Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The volume of a certain amount of a gas at 25^@Cand at 100 cm pressure is 80 mL. The gas is expanded to 140.5 mL keeping temperature constant. Calculate the pressure of the expanded gas. |
|
Answer» Solution :In the present CASE, `P_1 = 100 cm Hg, ""P_2` = ? `V_1 = 80 mL, "" V_2 = 140.5 mL` According to Boyle.s law, at constant TEMPERATURE `P_1 V_1 = P_2 V_2` or `P_2 = (P_1 V_1)/(V_2) = (100xx80)/(140.5)=56.94 cm` Hg Hence, the pressure of the expanded gas will be 56.94 cm Hg. |
|
| 2. |
Which one is non-biodegradable substance ? |
|
Answer» Paper |
|
| 3. |
X and Y represent |
|
Answer» `H_(2)//Pt, KMnO_(4)` |
|
| 4. |
Write the name and deduce the atomic number of the following element(i) The second alkali metal(ii) The fourth noble gas(iii) The third halogen(iv)The first transilation element |
|
Answer» Solution :(i)SODIUM (Z=11) (II) KRYPTON (Z=36) (iii) BORMINE (Z=35) (iv)SCANDIUM (Z=21) |
|
| 5. |
Which gives blue bead in borax bead test ? |
|
Answer» `CR^(3+)` |
|
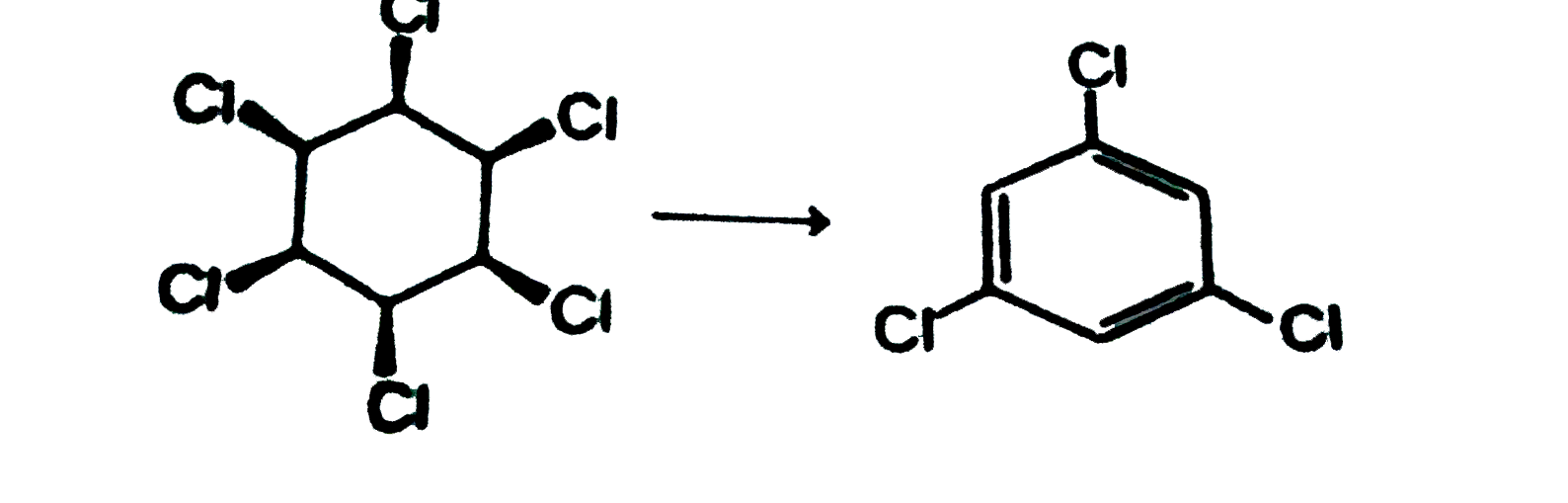
| 6. |
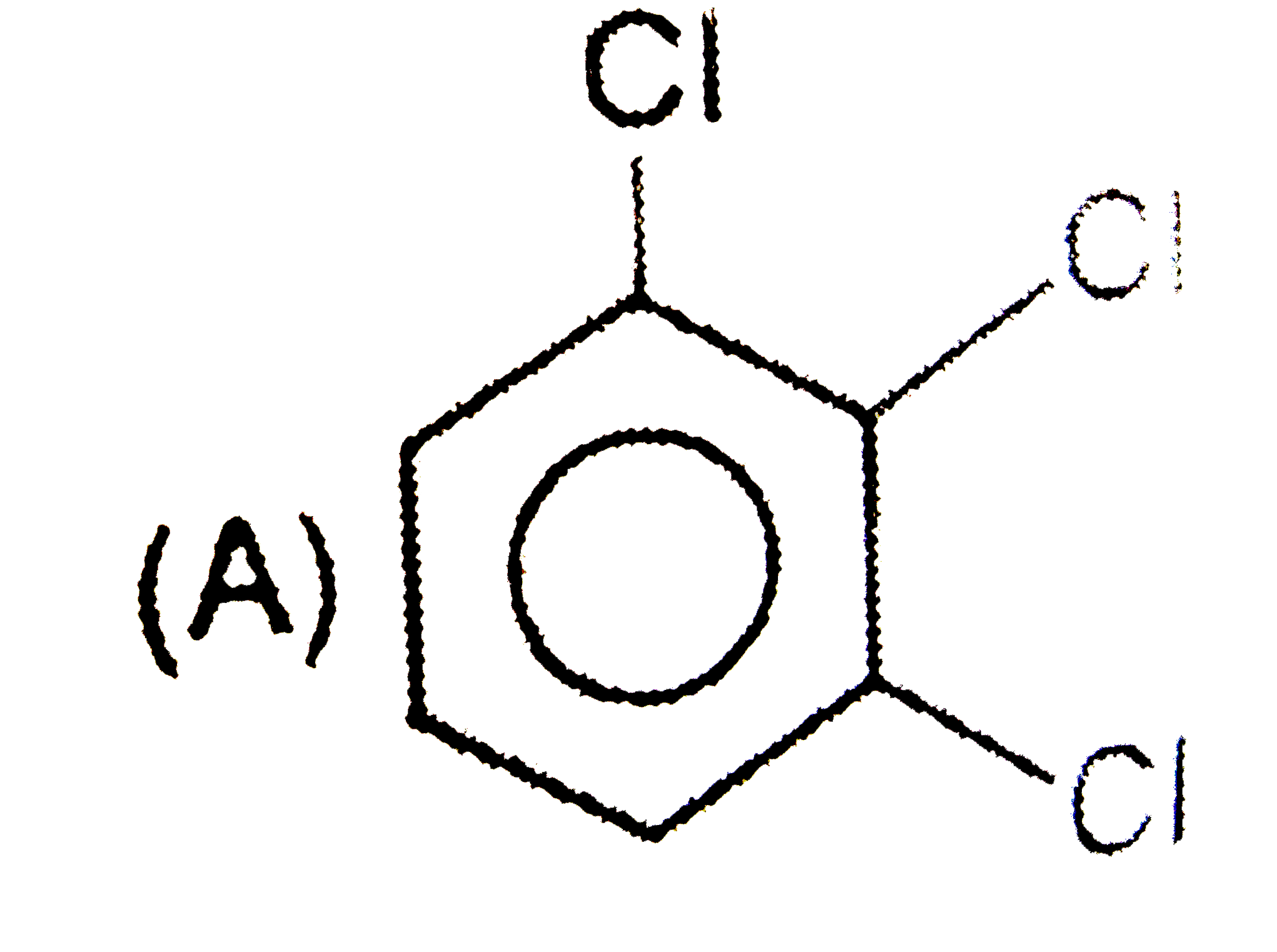
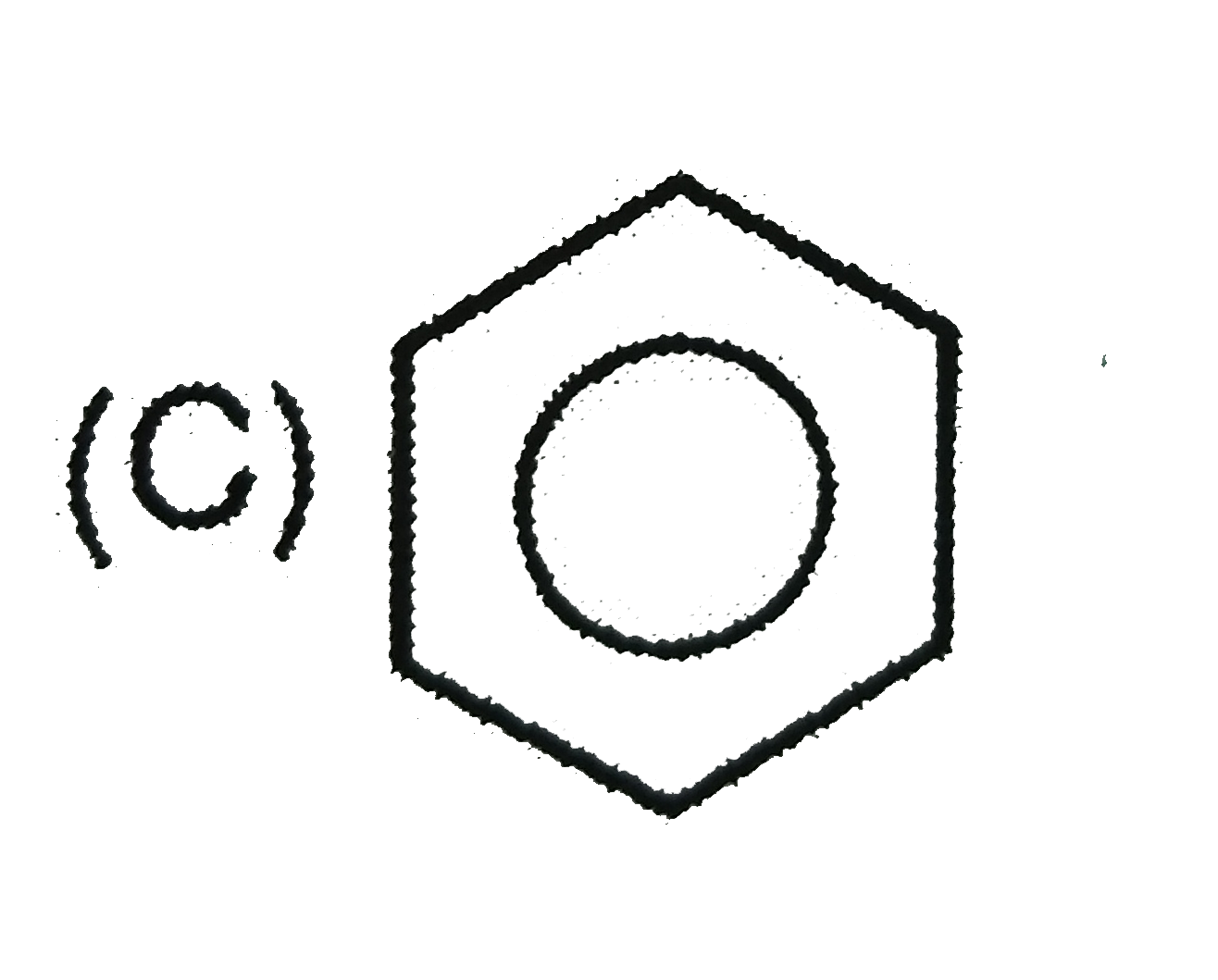
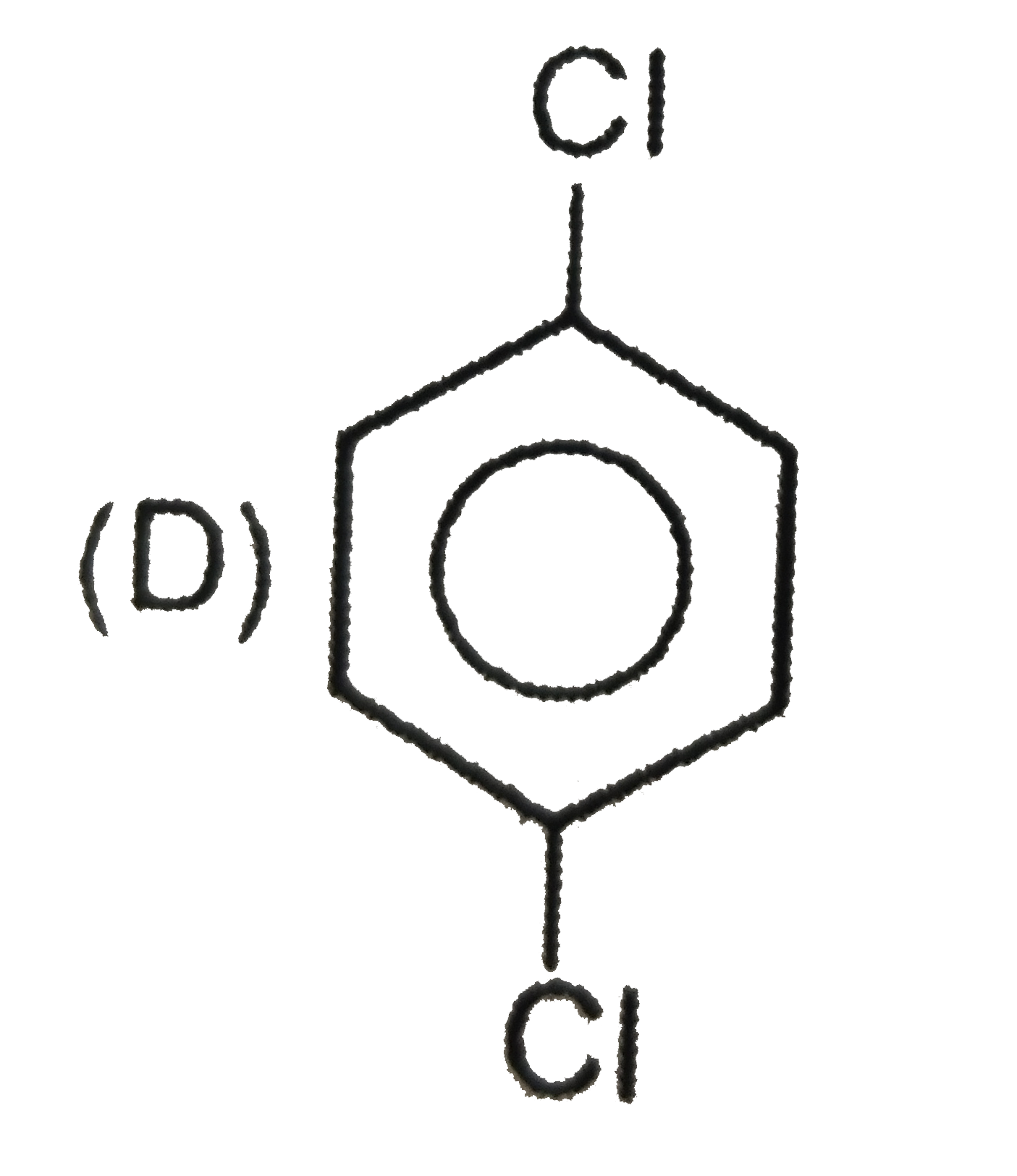
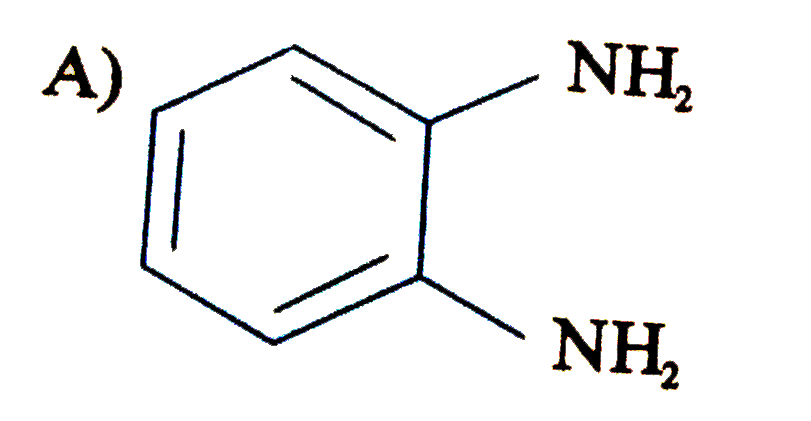
When the all-cis isomer of C_6H_6Cl_6 (1,2,3,4,5,6 -Hexachlorohexane) is heated with alc. KOH, the most probable product is : |
|
Answer»
|
|
| 7. |
What will be the value of K and Delta G^(@) for the process of transformation of ice into water at room temperature ? |
|
Answer» `K=1, Delta G^(@)` Zero `therefore` According to second law of thermodynamics value of `Delta G^(@)` will be negative. Moreover the amount of product is HIGHER by forward reaction. So `K= ("[product]")/( "[reactant]") gt 1` |
|
| 8. |
The time for complete evaporation depends on which factors ? Explain. |
|
Answer» Solution :The LIQUID eventually disappears and the time taken for complete evaporation depends on three factor. The nature of the liquid : The rate of complete vaporisation of liquid LIKE ethanol, ether, water, petrol, acetone at constant temperature is different. Equal AMOUNT of ether and alcohol REQUIRE less time for complete vaporisation compare to equal amount of water. Amount of liquid : At definite temperature the time for complete vaporisation of liquid depends on the amount of liquid. For example, 10 mL water required more time for complete vaporisation than 5 mL water. Temperature : As a temperature increase, rate of reaction increases. So, at 1 atmosphere definite amount of water require less time for complete vaporisation at high temperature. |
