Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Urea is prepared by the reaction between ammonia and carbon dioxide.2NH_(3_((g)))+CO_(2_((g)))rarr(NH_(4))_(2)CO_(aq)+H_(2)O_((l))In one process, 637.2 g of NH_(3)are allowed to react with 1142 g of CO_(2)(a) Which of the two reactants is the limiting reagent? 2NH_(3_((g)))+CO_(2_((g)))rarr(NH_(4))_(2)CO_(aq)+H_(2)O_((l))(b) Calculate the mass of (NH_(4))_(2)CO formed. 2NH_(3_((g)))+CO_(2_((g)))rarr(NH_(4))_(2)CO_(aq)+H_(2)O_((l))(c) How much of the excess reagent in grams is left at the end of the reaction? |
|
Answer» Solution :(a) `2NH_(3_((g))) + CO_(2((g))) rarr (NH_(4))_(2)CO_(aq) + H_(2)O_((l))` No. of moles of ammonia =`637.3/17` = 37.45 MOLE No. of moles of `CO_(2)` =`1142/44`=25.95 moles As per the balanced equation, one mole of `CO_(2)` requires 2 moles of ammonia. `:.` No. of moles of `NH_(3)` REQUIRED to react with 25.95 moles of `CO_(2)` is = `2/1 xx 25.95` = 51.90 moles. `:.` 37.45 moles of `NH_(3)` is not ENOUGH to completely react with `CO_(2)` (25.95 moles). HENCE, `NH_(3)` must be the limiting reagent, and `CO_(2)` is excess reagent. (b) 2 moles of ammonia produce 1 mole of urea. `:.` Limiting reagent 37.45 moles of `NH_(3)` can produce `1/2 xx 37.45`moles of urea = 18.725 moles of urea. The mass of 18.725 moles of urea = No. of moles `xx` Molar mass = 18.725 `xx` 60 = 1123.5 g of urea. (C) 2 moles of ammonia requires 1 mole of `CO_(2)` `:.` Limiting reagent 37.45 moles of `NH_(3)` will require` 1/2 xx 37.45` moles of `CO_(2)` = 18.725 moles of `CO_(2)` `:.` No. of moles of the excess reagent `(CO_(2))` left = 25.95- 18.725 — 7.225 The mass of the excess reagent `(CO_(2))` left = `7.225xx44` = 317.9 g of `CO_(2)` . |
|
| 2. |
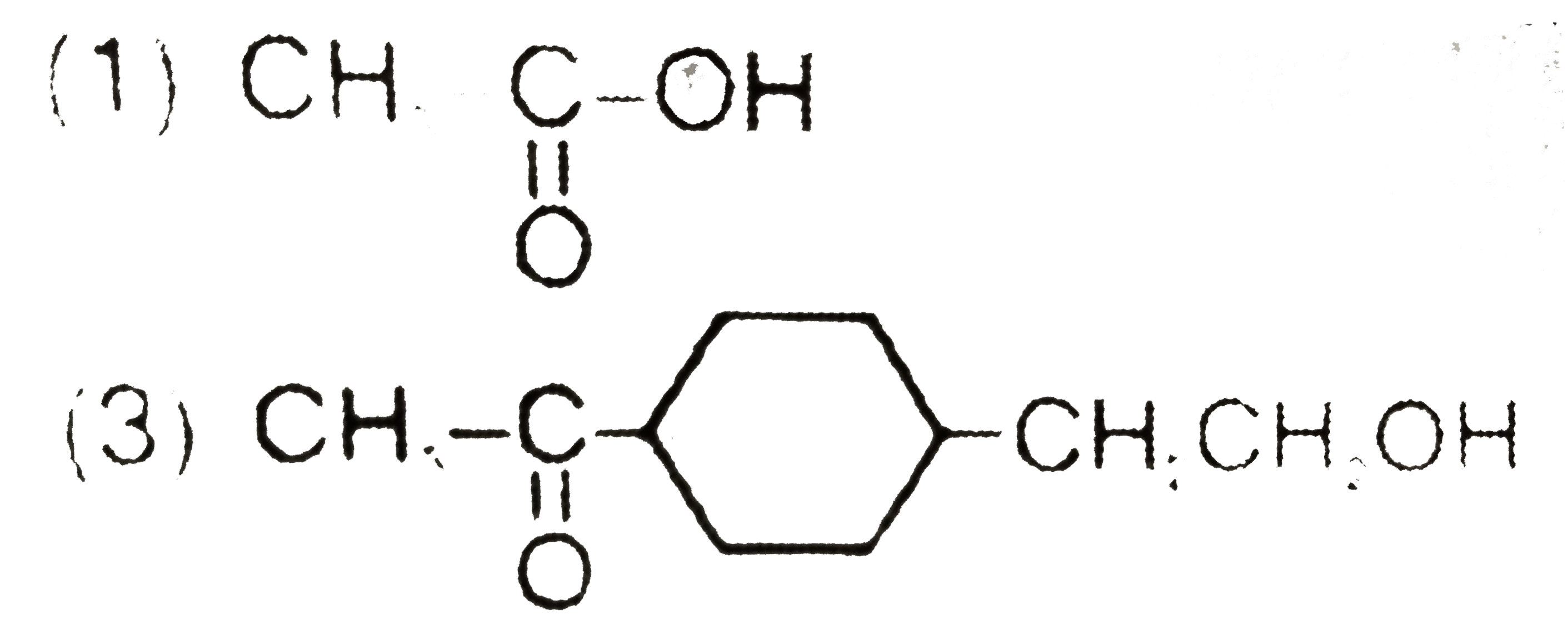
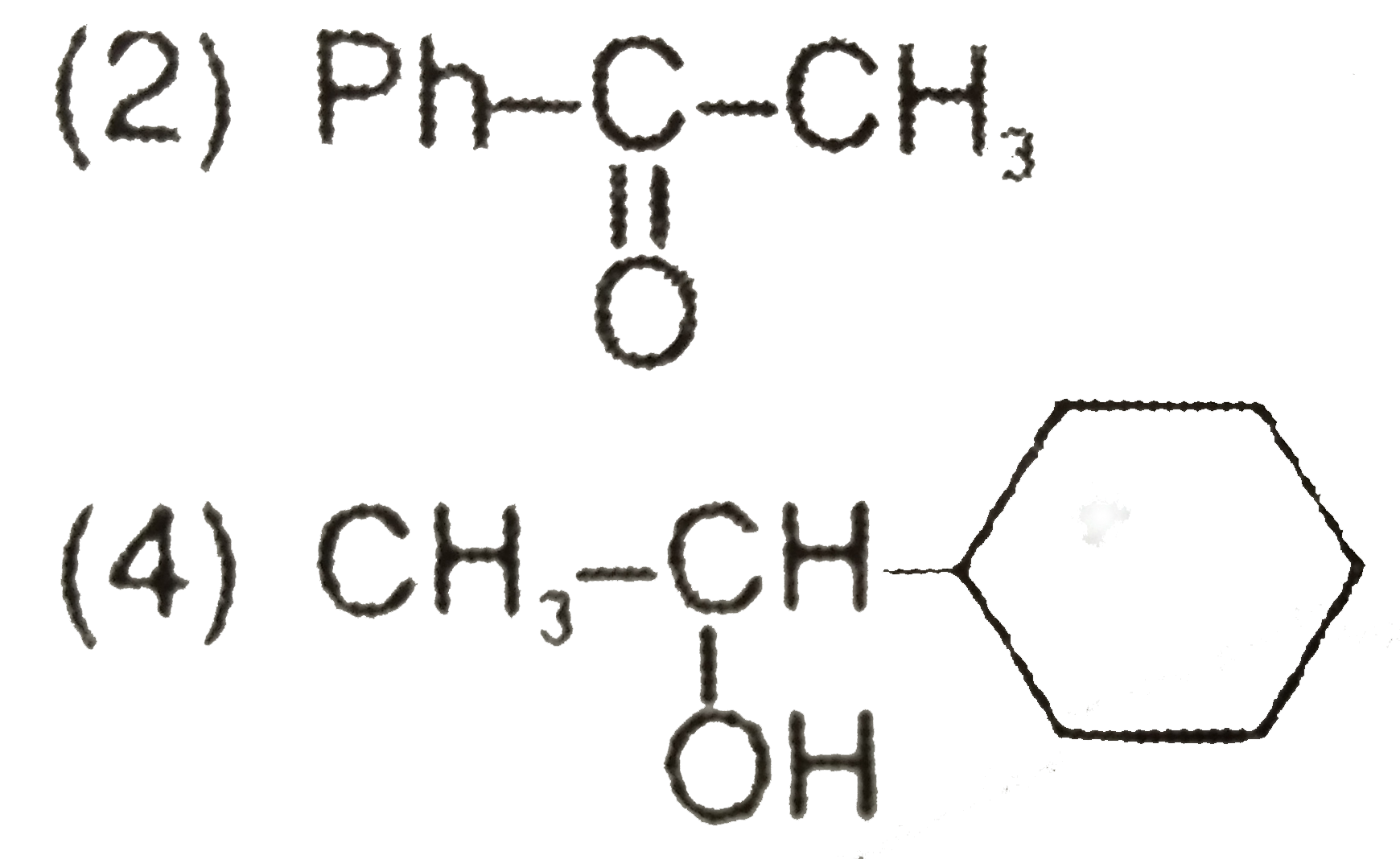
Which of the following alcohols would undergo esterification most readily? |
|
Answer» `CH_(3)OH` |
|
| 3. |
Which substance is used in refrigerator instead of CFC ? |
|
Answer»
|
|
| 4. |
Which one of the following ionic species has the greatest proton affinity to form stable compound ? |
|
Answer» `I^(-)` |
|
| 5. |
Which of the following statement(s) is(are) correct? |
|
Answer» vander Waals radius of iodine is more than its covalent radius |
|
| 6. |
When 2-butyne is treated with H_(2)/Lindlar's catalyst, compound X is produced as the major product and when treated with Na/liq. NH_(3) it produces Y as the major product. Which of the following statements is correct ? |
