Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which one of the following is the weakest base? |
|
Answer» `(C_(2)H_(5))_(3)N` |
|
| 2. |
The total number of contributing structures showing hyperconjugation (involving C-H bonds) for the carbocation given on R.H.S. is |
Answer» 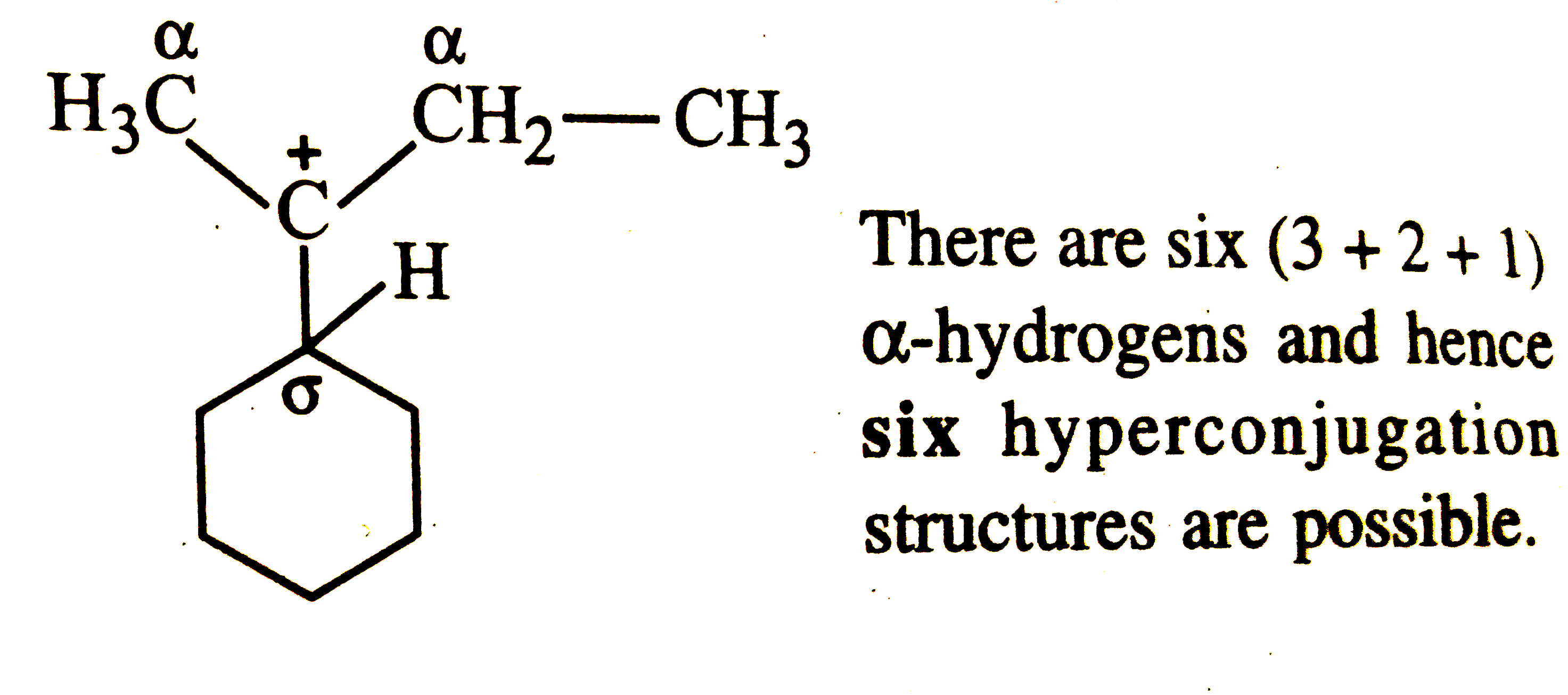 There are six (3+2+1) `ALPHA`-HYDROGENS and HENCE six hyperconjugation structures are POSSIBLE. |
|
| 4. |
Which statement is not correct about alcohol? |
|
Answer» Ethyl alcohol is heavier than water |
|
| 5. |
Which of the following metal oxides is anti-ferromagnetic in nature ? |
|
Answer» `MnO_(2)` |
|
| 6. |
What is ideal pressure and volume of real gas ? |
|
Answer» Solution :`(("Ideal GAS or"),("Real gas"))=(("Practically"),("measured"),("pressure"))+(("Correction in"),("pressure due"),("to attraction"))` `p_("Ideal")=p_("real")+(an^(2))/(V^(2))` `(("Ideal volume"),("of real gas"))=(("Practically"),("volume of"),("real gas"))-(("Correction"),("of"),("volume"))` `V_("Ideal")=(V_("Real")-NB)` |
|
| 7. |
Which of the following will have zero dipole moment? |
|
Answer» 1, 1 -DICHLOROETHYLENE |
|
| 8. |
Which of the following may not be present in all galvanic cells |
|
Answer» ELECTROLYTE |
|
| 9. |
Which compound is used for the manufacture of chemicals like sodium perborate and percarbonate which are used in high quality detergents. |
|
Answer» `H_(2)S` |
|
| 10. |
Value of R is depend upon ? What is its unit ? |
| Answer» Solution :Ideal GAS eq. pV = nRT `therefore R=(PV)/(NT)rArr` the VALUE of R depends upon UNITS in which p, V, n and T are MEASURED. | |
| 11. |
Which of the following have P-O-P bond ? |
|
Answer» HYPO PHOSPHOROUS acid |
|
| 12. |
What values of magnetic quantum number, m are permitted for an electron having angular momentum quantum number value, l = 2? |
| Answer» SOLUTION :`-2, -1, 0, +1, +2` | |
| 13. |
Which is correct statement about X and Y. |
|
Answer» X is product of ionic REACTION and Y is product of RADICAL reaction |
|
| 14. |
