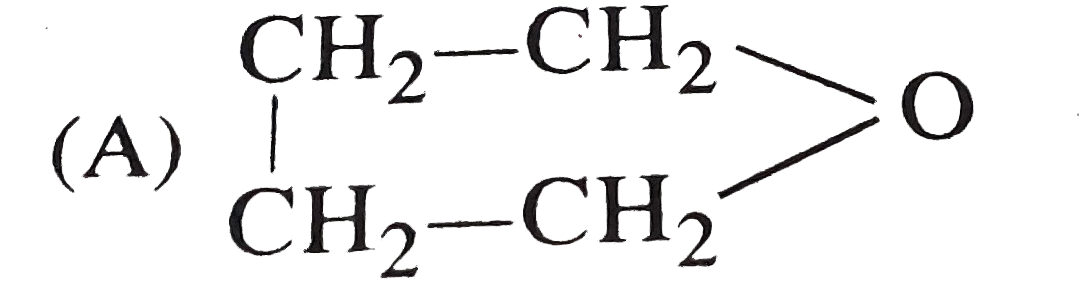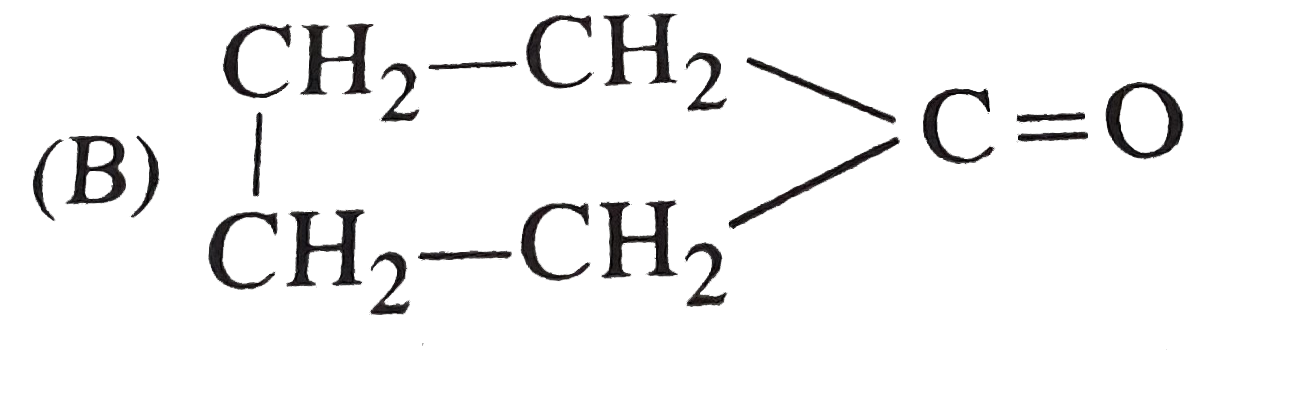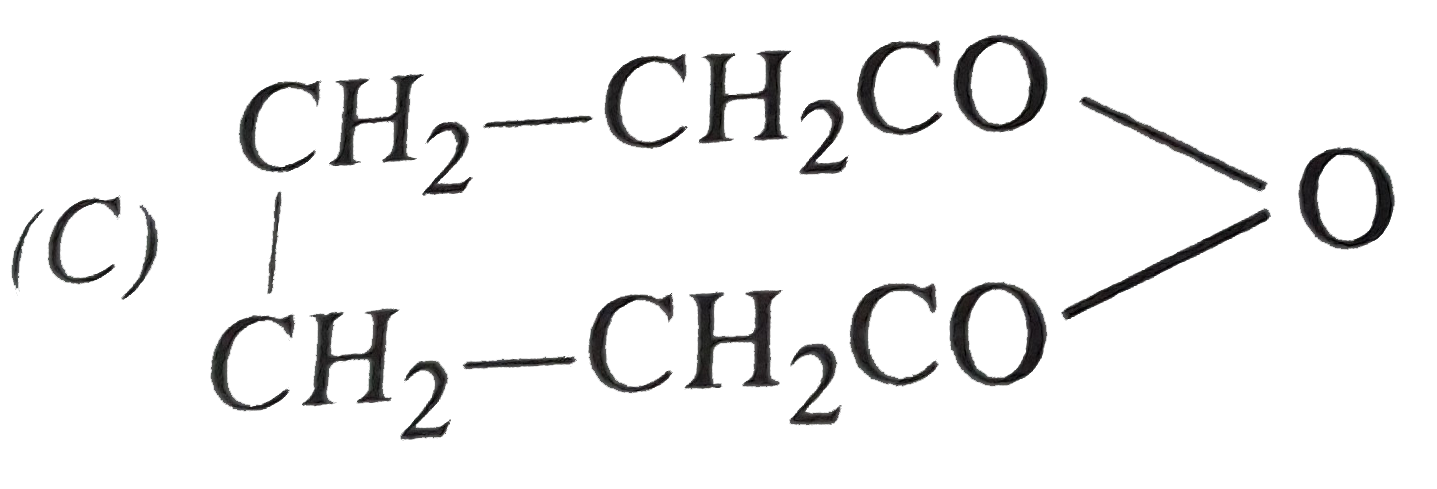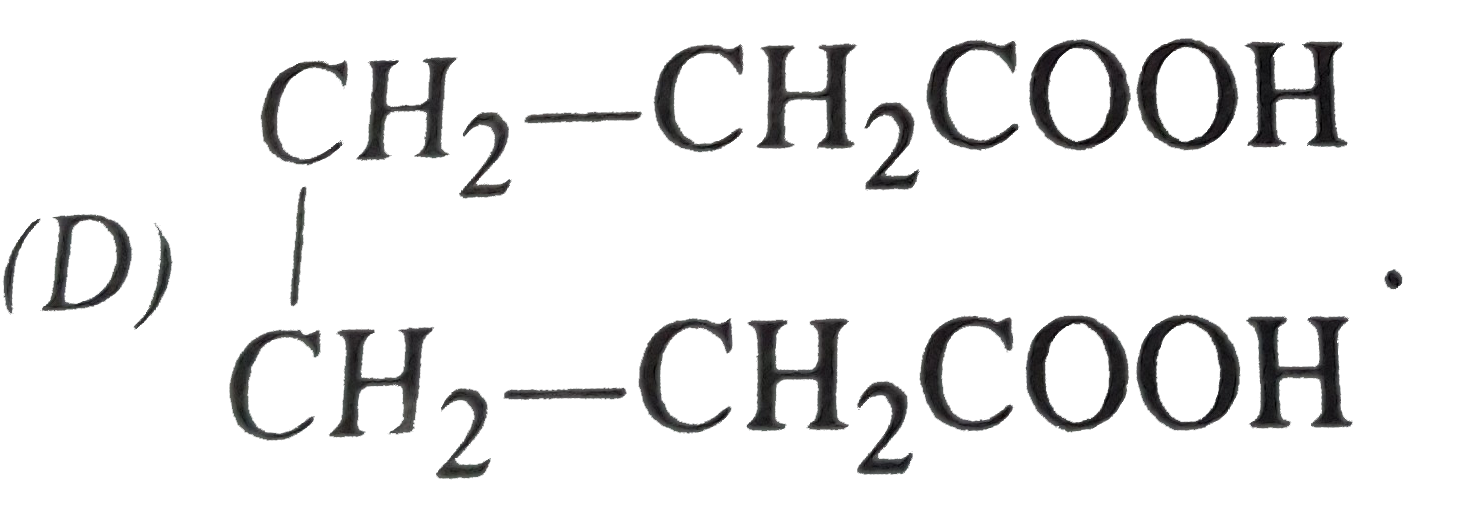Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
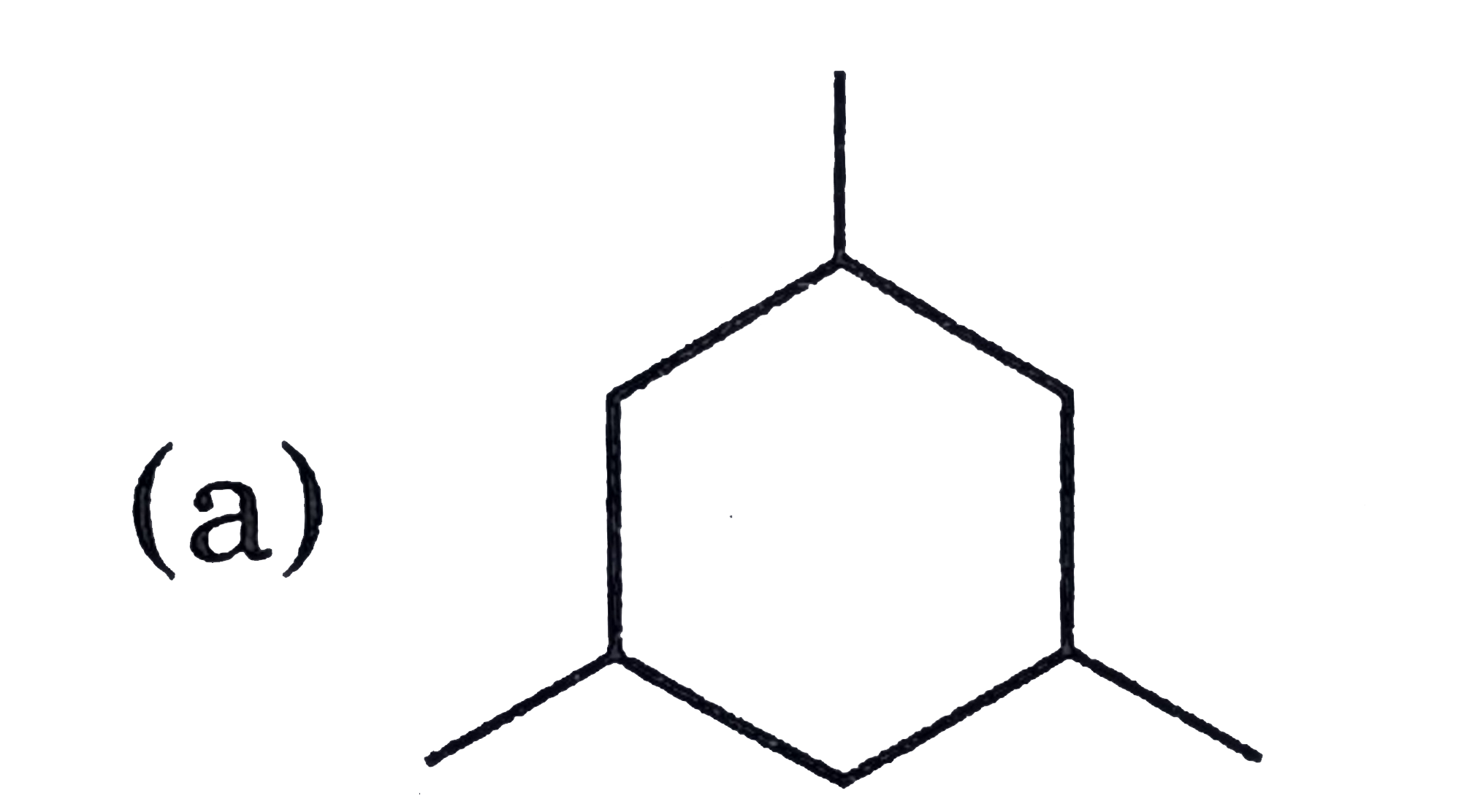
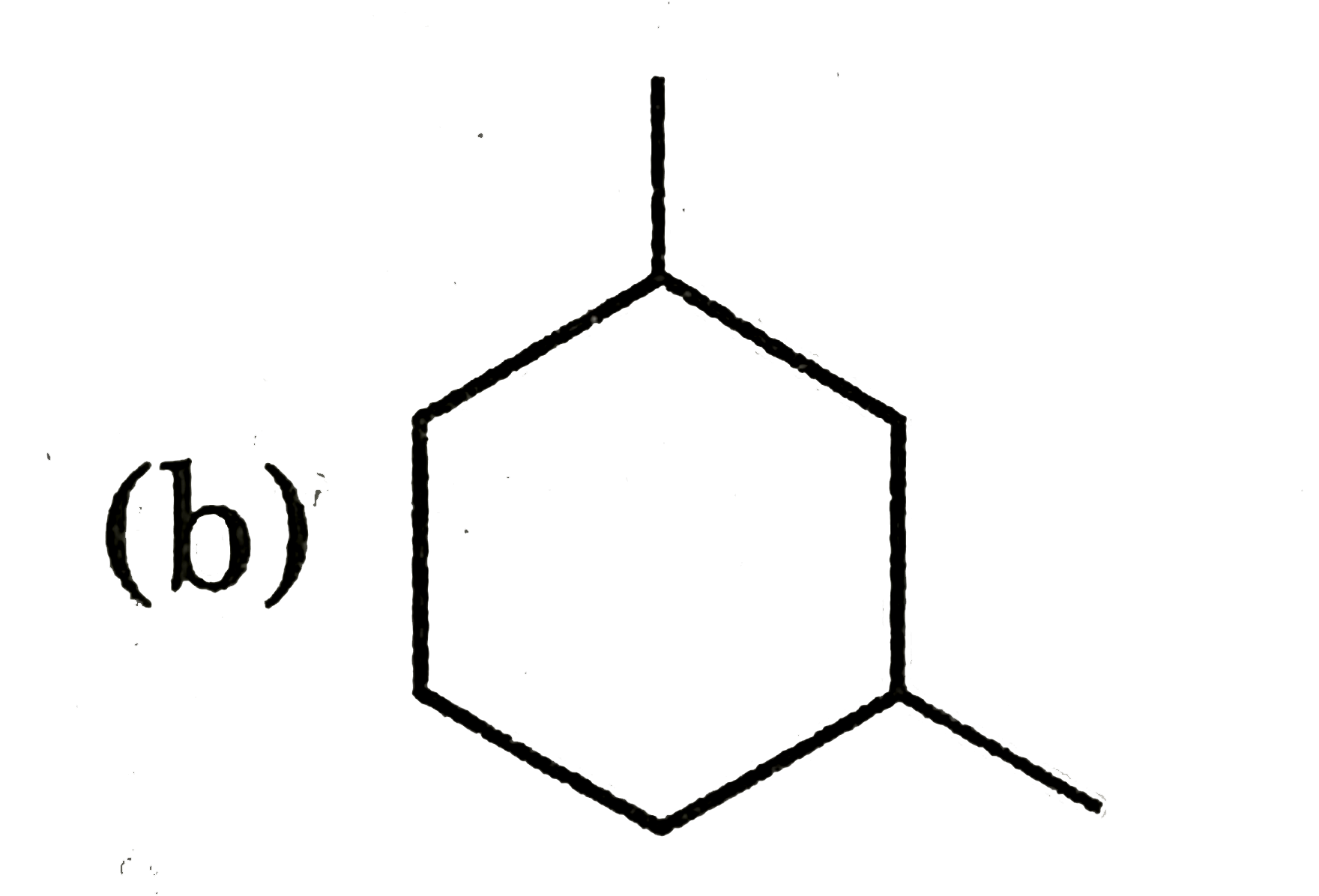
What is true and false for the ethene from the following ? (i) Ethene molecule is three dimensional (ii) Ethene is planar (iii) Ethene has four hydrogen (iv) Ethene has sp carbon. |
|
Answer» |
|
| 2. |
Trans-2 Butene has higher melting point than cis-2-butene, why? |
| Answer» Solution :Because more CLOSE packing of molecules is possible in trans-isomer due to more SYMMETRY. | |
| 3. |
What is the pH of a 0.05 M solution of formic acid? (K_(a)=1.8xx10^(-4)) |
|
Answer» Solution :`[H^(+)]=SQRT(K_(a)*C)=sqrt(1.8xx10^(-4)xx0.05)=3.00xx10^(-3)"mol"dm^(-3)` `pH=-log[H^(+)]=-log(3.0xx10^(-3))=2.52`. |
|
| 4. |
Which of the following is like copper in electrical conductivity and appearance ? |
|
Answer» Silver |
|
| 5. |
Which of the following compound will show optical as well as geometrical isomerism? |
|
Answer»
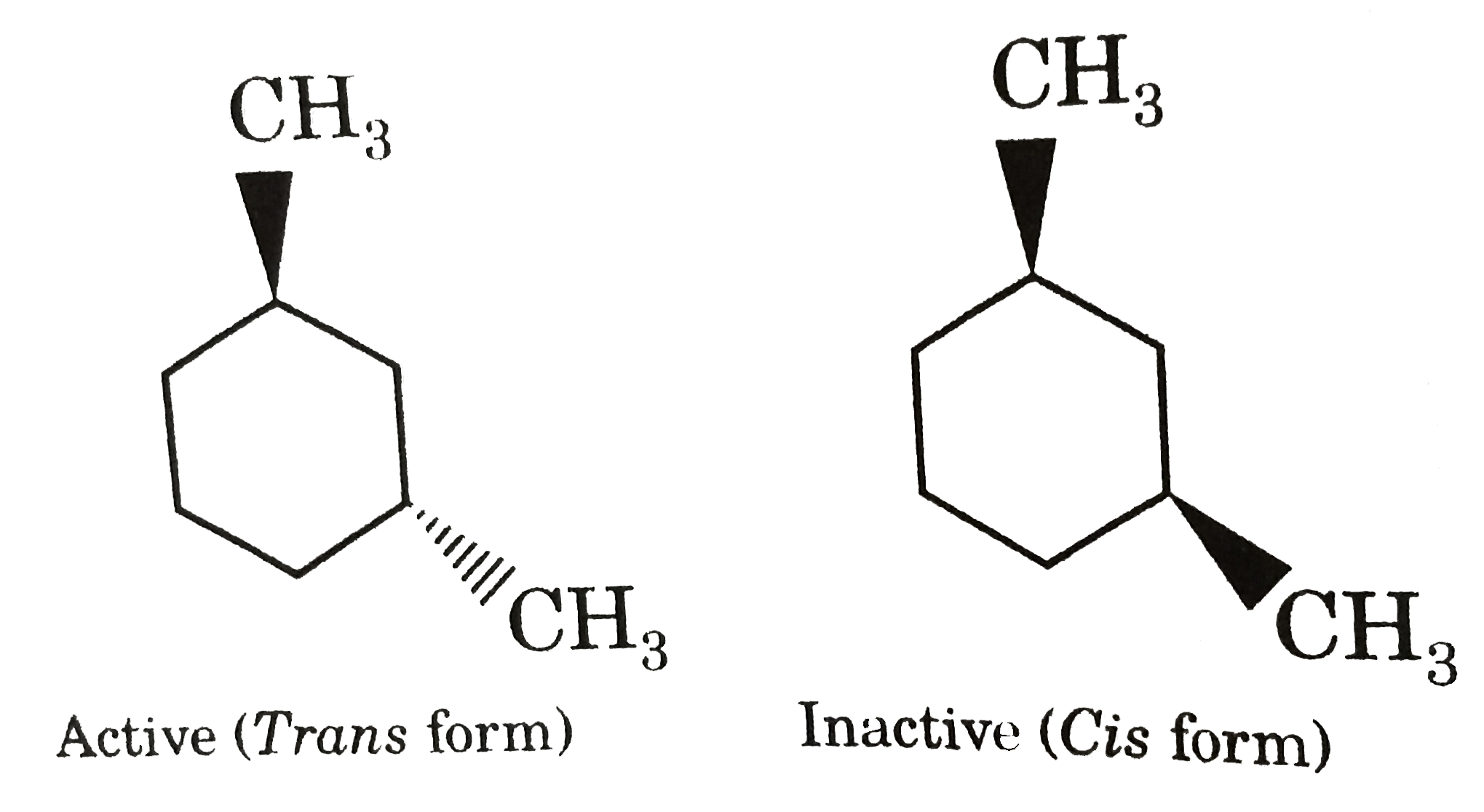
|
|
| 6. |
What is the equlivalent weight of methane during its Combustion? |
|
Answer» EQ. wt of `CH_(4)=(16)/(8)=2` |
|
