Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
{:(ul"Compound",ul"Ozonolysis products"),("A) Acetylene","1) Formaldehyde and Acetaldehyde"),("B) Ethylene","2) Acetaldehyde"),("C) Benzene","3) One mole of glyoxal"),("D) 2-Butene","4) 3 moles of glyoxal"),(,"5) Formaldehyde"):} The correct match is |
|
Answer» `{:(ULA,ULB,ulC,ULD),(3,2,5,4):}` |
|
| 2. |
Which of the following statements(s) is/are correct regarding the structure of borax ? |
|
Answer» NUMBER of B - B bonds are ZERO 
|
|
| 3. |
What is the total number of orbitals present in the shell with the principal quantum number, n = 3 |
|
Answer» Solution :For n = 3, the possible VALUES of l are 0,1 and 2. Thus, there is one 3S orbital (n = 3, l = 0 and `m_l` = 0), there are three p orbitals (n = 3, l = 1 and ml = -1, 0, 1) there are five 3D orbitals (n = 3, l = 2, ml = -2, -1, 0, 1, 2). Therefore, the total number of orbitals is 1+3+5 = 9. |
|
| 4. |
What is the effect of pressure on (i) visocity, (ii) surface tension,(iii) density of liquid ? |
| Answer» Solution :Increase in PRESSURE increases viscoity, surface tension and density becauseinter molecular FORCE of ATTRACTION increases. | |
| 5. |
What will be the products of electrolysis of AgNO_(3) solution in water with platinum electrodes ? |
|
Answer» Ag is liberatedat cathode and Ag is deposited in anode. |
|
| 6. |
Which of the following reactant can react with benzene ? |
|
Answer» `I_(2)` and sunlight (NORMAL temperature) (B) Cold `KMnO_(4)` reaction is UNSATURATION bayer test. (C) Bromine water or `Br_(2)(Cl_(4))` is also unsaturation test. Benzene has characteristic stability so it does not GIVE unsaturation test (B) and (C). (D) Benzene react with `Br_(2)` in presence of Lewis acid `FeBr_(3)` and form `Br^(+)` which can form bromobenzene from benzene. |
|
| 7. |
What is coefficient of Viscosity ? |
|
Answer» Solution :A force is required of MAINTAIN the flow of layers. This force is proportional to the area of CONTACT of layers and velocity gradient `F = eta A (dy)/(DZ), eta = (F)/(A((dz)/(dz)))` `.eta.` is proportionality constant and is called COEFFICIENT of viscosity. If contact surface area = unit Area (1) and viscosity velocity gradient `((dy)/(dz))=1` then `eta = 1`. Viscocity CO - efficient is the force when velocity gradient is unity and the area of constant is unit area. This `.eta.` is measure of viscocity. |
