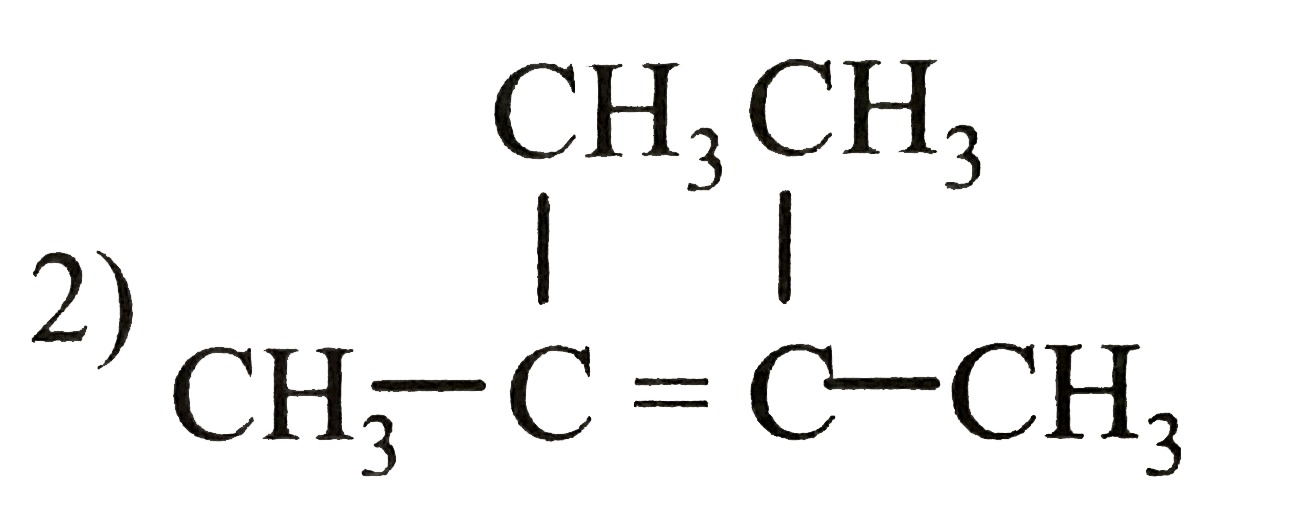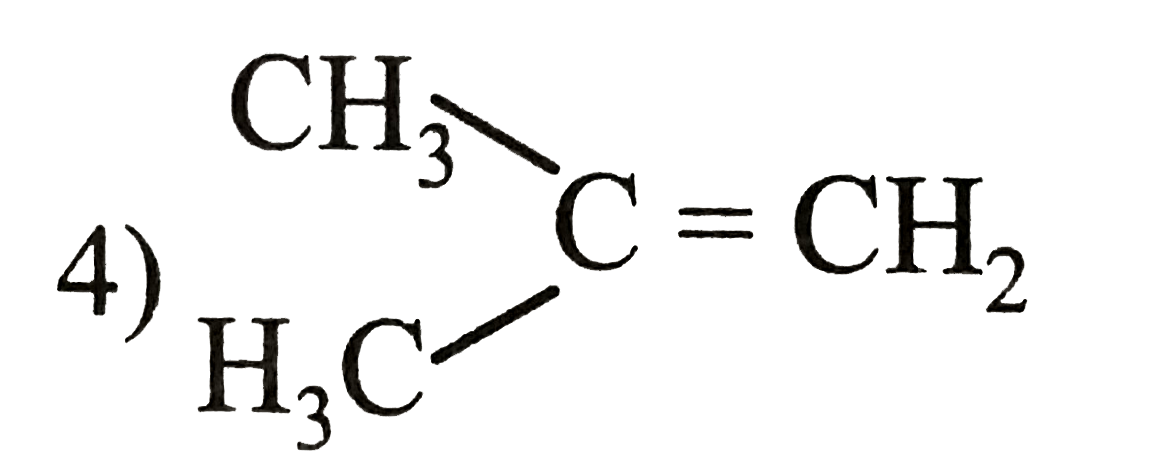Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
When TLC solvent get 10cm height then compound (A) gets 8cm and (B) gets 6cm height calculate R_(f) of A and B |
| Answer» SOLUTION :(A) 0.8 and (B) 0.6 | |
| 2. |
The salt Al(OH)_(3) is involved in the following two equilibria, Al(OH)_(3(s)) hArr Al_((aq))^(3+) + 3OH_((aq))^(o.), ksp Al(OH)_(3(s)) + OH_((aq))^(-) hArr [Al(OH)_(4)^(-)]_((aq)), kc Which of the following relationship is correct at which solubility is minimum? |
|
Answer» `[OH^(-)] = ((Ksp)/(Kc))^(1//3)` `S = (Ksp)/([OH^(-)]^(3) + Kc[OH^(-)]` for MINIMUM SOLUBILITY `(ds)/(d(OH^(-))) = 0` `:. - (Ksp XX 3 )/([OH^(-)]^(4)) + Kc = 0,[OH^(-)] = ((3Ksp)/(Kc))^(1//4)` |
|
| 3. |
Write expressions for Boyle temperature and critical temperature in terms of van der Waals constants. Which one is greater for a particular gas ? |
|
Answer» Solution :Boyle temperature, `T_(B)=(a)/(Rb)` Critical temperature, `T_(C )=(8A)/(27" Rb)` Thus, `T_(B) GT T_(C )`. |
|
| 4. |
Which forces are present between H_(2) and HCl molecules ? Why ? |
| Answer» Solution :(i) LONDON forces : Because it is universal. (ii) Dipole - INDUCED Dipole forces : Because HCl is PERMANENTLY polar but`H_(2)` is non polar molecule (Deficiency of permanently dipole). | |
| 5. |
Which noble gas effuses approximately twice are fast as Kr? |
|
Answer» Ne |
|
| 6. |
Which of the following salt act as strong oxidizing agent? |
|
Answer» `TI^(+3)` |
|
| 7. |
Which of the following reactions represents DeltaH_f^@ |
|
Answer» `C_(("diamond")) + O_(2(g)) rarr CO_(2(g))` |
|
| 8. |
What happens when chloroform reacts with oxygen in the presence ot sunlignt ? |
|
Answer» Solution :Chloroform undergoes oxidation in the presence of sunlight and AIR to form phosgene (carboxyl chloride) `underset("Chloroform")(CHCl_3) + 1/2O_2 underset("light")OVERSET("air")to underset("Phosgene")(COCl_2)+HCl` SINCE phosgene is very poisonous, therefore its presence makes chloroform unfit for USE as anaesthetic. |