|
| 9. |
Water is an amphoteric oxide. Justify this statement. |
|
Answer» Solution :WATER is an amphoteric oxide. It has the ability to accept as well as donate PROTONS and hence it can act , as an ACID or a base. For example, in the reaction with HCL it accepts proton where as in the reaction with WEAK base ammonia it donates proton. `NH_3+H_2OtoNH_4^++OH^-` `HCl+H_2OtoH_3O^++Cl^-` |
|
| 10. |
Which ion is present in bones and teeth as apatite ? |
|
Answer» `MG^(+2)` |
|
| 11. |
Treatment of compound O with KMnO_(4)//H^(+) have P, which on heating with ammonia gave Q. The compound Q on treatment with Br_(2)//NaOH produced R. On strong heating, Q gave S, which on further treatment with ethyl 2-bromopropanoate in the presence of KOH followed by acidification, gave a compound T. Q. The compound R is |
|
Answer»
|
|
| 12. |
Treatment of compound O with KMnO_(4)//H^(+) have P, which on heating with ammonia gave Q. The compound Q on treatment with Br_(2)//NaOH produced R. On strong heating, Q gave S, which on further treatment with ethyl 2-bromopropanoate in the presence of KOH followed by acidification, gave a compound T. Q. The compound T is |
|
Answer» glycine 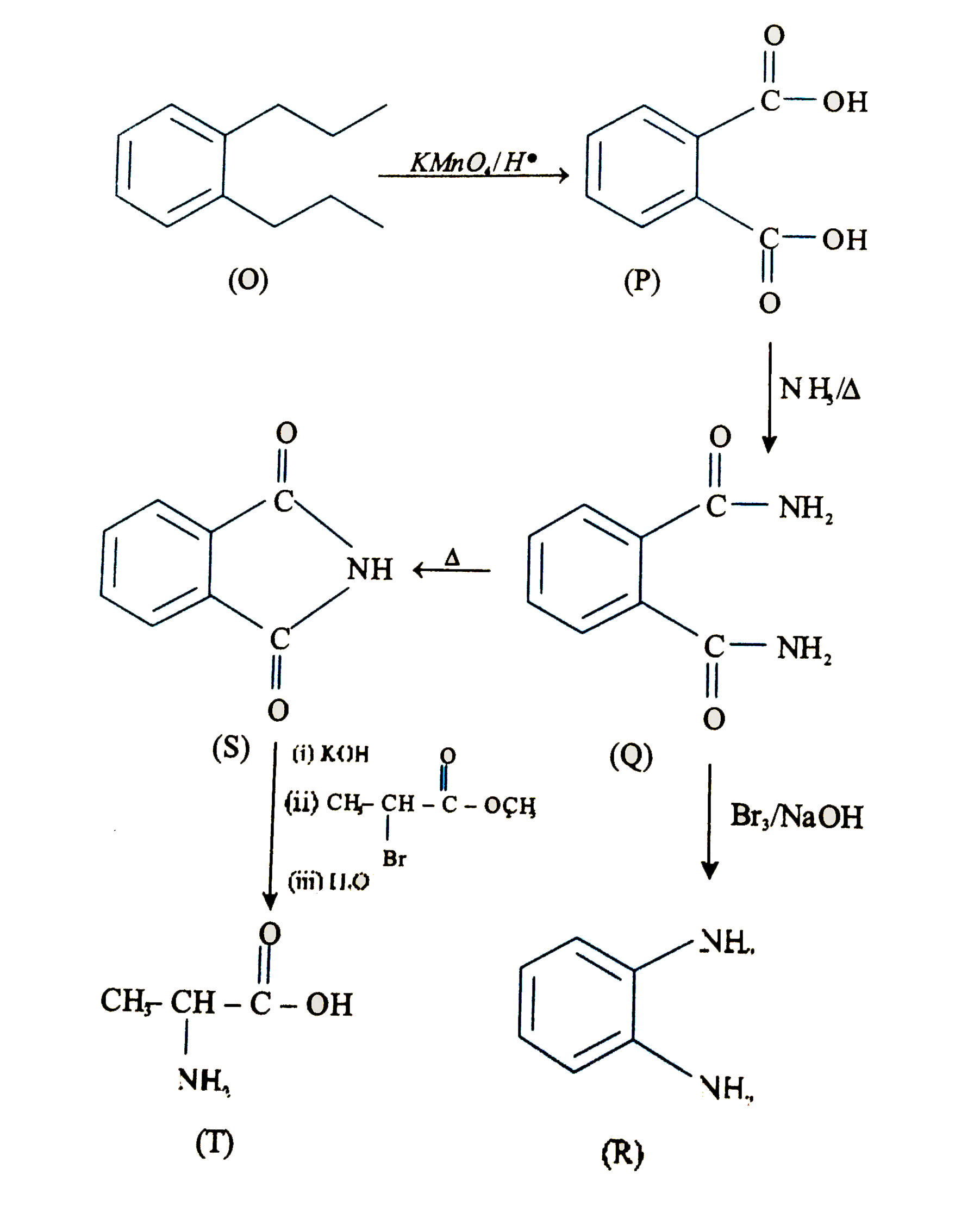
|
|
| 13. |
Write ionic equilibrium reactions of aqueous solution of weak acid HNO_2 and HClO. |
|
Answer» Solution :`HNO_(2(aq)) + H_2O_((L)) HARR H_3O_((aq))^(+) + NO_(2(aq))^(-)` `HClO_((aq))+ H_2O_((l)) hArr H_3O_((aq))^(+) + ClO_((aq))^(-)` |
|
| 14. |
Which ion has the stable electronic configuration? Ni^(2+) or Fe^(3+). |
|
Answer» SOLUTION :`Ni(Z=28) . 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^8` `Ni^(2+)` electronic configuration `=1s^2 2s^2 2p^6 3s^2 3p^6 3d^8` `Fe (Z=26) . 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^6` `Fe^(3+) 1s^2 2s^2 2p^6 3s^2 3p^6 3d^5` If d orbital is half FILLED, according to Aufbau principle , it is more stable . So `Fe^(3+)` is more stable than `Ni^(2+)`. |
|
| 15. |
Write the significance of plus and minus signs in representing the orbitals |
|
Answer» Solution :PLUS and MINUS signs have been GIVEN to identify the nature of the electron waves. Plus sign DENOTES crest while minus sign denotes trough. |
|
| 16. |
Which of the following reaction shows the oxidising nature of H_2SO_4 ? |
|
Answer» `CA(OH)_2 + H_2SO_4 to CaSO_4 + 2H_2O` |
|
| 17. |
What isthe chargemassitsratioelectron ? |
|
Answer» SOLUTION :CHARGE(E ) = 1.6022`xx 10^(19) C` mass `m_(e )= 9 .10939 xx 10^(31) KG` the ratioof chargeand mass `=(e )/(m_(e ))= 1.758820 xx 10^(11)C kg ^(-1)` |
|
| 18. |
