Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What do you understand by the term formality? |
|
Answer» SOLUTION :Formality (F) is defined as the number of formula weight of solute present IN1 LITRE of the solution . Formality (F) = `("Number of formula weight of solute")/("volume of the solution (in L)")` |
|
| 2. |
Which of the following reagent is not used to convert alcohol to haloalkane? |
|
Answer» `H - X` |
|
| 3. |
The volume of water to be added to 100cm^(3) of 0.5 N H_(2)SO_(4) to get decinormal concentration, is |
|
Answer» 100 `cm^(3)` i.e.`0.5xx100=0.1xxV_(2)orV_(2)=500cm^(3)` `therefore` WATER to be added to `100CM^(3)` solution `= 500-100=400cm^(3)` |
|
| 4. |
Which of the following sets of atomic number belong to that of alkali metals ? |
|
Answer» `1, 12, 30, 4, 62` |
|
| 5. |
Use the periodic table to answer the following questions. (a) Identify an element with five electrons in the outer subshell.(b) Identify an element that would tend to lose two electrons.(c) Identify an element that would tend to gain two electrons. (d) Identify the group having metal, non metal, liquid as well as gas at the room temperature. |
|
Answer» Solution :(a) Chromium `(Z = 24)`: It have five electrons in the outer 3d subshell. (b) Magnesium (Mg) (Z = 12): Can lose TWO electrons readily. (c) Oxygen (O) (Z = 8): Can gain two electrons. (d) Halogens (Group 17): It has metal (iodine), non-metals (F, CI, Br), liquid bromine and GASES. |
|
| 6. |
The wave function of 2s electron is given by Psi_(2s) = (1)/(4 sqrt(2pi)) ((1)/(a_(0)))^(3//2) (2 - (r)/(a_(0))) e^(-r//a_(0)) It has a node at r = r_(0). Find the relation between r_(0) and a_(0) |
|
Answer» Solution :The PROBABILITY of finding 2s ELECTRON will be : `Psi_(2s)^(2) = (1)/(32pi) ((1)/(a_(0)))^(3) (2- (r_(0))/(a_(0)))^(2)e^(-2R//a_(0))` Node is the point at which probability of finding electron is zero. Thus, `Psi_(2s)^(2) = 0` when `R = r_(0)` `:. (1)/(32pi) ((1)/(a_(0)))^(2) (2 - (r_(0))/(a_(0)))^(2) e^(-2r_(0)//a_(0)) = 0` In this expression, the only factor that can be zero is `(2 - (r_(0))/(a_(0)))` Thus, `2 - (r_(0))/(a_(0)) = 0 or r_(0) = 2 a_(0)` |
|
| 7. |
What do you understand by the term non stoichiometric hydrides? Do you expect these types of hydries t be formed from alkali metals? Justify answer. |
| Answer» SOLUTION :For non stoichiometric, consult section 9.6. Alkali metals do not form these type of HYDRIDES because in their crystal lattices,atoms of hydrogen, which are very small in size do not FIT in. | |
| 8. |
What happence to the equilibriumPCl_(5) (g) hArr PCl_(3) (g) + Cl_(2) (g), if nitrogen gas is added to it (i)at constant volume(ii) at constant pressure ? Give reasons. |
|
Answer» Solution :(i) The state of EQUILIBRIUM remains unaffected (ii) DISSOCIATION INCREASES (i.e., equilibrium shifts FORWARD ).For REASON |
|
| 9. |
Which one of the following has found to have abnormal molar mass ? |
| Answer» Answer :D | |
| 10. |
When iodine is heated under atmospheric pressure conditions it transforms to vapours without passing through the liquid state . If he triple point pressure of a system is high and unless external pressure is applied to exceed the triple-point pressure sublimation will take place . |
| Answer» SOLUTION :EXPLANTION is CORRECT REASON for STATEMENT | |
| 11. |
What will be the molecular orbital order of CO molecule ? |
|
Answer» `pi 2p_(X) = pi 2p_(y) gt sigma 2p_(z)` |
|
| 12. |
Why akali metals are normally kept in kerosene oil ? |
|
Answer» SOLUTION :This is because in the air they are easily oxidized to oxide which may dissolve in the moisture of the air to form hydroxides or they also combine directly with water VAPOURS present in moisture to form hydroxides . The hydroxides , in turn , may combine with `CO_(2)` of the atmosphere to form carbonates. `4 M + O_(2) to 2 M_(2)O , M_(2)O + H_(2) O to 2 MOH` `2 MOH + CO_(2) to M_(2) CO_(3) + H_(2)O` Therefore , to protect them from air and moisture , they are kept under kerosene OIL . |
|
| 13. |
Which two metal elements are used in photoelectric cell? |
|
Answer» Na and K |
|
| 14. |
Zn+CuSO_(4)rarrZnSO_(4)+Cu, Zn can act as |
|
Answer» Oxisiding agent |
|
| 15. |
What are Subshells? |
|
Answer» <P> Solution :Subshell concept is introduced in order to explain the give structure of hydrogen. i.e. EVERY shell consists of subshells which slightly DIFFER in their energies ex:- K shell has oniy one subshell (S - subshell), L shell has to subshells (Sand P subshells) M shell has THREE subshells (S, P and d subshells) |
|
| 16. |
Which and how many groups present in periodic table ? Explain the factors which depends upon it. |
|
Answer» Solution :There are four GROUP present in PERIODIC table which are s, p, d, f. Which is depend upon atomic ORBITALS which are filled by electrons. These four groups are SHOWN in figure. 
