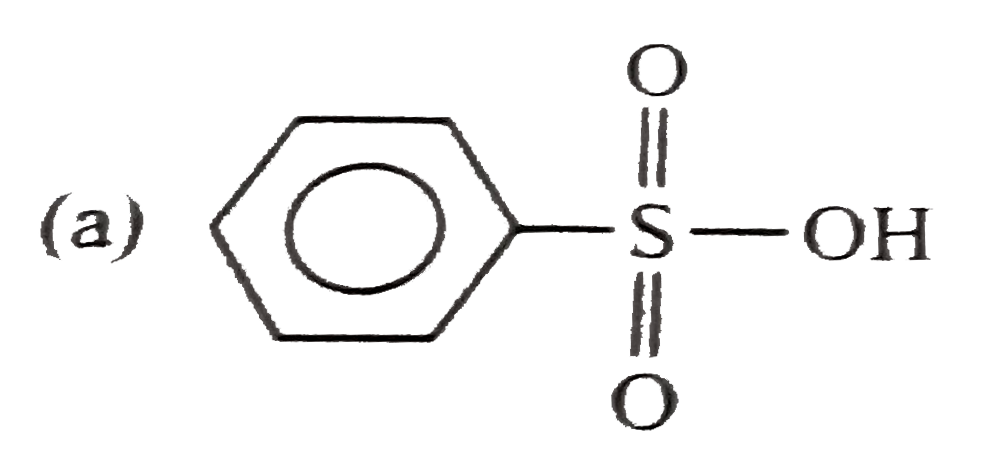
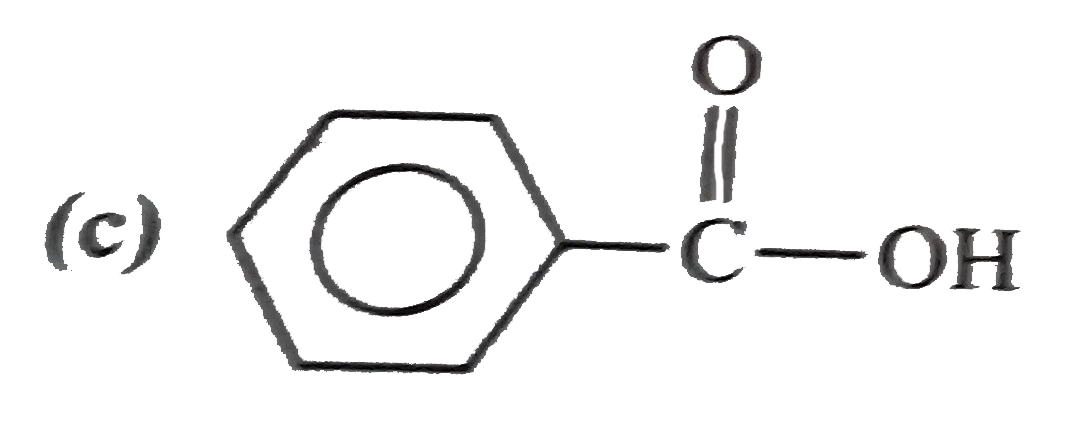
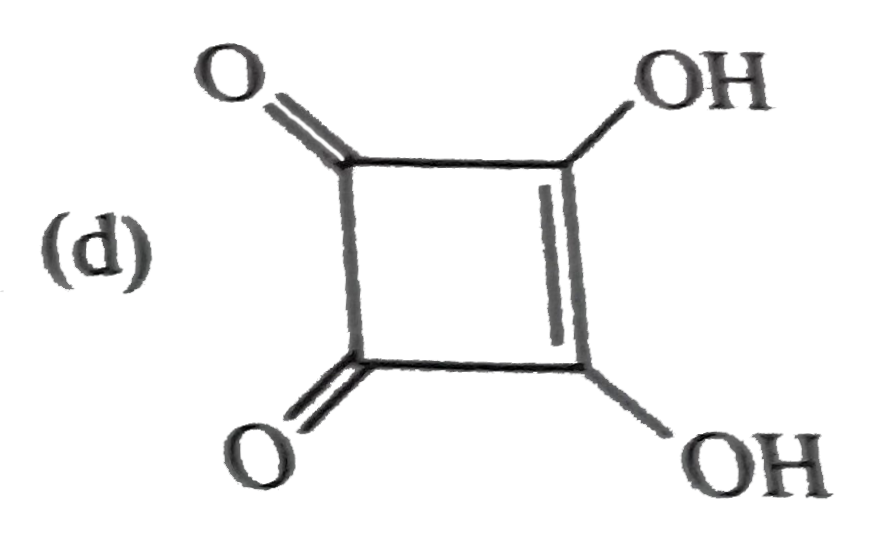
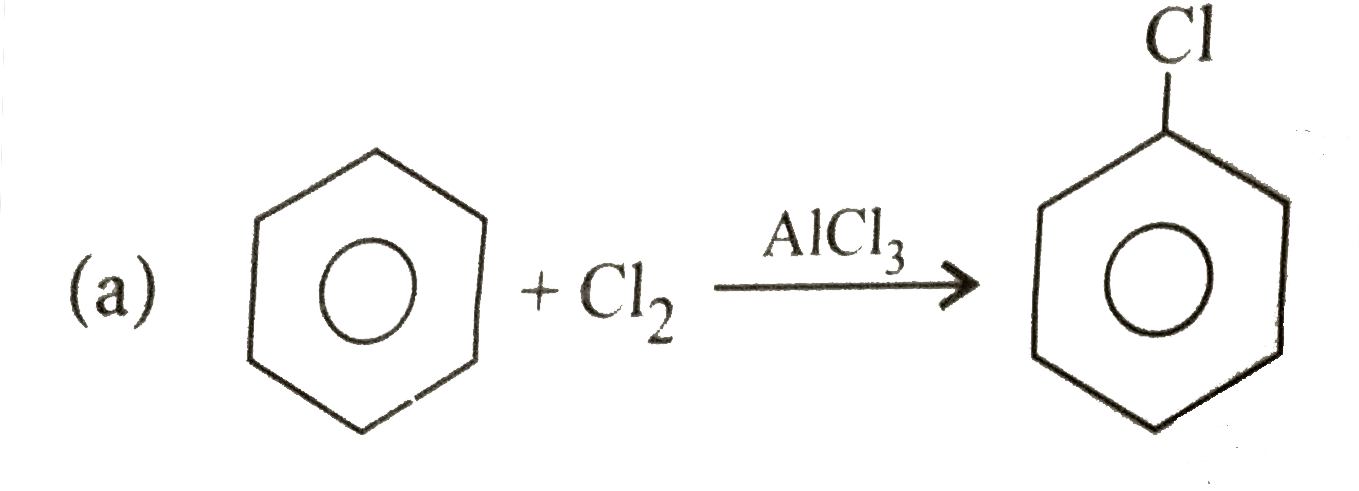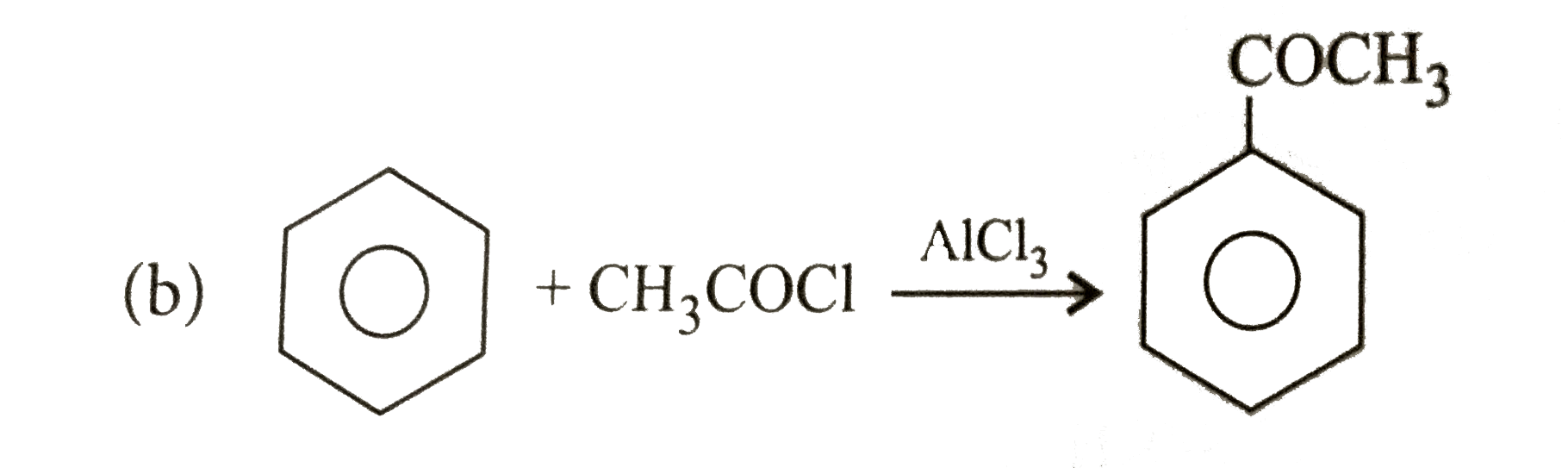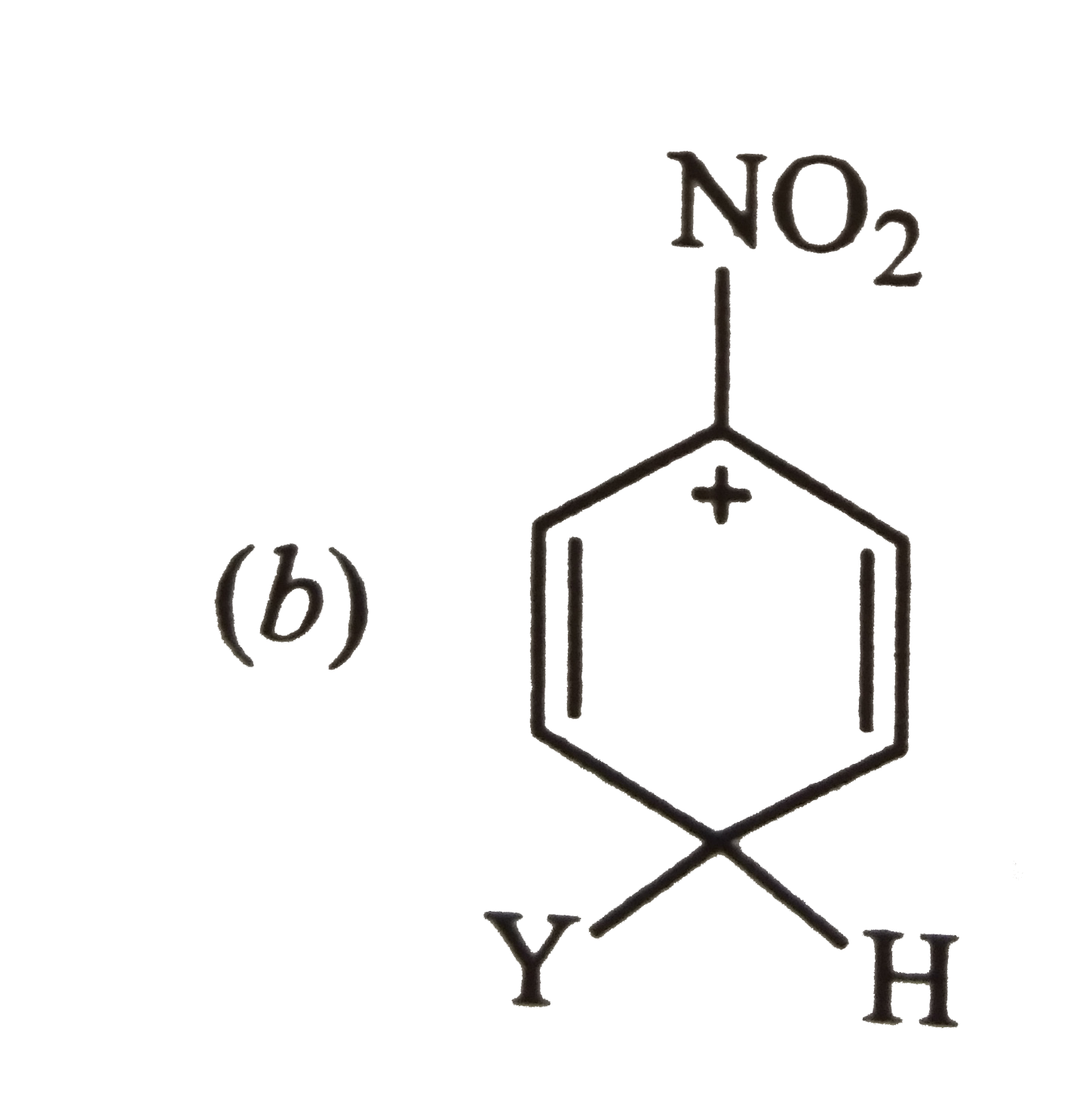Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The threshold frequency v_(0) for a metal is 7xx10^(14)s^(-1) . Calculate the kinetic energy of an electron emitted when radiation of frequency v = 1.0 xx 10^(15)s^(-1) hits the metal . |
|
Answer» SOLUTION :According to EINSTEIN's equation, K.E. of the photoelectron emitted `((1)/(2) mv^(2))` `= h (v - v_(0)) = (6.626 xx 10^(-34) Js) (1.0 xx 10^(15) s^(-1) - 7.0 xx 10^(14) s^(-1))` `= (6.626 xx 10^(-34)Js) (3 xx 10^(14) s^(-1)) = 19.88 xx 10^(-20) J or 1.988 xx 10^(-19) J` |
|
| 2. |
Which of the following pair gives curdy white precipitate and yellow precipitate respectivety in their Lassaignes test? |
|
Answer» `C_2H_5I andC_2 H_5 BR` |
|
| 3. |
Which of the follwoing correct option for free expansion of an ideal gas under adiabatic condition? |
|
Answer» `q = 0, DeltaT NE 0, w = 0` |
|
| 4. |
What is the significance of the term '15 volume' H_(2)O_(2) solution |
| Answer» Solution :This means that one VOLUME of `H_(2)O_(2)` at N.T.P EVOLVES 15 volumes of `O_(2)` | |
| 5. |
The shape of an orbital is given by the quantum number |
|
Answer» n |
|
| 6. |
Use the informationand data given below to answer the questions (a)to (c ) : Stronger intermolecular forces result in higher boiling point. Strength of London forces increases with the number of electrons in the molecule. Boiling point of HF ,HCl HBr and HI are 293 K, 189K, 206 K and 238 K respectively. (a) Which type of intermolecular forces are present in the