What weight of O_(2) is obtained when 0.245gm of KCIO_(3) decomposes completely to KCl and O_(2): |
|
Answer» |
|
| 19. |
What happenes whencalcium nitrate is heated? |
| Answer» SOLUTION :`2CA(NO_3)_2 RARR 2CaO+4NO_2+O_2` | |
| 20. |
What is gem-dihalide? Give one example and explain its preparation. |
|
Answer» Solution :(i) If two halogen atoms are attached to one carbon atom of alkyl HALIDE . It is named as gemdialide e.g. , `CH_(3) - CH Cl_2` (ethylidene dichloride ) (II) Preparation of `CH_(3) - CHCl_(2)` `underset("Acetaldehyde") (CH_(3) CHO) + PCl_(5) to CH_(2) = underset(Cl) underset(|)(C)HCl + POCl_(3)` |
|
| 21. |
Which hybridisation of carbon is same in the following? Give type of hybridisation (a) CH_(2) = C = CH_(2) (b) CH_(2) = CH - CH = CH - CH = CH_(2) (c ) HC -= C - C -= C - C -= CH (d) CH_(2) = CH - CH = CH_(2) (e ) CH_(3)C -= C- CH_(3) (f) C_(6) H_(6) (g) C_(6)H_(12) (cyclic comp) (h) CH_(3) - CH = CH - CH= CH_(3) |
| Answer» SOLUTION :(B) all C in `sp^(2)` hybridisation (c ) all C in sp hybridisation (d) all C in `sp^(2)` hybridisation (f) all C in `sp^(2)` hybridisation (g) all C in `sp^(2)` hybridisation | |
| 22. |
Which of the following reactions is/are represented correctly? |
|
Answer» `{:(Pb(OAc)_(2)+dil.H_(2)SO_(4)rarrunderset(("white ppt"))('A'darr)),(""darr),("""Soluble in conc." H_(2)SO_(4)):}` |
|
| 23. |
When the temperature is raised, viscosity o the liquid decreases. This is because |
|
Answer» volume of the SOLUTION decreases |
|
| 24. |
Which of the following gas is highly water soluble |
| Answer» ANSWER :A | |
| 25. |
Which of the following calcium halide has more melting point. |
|
Answer» `CaF_(2)` `CaF_(2) gt CaCl_(2) gt CaBr_(2) gt CaI_(2)` As the size of the anion increases, the COVALENT character increases hence the DECREASE melting point. |
|
| 26. |
Which element is most electropositive and most electronegative respectively ? |
|
Answer» K, Cl  Na, K, Cs are ALKALI metals of group ONE. Among the three Cs has least electronegativity and is most electropositive. Cl and F are halogen element of group seven. Among the two F is more ELECTRONEGATIVE and F is most electronegative, among all. |
|
| 27. |
Thermal decomposition of gaseous X_(2) to gaseousX at 298 K takes place according to the equation : X_(2) (g) hArr 2X (g) The standard reaction Gibbs energy , Delta_(r)G^(@) of this reaction is positive . At the start of the reaction, there is positive . At the start of the reaction , there is one mole of X_(2) andno. As the reaction proceeds, the number of moles of X formed is given by beta." Thus " beta _(equilbrium )is the number ofmoles of X formed at equilibrium . The reaction is carried out at a constant total pressure of 2 bar . Consider the gases to behave ideally . ( Given : R = 0* 0833 " L bar " K^(-1)mol^(-1)). The INCORRECT statement among the following for this reaction is |
