Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The number of nucleons present in chlorine -37 is |
|
Answer» 17 |
|
| 2. |
The number of nucleons in the isotope of an atom ""_(z)X^(m) are |
|
Answer» m |
|
| 3. |
The number of nucleons in chlorine - 37 is |
|
Answer» 17 |
|
| 5. |
The number of nodes and nodal planes in 4p orbital are respectively |
|
Answer» 2, 1 NODES = n - l - 1 for 4p orbital, n = 4 , l=1 |
|
| 6. |
The number of nodes in radial distribution curve of 4s sublevel is |
|
Answer» 1 |
|
| 7. |
The number of nodal planes in p_(x)-obital is: |
|
Answer» one |
|
| 8. |
The number of nodal planes for P_(x) orbital is |
|
Answer» 1 |
|
| 9. |
The number of nodal planes .d. orbital has |
|
Answer» Zero |
|
| 10. |
The number of nodal planes '5d' orbital has, is |
|
Answer» zero |
|
| 11. |
The number of nitrogen molecules present in lc.c of gas at NTP is |
|
Answer» `2.67 xx 10^(22)` |
|
| 12. |
The number of neutrons in the dipositive zinc ion (Mass no. of Zn = 65) |
|
Answer» 35 |
|
| 13. |
The number of neutron in 540 gm water |
|
Answer» `240xx N_(A)` |
|
| 14. |
The number of neutorn(s) present in deuterium is |
|
Answer» 0 |
|
| 15. |
The number of moles of sulphate ions present in the general formula of 1 mole of alum ? |
|
Answer» |
|
| 16. |
The number of moles of oxygen required to prepare 1 mole of water is |
|
Answer» 1 MOLE 0.5 mole of OXYGEN is required to PREPARE 1 mole of `H_(2)O` |
|
| 17. |
The number of moles of oxygen in 1L of air which contains 21% oxygen by volume under standard conditions is : |
|
Answer» 0.186 MOL `0.21 L` of `O_(2) = (0.21)/(22.4)= 0.0093` mol |
|
| 18. |
The number of moles of MnO_(4)^(-) and Cr_(2)O_(7)^(-2) separately required to oxidise 1 mole of FeC_(2)O_(4) each in acidic medium respectively |