|
Answer» X will have HIGHER dipole MOMENT and higher BOILING POINT than Y. |
|
| 7. |
When H_(2)S gas is passed through aqueous solution of CuCl_(2), HgCl_(2), BiCl_(3) and CoCl_(2) in the presence of excess of dilute HCl, it fails to precipitate |
|
Answer» `CuS` |
|
| 8. |
Which of the following is true about an aqeous solution (saturated) of AgCl? |
|
Answer» Adding `NaCl` loweres the solubility of `AGCL`. |
|
| 9. |
What is the relation between reactivity of compound, bond strength and bond order between two carbon atoms ? |
| Answer» Solution :As the BOND ORDER between C-C INCREASES bond ENTHALPY increases and bond STRENGTH increases. But as the bond order increases, reactivity of compound increases. | |
| 10. |
Two glass bulbs A and B are connected by very small tube having a stop cock. Bulb A has a volume of 100 ml and contained a gas while bulb B is empty and has a volume of 150 ml. On opening the stop cock, the pressure of the gas in bulb A will fall down to : |
Answer» Solution : Total moles INITIALLY and finally is constant `:. 100 xx p^@ + O = 100 xx P + 150 xx P` `implies P (100 xx P^@)/(250) = 40%` of `P^@.` |
|
| 11. |
Those stereoisomers are called configurational isomers if they differ in spatial arrangement of atom or group and are not interconvertible without cleaving a bond. Geometrical isomer arises due to restricted rotation about a bond or centre. The number of G.I is 2^(n) where n= number of stereoisomers, in some cases the number of G.I is less than 2^(n) in case where either side nomenclature will be possible. Which of the following compounds can show geometrical isomerism. |
|
Answer»
|
|
| 12. |
Which of the bicarbonate does not exist in solid state? |
|
Answer» `NaHCO_(3)` |
|
| 13. |
While filling electrons in pi_(2p_(x)) and pi_(2p_(y)), the electronic configuration rules that one to be followed is |
|
Answer» PAULI's exclusion PRINCIPLE |
|
| 14. |
Which of the following is electron donor? |
|
Answer» `-COOH` |
|
| 15. |
Which of the followinte test will not be given by (Squaric acid) |
|
Answer» `Br_(2)` water test |
|
| 16. |
Which one of the following statement is false ? |
|
Answer» Photochemical SMOG CAUSES IRRITATION in eyes |
|
| 17. |
Two identical vessels are filled with 44g of Hydrogen and 44g of carbon dioxide at the same temperature. If the pressure of CO_(2)is 2 atm, the pressure of Hydrogen is |