|
| 8. |
Which one is Charle.s formula ? |
|
Answer» `V_(1)T_(2)=T_(2)V_(1)` |
|
| 9. |
What is tin plague ? |
|
Answer» Solution :Tin exist in three ALLOTROPIC forms, i.e,Prey, white and rhombic, `"Grey" UNDERSET(larr)overset(291K)(rarr)"White"underset(larr)overset(434K)(rarr)"Rhombic"` In cold countries, the conversion of white tin to grey tin occurs. Since grey tin is quite brittle, it starts CRUMBLING down and hence tin gets corroded. This corroding PROCESS of tin is called tin disease or tin PEST or plague. |
|
| 10. |
When ethylene glycol is treated with conc. H_(2)SO_(4) the compound formed is |
|
Answer» ketal 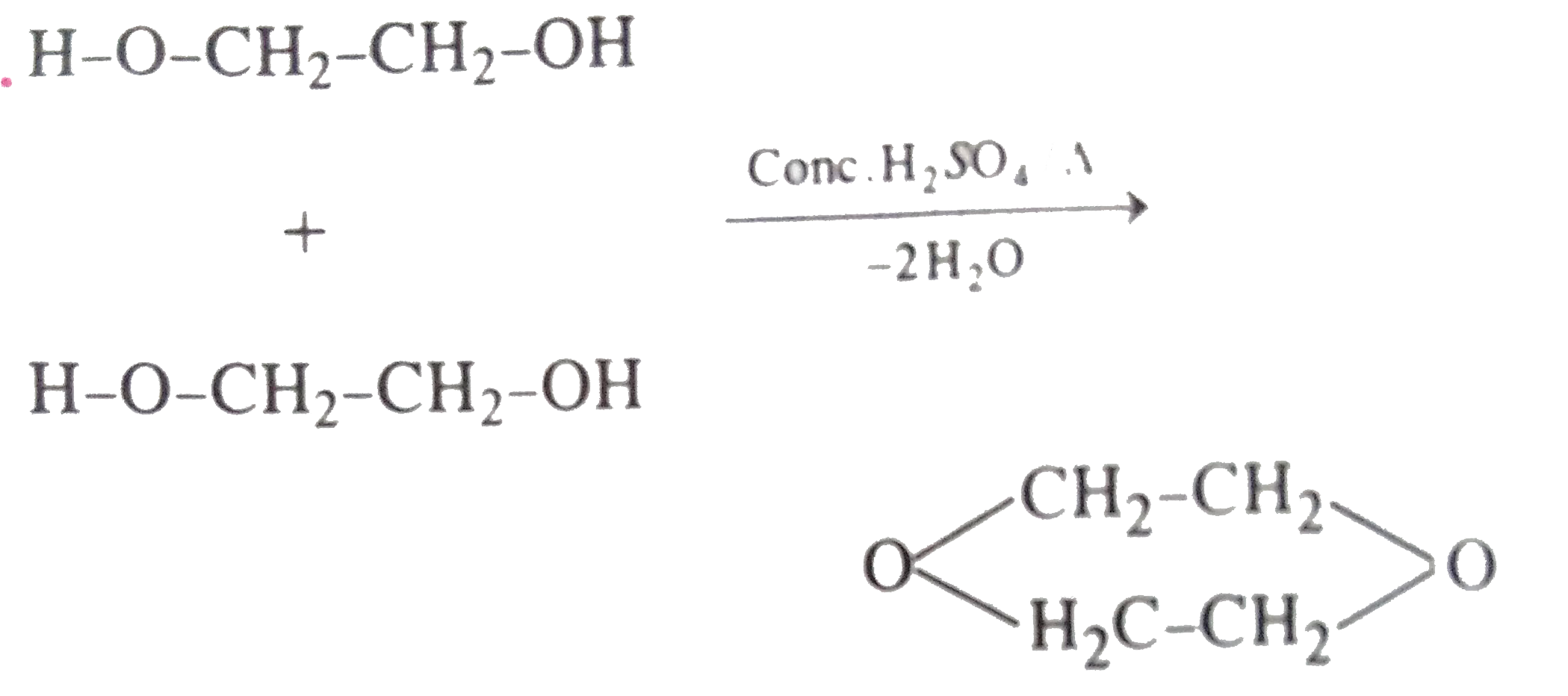
|
|
| 11. |
Writethe complete symbol for theatom with thegivenatomicnumber(z ) andatomicmass ( A) (i) Z= 17 ,A= 35(ii) Z= 92 , A= 233 (iii) Z=4, A=9 |
Answer» SOLUTION :CI forZ= 17u For Z = 92Be for Z= 4
|
|
| 12. |
Which o f the following species is not paramagnetic ? |
|
Answer» NO Total number of electrons in CO = 6 + 8 = 14 THEREFORE molecular ELECTRONIC CONFIGURATION = `sigma 1s^(2), sigma^(**) 1s^(2), sigma 2s^(2), sigma^(**) 2s^(2) (pi 2p_(x)^(2)= pi 2p_(y)^(2)) sigma 2P_(2)^(2)` Therefore all the electrons are paired, So it is diamagnetic rest of all have minimum ONE electron is unpaired |
|
| 13. |
Write the IUPAC names of the following compounds. (i) CH_(3)-underset(OH)underset(|)(CH)-CH_(2)-CH=CH-CH_(3) (ii) CH_(3)-CH_(2)-underset(CH_(3))underset(|)(CH)-CH_(2)-overset(C_(2)H_(5))overset(|)(CH)-CH_(2)-CH_(3) |
|
Answer» SOLUTION :(i) Her-4-ene-2-ol (II) 3-ethyl-5-methyl leptane |
|
| 14. |
The relation between pH, pK_(a) and concentration c of the solution of a weak acid is ........... |
|
Answer» |
|
| 15. |
What are the molecular mass and molar mass, if each molecule of a substance contains 9 carbon and 13 hydrogen atom? |
|
Answer» |
|
| 16. |
What is the total number of ions present in one unit cell of sodium chloride lattice ? |
|
Answer» 2 |
|
| 17. |
What are transuranium elements? |
| Answer» Solution :In PERIODIC table the ELEMENTS PLACED BEYOND uranium (Z=92) are all synthetic elements and are called as transuranium elements. | |
| 18. |
What happens when acetyl chloride is treated with excess of CH_3Mgl? |
Answer» Solution :When acetyl chloride is TREATED with excess of Grignard reagent, the PRODUCT formed is TERTIARY butyl ALCOHOL. 