|
Answer» Decrease in the total pressure will result in formation of more moles of gaseous X (b) At the start of the reaction , `Q LT V` Hence, reaction will proceed in the forward direction . So (b) is also correct. (c) If `beta_(eq.) = 0*7 , " then " K = (8 xx (0*7)^(2))/(4 - (0*7)^(2) ) gt 1 ` `Delta G^(@) =-RT "In" K_(p) :.Delta G^(@) "will be" - "ve but we are given that " Delta G^(@)" is " +" ve which can be so if " K_(p) lt 1. " Hence " beta_(eq.) !=0*7 , " i.e, (c) is incorrect . "` (d) `K_(p) = K_(c) (RT)^(Deltan_(g))= K_(c) (RT) or K_(c) = K_(p)/(RT)`. Thus, `K_(c) lt K_(p)` As `K_(p) lt 1, "therefore " K_(c) lt 1. ` Hence, (d) is also correct. |
|
| 28. |
Thermal decomposition of gaseous X_(2) to gaseousX at 298 K takes place according to the equation : X_(2) (g) hArr 2X (g) The standard reaction Gibbs energy , Delta_(r)G^(@) of this reaction is positive . At the start of the reaction, there is positive . At the start of the reaction , there is one mole of X_(2) andno. As the reaction proceeds, the number of moles of X formed is given by beta." Thus " beta _(equilbrium )is the number ofmoles of X formed at equilibrium . The reaction is carried out at a constant total pressure of 2 bar . Consider the gases to behave ideally . ( Given : R = 0* 0833 " L bar " K^(-1)mol^(-1)). Theequilibrium constantK_(p) for this reaction at 298 K, in terms ofbeta _(equilibrium ) , is |
|
Answer» `(8 beta_("equilibrium ")^(2))/(2 - beta _("equilibrium")` As the number of moles of X formed at equilibrium `= beta _(eq).` ` :. " No. of moles of " X_(2)" reacted at equilibrium " = (beta_(eq))/2 ` `{:(,X_(2)(g) ,hArr,2X(g)),(" INTIAL moles" ,1,,0),("Moles at eqm",1-beta_(eq)/2,,beta_(eq)/2):}` Total no. of moles at equilibrium `= 1- (beta_(eq.))/2 + beta_(eq.) =1+ beta_(eq.)/2` `:. p_(X_(2)) = (1-beta_(eq.)/2)/(1+beta_(eq)/2)xx2, p_(X) = beta_(eq.)/(1+(beta_eq)/2)xx2, p_(X) = beta_(eq.)/(1+(beta_(eq))/2)xx2` (as `P_(total ) = 2 "BAR")` ` :. K_(P) = (p_(X)^(2))/p_(X)^(2) = ((2beta_(eq.))/(1+(beta_(eq.))/2))^(2) xx ((1+(beta_(eq.))/2))/(2(1-(beta_(eq.))/2))` `=(4beta_(eq.)^(2))/(1+beta_(eq.)/2)xx1/(2(1-beta_(eq)/2)) ` ` (4beta_(eq).)/(2(1-beta_(eq)^(2)/4 ))= (2beta_(eq)^(2))/((4-beta _(eq)^(2))/4)=(8beta_(eq.)^(2))/(4-beta_(eq.)^(2))` |
|
| 30. |