molecules HF,HCl,HBr and HI ? (b) Looking at the trend of boiling points of HCl, HBr, and HI, explain out of dipole-dipole interaction and London interaction, which one is predominant here. (c ) Why is boiling point of hydrogen fluoride highest while that of hydrogen chloride lowest ? |
|
Answer» Solution :(a) All the given molecules viz. HF,HCl, HBR and HI have PERMANENT dipoles. Hence, all of them posses dipole-dipole and London forces. Hf in addition to dipole-dipole and London forces also has hydrogen bonding. (b) ELECTRONEGATIVITY of Cl, Br and I is in the order : `Cl gt Br gt I`. Therefore, polar character and hence dipole-dipole interactions should be in the order `HCl gt HBr gt HI`. But boiling POINTS are found to be in the order `HCl lt HBr lt HI`. This shows that London forces are predominant. This is because London forces INCREASES as the number of electrons in the molecule increases. In this CASE, the number of electrons increases from HCl to HI. (c ) Due to very high elctronegativity of F, HF is most polar and also there is hydrogen bonding present it. Hence, it has the highest boiling point. |
|
| 7. |
Which of the following possess highest melting point |
|
Answer» cis-2-butene |
|
| 8. |
Write the reaction products of 2-pentene when oxidised with acidified potassium permanganate at 100^(@)C. |
| Answer» Solution :`underset("2-Pentene")(CH_(3)-CH_(2))-CH=CH-CH_(3)+4(O)underset("Acid")overset(KMnO_(6))tounderset("Propanoic acid")(CH_(3)CH_(2)COOH)+underset("Ethanoic acid")(CH_(3)COOH)` | |
| 9. |
The total pressure of a mixture of 6.4 grams of oxygen and 5.6 grams of nitrogen present in a 2 lit vessel is 1200mm. What is the partial pressure of nitrogen in mm? |
|
Answer» 1200 |
|
| 10. |