|
|
| 17. |
What is standard electrode potential ? |
| Answer» SOLUTION :At 298 K TEMPERATURE and unit concentration POTENTIAL of electrode potential. | |
| 18. |
Which statement about solubility product is/are correct? |
|
Answer» It is the product of ionic concentration of a slightlysoluble salt in its saturated solution at a certain temp. |
|
| 19. |
The reason for small radius of Ga compared to Al is …… |
|
Answer» poor screening effect of d and f-orbitals. |
|
| 20. |
Which is/are true statement/reactions? |
|
Answer» `Al_(4)C_(3)+H_(2)OtoCH_(4)` |
|
| 21. |
Which of the following reactions gives H_2C = C = C = CH_2 |
|
Answer» `CH_2Br- CHBr = CH_2 overset(Zn// CH_3OH)to` b) `HC -= C - CH_2 - COOH overset(aq. Na_2CO_3)to` `HC-=C-CH_2-COONa overset(DELTA)to HC -= C - CH_3` C) `BrCH_3 - C -= C-CH_2 Br overset("Zn, heat")to H_2C = C = CH_2+ ZnBr_2` D) `2CH_2 = CH - CH_2I + Zn overset("heat")to` `CH_2 = CH-CH_2 - CH_2 - CH = CH_2 + ZnI_2` |
|
| 22. |
The rms velocity of an ideal gas at 27° is 0.3 m/s. Its rms velocity at 927° C in m/s is |
| Answer» | |
| 23. |
Which is not a method of preparing carbon monoxide on a commercial scale? |
|
Answer» `C_(S) + H_(2) O_((G)) OVERSET(473 - 1273 K)(to) CO_((g)) + H_(2(g))` |
|
| 24. |
Which of the following compounds will not exist as resonance hybrid. Give reason for your answer : (i) CH_(3)OH (ii) R-CONH_(2) (iii) CH_(3)CH=CHCH_(2)NH_(2) |
|
Answer» Solution :(i) `CH_(3)-OH` does not contain `pi`-electrons and HENCE it cannot be regarded as a resonance hybrid. (II) Due to the presence of `pi`-electrons in C = O bond and n-electrons on N, amide can be represented as a resonance hybrid of the following THREE resonating structures. 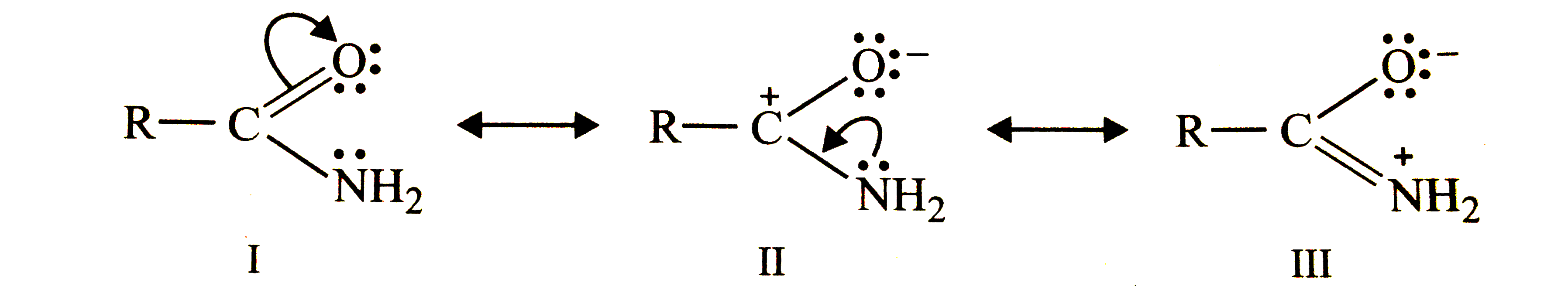 (iii) Since the lone pair of electrons on the N atom is not conjugated with the `pi`-electrons of the double bond, therefore, resonance is not possible. `CH_(3)-CH = CH - CH_(2) - overset(..)(N)H_(2)` Resonance not possible. |
|
| 25. |
Which of the following are correctly matched |
|
Answer» `Be(OH)_(2) lt Mg (OH)_(2) GT Ca(OH)_(2) gt Ba(OH)_(2)` [Basic strength] The solubility of sulphides in water decreases from Be to Ba Basic strength of II group metal oxides increase down the group |
|
| 26. |
Which of the following configuration may represent the ground state of nitrogen atom ? |
|
Answer»
|
|
| 27. |
What will be the minimum pressure required to compress 500 dm^(3) of air at 1 bar to 200 dm^(3) at 30^(@)C? |
|
Answer» <P> Solution :THUS, `P_(1)= 1 "bar" P_(2) = ?``v_(1)=500 dm^(3) "" v_(2)=200 dm^(3)` TEMPERATURE remain constant According to Boyle.s law `P_(1)v_(1)=P_(2)v_(2) or P_(2) =(P_(1)v_(1))/(v_(2))` `p_(2)=(1 "bar" xx500 dm^(3))/(200 dm^(3))=2*5 "bar"` |
|
| 28. |
Thermal decomposition of (NH_(4))_(2)Cr_(2)O_(7) involves. |
|
Answer» OXIDATION of N Oxidation of N. Reduction of Cr. Intermolecular redox process. |
|
| 29. |
Which of the following causes temporary hardness of water? |
|
Answer» `MgCI_(2)` |
|
| 30. |
What is the wave length in meters of an FM radio wave with frequency 102.5 MHz ? |