|
| 9. |
What role does the molecular interaction play in solution of alcohol and water? |
| Answer» Solution :There is strong hydrogen bonding in alcohol molecules as well as water molecules. The intermolecular forces both in alcohol ANS water are H-bonds. When alcohol ans water are mixed. They from solution because of formation of H-bonds between alcohol and `H_(2)O` molecules but these interactions are WEAKER and less EXTENSIVE than those in pure water. Hence, they show IVE DEVIATION from ideal behaviour. | |
| 10. |
The species having bond order different from that in CO is |
|
Answer» `NO^(-)` (containing 14 ELECTRONS each) . Hence , all of tham have the same BOND order , VIZ ., `3 NO^(-)`has 16 elelctrons with the same configuration as that `O_(2)` Hence , its bond order = 2 |
|
| 11. |
Which period contain Na and F elements ? What is order of atomic radius? |
|
Answer» SOLUTION : F and Na elements are present third period. ORDER of radius: `Na gt F` ` (.:. " Na REDIUS of 3rd period" )gt ("F radius of " 2^(nd)"period" ) ` |
|
| 12. |
The science for environmentally favourable synthesis is known as ....... |
|
Answer» BIOCHEMISTRY |
|
| 13. |
What is the concentration of sugar (C_(12)H_(22)O_(11)) in mol L^(-1) if its 20g are dissolved in enough water to make a final volume up to 2L ? |
|
Answer» `m = (12xx12.01) + (22xx1.0079) + (11 xx 16.00)` `= 342.2938 ~=342` g/mole Weight of solute `= 20g` Volume of solution `= 2L` `:.` MOLARITY `= ("Weight of solute")/("Molecular mass of solution" xx "Volume of solution")` `=(20)/(342xx2) = 0.0292M` |
|
| 14. |
What aremajordifferencesbetweenmetals andnon-metals ? |
|