|
|
| 19. |
What is physical equilibrium ? |
| Answer» Solution :The TYPE of equilibrium in which only PHYSICAL state CHANGE but the COMPOSITION of system does not change is called physical equilibrium. | |
| 20. |
Which reaction is not feasible? |
|
Answer» `HCequivCH+LirarrLi^(+)OVERSET(-)Cequivoverset(-)CLi^(+)+H_(2)` (b) `2NaNO_(3)overset(Delta)rarr2NaNO_(2)+O_(2)` |
|
| 21. |
What will be the minimum pressure required to compress 500 dm^(3) of air at 1 bar to 200 dm^(3)" at "30^(@)C. |
| Answer» SOLUTION :`2.5` BAR | |
| 22. |
Which of the following is a roleof Ca^(+2) in biology |
|
Answer» `Ca^(+2)` ions are necessaray for blood CLOTHING |
|
| 23. |
What happens when water is added to ethene and propene? |
|
Answer» Solution :When WATER is added to ETHENE and propene in presence of sulphuric acid gives alcohols, ACCORDING to Markownikoff RULE. `underset("ethane")(CH_(2) = CH_(2)) + underset("water")(H_(2)O)rarr underset("ethanol(ethyl alcohol)")(CH_(3) - CH_(2)) - OH` `CH_(3) - underset("propene")(CH = CH_(2)) + underset("water")(H_(2)O) rarr CH_3 - underset("Propan - 2-01[Isopropy alcohol]")underset(OH)underset(|)(CH) - CH_(3)` |
|
| 24. |
Which of the following represent a set of nuclephiles ? |
|
Answer» ` BF_(3), H_(2) O, NH^(2-)` |
|
| 25. |
What is value of standard hydrogen potential ? |
| Answer» SOLUTION :VALUE of standard HYDROGEN potential is ZERO. | |
| 26. |
Which contains more number of molecules? |
|
Answer» 1 mole of CARBON dioxide |
|
| 27. |
Which of the following is correctly matched |
|
Answer» `{:("Element",,"Calcium in flame TEST"),(Ca,HARR,"Apple green"):}` `Na harr` Golden yellow spanges |
|
| 28. |
What is [Ag^(+) ]in a solution made by dissolving both Ag_2 CrO_4 and Ag_2C_2O_4 until saturation is reached with respect to both salts ?[K_(sp)(Ag_2C_2O_4) =2xx 10 ^(-11), K_(sp)(Ag_2CrO_4) - 2xx10 ^(-12))] |
|
Answer» ` 2.80 xx 10^(-4)` ` "" 2X+2Y"" x` ` Ag_2 C_2O_4hArr 2Ag^(+) C_2O_4^(-2)` ` "" 2y+2x"" y` ` (K_(s_(p1)))/(K_(s_(p2)))=(x)/(y)=(2xx 10^(-12))/(2xx 10^(-11)) =0.1` ` 2xx 10 ^(-11) =(2x + 2y)^(2) .y` ` 2 xx 10^(-11)=4.89 y^(3) ` ` "" y= 1.6 xx 10^(4)rArr x= 0.16 xx 10^(-14) ` Total ` [AG^(+) ]=2x +2y =3.52 xx 10 ^(-4)` |
|
| 29. |
what is the need for second Law of thermodynamics. |
|
Answer» Solution :(i) The second law of THERMODYNAMICS helps us to predict whether the reaction is feasible or not (ii) It tells about the direction of the flow of heat. (iii) It ALSO tells that energy cannot be COMPLETELY converted into equivalent WORK. |
|
| 30. |
What do you mean by strength and reactivity of bond ? |
| Answer» SOLUTION :Bond STRENGTH MEANS ENERGY REQUIRED to break the bond or bond formation enthalpy. Reactivity means easiness of reaction by broken down one bond. | |
| 31. |
xCr^(+3) + yH_(2)O_(2)+mOH^(-)rarr zCO_(4)^(-2) +nH_(2)O In the above balance the equation , what is the value of x+y+z is : |
|
Answer» |
|
| 32. |
The species formed in the depletion of ozone layer by chlorofluorocarbons in free radial mechanism is |
|
Answer» `CLO^(*)` |
|
| 33. |
What do you mean by non-stoichiometric hydrides? |