Which of the following statemets are false ? Assign suitable explanation wherever possible. (a) Beilstein test is a reliable test for halogens in organic compounds. (b) The presence of nitrogen in Lassaigne's test can be shown with the help of Prussian blue colour. (c) Lassaigne's test for the detection of sulphur fails if the compound is of volatile nature. (d) If the compound contains both N and S, then a brick red colour is obtained in the Lassaigne's test. (e) Methods are available to detect oxygen in a compound. (f) Naphthalene can be separated from ammonium chloride by sublimation method. (g) The coloured impurities present in an organic compound can be removed with the help of animal charocal. (h) A mixture of o-nitrophenol and p-nitrophenol can be separated by steam distillation (i) Chromatographic methods can be used to purify impure compounds available in any amount. (j) The purpose of boiling Lassaigne's extract with conc. HNO_(3) before testing for halogens is to destroy any Na_(2)S or NaCN if formed in the reaction |
|
Answer» Solution :(a) Beilstein test is not a reliable test for halogens because many compounds which do not contain any HALOGEN also give this test. (d) If the compound contains both `N` and `S`, then a blood red colour is obtained in the LASSAIGNE's test (f) Sublimation method cannot be USED as both these substances are of volatile nature. (i) Chromatographic methods can be used to PURIFY impure compounds available in small amounts only. |
|
| 11. |
Write IUPAC names of the following compounds (i)(CH_3)_3C CH_2C (CH_3)_3 , (ii)(CH_3)_2C(C_2H_5)_2 (iii)Terta-tert-butylmetane. |
|