Answer» Solution :ELEMENTS whichhave astrongtendencyto loseelectronsto formcations are calledmetalswhilethose whichhave a strongtendencyto accept electronsto formanions ARECALLED NON- METALS. Thusmetalsare strong reducing agentstheyhave lowionizationenthalpies have lessnegative electrongain ENTHALPIES, low electronegativityformbasicoxides and ioniccompounds. Non- metalson theother hand are strongoxidisingagentsthey havehighionizationenthalpieshave highnegativeelectrongain enthalpieshigh electronegatively, formacidicoxides andcovalentcompounds. |
|
| 15. |
What is vertical columns and horizontal rows known as in periodic table ? What does it signifies ? |
|
Answer» SOLUTION :Horizontal rows known as period it indicates MAXIMUM QUANTUM number. Vertical column known as group. Group CONTAINS same electrons in its. COUNTER most orbital. |
|
| 16. |
What are the salient features of Newland's law of octaves? |
|
Answer» SOLUTION :(i) This law is quite well for lighter elements but not supported to heavier elements. (ii) Elements were arranged in increasing atomic masses without TAKING an account on the properties of elements. (iii) This law was SEEMED to be applicable only for elements upto calcium. |
|
| 17. |
When NaNO_3 is heated in a closed vessel, oxygen is liberated and NaNO_2 is behind. At equilibrium, NaNO_3(s)hArrNaNO_2(g)+(1)/(2)O(g): |
|
Answer» ADDITION of `NaNO_2` favours reverse reaction |
|
| 18. |
What is formal charge ? Explain by example. |
|
Answer» Solution :When molecule or ion presented by Lewis dot structure than the charge on atom is called formal charge. If an atom has MOVE ELECTRON than valence electron than is has negative formal charge and if less electron than valence electron than it has POSITIVE formal charge. Calculation of formal charge is express by following equation. `[{:("Formal"),("charge"),("(F.C) on an"),("atom in"),("a Lewis"),("structure"):}] = [{:("Total"),("number of"),("valence"),("ELECTRONS"),("in the free"),("atom"):}] - [{:("Total"),("number"),("of non"),("bonding"),("(ion pair"),("electrons"):}] - (1)/(2) [{:("total"),("number"),("of"),("bonding"),("(shered"),("elecrtons"):}]` Formal charge is not real charge in molecule but it give position and arrangement of election. e.g. Formal charge of oxygen in ozone molecule.  