|
Answer» 0.5 , 0.6 |
|
| 19. |
The number of moles of lead nitrate needed to coagulate 2 mol of colloidal [AgI]I^(-) is |
|
Answer» 2 THUS 2 moles of `[AgI]I^(-)` are coagulated by 1 mole of `Pb^(+)` i.e., 1 mole of Pb `(NO_(3))_(2)` |
|
| 20. |
The number of moles of KNnO_(4) that will be needed to react with one mole of sulphite ion in acidic solution is : |
|
Answer» Solution :The ionic equation for the reaction is : `2MnO_(4)^(-)+6H^(+)+5SO_(3)^(2-)rarr2Mn^(2+)+5SO_(4)^(2-)+3H_(2)O` 5 mole of `SO_(3)^(2-)` IONS are oxidised by `MnO_(4)^(-)` ions = 2 MOL 1 mole of `SO_(3)^(2-)` ions is oxidised by `MnO_(4)^(-)` ions = 2/5 mol. |
|
| 21. |
The number of moles of KMnO_(4) that will need to react completely with one mole of ferrous oxalate in acidic solution is : |
|
Answer» `2//5` |
|
| 22. |
The number of moles of KMnO_4 that will be needed to react completely with one mole of ferrous oxalate in acidic solution is |
|
Answer» Solution :`MnO_(4)^(-)+SO_(3)^(-2) RARR Mn^(+2)+SO_(4)^(2-)` change in oxidation of Mn = 5 change in oxidation of S= 2 `2MnO_(4)^(-)+5SO_(3)rarr2Mn^(+2)+5SO_(4)^(2-)` `:.`1 mol of `SO_(3)^(-2) `require `2/5` MOLES `KMnO_4` |
|
| 23. |
The number of moles of KMnO_(4) that will be needed to react with one mole of sulphite ion in acidic solution is : |
|
Answer» `(4)/(5)` `2MnO_(4)^(-)+5SO_(3)^(2-)+6H^(+) to 2Mn^(2+) + 5SO_(4)^(2-)+3H_(2)O` 1 mole `SO_(3)^(2-)` will be oxidised by `(2)/(5)` mol of `MnO_(4)^(-)` |
|
| 24. |
The number of moles of KMnO_(4) needed to react with one mole of SO_(3)^(2-) in acidic solution is |
|