Answer» Solution :The expanded strucures of these ALKANES are : (i)`UNDERSET"2,2,4,4-Tetramethylpentane"(overset1CH_3overset2-undersetunderset(CH_3)|oversetoverset(CH_3)|C-overset3CH_2overset4-undersetunderset(CH_3)|oversetoverset(CH_3)|C-overset5CH_3)` (ii) `{:(""overset(2)(C)H_(2) overset(1)(C)H_(3)),("|"),(CH_(3)-.^(3)C-overset(4)(C)H_(2)overset(5)(C)H_(3)),("|"),(""CH_(3)):}` 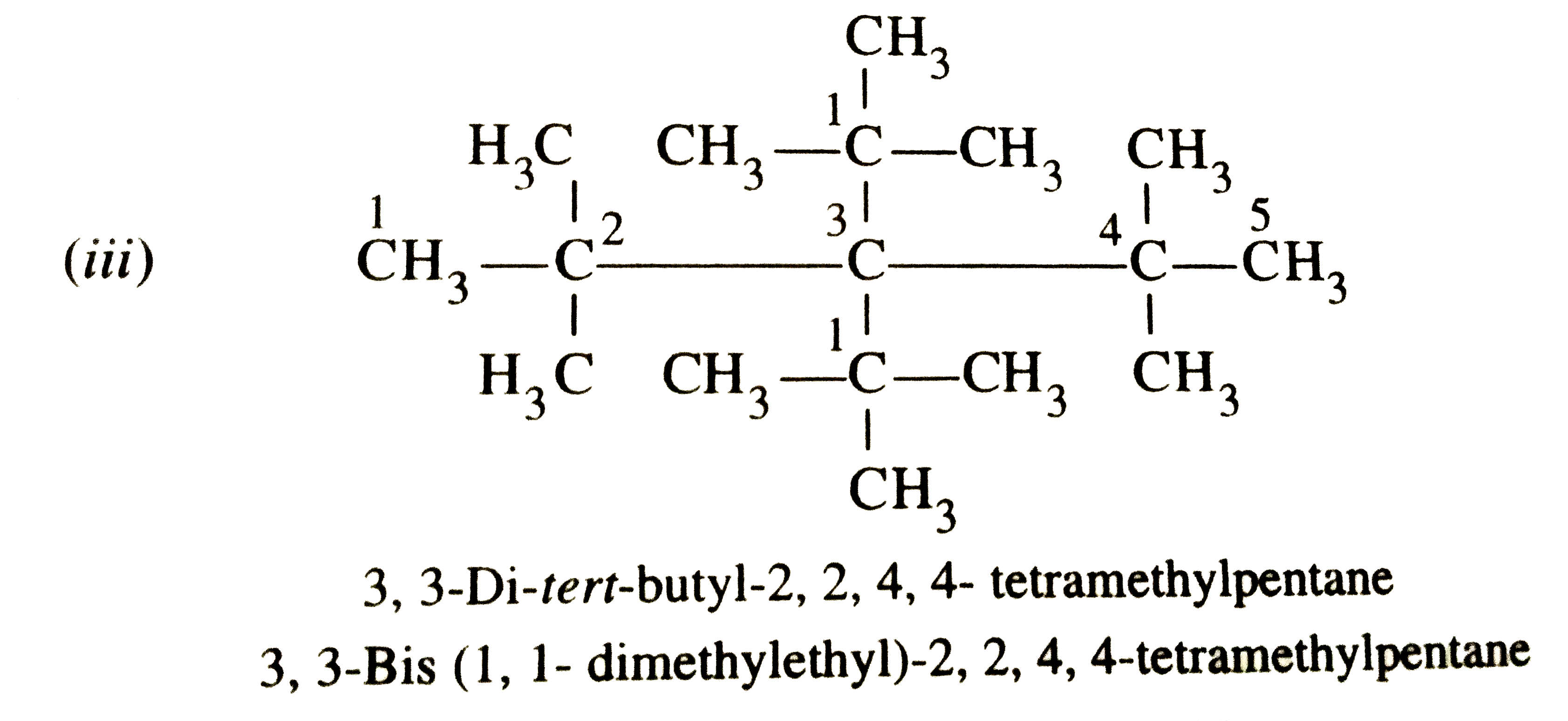
|
|
| 13. |
What is the actual configuration of copper (Z = 29)? Explain about its stability. |
|
Answer» Solution :Copper (Z = 29) Expected CONFIGURATION : `1s^(2)2s^(2)2p^(6)3S^(2)3p^(6)3d^(9)4s^(2)` Actual configuration : `1s^(2)2s^(2)2p^(6)3s^(2)3p^(6)3d^(10)4s^(1)` The reason is that fully filled orbitals have been found to have extra stability. Copper has the electronic configuration `[Ar]3d^(10)4s^(1)` and not `[Ar]3d^(9)4s^(2)` due the symmetrical DISTRIBUTION and exchange energies of d ELECTRONS. Symmetry leads to stability. The full filled configuration have symmetrical distribution of electrons and hence they are more stable than unsymmetrical configuration. |
|
| 14. |
Which of the following acid processes oxidising reducing and complex forming properties? |
|
Answer» `HNO_(3)` |
|
| 15. |
Total number of sulphate ions present in 3.92 g of chromic sulphate is (Cr=52, S=32, O=16) |
|
Answer» `1.8 XX 10^(22)` |
|
| 16. |
Which of the following compounds is/are optically inactive |
|
Answer» `(CH_(3))_(2) CHCH_(2)OH` |
|
| 17. |
Which of the following compound is used to understand the reaction mechanism and exchange of ions ? |
|
Answer» `H_2O` |
|
| 18. |
When an ideal gas undergoes unrestricted expansion |
|
Answer» cooling OCCURS because the molecules LIE above INVERSION TEMP. |
|
| 19. |
Which one is not lyophilic in nature? |
|
Answer» Gelatine sol |
|
| 20. |
Which of the following is (n,) type rection ? |