The Vividh Bharati station of All India Radio, Delhi, broadcasts on a frequency of 1,368 kHz (kilo hertz). Calculate the wavelength of the electromagnetic radiation emitted by transmitter. Which part of the electromagnetic spectrum does it belong to? |
| Answer» Solution :The wavelength, `lambda`, is equal to C/V, where c is the speed of electromagnetic radiation in vacuum and V is the frequency. Substituting the GIVEN VALUES, we have `V=c/V=(3.00xx10^8ms^(-1))/(1368 kHz)=(3.00xx10^8ms^-1)/(1368xx10^3s^(-1))=219.3m`It belongs to the radiowave region. | |
| 15. |
X,Y and Zare three members of a Doboreiner's triad . If the atomic mass of X is 7 and that of Z is 39 , what in the atomic mass of Y? |
|
Answer» 23 |
|
| 16. |
The stability order of the following anions : |
|
Answer» `IV GT III gt II gt I` |
|
| 17. |
Under similar conditions, what is the ratio of RMS velocity of ozone molecules and most probable velocity of oxygen molecules? |
|
Answer» Solution :RMS velocity of `O_3`molecules = ` SQRT((3 RT)/(M ) = sqrt((3RT)/(48))` Mostprobablevelocityof `O_2` molecules= ` sqrt((2 RT)/(M))= sqrt((2RT)/(32))` RATIOOF thevelocities`= sqrt((3 )/( 48 )) : sqrt((2 )/(32)) = 1:1` |
|
| 18. |
Which one is not lyophobic in nature ? |
|
Answer» STARCH SOL. |
|
| 19. |
Write strcutural formulas of the following compounds : (i) 3,4,4,5-Tetramethylheptane (ii) 2,5-Dimethylhexane |
|
Answer» Solution :(i) 3,4,4,5-Tetramethylheptane DETAILED structure as follows : `overset(1)CH_(3)-overset(2)CH_(2)-overset(CH_(3))overset(|)(""^(3)CH)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(""^(4)C)-overset(CH_(3))overset(|)(""^(5)C)-overset(6)CH_(2)-overset(7)CH_(3)` (II) 2,5-Dimethylhexane detailed structure as follows : `overset(1)CH_(3)-overset(CH_(3))overset(|)(""^(2)CH)-overset(3)(CH_(2))-overset(4)(CH_(2))-overset(CH_(3))overset(|)(""^(5)CH)-overset(6)CH_(3)` Or 
|
|
| 20. |
Which of the following is present in maximum amount in acid rain? |
|
Answer» `HNO_3` |
|
| 21. |
What is the oxidation number of nitrogen in (a) nitric acid (b) nitrous acid (c) nitric oxide (d) nitrous oxide (e) ammonia (f) N_2 ? |
|
Answer» |
|
| 22. |
Which of thefollowingremainsunchangedon descending agroupin theperiodictable ? |
|
Answer» Valenceelectrons |