Answer» `4//5` `MnO_(4)^(-)+8H^(+)+5e^(-)rarrMn^(2+)+4H_(2)O` OXDIATION half reaction `SO_(3)^(2-=)+H_(2)OrarrSO_(4)^(2-)+2H^(+)+2e^(-)` sinc 1 mole of `SO_(3)^(2-)` gives `2e^(-)` butreduction of 1 mole of `KmnO_(4)` k requires `5 e^(-)` therefore no of MLES of `KMnO_(4)` krequired to react with 1 mole of `SO_(3)^(2-)=2//5` |
|
| 25. |
The number of moles of KMnO_(4) reduced by one mole of KI in neutral medium is ("Hint "KI to IO_(3)^(-)) |
|
Answer» ONE |
|
| 26. |
The number of moles of KI required to produce 0.4 mole K_(2)HgI_(4) is |
|
Answer» `1.6` |
|
| 27. |
Number of moles of KI (potassium iodide) required to produce 0.1 mole of K_2Hg I_(4) is |
|
Answer» 1.6 |
|
| 28. |
The number of moles of H_(2)O_(2) needed to reduce 2 mole of KMnO_(4) in acidic medium is. |
|
Answer» 2 |
|
| 29. |
The number of moles of H_(2) in 2.24 litre of hydrogen gas at STP |
|
Answer» 0.1 MOLE 2.24 litre =`1/22.4 XX 2.24` = 0.1 mole |
|
| 30. |
The number of moles of H_(2) in 0.224 litre of hydrogen gas at STP is |
|
Answer» `:.` 0.224 litre of hydrogen gas at STP will contain `1/22.4xx0.224 = 0.01` |
|
| 31. |
The number of moles of ethanol chloride will produce 10.2 g of ethanoic anhydride on reaction with sodium acetate |
|
Answer» 0.51 mol 10.2 g of ethanoic anhydride will be OBTAINED from ACETYL chloride `= (1)/(102) xx 10.2 = 0.10 mol` |
|
| 32. |
The number of moles of Fe_(2)O_(3) formed when 5.6 lit of O_(2) reacts with 5.6g of Fe? |
|
Answer» 0.125 |
|
| 33. |
The number of moles of electrons involved in the manufacture of 1 mole of H_2O_2from 50% H_2SO_4 |
|
Answer» 2 |
|
| 34. |
The number of moles of CO_(2) produced when 3 moles of HCl react with excess of CaCO_(3) is |
|
Answer» 1 |
|
| 35. |
The number of moles of an acid or base added to one litre of the buffer solution so as to change its pH by one unit is called............of the buffer. |
|
Answer» |
|
| 36. |