| 7. |
What is the mass of H_2O_2 present in 1 litre of a 2M solution ? Calculate the volume of oxygen at S.T.P. liberated upon the complete decomposition of 100 cm^3 of the above solution. |
| Answer» SOLUTION :68 G , 2240 `cm^2` | |
| 8. |
What happen when diborane reacts with Lewis bases? |
|
Answer» It FORMS boron TRIHYDRIDE `(BH_(3))` due to cleavage. or `B_(2)H_(6) + 2NMe_(3) to 2NH_(3). Nme_(3)` |
|
| 9. |
Which halide has maxmum density ? |
|
Answer» `C_(3)H_(7)I`<BR>`C_(2)H_(5)I` |
|
| 10. |
Which of the followingrepresents thecorrectorderof increasingelectrongain enthalpywithnegativesign for aelement O, S , F and C1 ? |
|
Answer» `S lt O lt C1 lt F` |
|
| 11. |
Which of the following crystal does not exhibit Frenkel defect ? |
|
Answer» AgBr |
|
| 12. |
The term used to express the active ingredients present in therapeutics is …………. |
| Answer» SOLUTION :PERCENTAGE UNITS | |
| 13. |
The simplest formula of a compound containing 50% of element A (at.wt=10) and 50% of element B (at. wt = 20) is |
|
Answer» `AB` |
|
| 14. |
What different methods are used for softening the hard water ? Explain the principle used in each method ? (Write any two) |
|