|
| 23. |
Which postulation provide the basic for the modern concepts regarding ion formation by electron transfer and the formation opf ionic crystalline compounds? |
|
Answer» KOSSEL's postulations |
|
| 24. |
Why alkali metals are used as coolant in nuclear reactor ? |
|
Answer» Due to CONDUCTOR of heat |
|
| 25. |
Which is set of the following reagents would you select to convert C_(6)H_(5)COCH_(3) (acetophenone) to the following alcohol C_(6)H_(5)-underset(OH)underset(|)overset(CH_(3))overset(|)(C)-overset(Me)overset(|)(CH)-Me |
|
Answer» `CH_(3)CH_(2)CH_(2)MGBR` and ACID hydrolysis 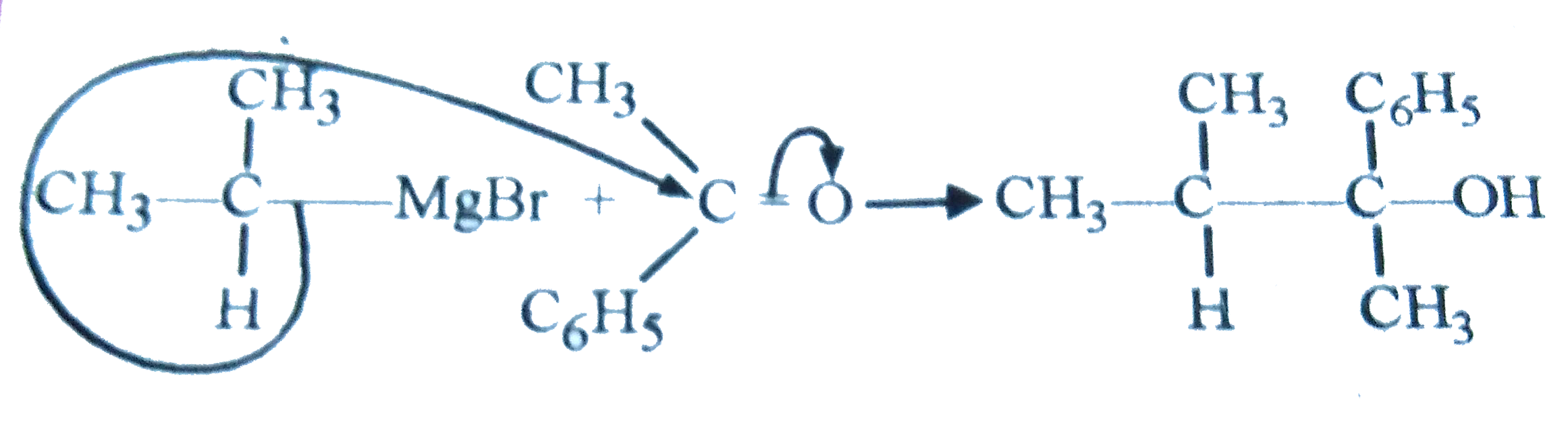
|
|
| 26. |
Which of the following reagents can not be used to distinguished between phenol and benzyl alcohol? |
|
Answer» `FeCl_(3)` |
|
| 27. |
When 10 ml of 5M H_2O_2aqueous solution is decomposed at the S.T.P. volume of oxygen obtained is |
|
Answer» 56ml |
|
| 28. |
Whichof the followinghas highestvalueof ionicradius ? |
| Answer» Solution :NA | |
| 29. |
Using electron transfer concept identify the oxidant and redcutant in the following redox reaction (a) Zn(s)+2H^(+)(sq)rarrZn^(2)(aq)+H_(2)(g) (b)2[Fe(CN_(3))]^(4-)(aq)+H_(2)O_(2)(aq)+2H^(+)(aq)rarr 2[Fe(CN)_(6)]^(3-)-(aq)+2H_(2)O(l) (c )2[Fe(CN)_(6)]^(3-)(aq)+2OH^(-)(aq)+H_(2)O(aq)rarr2[Fe(CN)_(6)]^(4)+(aq)+O_(2)+2 H_(2)O(l) (d) BrO_(3)^(-)(aq) +F_(2)(g)+2 OH^(-)+2 OH^(-)(aq) rarr BrO_(4)^(-)(aq)+H_(2)O(l) (e )2NaCI_(3)(Aq)+L_(2)(aq)rarr2NaIO_(3)rarr 2NaIO_(3)(aq)+CI_(2)(aq) |
|
Answer» SOLUTION : (a)`H^(+) (B)H_(2)O_(2) (c )[Fe(CN)]_(6)^(3-)(d)F_(2) (E )NaCIO_(3)` Reductants : (a) `Zn(b)[Fe(CN)]_(6)^(4-) (c )H_(2)O_(2)(d) BrO_(3)^(-) (e )I_(2)` |
|
| 30. |
What are the effects of photochemical smog? |
|
Answer» Solution :(i) The three main components of photochemical smog are NITROGEN oxide, ozone and oxidised hydrocarbons like `HCHO, CH_2 = CH-CHO` and PAN. (ii) Photochemical smog causes irritation to eyes, skin and lungs. It also increases the chance of asthma. (iii) High concentration of ozone and NO can cause nose and THROAT iritation, chest pain, difliculty in breathing (iv) PAN is toxie to plants, attack younger leaves and cause BRONZING and GLAZING of their surfaces. (V) It causes corrosion of metals, stones, building materials and painted surfaces. |