Write the resonánce structure for SO_(3), NO_(2) and NO_(3)^(-) |
Answer» SOLUTION :
|
|
| 31. |
Upon heating KClO_(3) in the presence of catalytic amount of MnO_(2), a gas W is formed. Excess amount of W reacts with white phosphorus to give X. The reaction of X with pure HNO_(3) gives Y and Z. Y and Z are, respectively |
|
Answer» `N_(2)O_(5)andHPO_(3)` `P_(4)+10O_(2)overset("Oxidation")tounderset((X))(P_(4)O_(10))` `P_(4)O_(10)` acts as dehydrating agent and removes `H_(2)O` from `HNO_(3)` to GIVE `N_(2)O_(5)` (Y) and itself gets converted into `HPO_(3)` (Z). Thus, X is `P_(4)O_(10)` Y is `N_(2)O_(5)`, and Z in `HPO_(3)` and W is `O_(2)`. `P_(4)O_(10)+4HNO_(3)tounderset((Y))(2N_(2)O_(5))+underset((Z))(4HPO_(3))` |
|
| 32. |
Upon heating KClO_(3) in the presence of catalytic amount of MnO_(2), a gas W is formed. Excess amount of W reacts with white phosphorus to give X. The reaction of X with pure HNO_(3) gives Y and Z. W and X are, respectively |
|
Answer» `O_(2)andP_(4)O_(6)` `P_(4)+10O_(2)overset("Oxidation")tounderset((X))(P_(4)O_(10))` `P_(4)O_(10)` ACTS as dehydrating agent and removes `H_(2)O` from `HNO_(3)` to give `N_(2)O_(5)` (Y) and itself gets converted into `HPO_(3)` (Z). Thus, X is `P_(4)O_(10)` Y is `N_(2)O_(5)`, and Z in `HPO_(3)` and W is `O_(2)`. `P_(4)O_(10)+4HNO_(3)tounderset((Y))(2N_(2)O_(5))+underset((Z))(4HPO_(3))` |
|
| 33. |
Which of the following is not an actinoid ? |
|
Answer» Curium (Z = 96) |
|
| 34. |
Which method is employed for the estimation of carbon and hydrogen organic compounds? |
|
Answer» |
|
| 35. |
Which of the following statements is not true ? |
|
Answer» Paramagnetic substances are weakly attacted by MAGNETIC field. |
|
| 36. |
What is the type of hybridisation of each carbon in CH_(3) - CH = CHCN |
Answer» SOLUTION :
|
|
| 37. |
What is second ionisation enthalpy? |
|
Answer» Solution :Energy required to lose 2 electron is known as second IONISATION enthalpy. The reaction is shown as second Ionisation enthalpy. `X_((g))^(+) to X_((g))^(2+) + e^(-) ....Delta_(i) H(2)` |
|
| 38. |
Wg of benzoic acid is completely burnt in bomb calorimeter in excess of O_2 so that KJ heat is released at 298K. Internal energy change for combustion of 1 mole benzoic acid (MW = M) will be (in KJ) |
|
Answer» `-Q` |
|
| 40. |
Which effect occurs on pressure if gas do not collide elastically ? |
| Answer» SOLUTION :If COLLIDE do not elastically than pressure DECREASES and becomes zero. | |
| 41. |
Two sparingly soluble salts AB and XY_(2) have the same solubility product. Which salt will be more soluble ? Explain . |
|