The atoms have been numbered as 1, 2 and 3. The formal charge on : The CENTRAL O atom marked 1 : = `({:("Velence"),("electron"),("of neutral"),("oxygen"):}) - ({:("non"),("bonding "),("electrons"),("of O - 1"):}) - (1)/(2) ({: ("bonding "),("electrons"),("of O - 2"):})` `= 6 - 2 - (1)/(2) (6) = + 1 ` the end O atom marked 2 : `({:("Velence"),("electron"),("of O"):}) - ({:("non"),("bonding"),("electrons"),("of O (2)"):}) - ({: ("bonding "),("electrons"),("of O (2)"):})` = 6 -4 - 2 = 0 The end O atom marked 3 : Hence, structure of `O_(3)` with formal charge : 
|
|
| 19. |
Which of the following exist together- |
|
Answer» NAOH+NaCl |
|
| 20. |
The volume of perhydrol which on decomposition gives 1.5 lit of O_(2) gas at STP is |
|
Answer» 25 ml 1.5 lit of `O_(2)` __________? = 15 ml |
|
| 21. |
underset(Cl_(2))overset(hv)rarr Number of mono (V) chloro dervatives obtained. underset("distillation")overset("fractional")rarr (X) Number of fractions |
Answer» 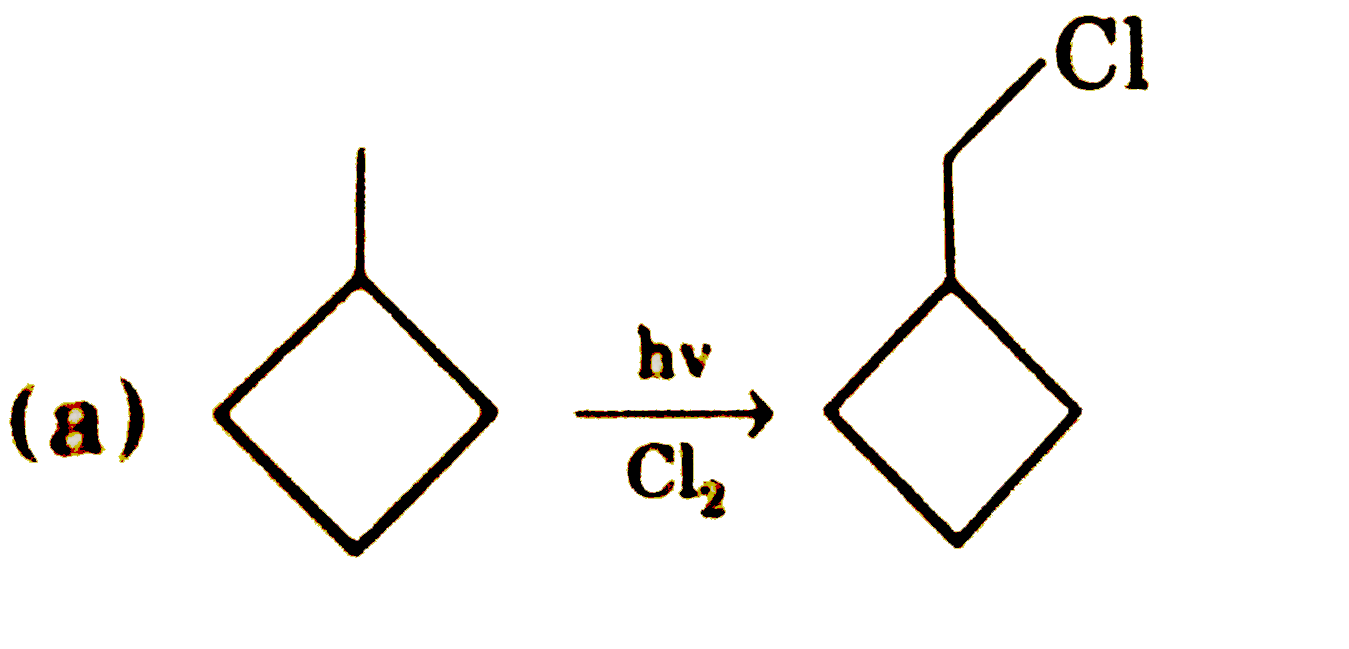 Total number of MONOHALOGEN derivative (V) =8 Total number of fractions (X) = 6 
|
|
| 22. |
The standard EMF of the cell : Ni|Ni^(2+)||Cu^(2+)|Cu is 0.59 volt The standsard elctrode potential (reduction potential of copper electrode is 0.34 volt . Calculate the standsard electrode potential of nickel electrode |
|
Answer» |
|
| 23. |
Two flasks A and B have equal volumes. A is maintained at 300 K and B at 600 K, while A contains H_(2)gas, B has an equal mass of CO_(2)gas. Find the ratio of total K.E. of gases in flask A to that of B. |
|
Answer» `1:2` |
|
| 24. |
The size of the crystals reflect the conditions of growth, rather than the internal constitution of thecrystal. NaCl crystals grown at the bottom of a beaker are squre plates whose thickness is never greater than half-their width. Thus the growth rate in either of the two horizontal orientations is twice that in the vertical direction. The appearance of NaCl crystal grown, suspended in a solution of Urea is |