|
| 31. |
Write Lewis symbol for the following atoms and ions : S and S^(2-),""Al and Al^(3+),""H and H^(-) |
| Answer» Solution :`:overset(..)UNDERSET(.)S.""overset(..)underset(..)S:^(2-),"".overset(.)Al. and Al^(3+),""overset(.)H and overset(..)H^(-)` | |
| 32. |
Write down the products of ozonolysis of 1,2-dimethylbenzene (o-xylene) . How does the result support Kekule structure of benzene ? |
Answer» SOLUTION :o-Xylene may be regarded as a resonance hybrid of the following two kekule structures. Ozonolysis of each one of these GIVES two productsas SHOWN below : 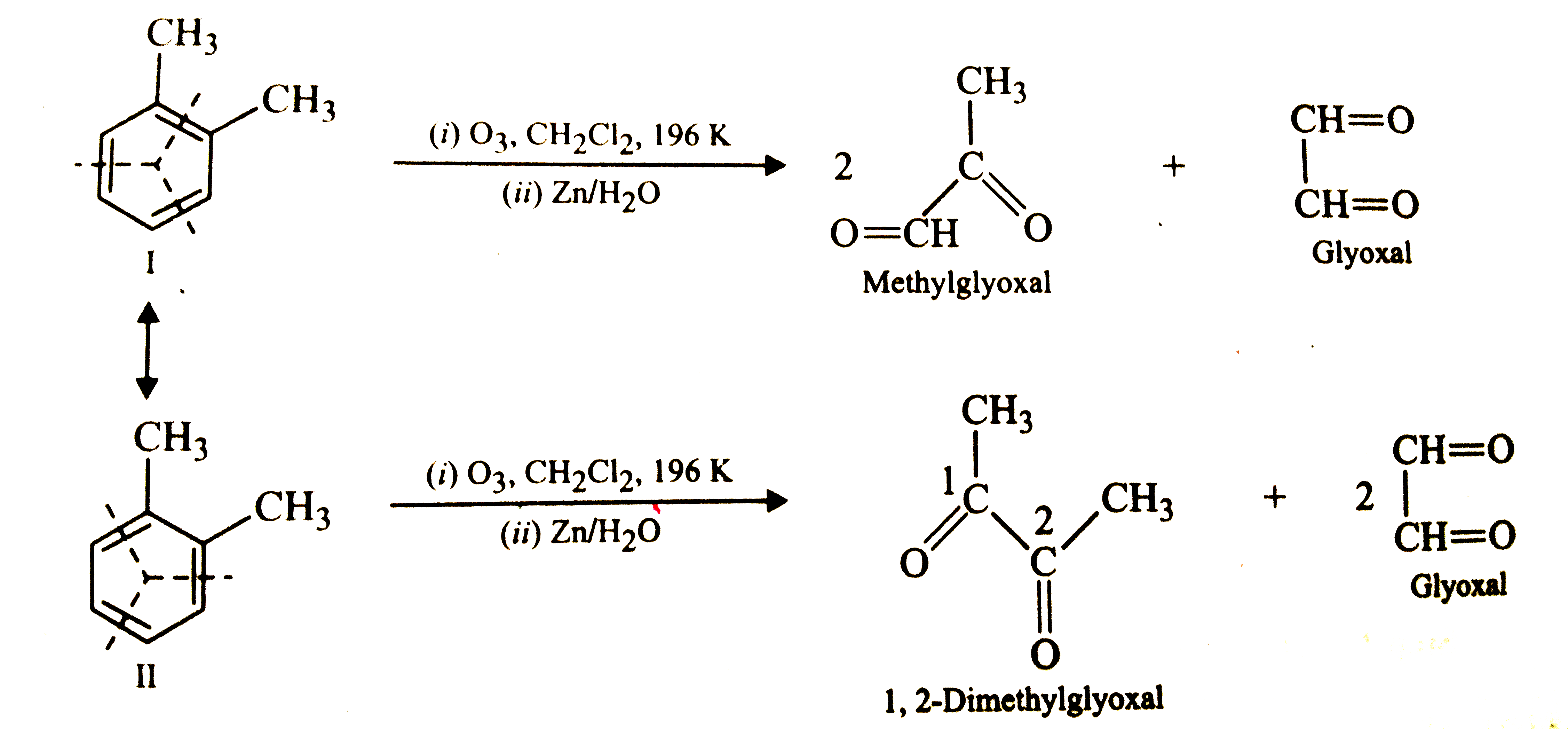 Thus, in all, THREE PRODUCTS are formed . Since, all the three products cannot be obtained from any one of the two Kekule structures, this shows that o-xylene is a resonance hybrid of the two Kekule structures (I and II) |
|
| 33. |
Total pressure observed after opening the stopcock is [neglecting the volume of the tube connecting the two bulbs] |
|
Answer» 1.33 ATM `(0.1 xx 2)/(RT) + (0.2 xx 1)/(RT) = (P xx 0.1)/(RT) + (P xx 0.2)/(RT)` `implies P = (0.4)/(0.3) = 1.33 atm.` |
|
| 34. |
Which of the following set of elements follows Dobereiner's law of triads ? |
| Answer» Answer :B | |
| 35. |
Which statement is correct about the critical point of a phase diagram? |
|
Answer» SOLID, LIQUID and gas are present in EQUILIBRIUM. |
|
| 36. |
Which of the following is the condition for a non spontaneous reaction at high temperature but spontaneous at low temperature ? |
|
Answer» `{:(DELTAH, DELTAS),(-,-):}` |
|
| 37. |
Which of the following options represents the correct bond order ? |
|
Answer» `O_(2)^(-) gt O_(2) gt O_(2)^(+)` According to MOLECULAR ORBITAL theory (MOT) ` ""O_(2)^(-) "" O_(2) "" O_(2)^(+)` Number of `E^(-) " :171615 " ` Bond order:1.522.5 |
|
| 38. |
What are the reacting conditions used for the formation of ethylene glycol from ethylene? |
|
Answer» Bromine water |
|
| 39. |
Zn_((s)) + Cu_((aq))^(2+) to Zn_((aq))^(2+) + Cu_((s)). In this reaction, which gets oxidised? |
