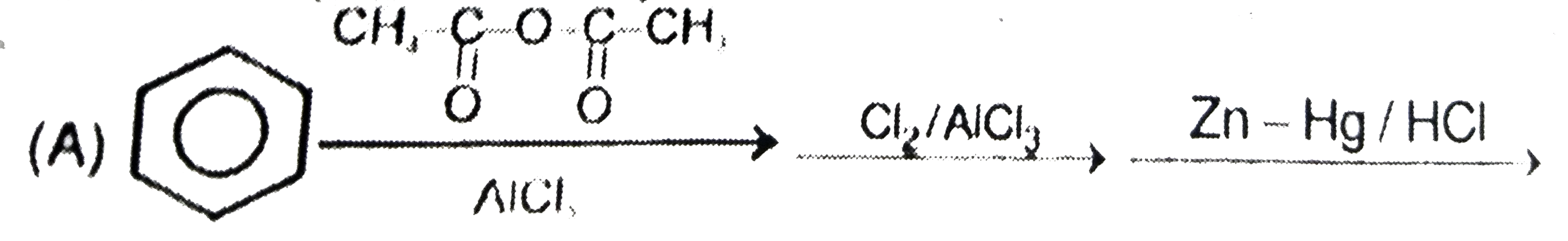
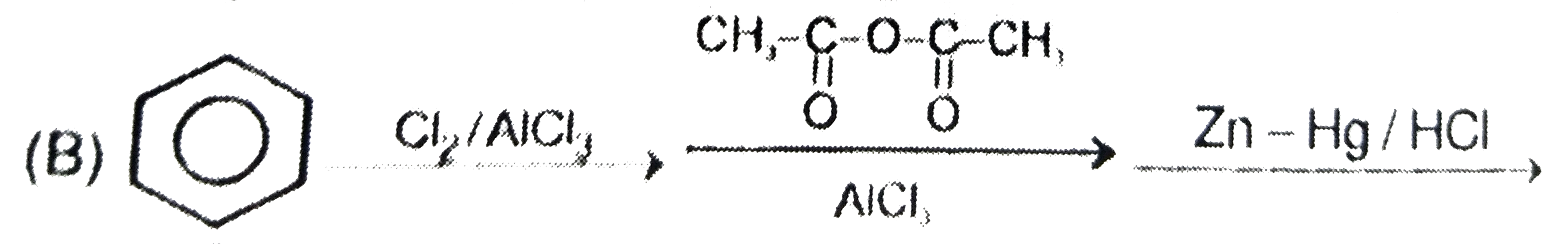
Which step is used to produce 1-Chloro-3-ethylbenzene |
|
Answer»
|
|
| 26. |
What is relation between compressibility factor and molar volume. |
| Answer» SOLUTION :`Z=(V_("REAL"))/(V_("IDEAL"))` | |
| 27. |
When the oxidation number of an element is maximum it can act only as……………… |
|
Answer» |
|
| 28. |
Which of the following carbocation is most stable ? (CH_(3))_(2)CH-overset(+)(C)HCH_(3) and (CH_(3))_(2)overset(+)(C)-CH_(2)CH_(3) |
| Answer» SOLUTION :`(CH_(3))_(2)OVERSET(+)(C)CH_(2)CH_(3)` is most stable , because it can FORM `3^(@)`-carbocation. | |
| 29. |
When an electron adds on F atom, It becomes __________ |
|
Answer» It acts as a REDUCING agent |
|
| 30. |
Which of the following compounds will notundergo friedal crafts reactioneasily? |
|
Answer» nitrobenzeen |
|
| 31. |
The tendency of alkali metal cations to undergo hydration............ in going from Li^(+) " to " Cs^(+). |
| Answer» SOLUTION :DECREASES | |
| 32. |