|
Answer» Cube |
|
| 25. |
The size of the crystals reflect the conditions of growth, rather than the internal constitution of thecrystal. NaCl crystals grown at the bottom of a beaker are squre plates whose thickness is never greater than half-their width. Thus the growth rate in either of the two horizontal orientations is twice that in the vertical direction. The angle between between two characteristic faces of octahedra and cube of NaCl crystal are theta_(1)and theta_(2) then correct relation is |
|
Answer» `theta_(1) gt theta_(2)` |
|
| 26. |
The size of the crystals reflect the conditions of growth, rather than the internal constitution of thecrystal. NaCl crystals grown at the bottom of a beaker are squre plates whose thickness is never greater than half-their width. Thus the growth rate in either of the two horizontal orientations is twice that in the vertical direction. The appearance of NaCl crystal thatare carefully grown by suspending a seed crystal in a slightly uspersaturated solution is |
|
Answer» Cube |
|
| 27. |
Which of the following order of energies ofmolecular orbitals of N_(2)is correct ? |
|
Answer» `(pi2p_(y)) lt (sigma 2p_(z)) lt (pi^(**) 2p_(X)) = (pi^(**)2p_(y))` |
|
| 28. |
Universalindicator shows green colour when pH of the solution is nearly........ |
|
Answer» 4 |
|
| 29. |
Which of the following represent a set of nucleophiles ? |
|
Answer» `BF_(3), H_(2)O, NH^(2-)` |
|
| 30. |
Which one of the following is/are acid slat/s ? |
|
Answer» `NaH_(2)PO_(2)` |
|
| 31. |
The specific gravity of 98% H_(2)SO_(4) is 1.8 g/cc. 50 ml of this solution is mixed with 1750 ml of pure water. Molarity of resulting solution is |
|
Answer» 0.2 M `IMPLIES 18xx50=M_(2)xx1800impliesM_(2)=0.5M` |
|
| 32. |
The volume of oxygen liberated at STP by decomposition of one L hydrogen peroxide solution with a strength of 17g per litre ? |