| Answer» Answer :D | |
| 18. |
Which of the following statements is not correct from the view point of molecular orbital ? |
|
Answer» ` Be_2` is not a stable molecule |
|
| 20. |
Which is the correct relationship between wavelength and momentum of particles |
|
Answer» `lambda=h/P` |
|
| 21. |
Those stereoisomers are called configurational isomers if they differ in spatial arrangement of atom or group and are not interconvertible without cleaving a bond. Geometrical isomer arises due to restricted rotation about a bond or centre. The number of G.I is 2^(n) where n= number of stereoisomers, in some cases the number of G.I is less than 2^(n) in case where either side nomenclature will be possible. Which of the following pairs has correct stability order : |
|
Answer»
|
|
| 22. |
Which of the following statements are correct about this reaction ? |
|
Answer» The given reaction follows `S_(N)2` MECHANISM. |
|
| 23. |
Write periodicity of ionization enthalpy of 1 to 60 elements of periodic table. |
|
Answer» Solution :Generally IONISATION ENTHALPY means first IONIZATION enthalpy. The first ionization enthalpies of elements having atomic numbers up to 60 are plotted in figure.  The periodicity of the graph is quite striking. You will find maxima at the noble gases which have CLOSED electron shells and very stable electron configurations. On the other HAND, minima occur at the alkali metals and their low ionization enthalpies can be correlated with their high reactivity. In period we go left to right ionization enthalpy increases. In group we go top to bottom ionization enthalpy decreases. |
|
| 24. |
What is the equilibrium concentrations of each of the substances in the equilibrium when the initial concentration of I Cl was 0.78 M ? 2I Cl(g) hArr I_2(g) + Cl_2(g) , K_c=0.14 |
|
Answer» Solution :Let at EQUILIBRIUM , `[I_2]=[Cl_2]="x mol L"^(-1)` `{:(,2ICl(g) hArr, I_2(g) +,Cl_2(g)),("Initial conc.",0.78 M, 0,0),("At EQU.", 0.78-2x, x ,x):}` `K_c=([I_2][Cl_2])/[IC l]^2` `0.14=(x xx x)/(0.78-2x)^2` or `x^2=0.14(0.78-2x)^2` or `x/(0.78-2x)=sqrt0.14=0.374` or x=0.292-0.748 x 1.748 x = 0.292 `therefore` x=0.167 Thus, at equilibrium , `[I_2]` =0.167 M , `[Cl_2]` =0.167 M , [I Cl]=0.78 - 2 x 0.167 = 0.446 M |
|
| 25. |
Which of the following ions is more stable? Use resonance to explain your answer |