Answer» Solution :Hard water can be softened by the following methods depending upon the nature of hardness. (a) Temporary hardness : (i) By boiling. It can be removed by merely boiling the water. Boiling decomposes the bicarbonates to give carbon DIOXIDE and insoluble carbonates, which can be removed by filtratoin. `Ca(HCO_(3))_(3)overset("heat")tounderset("white ppt.")(CaCO_(3))+H_(2)O+CO_(2)` `Mg(HCO_(3))_(2)overset("heat")tounderset("white ppt.")(MgCO_(3))+H_(2)O+CO_(2)` II. Clark's process : Temporary hardness can be removed by the addition of a calculated amount of lime, where magnesium carbonate or calcium carbonate is precipated. `Ca(HCO_(3))_(2)+Ca(OH)_(2)to2CaCO_(3)+2H_(2)O` `Mg(HCO_(3))_(2)+Ca(OH)_(2)toCaCO_(3)+MgCO_(3)+2H_(2)O` b. Permanent hardness : With sodium carbonate. On treatment with washing soda, `Ca^(2+)andMg^(2+)` in hard water are precipitated. The precipitate of the insoluble carbonates thus formed is removed by filtration. `Ca^(2+)+CO_(3)^(2-)tounderset("white ppt")(CaCO_(3)),Mg^(2+)+CO_(3)^(2-)toMgCO_(3)` The anion may remain in solution but in this form it is not dangerous. ii. Ion-exchange methode. The common SUBSTANCES used for this process is zeolite which is hydrated sodium aluminium silicate, `NaA1(SiO_(2))_(2).