Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The number ofisomerspossiblefor disubstitutedborazine, B_(3)N_(3)H_(4)X_(2) is |
|
Answer» 3  
|
|
| 2. |
The total number of isomers (containing benzene ring) of molecular formula C_(7)H_(8)O is : |
|
Answer» 2 |
|
| 3. |
The number of isomers of the aromatic compound C_(8)H_(10) are : |
|
Answer» 3 
|
|
| 4. |
The number of isomers of octanc with five carbons in their principal chain is |
|
Answer» |
|
| 5. |
The number of isomers of C_(6)H_(14) is (MLNR) |
|
Answer» 4 |
|
| 6. |
The number of isomers including stereoisomers possible for the compound having molecular formula C_4 H_8 is |
|
Answer» one |
|
| 7. |
The number of isomers of C_(6)H_(14) are…….. |
|
Answer» 4 |
|
| 8. |
The number of isomers for the compound with molecular formula C_2BrClFI is |
|
Answer» 3 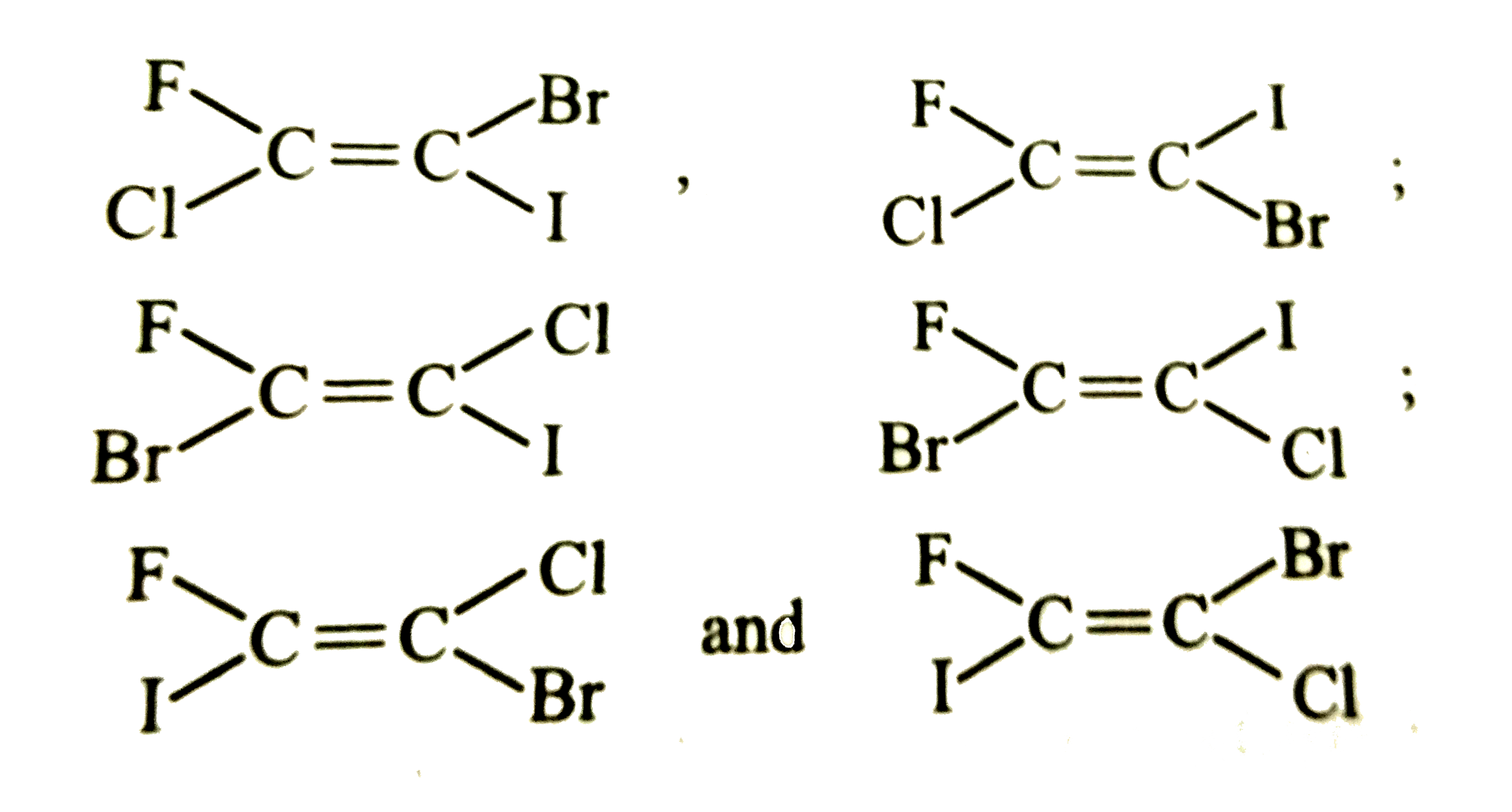
|
|
| 9. |
The number of isomeric primary amines possible for the formula C_(3)H_(9)N |
|
Answer» 2 
|
|
| 10. |
The number of isomeric pentyl alcohols possible are |
|
Answer» Two |
|
| 11. |
The number of isomeric compounds having a benzene ring and molecular formula C_7 H_9 N are_____ |
Answer»  
|
|
| 12. |
The number of isomeric alkyl group possible for C_4H_5- is |
|
Answer» FOUR |
|
| 13. |
The number of ions present in 1 ml of 0.1M CaCl_(2) solution is |
|
Answer» `1.8 XX 10^(20)` |
|
| 14. |
The number of ion pairs that constitute one unit cell of CsCI |
|
Answer» 4 |
|
| 15. |
The number of intensivepropertiesamong the following isTemperature, Pressure, Volume , Heatcapacity ,Density ,pH of a solution , EMF of a cell,Entropy, Free energy,Enthalpy , Surface busion, Viscosity , Boiling point |
|
Answer» |
|
| 16. |
The number of hyperconjugable hydrogen atoms of followingspecies are respectively : |
|
Answer» 3, 5, 9, 8 |
|
| 17. |
The number of hydroxide ions produced by one molecule of Na_(2)CO_(3) on hydrolysis |
|
Answer» 4 |
|
| 18. |
The number of hydrogen bonds that can be formed by each borax acid molecule. |
|
Answer» |
|
| 19. |
The number of hydrogen - boded and coordinated water molecule present in hydrated copper sulphate salt are respectively |
|
Answer» 1, 4 |
|
| 20. |
The number of hydrogen atoms present in 25.6 g of sucrose (C_(12)H_(22)O_(11))which has a molar mass of 342.3 g is (N_(A)=6.023xx10^(23)) |
|
Answer» `22xx10^(23)` 1 MOLE sucrose CONTAINS `=22xx6.023xx10^(23)` of H-ATOM 0.075 moles of sucrose contain `= 0.075xx6.023xx10^(23)xx22` `=9.91xx10^(23)` of H -atom |
|
| 21. |
The number of P-OH bonds present in pyrophosphoric acid and hypophosphoric acid is respectively. |
|
Answer» 120 |
|
| 22. |