The two ions A^(+) and B^(-) have radii 88 and 200 pm respectively. In the close packed crystal of compound AB, predict the corrdination number of A^(+) . |
|
Answer» Solution :`(r_(+))/(r_(-)) = (R(A^(+)))/(r(B^(-)))= (88"PM")/(200"pm") = 0.44` It lies in the range0.414 to 0.732 HENCE, the cordination number of ` A^(+) = 6` |
|
| 33. |
Which of the following compound is produced when alpha-chloroacetic acid is heated with Ag powder ? |
|
Answer» Acetic acid |
|
| 34. |
Which of the following statements are false regarding BeCl_(2) |
|
Answer» In SOLID state, `BeCl_(2)` exits in the form of linear structure |
|
| 35. |
Which one of the alkaline earth metal carbonates is thermally the most stable ? |
|
Answer» `MgCO_3` |
|
| 36. |
Why an ionic bond is formed between two elements having large difference in their electrongativity? |
|
Answer» Solution :The more electronegative element will attract the shared pair of electrons to such a large EXTENT extent than the other that it will amount to transfer of electron RESULTING in the FORMATION of ions . |
|
| 37. |
The solubility product of BaSO_(4) at 18^(0) C is 1.5xx10^(-9) . It solubility (mole lit^(-1)) at the same temperature is |