Answer» Solution :SUPPOSE SOLUBILITY of `AB = a "MOL " L^(-1) `. Then `AB hArr A^(+) + B^(-) , K_(sp)= [A^(+)][B^(-)]=axxa=a^(2)` `:.a=sqrt(K_(sp))` Suppose solubility of salt `XY_(2) = b "mol" L^(-1)`. Then `XY_(2) hArr X^(2)+2, Y^(-), K_(sp)=[X^(2+)][Y^(-)]^(2)=b(2b)^(2) i.e., 4b^(3)=K_(sp) or b= (K_(sp)//4)^(1//3)` |
|
| 42. |
What should be the pH at the equivalence point for the titration of 0.10M KH_(2)BO_(3) with 0.01M HCI ? (K_(a) of H_(3)BO_(3)= 7.2xx10^(-10)) |
|
Answer» |
|
| 43. |
When O_(2)^(+)from O_(2) and N_(2)^(+) from N_(2) are form then bond order is decrease or increase ? |
|
Answer» Solution :`O_(2) RARR O_(2)^(+)` 2.0 `rarr` 2.5So, INCREASE bond order `N_(2)rarr N_(2)^(+)` 3.0 `rarr` 2.5 So, DECREASE bondorder Note : As the ion is form by LOSS of one `e^(-)` from neutral atom bond order increase or decrease. |
|
| 44. |
Which type of rate is observed in dehydrohalgenation (beta-elimination) reactionof alkyl halide ? |
|
Answer» Solution :(i) Rate of DEHYDROHALOGENATION reaction for ALKYL group is `3^(@) gt 2^() gt 1^(@)` (ii) Order for halogen atom in alkyl halide is : IODINE `gt` Bromine `gt` Chlorine |
|
| 45. |
Which one of the following reacts with CH_3Mgl followed by hydrolysis and gives isopropyl alcohol? |
|
Answer» `CH_3COCH_3` |
|
| 46. |
Which of the following molecules is least resonance stabilized? |
|
Answer»
 Have MINIMUM RESONATE STABILITY because it is not AROMATIC compound rest of all are aromatic |
|
| 47. |
Though nitrogen exhibits +5 oxidation state, it does not form pentahalide. Give reason. |
|
Answer» Solution :Nitrogen shows an oxidation state of +5 in `N_(2)O_(5)` as calculated below. `2x+2(-2)+2(-2)+2(-1)=0` `("for "=O)+("for"toO)+2("for"-O-)=0` `:.2x-10=0orx=+5` It, however, does not form a pentabalide. This may be explained as follows: The electronic configuration of nitrogen is `1s^(2)2s^(2)2p_(x)^(1)2p_(y)^(1)2p_(z)^(1)`. It has three half-FILLED p-orbitals and hence can form a trihalide. To MAKE a pentahalied, we need five half filled orbitals. Since, nitrogen with n=2 CNA have olny s-and p-orbitals and no d-orbitals, it cannot expand its valence shell to show a covalency of 5. However, N has a vacant 3s orbital. If one of the 2s-electrons gets promoted to 3s orbital, we can still have five half-filled orbital. to form `NCl_(5)`. Since energy requiredto promote one 2s electron to 3s orbital is much more than the energy released during the formation of two ADDITIONAL bonds, therefore, such on excitation is thermodynamaically not feasible. Thus, N cannot have a covalency of 5. i.e., nitrogen does not form a pentahcalide.  