` The exchange occurs when on PASSING over the zeolite bed, sodium ions from zeolite are replaced by calcium and magnesium ions. Thus `2NaZe+Ca^(2+)to(Ze)_(2)+2Na^(+),2NaZe+Mg^(2+)to(Ze)_(2)Mg+2Na^(+)` When all the sodium ions of zeolite has replace, the zeolite is said to be EXHAUSTED. It can be regenerated by treatment with strong solution of sodium chloride. `2Na+(Ze)_(2)Cato2ZeNa+Ca^(2+)` |
|
| 15. |
Which of the following species has same number of unpaired electrons?(1) Cr^(3+) (2) Mn^(2+) (3) Fe^(3+) (4) Cu^(2+) |
| Answer» SOLUTION :(1) and (4) | |
| 16. |
What is the relationship between the standard oxidation potential and the standard reduction potential? |
| Answer» SOLUTION :Both are EQUAL in MAGNITUDE but OPPOSITE in SIGN. | |
| 17. |
Which of the following represents the correct IUPAC name for the compounds concerned? 2-Chloro-4-methylpentane or 4-Chloro 2-methylpentane |
|
Answer» |
|
| 18. |
When sulphur in the form of S_8 is beated at 900K, the initial pressure of 1 atm falls by 29% at equilibrium. This is because of conversion of S_8 to S_2. Calculate the equilibrium constant for the reaction. |
|
Answer» SINCE at equilibrium `alpha`=29%=0.29 `therefore p_(S_8)=1-alpha` =1-0.29=0.71 `p_(S_2)=4alpha` =4 x 0.29=1.16 `K_p=([p_(S_3)]^4)/p_(S_8)=(1.16)^4/0.71 = "2.55 ATM"^3` |
|
| 19. |
What type of hybridization does carbon undergo in diamond and graphite ? |