|
Answer» `CU^(2+)` Zn GETS oxidised |
|
| 40. |
Which is expected to have highest melting point: PH_3 , NH_3 , (CH_3)_3,N? Explain. |
| Answer» Solution :`NH_(3)` has the strongest intermolecular hydrogen bonding and it is expected to have the heighest melting point. Acutal melting points are: `NH_(3)=-77.7^(@)C,PH_(3)=-133^(2)C` and `(CH_(3))_(3)N=-117^(@)C`. | |
| 41. |
Write IUPAC names of the products obtained by addition reaction of HBr to hex-1-ene (i) in the absence of peroxide and (ii) in the presence of peroxide. |
Answer» SOLUTION :
|
|
| 42. |
What is meant by hctero (polar) covalent bond? |
| Answer» Solution :In a COVALENT BOND if the PAIR of electrons are shared UNEQUALLY (asymmetrically) between that ATOMS are called polar covalent bond. | |
| 43. |
underset((A)"major product" overset(HBr)(to)(B)"major product")underset(darrH^(+)//heat)(CH_(3)-CH(CH_(3))-CH(OH)-CH_(3)) Identify A and B |
Answer» Solution :`CH_(3)-underset(CH_(3))underset(|)(C) H-underset(OH)underset(|)(C )H-CH_(3)OVERSET(H^(+))underset(Delta)(to)`  `CH_(3)-underset(CH_(3))underset(|)(C )H-CH=CH_(2)` 
|
|
| 44. |
Whichof thefollowingmetalis usedtoremovesulphurfrompetroleum? |
|
Answer» BERYLLIUM |
|
| 45. |
Which of the following compounds may give blood red colouration while performing Lassaigne's test for nitrogen? |
|
Answer» `(NH_(2))_(2)CO` |
|
| 46. |
Which quantum number does not follow from the solution of Schrodinger wave equation ? |
| Answer» SOLUTION :SPIN QUANTUM NUMBER | |
| 47. |
Which of the following pair will diffuse at the same rate? |
|
Answer» `CO_2` and `N_2O` |
|
| 48. |
What will happen if LiAlH_(4) is added to an ester ? |
|
Answer» Two units of alcohol are obtained |
|
| 49. |
Which of the following hydroxide is partially soluble in water ? |
|
Answer» LIOH |
|
| 50. |
What indicated by following figure ? |
|
Answer» <P> Solution :As per FIG equilibrium is established between water and vapour. It is physical equilibrium `H_2O_((l)) hArr H_2O_((g))`at constant TEMPERATURE and volume `P_(H_2O)` = difference of height of mercury in MANOMETER. |
|