Answer» Solution : Out of the FOLLOWING two ions, carbocation (A) is more stable than carbocation (B) is LESS planar and hence does not undergo resonance. Alternativity, since and endocyclic (within the RING) double bond is more stable than an exocyclic (outside the ring) double bond, THEREFORE carbocation (A) is more stable than carbocation (B) |
|
| 26. |
When alpha particles are sent through a thin metal foil most of them go straight through the foil because |
|
Answer» ALPHA particles are much heavier than electrons |
|
| 27. |
Which resonance form in each of the following sets is the major contributor to the real structure? (i) underset((A))(CH_(2)-underset(CH_(3))underset(|)(C)-CH)=CH_(2)harrunderset((B))(overset(+)(CH_(2))-underset(CH_(3))underset(|)(C)=CH)-overset(-)(CH_(2)) harr underset((C))(overset(-)(CH_(2))-underset(CH_(3))underset(|)(C)=CH)-overset(+)CH_(2) (ii) underset((A))(CH_(3)-overset(O)overset(||)(C)-CH)=CH_(2)harrunderset((B))(CH_(3)-overset(O^(-))overset(|)underset(+)(C)-CH)=CH_(2)harr underset((C))(CH_(3)-overset(O^(-))overset(|)(C)=CH)-overset(+)(CH_(2)) (iii) underset((A))(CH_(2)=CH-CH=CH_(2))harrunderset((B))(overset(+)(C)H_(2)-CH=CH)-overset(-)CH_(2)harrunderset((C))(overset(-)(CH_(2))-CH=CH-overset(+)(CH_(2)) |
|
Answer» |
|
| 28. |
which of the following compound cannot give iodoform when react with IO^(-) (hypolodite) |
|
Answer» `CH_(3)-underset(O)underset(||)(C)-OH` |
|
| 29. |
What type of solids are eletrical conductors, Malleable and ductile ? |
| Answer» SOLUTION :METALLIC SOLIDS. | |
| 30. |
Which of the following compound changes the salmon-coloured precipitate of zinc nitroprusside into red? |
|
Answer» `Na_(2)CO_(3)` |
|
| 31. |
Which one of the following is a better reducing agent for the following reduction. RCOOH rarr RCH_(2)OH |
|
Answer» `SnCl_(2)//HCL` |
|
| 32. |
The Van't Hoff factor (i) for a dilute aqueous solution of the strong elecrolyte barium hydroxide is |
|
Answer» Solution :`Ba (OH)_(2)` dissociates of form `Ba ^(2+) and 2OH^(-)` ION `alpha = ((i -1))/((N-1))` ` i = alpha ( n -1) +1` `THEREFORE n = i =3 ` (for `Ba (OH)_(2), alpha =1`) |
|
| 33. |
What will be value of 40""^(@)C in Fahrenheit scale ? |
|
Answer» `183""^(@)F` |
|
| 34. |
Write the mechanism for followingreaction underset("Propane")(CH_(3)-CH=CH_(2))+Hbroverset(C_(6)H_(5)CO_(2)O_(2))rarr underset("1-bromopropane")(CH_(3)-CH_(2)- Br) |
|