|
Answer» `""_(5)C^(13) +""_(1)H^(1) to ""_(6)C^(14)` |
|
| 21. |
What is the electrophile in the electrophilic substitution reaction of acetyl chloride (CH_3COCI) andAlCl_3 reacting with benzene? |
|
Answer» `CL^(+)` |
|
| 22. |
Which of the following sets contain only isoelectronic ions ? |
|
Answer» `Zn ^(2+), Ca ^(2+), Ca ^(2+), Al ^(3+)` `(31-3 =28) , Al ^(3+) , Al ^(3+) (13 -3=10)` are not isoelectronic, (b) `K ^(+) (19-1=18), Ca ^(2+) (20-2=18), Se ^(3+) (21 -3=18),` `Cl^(-) (17 + 1=18) ` are isoelectronic. (c )` P ^(3-) (15 +3 =18), S ^(2-) (16 +2 =18), Cl^(-)(17 + 1=18),` `K ^(+)(19-1 =18) ` are isoelectronic. (d) `Ti ^(4+) (22 -4=18), Ar (18), Cr ^(2+) (24-3=21),` `V ^(5+) (23-5=18)` are not isoelectronic. |
|
| 23. |
……type of 3-D model indicate the atomic volume but it is not indicate……. |
| Answer» SOLUTION :SPACE FILLING, BOND | |
| 24. |
Write formula for the following compound: Chromium (III) oxide |
| Answer» SOLUTION :`Cr_2` (III) `O_3` | |
| 25. |
When propionic acid is treated with aqueous sodium bicarbonate CO_(2) is liberated. The 'C' of CO_(2) comes from |
|
Answer» methyl group |
|
| 26. |
Which one of the following statement is wrong in case of enzyme catalysis:- |
|
Answer» ENZYMES WORK BEST at an OPTIMUM temperature |
|
| 27. |
Which of the following compounds produce CO_(2) on reaction with NaHCO_(3) ? |
|
Answer»
|
|
| 28. |
What is chemical bond? When is it formed? |
| Answer» Solution :Chemical bond is the FORCE of attraction between atoms. It is FORMED when ATTRACTIVE FORCES and the repulsinve forces are balanced or at equilibrium. | |
| 29. |
The reagent needed for converting |
|
Answer» Cat. HYDROGENATION |
|
| 30. |