The number of hybrid orbitals involved in the formation of B_(2)H_(6), B_(3)N_(3)H_(6), BCl_(3),H_(3)BO_(3) are p, q, r & s, then the sum of (p+q+r+s) is 8y, then y = ? |
|
Answer» `B_(3)N_(3)H_(6)to 18 ["each "B, N-3sp^(2)]B_(3)N_(3)H_(6)rarr 18 ["each "B, N-3sp^(2)]` `BCl_(3)to 3 ["each "B-3sp^(2)]` `H_(3)BO_(3)to 3["each "B-3sp^(2)]` `P+Q+R+S rArr 8+18+3+3=32(8y)` `therefore y=4`. |
|
| 23. |
The number of hexagonal faces that are present in a truncated octahedron is |
Answer» 
|
|
| 24. |
The number of water molecule(s) derectly bonded to the metal centre in CuSO_4.5H_2O is |
|
Answer» `H_3PO_2` |
|
| 25. |
The number of grams of oxygen in 0.10 mol of Na_(2)CO_(3) .10H_(2)O is |
|
Answer» 1 mole of `Na_(2)CO_(3) .10H_(2)O` CONTAINS 13 OXYGEN ATOMS. Mass of 13 oxygen atoms = `13 xx 16 = 208` 1 mole of `Na_(2)CO_(3) .10H_(2)O` contains 208 G of oxygen. `:.` 0.10 mole of `Na_(2)CO_(3).10H_(2)O` contains `208/1xx0.1=20.8g` |
|
| 26. |
The number of grams of H_2SO_4 required to dissolve 5 g of CaCO_(3) is: |
|
Answer» 10.24 |
|
| 27. |
The number of gram - atoms of sulphur in 2 moles of peroxydisulphuric acid is |
|
Answer» 2 |
|
| 28. |
The number of gram atoms of hydrogen present in 1.5 mole of hydrogen sulphide is |
|
Answer» 3 |
|
| 29. |
The number of Glucose molecules present in 10 ml of decimolar solution is |
|
Answer» `6.0 XX 10^(20)` |
|
| 30. |
The number of isomers (geomctrical and optical) possible for the compound with the structure CH_3 CH=CH-CH=CH-CH_2 CHOHCH_3 is |
|
Answer» 2 |
|
| 31. |
The number of geometrical isomers of CH_3CH=CH-CH-CH=CH-Cl |
|
Answer» 2 |
|
| 33. |
The number of esters possible for the molecular formula C_5H_10O_2is ____ |
Answer»  
|
|
| 34. |
The number of equivalents of Na_(2)S_(2)O_(3) required for the volumetric estimation of one equivalent of Cu^(2+) is : |
|
Answer» `1//3` |
|
| 35. |
The number of enantiomers of the compound CH_(3)CHBrCHBrCOOH is |
|
Answer» 0  NUMBER of ENANTIOMERS ` = 2^n` n = number of CHIRAL centres `n = 2 rArr 2^2 = 4` |
|
| 36. |
The number ofelements present in 2nd, 3rd, 4th and 5th IJeriods of modern periodic table respectively are |
|
Answer» 2, 8, 8 & 18 |
|
| 37. |
The number of elliptical orbits excluding circular orbits in the N-shell of an atom is: |
|
Answer» 3 |
|
| 38. |
The number of elements in the first period is only 2. Give reason. |
| Answer» Solution :The first period n=1 ,it is the first energy level and has only 1 s orbital which can accommodate a maximum of 2 electrons HENCE the NUMBER of ELEMENTS in the first period is only 2. | |
| 39. |
The number of elements classified by Newland was ....... |
|
Answer» 72 |
|
| 40. |
The number of electrons in M-shell of an element with atomic number 24 is |
|