| Answer» SOLUTION :In DIAMOND `SP^(3)`, in graphite `sp^(2)`. | |
| 20. |
What is the effect of increasing pressure on the equilibrium of the equilibrium N_(2) + 3H_(2) hArr 2NH_(3)? |
| Answer» Solution :On INCREASING pressure, equilibrium shifts in the direction in which pressure decreases, i.e., having lesser number of moles, HENCE, equilibrium will SHIFT in the forward direction FORMING more of ammonia. | |
| 23. |
What is slaked lime? |
|
Answer» Solution :When SMALL water is added to calcium oxide make HISSING sound and produced large amount of heat which CONVENTS water into steam. This process is called slaking of lime and fine powder obtained is called slaked lime `CaO + to H_(2) O to underset(("slaked lime"))(Ca(OH)_(2))` |
|
| 24. |
The standed enthalpies of combustion of formaldehyde , H_(2)C=O(g), and formic acid HCOOH(l), are -563 and -270 Kj.mol^(-1) respectively . What Is DeltaH^(@) for the following reaction ? H_(2)C=O(g)+(1)/(2)O_(2)(g)to HCOOH(l) |
|
Answer» `-833KJ.mol^(-1)` |
|
| 25. |
Write Lewis symbols for the following atoms and ions S and S^(2-), Al and Al^(3+), H and H^(+) |
Answer» SOLUTION :
|
|
| 26. |
Which one is source of NO_x ? |
|
Answer» PHOTOSYNTHESIS |
|
| 27. |
Which of the following products is formed when adipic acid is heated : |
|
Answer»