The number of molesof an ideal gas that should be taken in a closed vessel of30Lcapacity at a temperature of 27^(@)C so that the pressure exertedby the gas on the walls of the container is 4.1 atmosphere is |
|
Answer» |
|
| 37. |
The number of moles of acidified KMnO_(4) required to oxidise one mole of ferrous oxalate (FeC_(2)O_(4)) is : |
|
Answer» 5 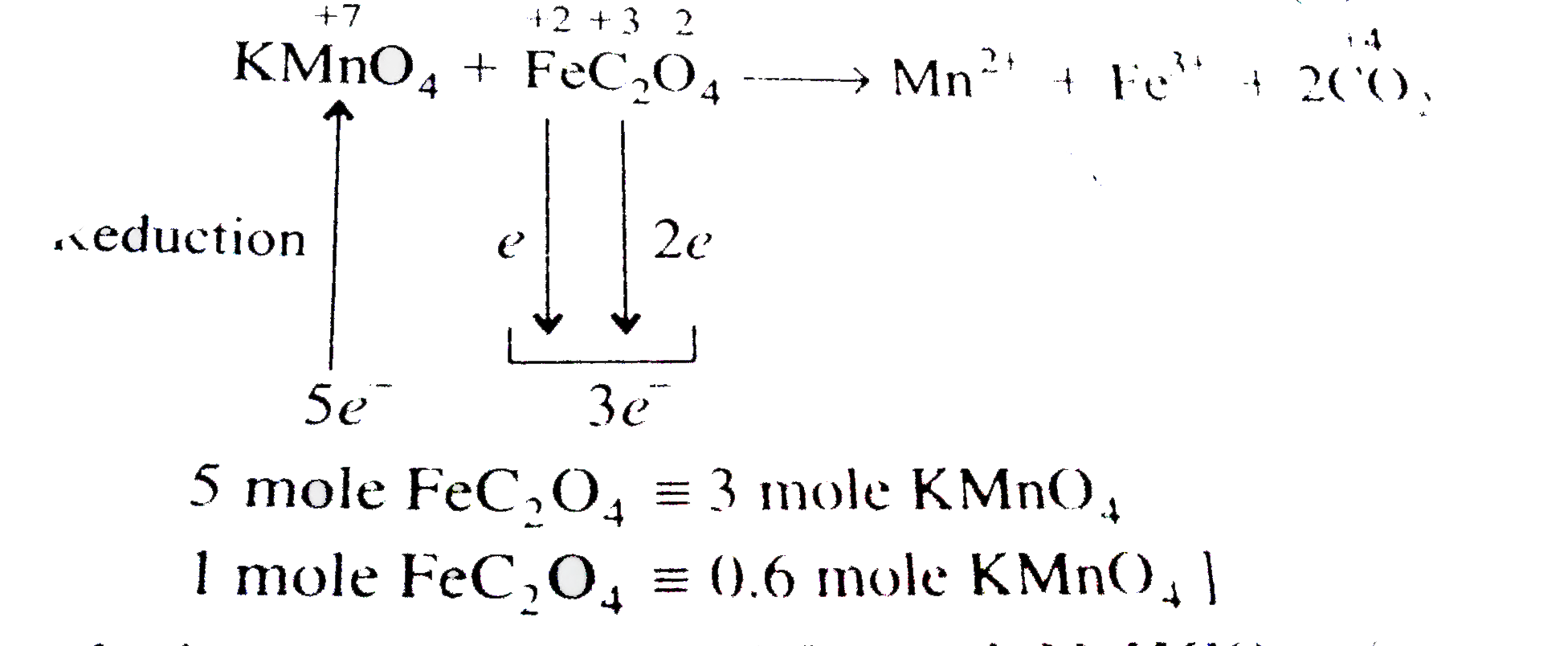 `therefore 5 " MOLE" FeC_(2)O_(4)-=3 " mole" KMnO_(4)` 1 mole `FeC_(2)O_(4)-= 0.6 " mole" KMnO_(4)` |
|
| 38. |
The number of moles in 0.44 g of CO_2 is ( C = 12, O = 16) |
|
Answer» 100 |
|
| 39. |
The number of molecules of CO_(2) liberated by complete combustion of 0.1 g of graphite in air is |
|
Answer» `3.01xx10^(22)` |
|
| 40. |
The number of molecules in one litre of water is (density of water = 1g/mL) |
|
Answer» `6XX10^(23)//22.4` |
|
| 41. |
The number of molecules in a drop of water (0.0018 ml) at room temperature is |
|
Answer» Number of molecules in 0.0018 g = `(6.023xx10^(23))/18 xx 0.0018 = 6.023xx10^(23)xx10^(-5)` `= 6.023xx10^(19)` molecules. [OR] DENSITY of water at `25^@` C=997.0479 g/L Mass of 0.0018 ml (or) `0.0018xx10^(-3)L` `=DXXV` `=997.05xx0.0018xx10^(-3)` `=1.795xx10^(-3)g` Molar mass of water = 18 g Mole=`(1.795xx10^(-3))/18=9.971xx10^(-5)` mole `:.`Number of molecules in 0.0018 ml= moles `xx` Avogadro number `9.971xx10^(-5)xx6.023xx10^(23)` `6xx10^(19)` molecules |
|
| 42. |
The number of molecules in 40 g of sodium hydroxide is …….. |
|
Answer» 40 g= 1 mole = `6.023 xx 10^(23)` |
|
| 43. |
The number of molecules in 16g of methane is : |
|
Answer» `3.0 xx 10^(23)` |
|
| 44. |
The number of molecules in 16g of methane is ............. |
|
Answer» Molecular MASS = 12 + 4=16 16 g of methane CONTAINS Avogadro number of molecules = `6.023 xx 10^(23)` molecules. |