When aqueous solution of sodium ethanoate electrolysed, the product(s) at anode is/are |
|
Answer» ETHANE 
|
|
| 31. |
Which of the following compounds will not give positive iodoform test. Acetophenone, Banzonphenone, 2-Pentanone,3 -pentanone, Acetaldehyde. CH_(3)CO CH_(3),(CH_(3))_(2)CHOH,(CH_(3)CH_(2))_(2)CH-OH,CH_(3)CO OH,CH_(3)CO NH_(2),CH_(3)CO OCH_(3),CH_(3)COCl |
|
Answer» |
|
| 32. |
Which is the most abundant alkaline earth metal in the earth's crust? |
| Answer» SOLUTION :CALCIUM (CA). | |
| 33. |
Which of the following reactions is not an example of electrophilic substitution in benzene ring? |
|
Answer»
|
|
| 34. |
Which is not correctly matched : 1) Basic strength of oxides : K_(2)O lt Na_(2)O lt Li_(2)O 2) Mobility of hydrated ions : Be^(+) lt Li^(+) lt Na^(+) lt K^(+) 3) Thermal stability of bicarbonates : NaHCO_(3) lt KHCO_(3) lt RbHCO_(3) 4) Melting point : NaF lt NaCl lt NaBr ltNaI |
|
Answer» 1&4 |
|
| 35. |
What is the general name for elements of group 1 ? |
| Answer» Solution :ELEMENTS of GROUP 1 are called alkali metals because their OXIDES DISSOLVES in WATER to form hydroxides which are strong bases or alkalies . | |
| 36. |
To study the decomposition of hydrogen iodide, a student fills an evacuated 3 litre flask with0.3 mole of HI gas and allows the reaction to proceed at 500^@C. At equilibrium he found the concentratin of HI which is equal to 0.05 M. Calculate K_C and K_P for this reaction. |
|
Answer» Solution :`V = 3L` `[HI]_("initial") = (0.3 mol)/(3L) = 0.1 M` `[HI]_(EQ)= 0.05 M` `2HI (g) hArr H_2(g) + I_2(g)`  `K_C = ([H_2][I_2])/([HI]^2) = (0.025 xx 0.025)/(0.05 xx 0.05)` `K_C = 0.25` `K_P = K_C (RT)^(Deltan_g)` `DELTA n_g = 2-2=0` `K_P = 0.25 (RT)^0` `K_P = 0.25` |
|
| 37. |
Which of the following is correct order of stability of alkene ? |