|
|
| 28. |
What is free energy ? |
| Answer» SOLUTION :FREE ENERGY of a system is its CAPACITY to do usefulwork or mathmetically, `G= H -TS`. | |
| 29. |
Which one of the following is a reversible reaction? |
|
Answer» Ripening of a banana |
|
| 30. |
Whichof the following is paramagnetic ? |
|
Answer» CO |
|
| 31. |
This quantity is the energy associated with a chemical that can be used to do work is |
|
Answer» entropy |
|
| 32. |
What is the pH of the resulting solution when equal volumes of 0.1 MNaOH and 0.01 M HCl are mixed? |
|
Answer» `7.0` 100ML of 0.1 M NaOH contain NaOH `=100xx0.1` MMOL = 10 mmol `100 mL` of0.01 M HCl contain HCl `=100xx0.01` mmol = 1 mmol 1 mmol of HCl will neutralize 1 mmol of NaOH `:.` NaOH LEFT unneutralized in the solution = 9 mmol Volume of solution after mixing = 200 mL `:.` Molarityof NaOH in the final solution `=(9)/(200) M = 0.045M ` `pOH = -log (0.045) = 1.35` `pH = 14-1.35 = 12.65` |
|
| 33. |
Wrong match is |
|
Answer» K-density LESS than sodium |
|
| 34. |
What are the possible values of I w hen n = 4 |
|
Answer» 0,1,2,3 |
|
| 35. |
which is not soluble in CH_(3)COOH ? |
|
Answer» `CaC_(2)O_(4)` |
|
| 36. |
What is the effect of catalyst on composition of system and the value of K ? |
| Answer» SOLUTION :No EFFECT | |