|
Answer» `1.12`L |
|
| 33. |
What qualitative information can you obtain from the magnitude of equilibrium constant ? |
|
Answer» Solution :(i) Large value of equilibrium constant `(gt 10^(3))` shows that forward reaction is favoured , i.e., concentration of PRODUCTS is MUCH larger than that of the REACTANTS at equilibrium. (II) Inermediate value of `K(10^(-3) "to" 10^(3))` showsthat the concentration of the reactants and products are comparable. (iii) oLow value of `K(lt10^(-3))` shows that backward reaction is favoured , i.e., concentration of reactants is much larger than that of the products. |
|
| 34. |
What is an adiabaticprocess ? |
| Answer» SOLUTION :A PROCESS during which no heat flows betweenthe SYSTEM and the surroundings is CALLED an adiabatic process. | |
| 35. |
Which curve shows Charle's law? |
|
Answer»
|
|
| 36. |
You have a mixture of three liquids A, B and C. There is a targe difference in the boiling points of A and rest of the two liquids i.e., B and C. Boiling point of liquids B and C are quite close. Liquid A boils at a higher temperature than B and C and boiling point B is lower than C. How will you separate the comonents of the mixture. Draw a diagram showing set up of the apparatus for the process |
|
Answer» Solution :Order of boiling point `A gt gt gt B gt C` The boiling point of LIQUID A is much higher than those of liquid B and C whose b.p are quite does therefore separate liquid A by simple distillation using the apparatus shown in figure as under. The b.p. of liquid B and C are quite close but are much lower than that of liquid A, therefore, MIXTURE of liquid B and C will DISTIL together leaving BEHIND liquid A. On further heating liquid A will distil.   Mixture B and C has less difference in their boiling point. So following arrangement is done for SEPARATION of B and C  Now, place the mixture of liquid B and C in a flask fitted with a fractionating column as shown in figures. Since the b.p of liquid B is lower than that of liquid C, therefore, on fractional distillation first liquid B will distil over and the liquid C. |