| Answer» Solution :Metallic hydrides whose compositions do not CORRESPOND to SIMPLE whole number ratio are CALLED non-stoichiometric hydrides. Hence alkali metals form only stoichiometric. | |
| 34. |
Which of the following is not a periodic property ? |
|
Answer» Valency |
|
| 35. |
Writenumberof nucleonsof the following (i) ._(17) ^(35) CI (ii) ._(19)^(40) K |
| Answer» SOLUTION :(i) 35 (II) (40) | |
| 36. |
What is the molality of a solution containing 200 mg of urea (molar mass = 60 g mol^(-1)) ? |
|
Answer» 0.0825 Mass of water in kg `=(40)/(1000)=0.04kg` Molality of solution `= (("0.0033 mol"))/(("0.04 kg"))` `= 0.0825 "mol Kg"^(-1)=0.0825`m . |
|
| 37. |
While filling electrons in pi2p x and pi 2py the electronic configuration rules that one to be followed is |
|
Answer» PAULI's EXCLUSION PRINCIPLE |
|
| 38. |
Unlike sodium carbonate , potassium carbonate cannot be prepared by Solvay ammonia process . Why is it so ? |
| Answer» Solution :The reason being that unlike sodium bicarbonate `(NaHCO_(3))` which is SPARINGLY soluble in water , potassium bicarbonate `(KHCO_(3))` is FAIRLY soluble in water . Thus , when `CO_(2)` is passed through AMMONICAL brine , `NaHCO_(3)` gets precipitated while `KHCO_(3)` does not precipitated when `CO_(2)` is passed through and ammoniacal solution of KCL . Hence , `K_(2)CO_(3)` cannot be prepared by Solvay-ammonia process . | |
| 39. |
The stability of +1 oxidation state in aqueous solution is in the order- AlIn>Tl Tl>In>Ga>Al Al>Tl>Ga>In Tl>Al>Ga>In |
|
Answer» `AL LT GA gt "In gt Tl` |
|
| 40. |
What is shape of the orbital with (ii) n = 2 and I = 1? |
| Answer» Solution :`n = 2 and I = 1`. The orbital is 2P. Its shape is DUMB bell. | |
| 41. |
The reagent with which both acetaldehyde and acetone react easily is |
|
Answer» TOLLEN's reagent |
|
| 42. |
Write the electronic configuration of the elements given below and predict their period, group, block and also name the class, i.e., representative, transition etc. to which they belong. ""_(74)E |
|
Answer» SOLUTION :`""_(74)E=[""_(54)Xe], 4f^(14) 5d^(4) 6S^(2)` Period=6, Block =d, Group =4+2=6 CLASS =Transition element |
|
| 43. |
What is dissociation constant of a weak acid (K_(a)). |
|
Answer» Solution :DISSOCIATION constant of a weak ACID is the ratio of the product of molar CONCENTRATIONS of the products of ionisation to the molar CONCENTRATION of the un-ionised acid medium. For a reaction `HA+H_(2)OhArrH_(3)O^(+)+A^(-)` `K_(a)=([H_(3)O^(+)][A^(-)])/([HA])`. |
|
| 44. |
Write the net ionic equation for the reaction of potassium dichromate (VI), K_(2)Cr_(2)O_(7) with sodium sulphite, Na_(2)SO_(3), in an acid solution to give chromium (III) ion and the sulphate ion. |
|
Answer» Solution :Step-1 : The skeletal ionic equation is : `Cr_(2)O_(7(aq))^(2-)+SO_(3(aq))^(2-)toCr_((aq))^(3+)+SO_(4(aq))^(2-)` Step-2 : Assign oxidation numbers for Cr and S. `OVERSET(+6,-2)(Cr_(2)O_(7(aq))^(2-))+overset(+4,-2)(SO_(3(aq))^(2-))tooverset(+3)(Cr_((aq))^(3+))+overset(+6,-2)(SO_(4(aq))^(2-))` This indicates that the dichromate ion is the oxidant and the sulphite ion is the reductant. Step-3 : Calculate the increase and decrease of oxidation number, and make them equal : `overset(+6,-2)(Cr_(2)O_(7(aq))^(2-))+overset(+4,-2)(3SO_(3(aq))^(2-))tooverset(+3)(2Cr_((aq))^(3+))+overset(+6,-2)(3SO_(4(aq))^(2-))` Step-4 : As the reaction occurs in the acidic medium, and further the ionic charges are not equal on both the sides, add `8H^(+)` on the left to make ionic charges equal `Cr_(2)O_(7(aq))^(2-)+3SO_(3(aq))^(2-)+8H^(+)to2Cr_((aq))^(3+)+3SO_(4(aq))^(2-)` Step-5 : Finally, count the hydrogen atoms, and add appropriate number of water molecules (i.e., `4H_(2)O`) on the right to achieve balanced REDOX change. `Cr_(2)O_(7(aq))^(2-)+3SO_(3(aq))^(2-)+8H_((aq))^(+)to2Cr_((aq))^(3+)+3SO_(4(aq))^(2-)+4H_(2)O_((L))` |
|
| 45. |
What is the difference between heats of reaction at constant volume and constant pressure for the reaction N_(2(g)) + O_(2(g)) rarr 2NO_((g)) |
|
Answer» |
|
| 46. |
Which method can be used to find out strength of reductant/oxidant in a solution ? Explain with an example. |
|
Answer» Solution :STANDARD half cell potential connected with hydrogen ELECTRODE then measure of completely cell potential `(E_("cell")^(@))`. If value of `E_("cell")^(@)` is positive obtained then half cell acts as reducing agent if value of `E_("cell")^(@)` is negative then half cell as an oxidising agent. Using of this type of method calculate half cell potential of another cell. Then calculate strength of oxidising agent and reducing agent. Ex. We can calculate standard electrode potential of `Zn^(+2)//Zn` connected with hydrogen half cell.  GIVEN, `E_("cell")^(@)=0.76"volt"` `E_("cell")^(@)=0.76=E_("cathode")^(@)-E_("anode")^(@)` `0.76=0-E_("anode")^(@)` `E_("anode")^(@)=-0.76"volt"` `E_(Zn^(+2)//Zn)^(@)=-0.76"volt"` |
|
| 47. |
Which of the following species is not paramagnetic ? |
|
Answer» `O_(2)` ` KK sigma (2S)^(2) sigma^(**)(2s)^(2)sigma (2p_(z))^(2) pi (2p_(X))^(2) pi(2p_(y))^(2)` `pi^(**) (2p_(x))^(1) pi^(**) (2p_(y))^(1) ` (paramagnetic) `B_(2) = 10 ` electrons. M. O. configuration is `KK sigma (2s)^(2) sigma^(**)(2s)^(2) pi (2p_(x))^(1) pi(2p_(y))^(1)` (paramagnetic) NO = 15 electrons .M.O. configuration is `KK sigma (2s)^(2) sigma^(**)(2s)^(2) sigma (2p_(z))^(2) pi (2p_(x))^(2) pi (2p_(y))^(2)` `pi^(**)(2p_(x))^(1) ` CO = 14 electrons . M.O. configuration is `KK sigma (2s)^(2) pi(2p_(x))^(2)pi(2p_(y))^(2) sigma (2p_(z))^(2) sigma ^(**) (2s)^(2)` (diamagnetic) |
|
| 48. |
The thermal dissociation equilibrium of CaCO_(3) (s) is studied under different conditions CaCO_(3) (s) hArr CaO(s) + CO_(2) (g) For this equilibrium , the correct statement (s) is (are) |
|
Answer» `DELTAH ` is dependent on T Equilibrium constant of a reactiondependsonly on temperature and is independent of the amount of `CaCO_(3)` Hence, (B) is correct . For the given reaction , ` K = p_(CO_(2))`at givenThus, K is independent of pressure . Hence , ( c) is incorrect. |
|
| 49. |
Which oxide is formed when potassium is heated in e3xcess of oxygen? |
|
Answer» `K_(2)O` |
|
| 50. |
using the data give below findout the strongest reducing agent E_(Cr_(2)O_(7)^(2-))//Cr^(3+)^(@)=1.33 V ,E_(Ci_(2)//CI^(@))=1.36 V E_(MnO_(4)^(-)//Mn^(2+)=1.51 V) , E_(Cr^(3+)//Cr)^(@)=-0.74 V |
|
Answer» `CI^(-)` |
|