|
Answer» SOLUTION :Given frequency `V=102.5MHz` `=102.5xx10^(6)Hz=102.5xx10^(6)s^(-1)` `"Velocity of light, c "= 3 xx 10^(8) ms^(-1)` `therefore lambda=(c )/(v )=(3XX10^(8)ms^(-1))/(102.5xx10^(6)s^(-1))=2.93m` `"The wave length is 2.93 m "` |
|
| 31. |
When sodium peroxide is treated with dilute sulphuric acid, we get ......... |
|
Answer» sodium sulphate and water `Na_2O_2`+dil. `H_2SO_4 to Na_2SO_4 + H_2O_2` |
|
| 32. |
The types of Carbon atoms present in isobutanc are ·········· . |
|
Answer» only `1^(@)` and `2^(@)` C-atoms |
|
| 33. |
Write on essay about soil pollution especially stating about the sources of pollutioin. |
|
Answer» Solution :Soil Pollution `:` (i) Soil is a thin layer of organic and inorganic material that convers the earth's rocky surface. Soil CONSTITUTES the upper crust of the earth, which supports land, plants and animals. (ii) Soil pollution is defined as the BUILDUP of persistent toxic compounds, radioactive materials, chemical salts and disease causing agents in soils which have harmful effects on plant growth and animal health. (iii) Soil pollution affects the structure and fertility of soil, groundwater quality and food chain in biological ecosystem. Source of soil pollution `:` The major sources of soil pollute the soil are discussed below (i) Artifical fertilizers `:` Soli nutrients are useful for growth of plants. Plants obtains carbon, hydrogen and oxygen from air or water, WHEREAS other essential nutrients like nitrogen, phosphorous, potassium , calsium , magnesium, sulphur are being absorbed from soil. Toremove the deficiency of nutrints in soil, farmers add artificial fertilizers. Increased USE of phosphate fertilizers orexcess use of artificial fertilizers like NPK in soil , results in reduced yield in that soil. (ii) Pesticides `:` Pesticides are the chemicals that are used to kill or stop the growth of unwanted organisms. But these pesticides can affect the health of human beings. These are further classified as (i) Insecticides `:` Insecticides like DDT, BHC, aldrin etc. can stay in soil for long period of time and are absorbed by soil. They contaminate root crops like carrot, raddish, etc. (ii) Fungicide`:` Organo mercury compounds are used as most common fungicide. They dissociate in soil to produce mercury which is highly toxic. (iii) Herbicides `:` Herbicides are the chemical compounds used to control unwanted plants. THey are otherwise known as weed killers. Example `:` Sodium chlorate `( NaClO_(3))` and sodium arsenite `( Na_(3)` As `O_(3)`) .Most of the herbicides are toxic to mammals. (iv) Industrial wastes `:` Industrial activities have been the biggest contributor to the soil pollution especially the mining and manufacturing activities. Large number of toxic wastes are released from industries. Industrial wastes include cyanides, chromates, acids, alkalis,and METALS like mercury , copper , zinc, cadmium and lead, etc . These industrial wastes in the soil surface lie for a long time and make it unsuitable for use. |