|
| 45. |
The number of molecules in 11 g of carbon dioxide approximately (C = 12, O = 16) |
|
Answer» `0.5 xx10^(23)` `=(6xx10^(23)xx11)/(44) = 1.5 xx10^(23)` |
|
| 46. |
The number of molecule in 4.4 gmCO_(2) .......... |
|
Answer» `6.022xx10^(21)` mole of `CO_(2) = (4.4)/(44) = 0.1` MOLECULS `= 0.1xx6.022xx10^(23)` `=6.022xx10^(22)` |
|
| 47. |
The number of mole of oxalate ions oxidised by one mole of MnO_4^(-) is |
|
Answer» Solution :`overset(+7)(MnO_4^(-))overset((5))rarrMn^(2+),overset(+3)(C_2)O_4^(2-)overset((2))rarr2overset(+4)(CO_2)` `1/5` moles `MnO_(4)^(-)-=1/2` moles `C_(2)O_(4)^(2-)` 1 mol ........ ? `=5/2` |
|
| 48. |
The number of moes of KNnO_(4) reduced by one mole of KI in alkaline medium is |
|
Answer» one `2KMnO_(4)+H_(2)O+H_(2)O+KIrarr2MnO_(2)+2KOH+KIO` `therefore 2 KMnO_(4)=KI` Thus2 MOLES of `KMnO_(4)`are reduced by 1 mole of KI i.e option (b) is CORRECT |
|
| 49. |
The number of millimoles of H_2SO_4 present in 5 litres of 0.2N H_2SO_4 solution is |
| Answer» ANSWER :A | |
| 50. |
The number of lone paris of electrons one the central atoms ofH_(2)O, SnCl_(2), PCl_(3) and XeF_(2) respectively are |
|
Answer» Solution :O is in Group16 . Last shell has 6 electrons . Two are shared with H-atoms  . No of lone . No of lonepairs on o = 2 Sn is in Group 14 . Last shell has 4 electrons . Two areshared 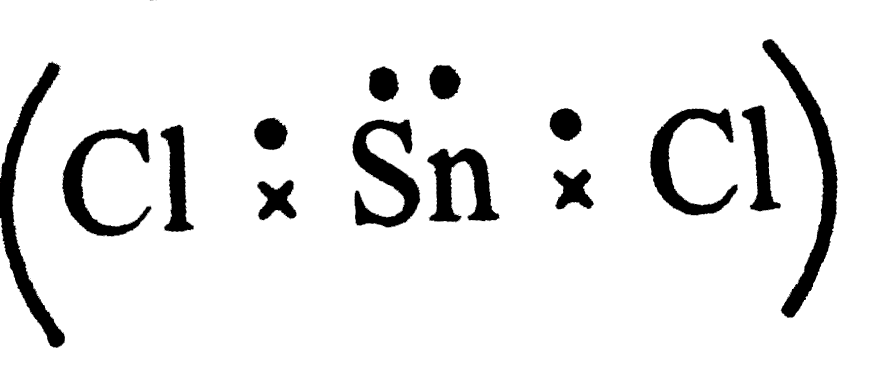 . No. of lone pairs on Sn = 1 . . No. of lone pairs on Sn = 1 .P is in Group 15 . shell has 5 electrons . Three are shared with Cl atoms 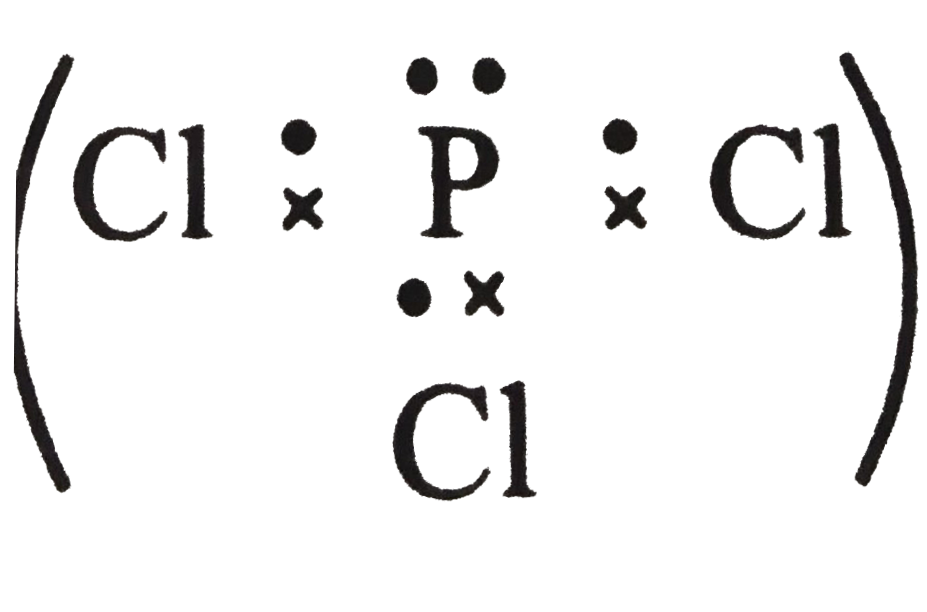 . No of . No oflone pair on P = 1 Xeis in Group 18 . Lastshell has 8 electrons. Two are shared with two F atoms . 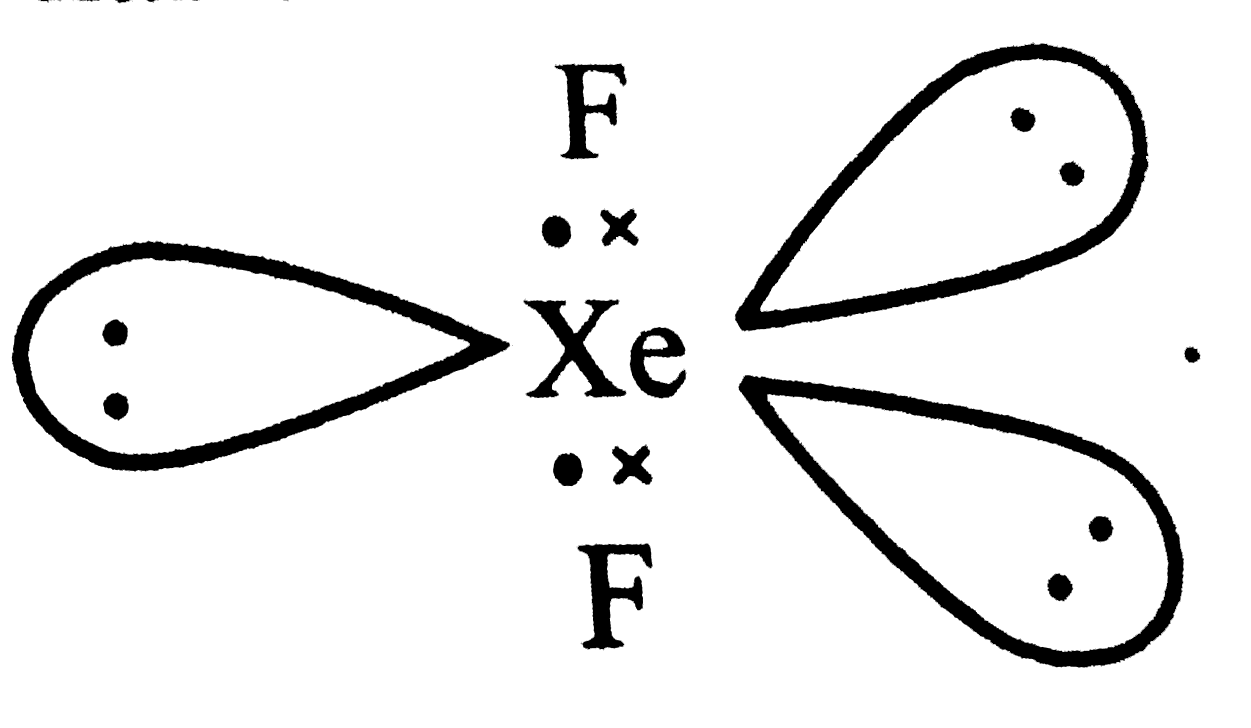 No . of lone pairs on Xe = 3 |
|