Answer» Each subshell has one ORBITAL with m = 0 `therefore ` No. of electrons `=2xx 7 +1 = 15` |
|
| 41. |
The number of electrons to balance the equation NO_(3)^(-)+4H^(+)+e^(-)to2H_(2)O+NO |
|
Answer» 5 |
|
| 42. |
The number of electrons that are involved in the reduction of permanganate to managanes (II) salt managanate and managanese dioxide respecitvely are |
|
Answer» 5,1,3 `overset(+7)MnO_(4)^(-)+e^(-)rarroverset(+6)MnO_(4)^(2-)` `overset(+7)MnO_(4)^(-)+3e^(-)rarroverset(+4)MnO_(2)` Thus option (a) is CORRECT |
|
| 43. |
The number of electrons shared between the two Boron atoms directly in the formation of bonds in diborane molecule |
|
Answer» 4 |
|
| 44. |
The number of electrons, protons and neutrons in a species are equal to 18, 16 and 16 respectively. Assign the proper symbol to the species. |
|
Answer» Solution :ELECTRONS = 18, PROTONS = 16, Neutrons = 16 ATOMIC number = No. of protons = 16 Mass number = No. of prtons + No. of neutrons `= 16 + 16 = 32` As the species has two electrons more than protons, it has two UNITS of -ve charge. Hence, the species is `._(16)^(32)S^(2-)` |
|
| 45. |
The number of electrons, protons and neutrons in a species are equal to 18,16 and 16 respectively. Assign the proper symbol to the species. |
| Answer» SOLUTION :ATOMIC number = 16. HENCE, the element is sulphur (S). Mass number = 16+16 = 32 CHARGE = 16 - 18 = -2 THEREFORE, the symbol is `""_16^33""s^(2-)` | |
| 46. |
Thenumberof electronsprotons and neutronsin aspeciesare equalto 18,16 and16 respectively . Assigh the proper symbolto the species. |
|
Answer» Solution :Numberof electron=18 Atomicnumber= numberof protons( z) = 16 Numberofneutron (n ) in sulphur = 16 A= massnumber= z+ n Numberof electron(18) `lt`numberproton(16) (18-16)=12electronisexcess chargeis -2so SYMBOL of NEGATIVE ION : `s^(2)` Symbolof species`._(16) ^(18) S^(2)` |
|
| 47. |
The number of electrons, protons and neutrons in a species are equal to 18, 16 and 16 respectively .Assign the proper symbol to the species. |
| Answer» Solution :The atomic number is EQUAL to number of protons = 16. The ELEMENT is sulphur (S). Mass number = number of protons + number of neutrons = 16+ 16 = 32 Species is not NEUTRAL as the number of protons is not equal to electrons It is anion, (negatively charged) with charge equal to excess electrons =18-16 = 2. SYMBOL is `""_16^32""s^(-2)` | |
| 48. |
The number of electrons per second which pass through a cross section of a copper wire carrying 10^(-16) A is |
|
Answer» `1.6xx10^(-3)` e/s CHARGE on one electron`=1.6xx10^(-19) C` Rate `=(10^(-16)C//s)/(1.6xx10^(-19))=625 C//s` |
|
| 49. |
The number of electrons lost or gained during the change, Fe + H_(2)O rarr Fe_(3)O_4 + H_2 |
|
Answer» 2 |
|