|
Answer»
|
|
| 38. |
What is elevation of boiling point? |
|
Answer» Solution :i. The boiling point of a liquid is the TEMPERATURE at whichits VAPOUR pressure becomes equal to the atmosphere pressure. ii. When a non-volatile solute is added to PURE solvent at its boiling point, the vapour pressure of the solution is LOWERED below 1 atm. To bring the vapour pressure again to 1 atm , the temperature of the solution has to be increased. iii. As a result , the solution boils at a higher temperature `(T_b)` then the boling point of pure solvent `(T_b^@)` . This INCREASE in the boiling point is known as elevation of boiling point . |
|
| 39. |
StateClausius form of thesecondlaw ofthermodynamics. |
| Answer» Solution :CLAUSIUS STATEMENT: Heat flows spontaneously from hot objects to cold objects and to re FLOW in the opposite DIRECTION, we have to spend some WORK. | |
| 40. |
Which of the following represents the chemical equation involved in the preparation of H_(2)O_(2) from barium peroxide ? |
|
Answer» `BaO_(2).8H_(2)O + H_(2)SO_(4) to BaSO_(4) + H_(2)O_(2) + 8H_(2)O` |
|
| 41. |
UV radiation from sun causes a reaction That produces |
| Answer» Answer :D | |
| 42. |
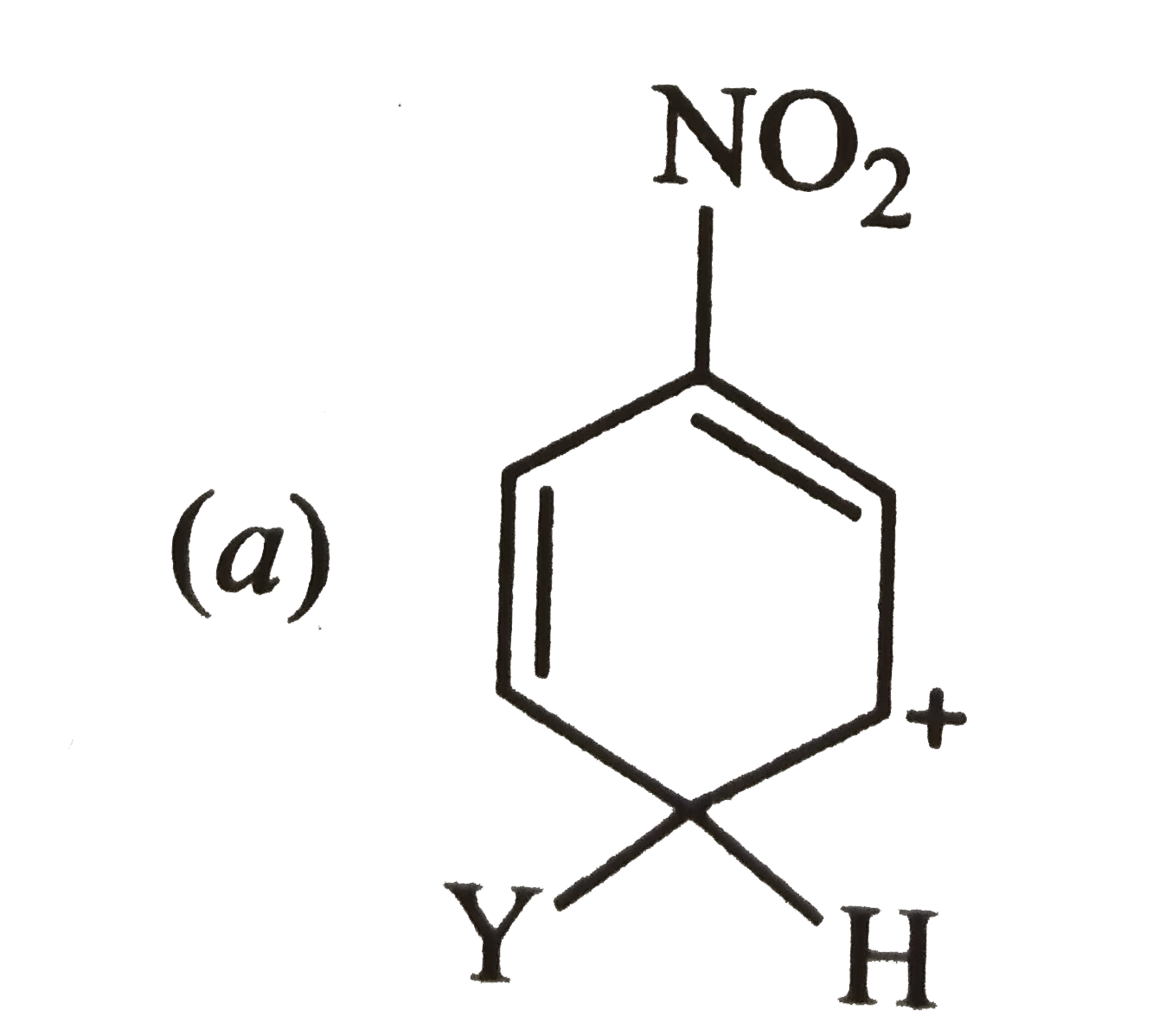
Which of the following carbocation is expected to be most stable ? |
|
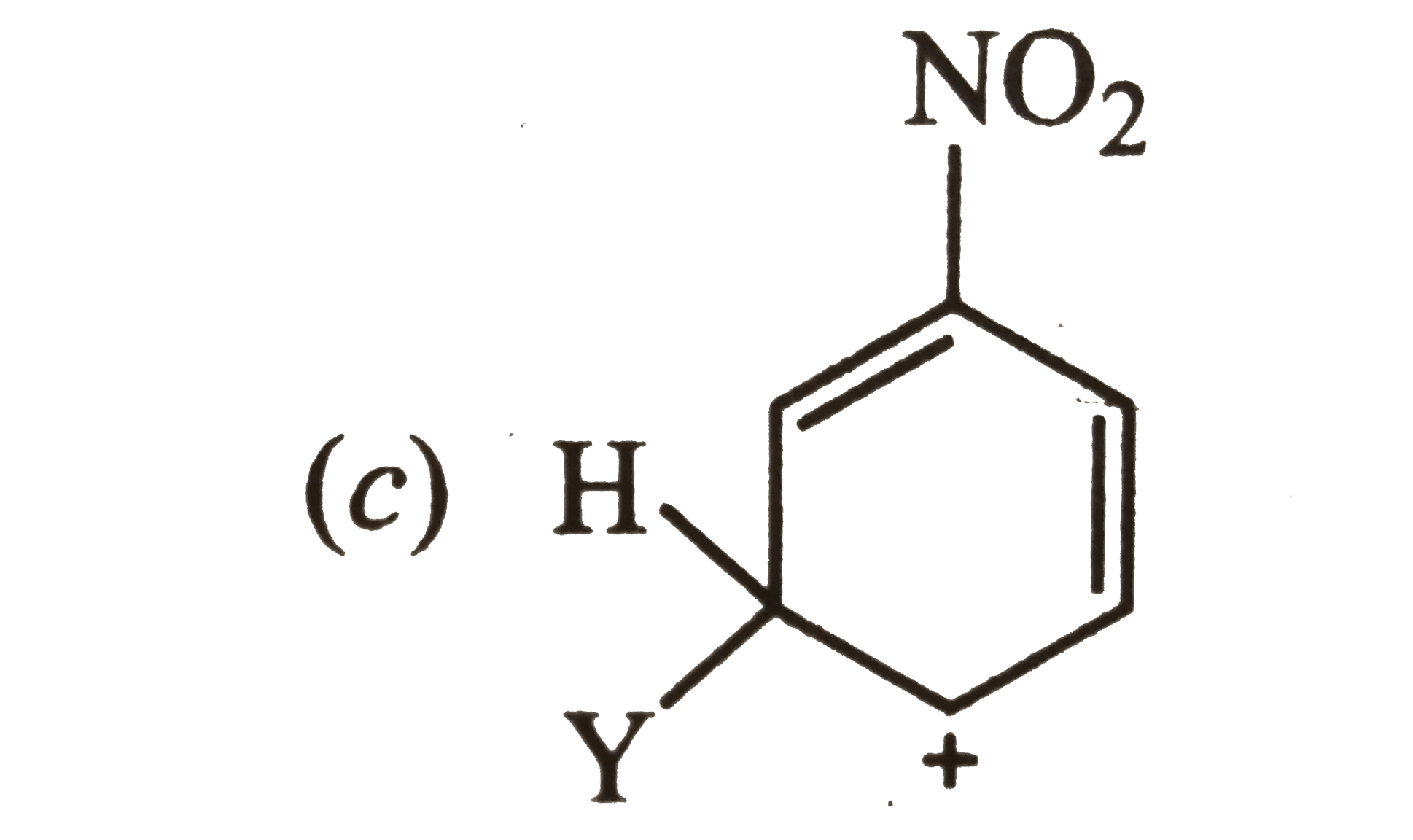
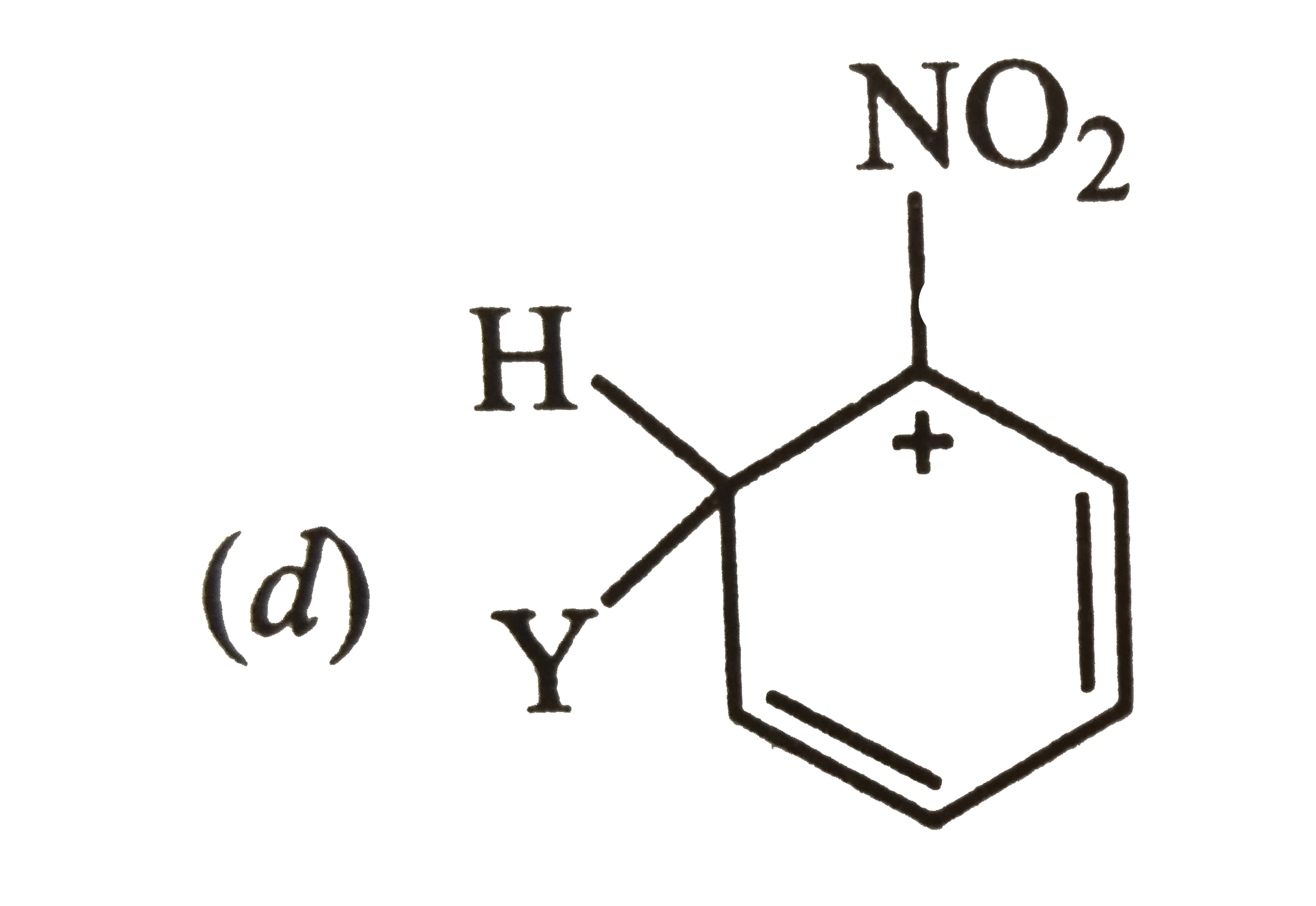
Answer»
Carbocation (a) is also destablized because in one of its resonating structures shown below, +ve charge is present on the carbon atom to which `-NO_(2)` group is attached. Thus, carbocation (a) is also destabilized. 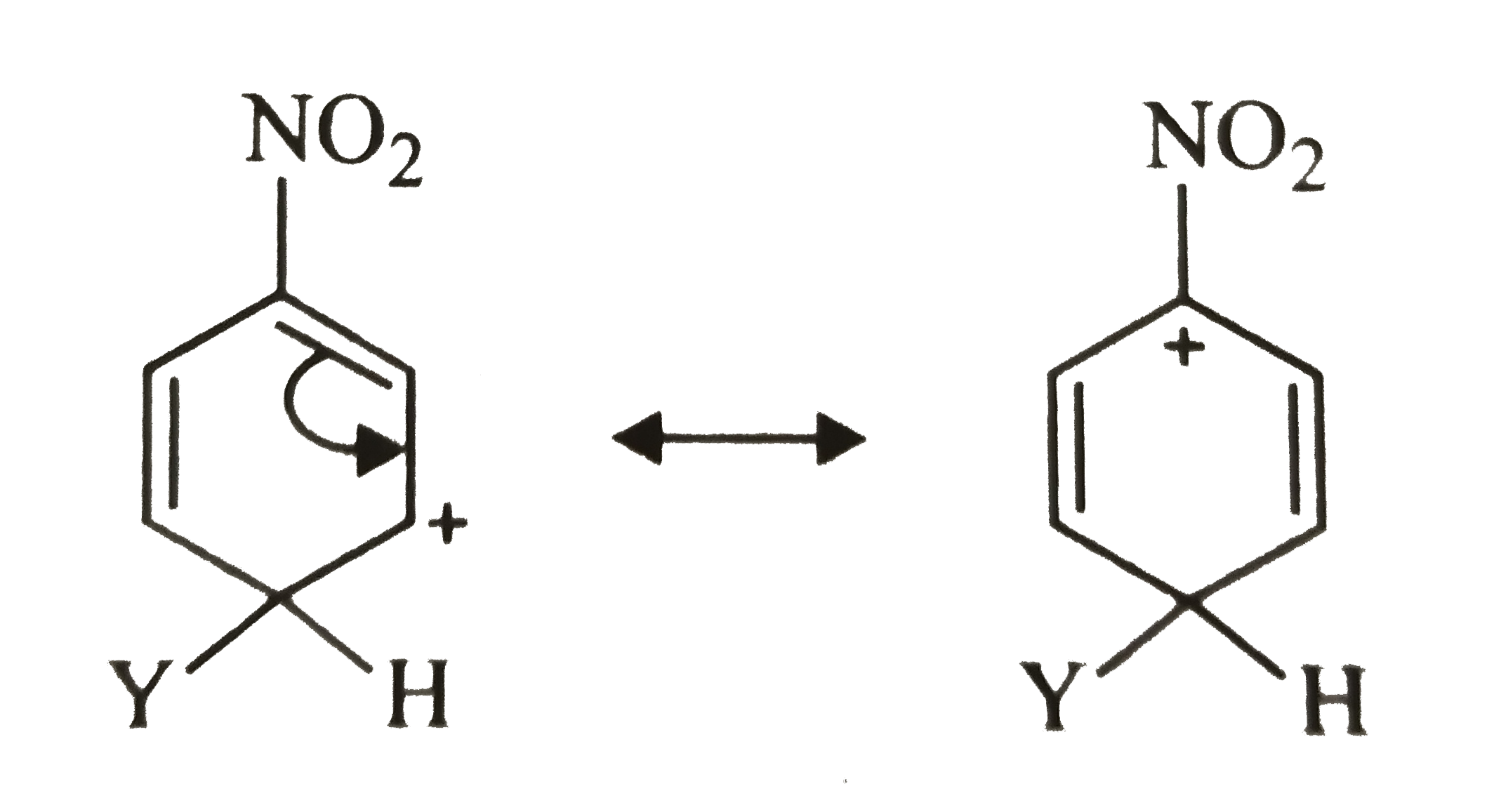 CONSEQUENTLY, carbocation (c) is expected to be most stable since in more of its resonating structures, +ve charge moves to that carbon which is attached to `-NO_(2)` group. 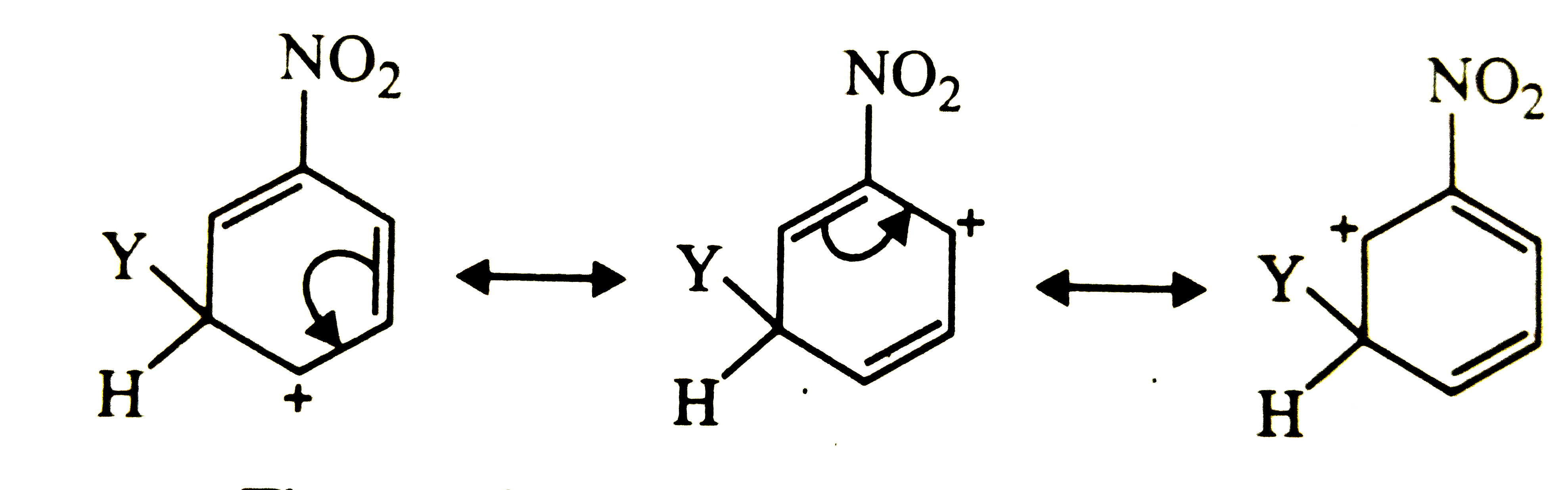 Thus, option (c) is correct. |