Answer» Solution :`CH_(3)-CH = CH_(2)+ HBroverset(C_(2) H_(5)CO_(2)O_(2))(to)UNDERSET("1-bromopropane")(CH_(3)-CH_(2)- Br)` thereactionproceeds throughfree radicalmechanism Step 1 TheweakO- Osinglebondlinkageof peroxideundergoeshomolyliceleavagetogenerate FREE RADICAL  Step :2TheradicalsabstractshydrogenfromHBrthusgeneratingbrominefree radical  Step : TheBrominefree radicaladds to the doublebond in the wayso asto formthemorestablealkylfreeradical  Step : 4Addtionof HBrsecondaryfreeradical 
|
|
| 35. |
Which of the following quantities refers to a saturated solution ? |
|
Answer» Mol solubility |
|
| 36. |
The solubility of CaF_2 in water is 1.7xx10^(-3) g/100 mL at 298 K temp. Calculate solubility product of CaF_2. |
| Answer» SOLUTION :`4.14xx10^(-11)` | |
| 37. |
The value of (n_2 + n_1) and (n_2^2 - n_1^2) for He^+ ion in atomic spectrum are 4 and 8 respectively . The wavelength of emitted photon when electron jump from n_2to n_1is |
|
Answer» `(32)/(9) R_H` `IMPLIES((n_2 - n_1)(n_2 + n_1))/((n_2 + n_1)) = (n_2 + n_1) = 2` `therefore n_2 = 3 , n_1 = 1 impliesbar(upsilon) = Z^2 R[1/(n_1^2)- 1/(n_2^2)]` `=(2)^2 R (1/((1)^2) - 1/((3)^2))= (32R)/(9) , LAMBDA =(9)/(32R)` |
|
| 38. |
When metals X is treatedwith sodiumhydroixdes , a whiteprecipitate (A) isobtained , which is solublein excess of NaOHto give soublecomplex (B). Compound(A)is soublein diluteHCl to form compound (C ). Thecompound (A)when heatedstronglygives (D), which isused to extractmetal.Identify(X), (A), (B), (C ) and (D).Write suitable equationsto supporttheir identities. |
|
Answer» Solution :Since metal X reacts with `NaOH`to first GIVE a white ppt. (A) which dissolves in excess of NaOH to give a soluble complex (B), therefore , metal (X) must beAl , ppt (A)must be `Al(OH)_(3)`and complex (B) must be sodium tetrahydroxoaluminate (III). `{:(underset((X))(2Al) + 3NaOH rarr underset(" Aluminium hydroxide (ppt.) ")(Al(OH)_(3)darr) + 3Na^(+)),(underset((A))(Al(OH)_(3))+NaOHrarr underset("Sod. tetrahydroxoaluminate (III)")underset((B))(NA^(+)[Al(OH)_(4)]^(-))):}` Since (A), i.e,`Al(OH)_(3)` react with DIL. `HCl` to give compound(C), therefore, (C ) must be `AlCl_(3)`. `underset((A))(Al(OH)_(3)) + 3HCl rarr underset((C ))(AlCl_(3)) + 3H_(2)O` Since (A)on heatinggives (D)which is used to extract metal (i.e, electrolysis of `Al_(2)O_(3)` gives Al metal), therefore , (D) must bealumina `(Al_(2)O_(3))` `underset((A))(2Al (OH)_(3)) OVERSET(Delta)rarr underset((D)) (Al_(2)O_(3)) + 3H_(2)O` |
|
| 39. |
What is hydrogen bond ? |