|
|
| 48. |
Which type of relation of K_p and K_c when Deltan=0, Deltan gt 0 and Deltan lt 0 |
|
Answer» Solution :If `Deltan=0`, So `K_p=K_c` Its mean, total no. of moles of products and reactants are equal e.g.: `H_(2(g)) + I_(2(g)) hArr 2HI_((g)) , K_p= K_c` If `Deltan gt 0` so, (c+d) `gt` (a+b) and `K_p gt K_c` Its mean , total no. of moles of products are more than reactants so, `K_p gt K_c` and `Deltan` = positive no. e.g.: `PCl_(5(g)) hArr PCl_(3(g)) + Cl_(2(g))` `Deltan`= Mole of product - Mole of REACTANT =2-1=+1 So, `K_p gt K_c` If `Deltan lt 0`, (c+d) `lt` (a+b) and `K_p lt K_c` Its mean, total no. of moles of products are less than reactants so , `K_p lt K_c` and `Deltan`=negative no. e.g. : `{:(N_(2(g))+ , 3H_(2(g)) hArr , 2NH_(3(g))),(1,3,2):}` For (a+b)=(1+3)=4 and for product (c+d)=2 `Deltan`=(c+d)-(a+b)=2-4=-1 `Deltan`=negative no. `THEREFORE Deltan lt 0` `therefore K_p lt K_c` for this reaction. |
|
| 49. |
What happens when : (i) Carbon dioxideis passedthroughan aqueous solution of sodium metaborate. (ii) Boric acid is heated with ethyl alcohol in presence ofconcentrated sulphuricacid. |