|
| 43. |
The statments regarding the compounds of sodium are (i) The mixture of NaOH and CaO is used as decarboxylating agent (ii) aq. NaOH reacts with silicon to form water glass |
|
Answer» only (i) is CORRECT |
|
| 44. |
Which of the following representation of the Fischer projection formula is the meso isomer of 2, 3 dichlorobutane |
|
Answer»

|
|
| 45. |
Which of the following process is employed to convert alkyl halide into alcohol? |
|
Answer» Addition |
|
| 46. |
What is abnormal molar mass? |
| Answer» Solution :The dissociation or association of SOLUTE MOLECULES would alter the total NUMBER of particles present in the solution and HENCE affect the results of measured colligative PROPERTIES . In such solutions, the value of the molar mass of the solute determined using colligative properties would be different from the actual molar mass, and it is called abnormal molar mass. s the number of solute involved in association. | |
| 47. |
What is the type of hybridisation of each carbon in the following compounds? CH_(3)-CH_(3) |
| Answer» SOLUTION :`DeltaS=37.10JK^(-1)` | |
| 48. |
Which of the following is true about catalyst |
|
Answer» It INITIATES REACTION |
|
| 49. |
Which of the followng process does not involve oxidation of iron ? |
|
Answer» formation of `Fe(CO)_(5)` from Fe `overset(0)Fe+5cOrarroverset(0)Fe(CO)_(5)` In all other cases oxidation of Fe occurs since its O.N increases (b) `3Fe(s)+4H_(2)O(g)rarrFe_(3)O_(4)(s)+4H_(2)(g)` (C )`4Fe+3O_(2)+xH_(2)Orarr2Fe_(2)O_(3)xH_(2)O` (d) `CuSO_(4)+FerarrFeSO_(4)+cu` |
|