| Answer» SOLUTION :The attractive FORCE which binds hydrogen atom of one MOLECULE with electronegtive atom PRESENT is the same or other molecule is called hydrogen bond. | |
| 40. |
Which of the following pairs are not functional group isomers ? |
|
Answer» II and III |
|
| 41. |
What is the [HF] in 0.02 M HF solution ? |
|
Answer» Solution :`(K_a=3.2xx10^(-4))` HF is weak ACID, its equilibrium established in aqueous solution is as under. `{:(,HF_((aq))+H_2O_((l)) hArr , H_3O_((aq))^(+)+, F_((aq))^(-)),("Initiall",0.02M,0,0),("At equilibrium",(0.02- CALPHA),calpha,C alpha):}` If `alpha`=degree of dissociation there so , decrease in concentration of HF = `c alpha` In solution [HF] =(0.02-0.02 `alpha`)M =0.02 (1-`alpha`) but value of `alpha` is much less , so neglible . `(1-alpha)=1` Thus, [HF]=0.02(1)=0.02 M So, most of weak acid remain as a undissociate molecule in solution and therefore its concentration in solution can be taken of INITIAL concentration C. |
|
| 42. |
Why C Cl_(4) is added in bromination of alkene ? |
| Answer» Solution :`C Cl_(4)` is liquid solvent, which is non-polar solvent. Bromine is soluble in non-polar liquid. So, on formation of bromine solution it become CAPABLE to REACT with non-polar alkene. | |
| 43. |
When density of ideal gas is maximum ? |
|
Answer» <P> SOLUTION :At HIGH pressure and low temperature.Because `d=(Mp)/(RT), d prop p prop (1)/(T)` |
|
| 44. |
What is the ratio of coefficients of caustic soda and zinc metal in the reaction between zinc and NaOH? |
|
Answer» Solution :The SKELETON equation is `NaOH+Zn to Na_(2)ZnO_(2)+H_(2)` The balanced equation is `2NaOH+Zn to Na_(2)ZnO_(2)+H_(2)` The RATIO of coefficent of NaOH and Zn=2:1 |
|
| 45. |
Which occupy peak of the atomic volume curve- |
|
Answer» TRANSITION element |
|
| 46. |
What is dehydrohalogenation ? |
| Answer» SOLUTION :REMOVAL of halogen acid (HX) from the ALKYL halide is CALLED dehydrohalogenation. | |
| 47. |
Which of the following does not have an active methylene group ? |
|
Answer» `CH_(3)CH_(2)NO_(2)` |
|
| 48. |
Which of the following option is incorrect? |
|
Answer»
|
|
| 49. |
Two liters of an ideal gas at a pressure of 10 atm. expands isothermally into a vacuum until its total volume is 10 liters. How much heat is absorbed and how much work is done in the expansion ? |
|
Answer» <P> Solution :We have `q=-w= p_(ex) (10-2) = 0(8) =0` no WORK is done, no HEAT is absorbed |
|