|
| 34. |
Which of the following is a chiral molecule |
|
Answer»

|
|
| 35. |
What is meant by work ? Give its unit. |
|
Answer» Solution :(i)Work is DEFINED as the force (F) multiplied by the displacement (x). (II) -w=F. x (III)Minus sign indicates the work done by the system (iv)UNIT of work: The SI unit of work is Joule (J) |
|
| 36. |
When kept open in air, the crystals of washing soda lose 9 molecules off water to form a monohydrate. Na_(2)CO_(3)*10H_(2)O underset("to air")overset("exposed")toNa_(2)CO_(3)*H_(2)O+9H_(2)O this process is called |
|
Answer» efflorescence |
|
| 37. |
Troposphere is spread upto 25 kms from sea level. |
| Answer» Solution :False statement (Troposphere is SPREAD upto 10 KM from sea LEVEL.) | |
| 38. |
Which of the following is an amphoteric hydroxide?(a)Mg(OH)_2(b)Be(OH)_2(c)Sr(OH)_2(d)Ca(OH)_2 |
| Answer» Solution :`Be(OH)_2` | |
| 39. |
Viscosity is depend upon ? Why ? |
|
Answer» Solution :(i) Viscosity of liquid `prop(1)/("FLUIDITY of liquid")` means, viscosity of liquid is more than liquid fluid flow. (II) Viscosity `prop (1)/("Temperature")` means viscosity decreases with temperature. Kinetic enegy of molecules increases with energy of mlecules increases with temperature and can over come the intermolecular forces to slip PAST one another between layers. (III) Viscosity `prop` Intermolecular attraction H-bond and Van der Waal forces. |
|
| 40. |
Which of the following is 'borane compound' ? |
|
Answer» Boron halide |
|
| 41. |
Which one of the following statements is correct ? |
|
Answer» 2S ORBITAL is SPHERICAL with two nodal planes |
|
| 42. |
What would be the value of van't Hoff factor for a dilute solution of K_(2) SO_(4) in water. |
|
Answer» 3 SINCE `K _(2) SO_(4) to 2K^(+) +SO _(4) ^(2-)` `K _(2) SO _(4)` is completely dissociated so `prop = (i-1)/(n -1) = (i-1)/(3-1) =1` `i - 1 =1 xx2` `i -1 =2` `therefore i =2 + 1 =3` |
|
| 43. |
What is the difference between anti-ferromagnetic and ferrimagnetic substances ? What is the cause of this difference ? |
| Answer» Solution :Anti-ferromagnetic substances possess zero net magnetic MOMENT but FERRIMAGNETIC possess small net magnetic moment but not zero . This is because the former contain equal number of electrons with opposite spin but the LATTER have UNEQUAL number. | |
| 44. |
What is the pH of 0.005 M H_2SO_4 solution ? |
|
Answer» SOLUTION :`[H^+]` = 2(0.005)=0.01 THEREFORE pH=2 |
|
| 45. |
What volume of water is to be a added to 100cm^(3) of 0.5 M NaOH solution to make it 0.1 M solution? |
|
Answer» 200 `CM^(3)` `0.5xx100=0.1xxV_(2)V_(2)=500cm^(3)` VOLUME of WATER to be added to `100cm^(3)` of solution `=500-100 =400 cm^(3)` |
|
| 46. |
What is the product of the following reaction? |
|
Answer»
|
|
| 47. |
Which one of the following has inductive, mesomeric and hyperconjugation effect ? |
|
Answer» `CH_(3)CL` |
|
| 48. |
What is the product of the following reaction? |
|
Answer»
|
|
| 49. |
Which does not represent correct method |
|
Answer» `TiCl_2` + Mg `rarr` Ti + MGCL : KROLL |
|