|
| 37. |
What are the constituents of soil? |
| Answer» SOLUTION :Soil is a THIN LAYER of organic and inorganic material that COVERS the earth.s rocky surface. Soil constitutes the upper crust of the earth, which supports LAND, plants and animals | |
| 38. |
The tetrahalides of carbon do not form complexes, whereas the tetrahalides of the other elements of group 14 do so. Comment on the statement and explain. |
| Answer» Solution :The STATEMENT is TRUE. This is due to the ABSENCE of vacant d-orbitals in C. | |
| 39. |
Which of the following orbitals are not possible? 7s, 2d, 3f and 1p |
| Answer» SOLUTION :2D, 3F and 1P | |
| 40. |
Which is true statement ? |
|
Answer» Nascent HYDROGEN is more POWERFUL oxidant than NORMAL hydrogen |
|
| 41. |
Which among the following alkenes will be most reaction during hydrogenation reaction. |
| Answer» Answer :C | |
| 42. |
What do you understand by the term "non-stoichiometric hydrides" ? Do you expect this type of the hydrides to be formed by alkali metals? Justify your answer. |
|
Answer» SOLUTION :Hydrides which are dificient in hydrogen and in which the ratio of the metal to hydrogen in fractional are called non-stoichiometric hydrides. Furthermore, even this fractional ratio is not fixed but VARIES with the temperature and the pressure . This type of hydrides are formed by d-and f-block elements. In these hydrides , the hydrogen atoms occupy holes in the metal lattice. Usually some holes always REMAIN unoccupiedand hence these metals form non-stoichiometric hydrides. Since ALKALI metals are highly reducing, they lone electron to the H atom , THEREBY forming `H^(-)` ions. In other words, alkali metal hydrides are ionic in which `H^(-)` ions occupy holes in the lattice . Since a hydride ion is formed by complete transfer of an electron, therefore, the ratio of metal to hydrogen is always fixed and hence alkali metals form only stoichiometric hydrides. In other words, alkali metal do not form non-stoichiometric hydrides. |