|
Answer» Solution :(i) A mixtureof sodiumtetraborate and SODIUM carbonatea formed. `underset(" SOD.Metaborate ")(4NaBO_(2))+CO_(2) rarr underset(" Sod. Tetraborate ")(Na_(2)B_(4)O_(7)) + underset(" Sod. Carbonate ")(Na_(2)CO_(2))` (II) Triethylborate is formed. `H_(3)NO_(3)+3C_(2)H_(5)OH overset("Conc." H_(2)SO_(4))rarrunderset("Triethylborate")(B(OC_(2)H_(5))_(3)) + 3H_(2)O` |
|
| 50. |
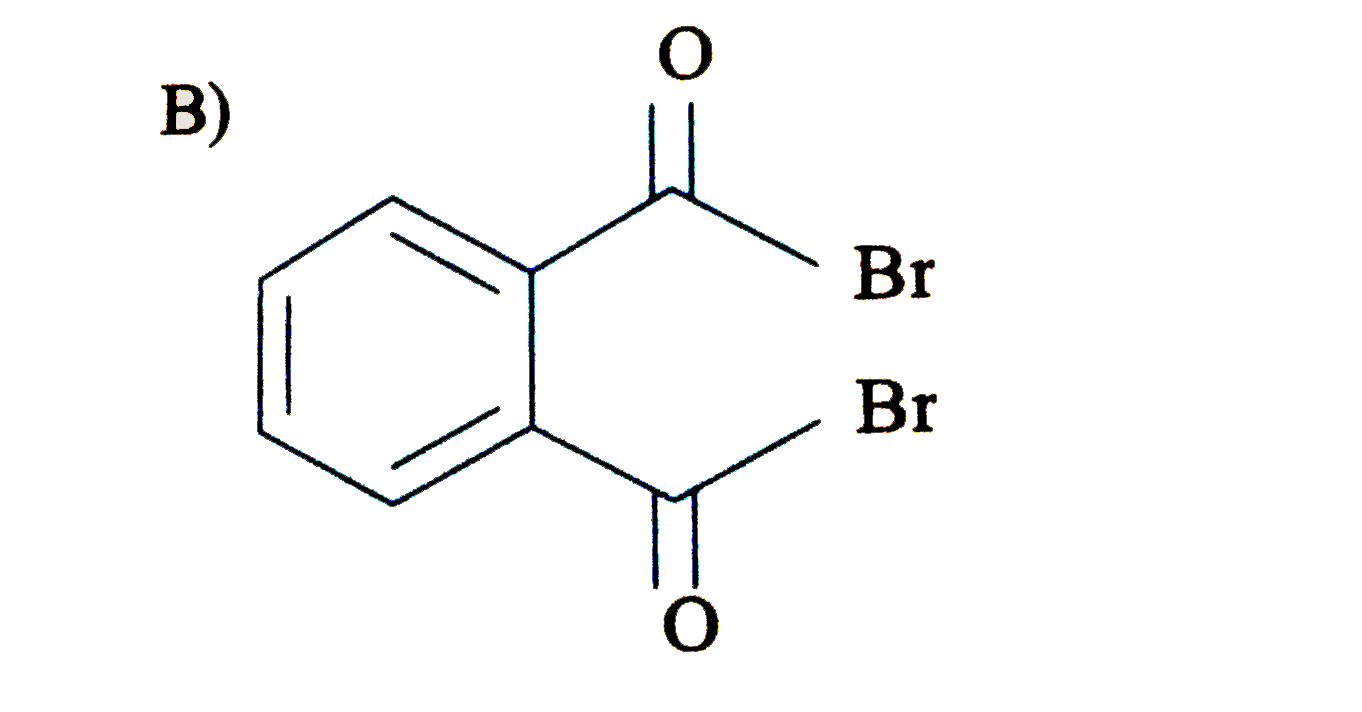
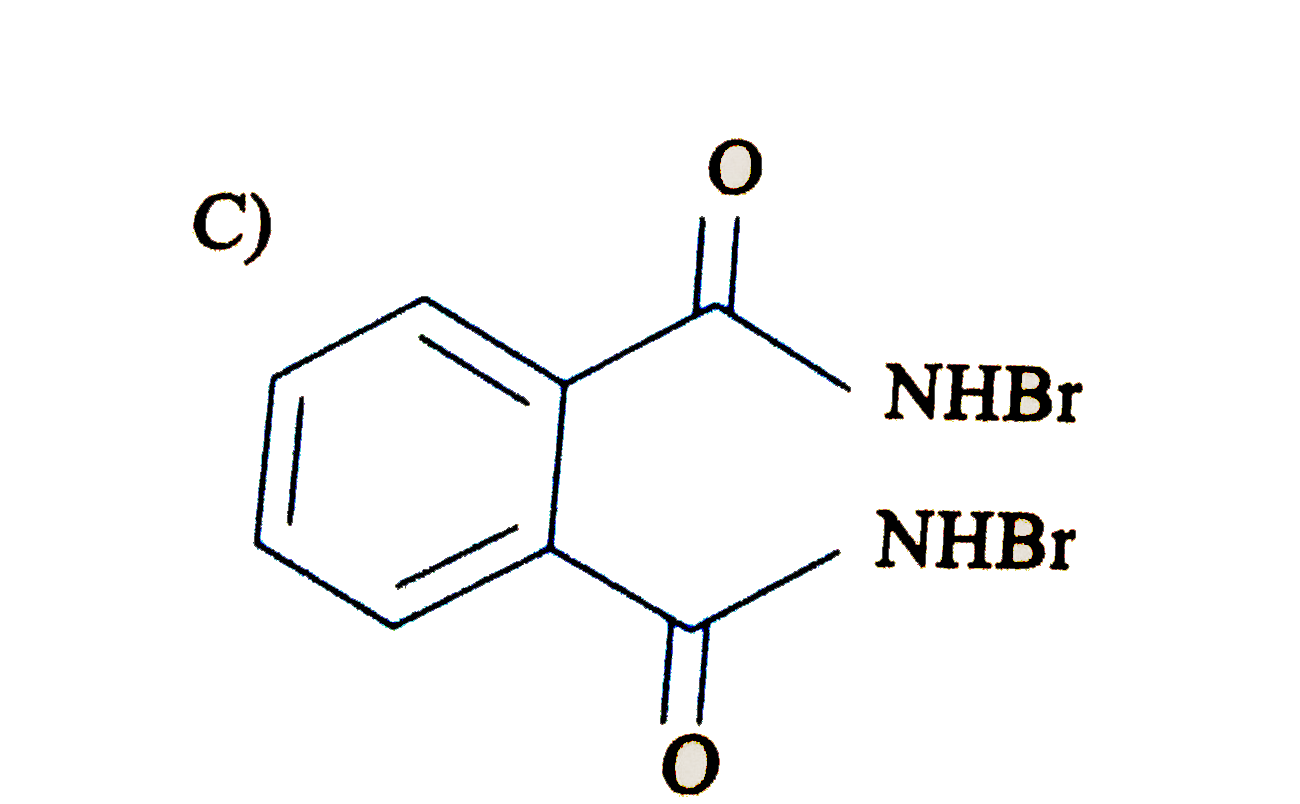
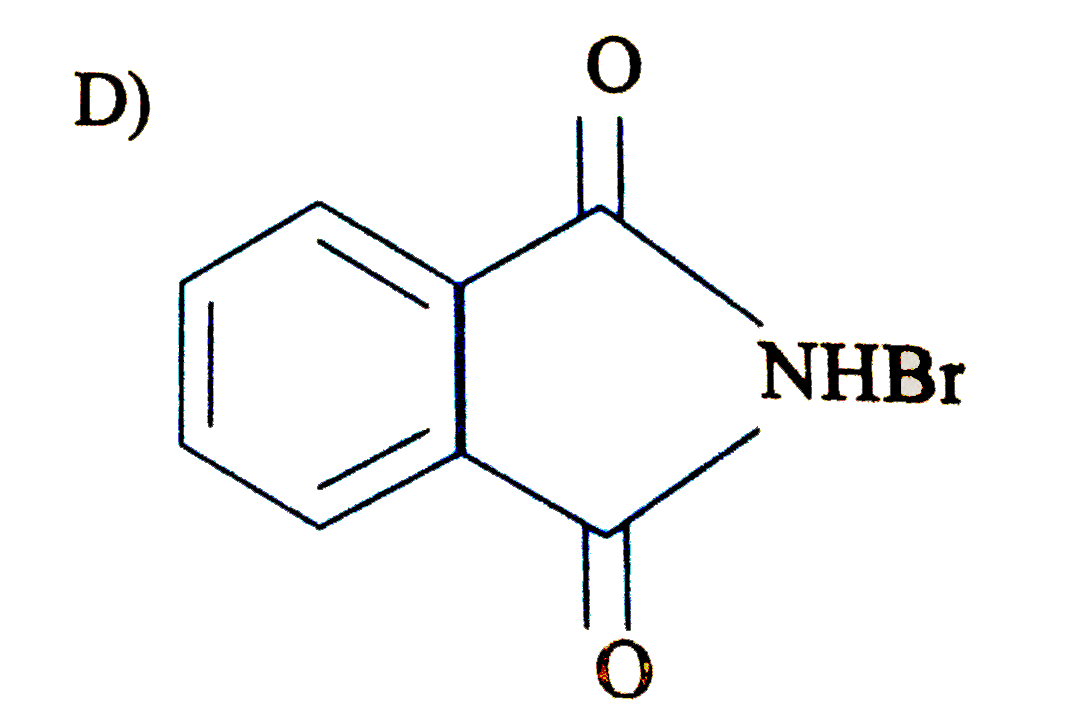
Write down the products of ozonolysis of 1, 2-dimethylbenzene (o-xylene). How does the result support Kekulé structure for benzene? |
Answer» Solution : 1,2-dimethylbenzene (o-xylene) may be regarded as a resonance hybrid of the following Kekule structures I and II. Their ozonolysis takes place as SHOWN below: Thus, in all three products are obtained. These cannot be obtained by any ONE of the Kekule.s structures. This shows that toluene and hence benzene is a resonance hybrid of the TWO resonating structures. |
|