|
Answer» `1.5 XX 10^(-9) ` `S = sqrt( K_(sp) ) = sqrt( 1.5 xx 10 ^(-9) )= 3.9 xx 10 ^(-5) M` |
|
| 38. |
Treatment of phenol with acetyl chloride produces |
|
Answer» METHYL benzoate 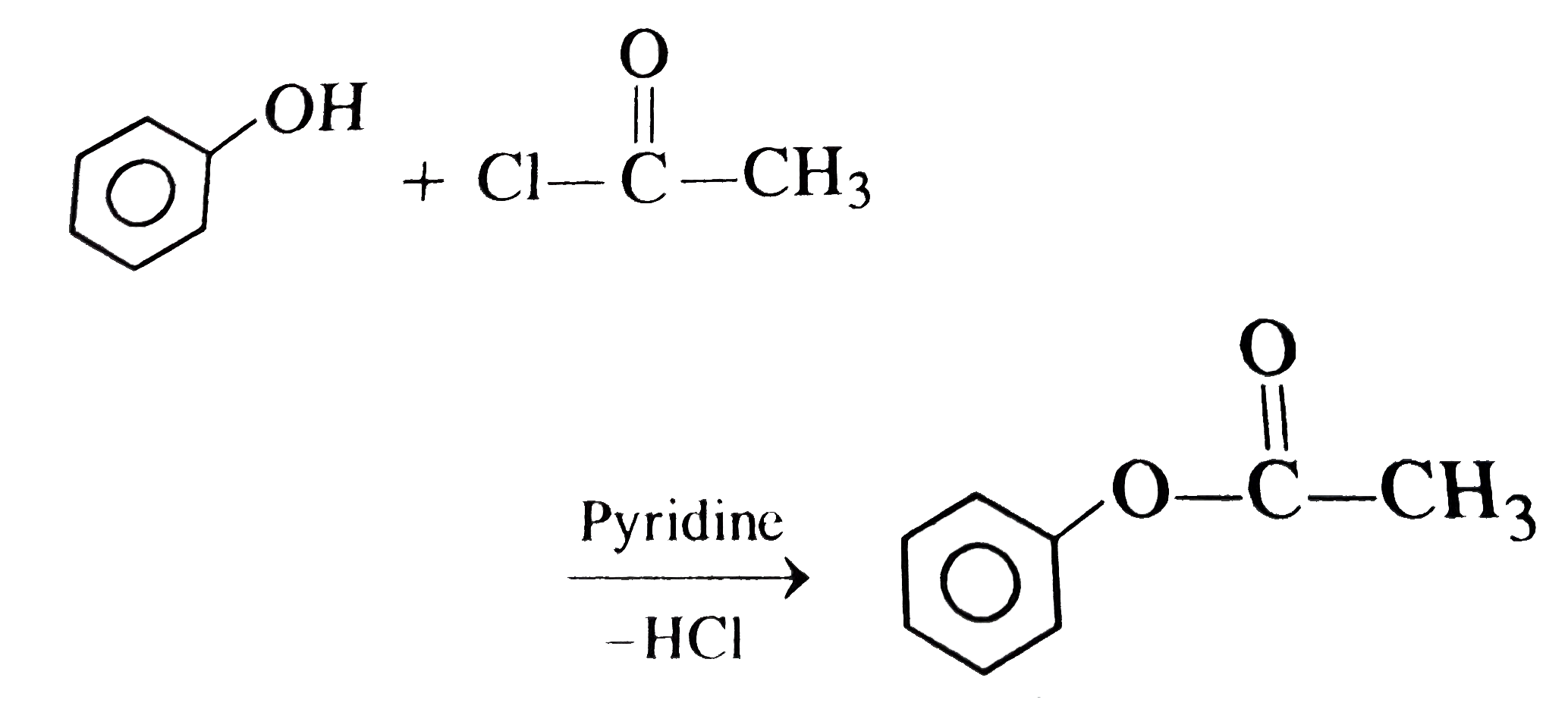
|
|
| 39. |
Who developedquantummechanics ? |
| Answer» SOLUTION :Quantummechanicswasdevelopedindependentyin 1926 byWernerHeisenbergand ErwinSchrodinger . | |
| 40. |
Which one of the following pairs do not impart colour to the flame? |
|
Answer» `BeCl_(2) and SrCl_(2)` |
|
| 41. |
Write the products of following reactions (a) CH_(3)-C-=N overset(H_(3)O^(-))rarr(b) CH_(3)NC overset(H_(3)O^(+))rarr |
|
Answer» |
|
| 42. |
What do you understand by the term .auto protolysis. of water? What is its significance ? |
|
Answer» Solution :Autoprotolysis means self ionisation of water. It may be represented of `underset("Acid"_1)(H_2O_((l)))+ underset("BASE"_2)(H_2O_((l))) hArr underset("Acid"_2)(H_3O_((aq))^(+)) +underset("Base"_1)(OH_((aq))^(-))` Due to autөprotolysis, water is amphoteric in NATURE. i.e. it reacts with