|
| 43. |
Which of the following oxides are basic ? |
|
Answer» `B_2O_3` |
|
| 45. |
Which of the following is not a characteristic of alcohol? |
|
Answer» They are lighter than water |
|
| 46. |
What indicator should be used for the titration of 1.0M KH_(2)BO_(2) with 1.10M HCI? |
|
Answer» Solution :`H_(2)BO_(3)^(Θ) + H_(3)O^(o+) rarr H_(3)BO^(3) + H_(2)O` At equivalence point, `0.05 M H_(3)BO_(3)` would be produced. Only the first IONISATION step of `H_(3)BO_(3)` is important to the `pH`. `H_(2)BO_(3)^(Θ) + H_(2)O rarr H_(2)BO_(3)^(Θ) + H_(3)O^(o+)` `K_(a) = ([H_(3)O^(o+)][H_(2)BO_(3)^(o+)])/([H_(3)BO_(3)]) =(x^(2))/(0.050) = 7.3 XX 10^(-10)` Thus `x = 6.0 xx 10^(-6)` and `pH = 5.22` `pH 5.22` is in the middle of the range of methy`1` red which would thereofre be SUITABLE. |
|
| 47. |
When alpha-particlesare madeincidenton a thinmetalfoil mostof thempassstraightthroughthe foil because . |
|
Answer» mostpartof THEATOM ISEMPTY |
|
| 48. |
Which points required in resonance structure? |
|
Answer» SOLUTION :TWO or more than two atoms and double bond or triple bond possessing molecular of ion or central atom has tone pair than such molecule possess resonance structure. For all resonance structure is same. Molecules or IONS have same electron so then all are iso electronic. In resonance structure POSITION of atom are same. |
|
| 49. |
Which of the following may show fixed equivalent weights? |
|
Answer» Mg |
|
| 50. |
Which of the following is not an example of disproportionation reaction ? |
|
Answer» `4ClO_(3)^(-)rarr Cl^(-)+3ClO_(4)^(-)` 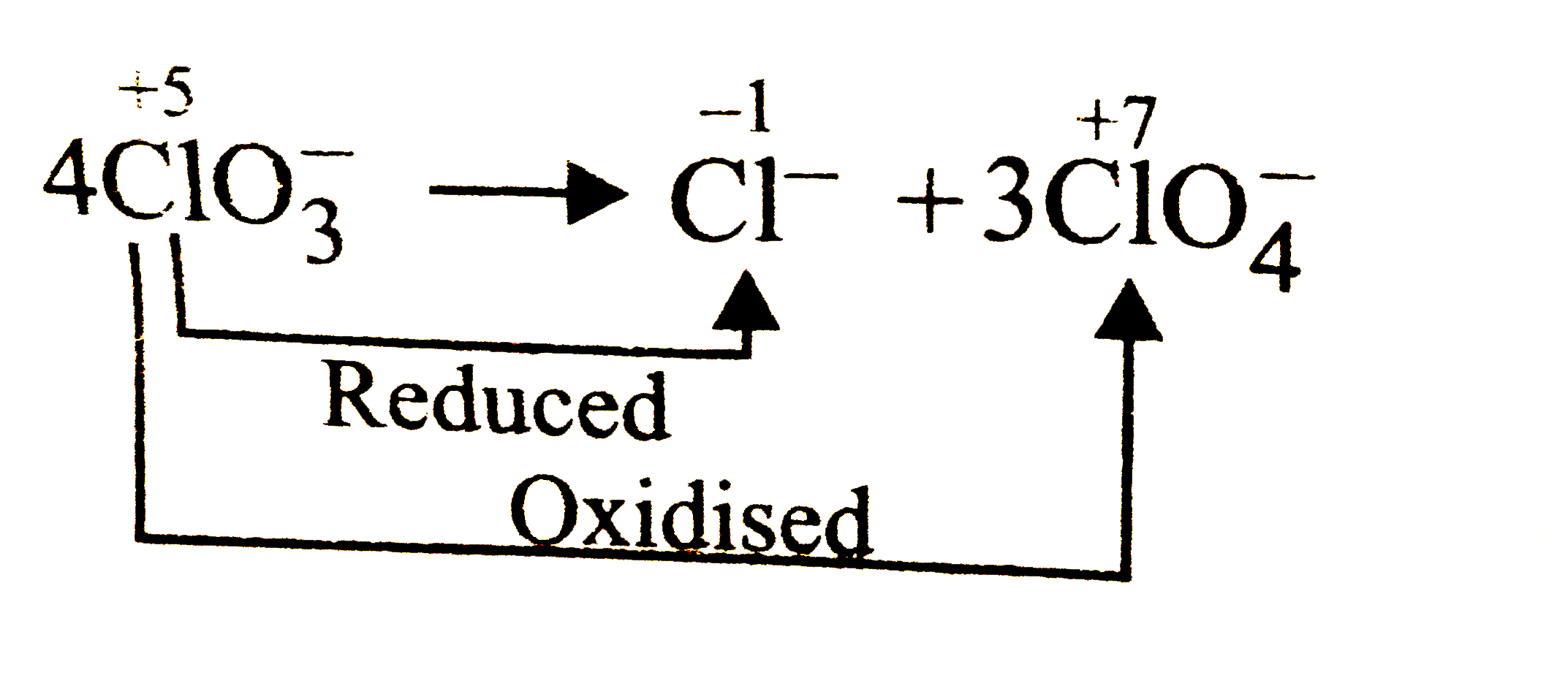 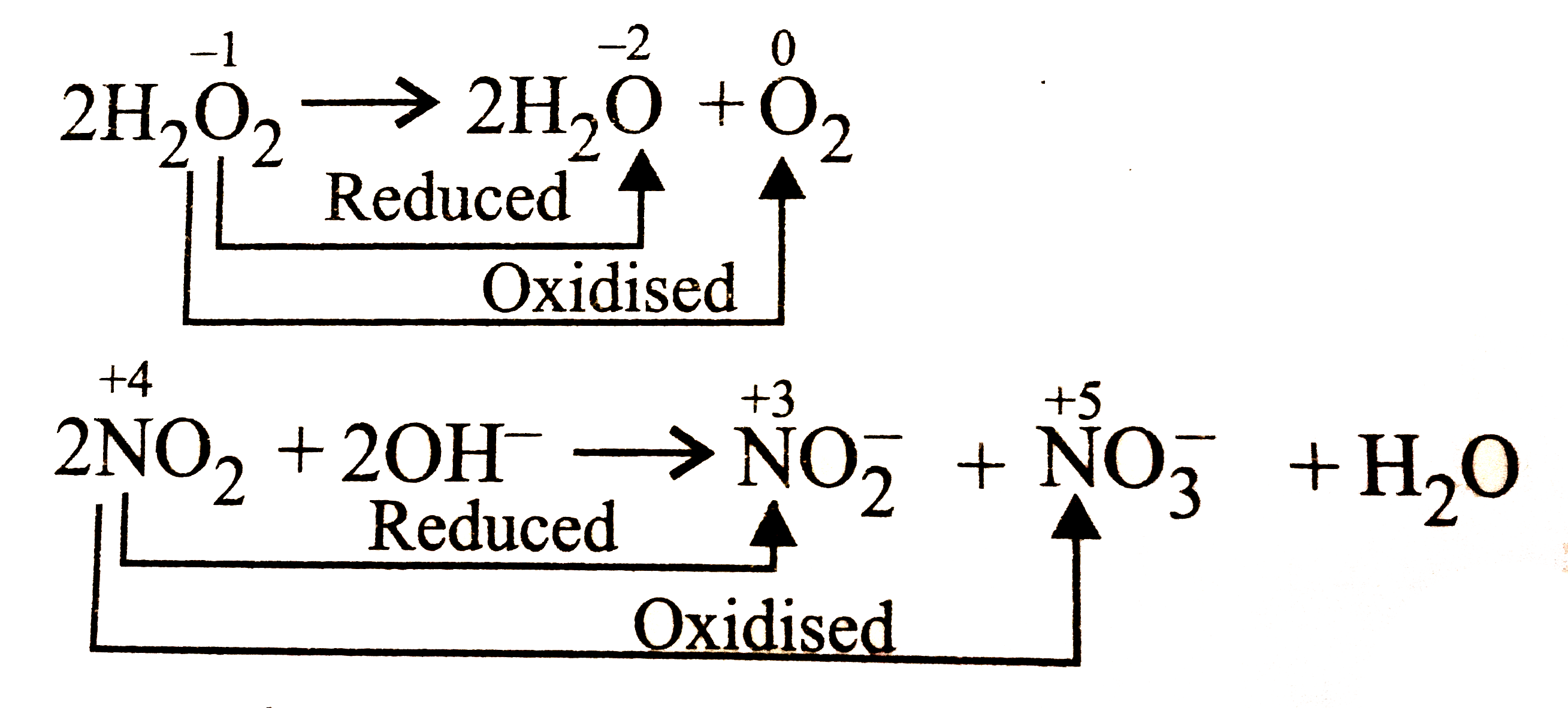 `overset(+4)(Ti)Cl_(4)+2overset(0)(M)grarroverset(0)Ti+2overset(+2)(Mg)Cl_(2)` Displacement reaction |
|