both acids and BASES. If acts as a base towards acids stronger than itself and as an acid towards base stronger then itself. For example: `underset("Acid"_1)(H_2O_((l)))+ underset("Base"_2)(NH_(3(aq))) to underset("Acid"_2)(NH_(4(aq))^(+)) +underset("Base"_1)(OH_((aq))^(-))` `underset("Base"_1)(H_2O_((l)))+ underset("Acid"_2)(H_2S_((aq)))to underset("Acid"_1)(H_3O^(+))+ underset("Base"_2)(HS^-)` |
|
| 43. |
What happeas when alkali metals react with halogens? |
|
Answer» SOLUTION :Alkali metals reacts with HALOGENS vigorously to from metal halides. `2 M + X_(2) to 2M^(+) X^(-)` (metals HALIDE) EX: `2Na + CI_(2) (g) overset(Delta)(to) underset("Sodium chloride")(2Na^(+) CI^(-))` |
|
| 44. |
The value of carbon-carbon double bond length is …………………… |
|
Answer» 1.43Å |
|
| 45. |
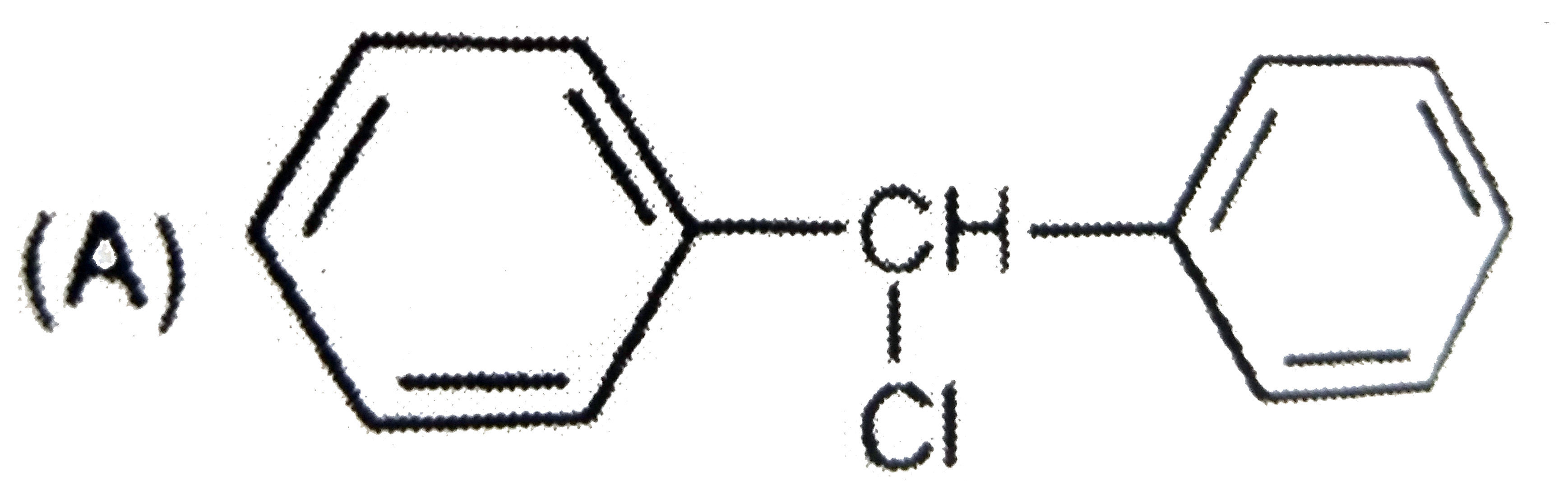
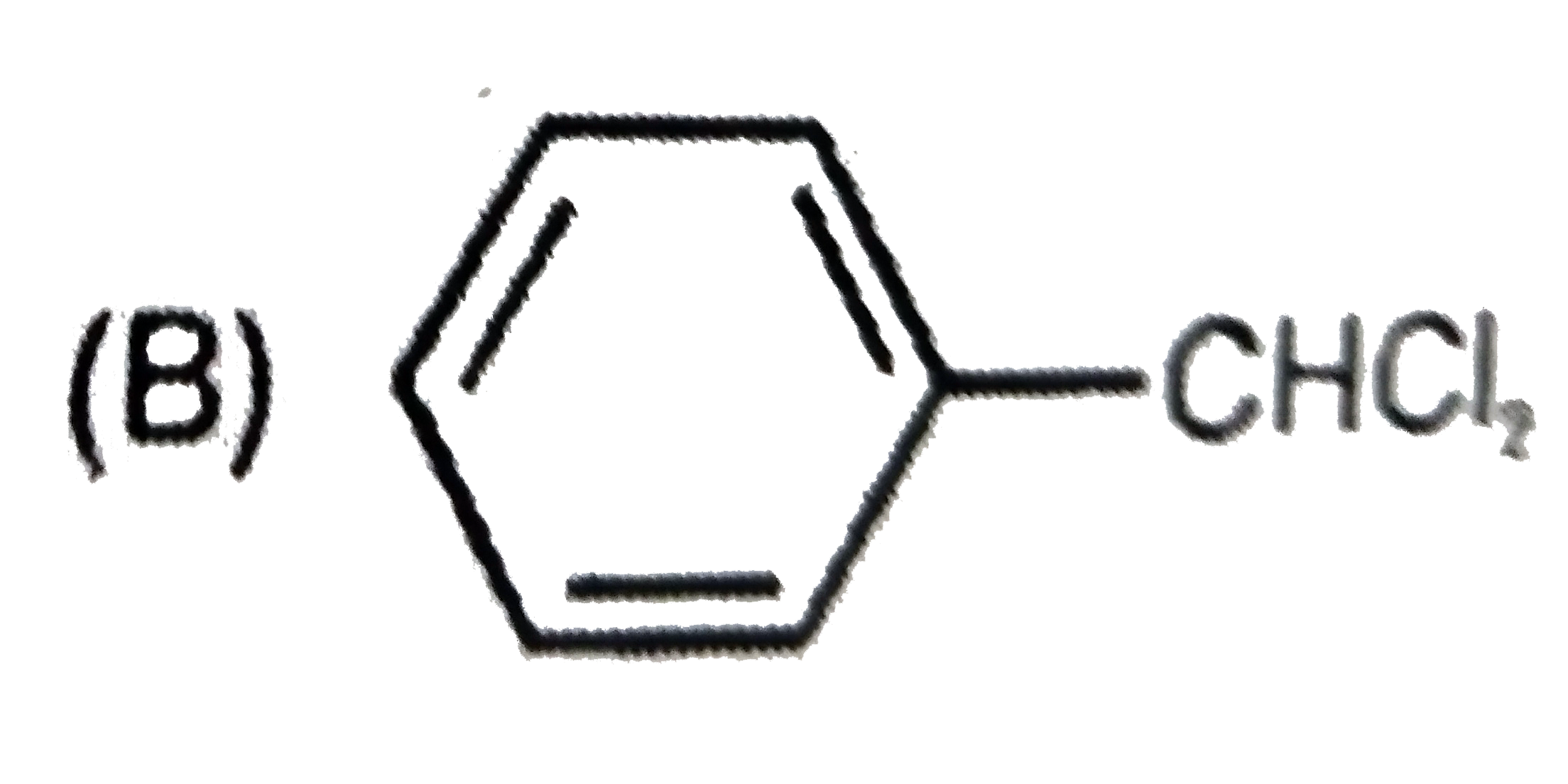
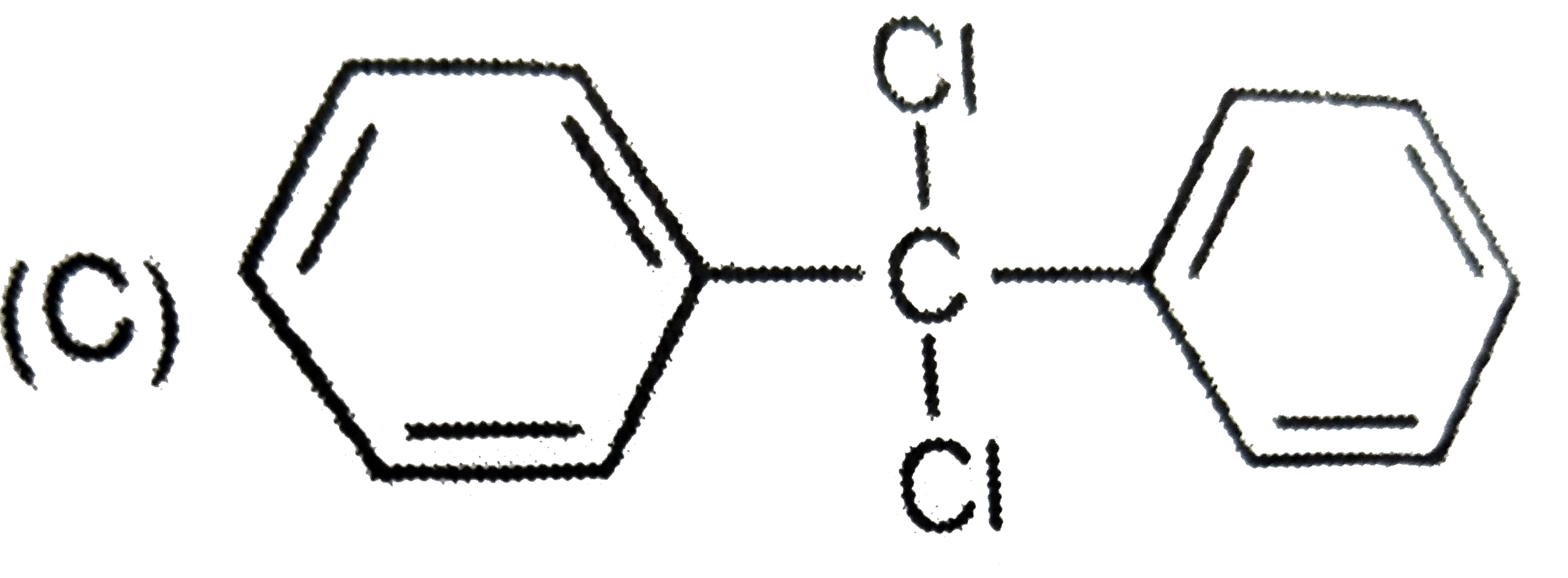
Which of the following structures correspond to the product expected , when excess of C_6H_6 reacts with CH_2Cl_2 in presence of anhydrous AlCl_3. |
|
Answer»
|
|
| 46. |
The uncertainty in the position of a flying dust particle of mass 0.1 mg is found to be 10^(-4)m. The uncertainty in the position of another flying dust particle of mass 0.5 mg is also found to be same The uncertainty in the velocity of the lighter dust particle is nearly |
|
Answer» `5 xx 10^(-24) ms^(-1)` `Delta x = 10^(-4) m` `Delta x.(m Delta v) = (H)/(4pi)` `:. 10^(-4) m (0.1 xx 10^(-6) kg xx Delta v)` `= (6.6 xx 10^(-34) kg m^(2) s^(-1))/(4 xx 3.14)` or `Delta v = 5.25 xx 10^(-24) ms^(-1)` |
|
| 47. |
The uncertainty in the position of a flying dust particle of mass 0.1 mg is found to be 10^(-4)m. The uncertainty in the position of another flying dust particle of mass 0.5 mg is also found to be same If a third flying dust particle of mass 1 mg had the same uncertainty in position as in its velocity, the uncertainty in its momentum would be |
|
Answer» `5.2 xx 10^(-28) kg ms^(-1)` Hence, `Delta x (m Delta v) = (h)/(4pi)` gives `x xx (10^(-6) kg xx x) = (6.6 xx 10^(-34) g m^(2) s^(2))/(4 xx 3.14)` or `x^(2) = 52.5 xx 10^(-30) or x = 7.25 xx 10^(-15)` `:. Delta p = m Delta v = 10^(-6) kg xx 7.25 xx 10^(-15) ms^(-1)` `= 7.25 xx 10^(-21) kg m s^(-1)` |
|
| 48. |
The uncertainty in the position of a flying dust particle of mass 0.1 mg is found to be 10^(-4)m. The uncertainty in the position of another flying dust particle of mass 0.5 mg is also found to be same The uncertainty in the velocity of the heavier dust particle as compared to the lighter dust particle is |
|
Answer» 5 times As `Delta x = Delta x' :. m Delta v = m' xx Delta v'` or `(Delta v')/(Delta v) = (m)/(m') = (0.15mg)/(0.5mg) = (1)/(5)` |
|
| 49. |
Which hydrated halide compound shows hydrolysis? |
|
Answer» `MgCl_(2)*8H_(2)O` |
|
| 50. |
What is the difference between the terms 'hydrolysis' and 'hydration'? . |
|
Answer» Solution :Hydrolysis is a CHEMICAL reaction in which a substance reacts with water under neutral, ACIDIC or alkaline conditions. `Na_(21)CO_(3)+2H_(2)Oto2NaOH+H_(2)CO_(3)` SALT 1 Base 2 Acid 1 Hydration on the other hand is the property of a chemical compound to take up molecules of water of crystallisation and get HYDRATED. `CuSO_(4(s))+5H_(2)O_((l))+CuSO_(4)5H_(2)O_(s)` |
|