| 37. |
The vapour density of a gas is 35.5. The volume occupied by 3.55 g of the gas at S.T.P is? |
|
Answer» 1.12 LITRES |
|
| 39. |
Which of the following products is obtain when ethene react with hydrogen in presence of raney nikel at 390 K ? |
|
Answer» Ethane |
|
| 40. |
Write two main differences between nascent hydrogen and atomic hydrogen. |
|
Answer» Solution :Nascent HYDROGEN can never be isolated, but ATOMIC hydrogen can be isolated. Nascent hydrogen can be PRODUCED at room TEMPERATURE, but atomic hydrogen is produced only at elevated temperature. |
|
| 41. |
Xenon crystallises in the face-centred cubic lattice and the edge of the unit cell is 620 pm. What is the nearest neighbour distance and what is the nearest neighbour distance and what is the radius of xenon atom ? |
|
Answer» SOLUTION :Here, a=620 PM , d=? , R=? For the face-centred CUBIC `d=a/sqrt2=620/1.414`=438.5 pm , `r=d/2=438.5/2` =219.25 pm |
|
| 42. |
Which of the following can act as sink for CO_(2)and SO_(2) |
| Answer» ANSWER :C | |
| 43. |
Which of the following is not an ore of lithium ? |
|
Answer» Spodumine |
|
| 44. |
Three moles of helium gas at one atmosphere are compressed reversibly and isothermally at 127^(@)C to 5 atmospheres. Calculate the work done. |
|
Answer» <P> Solution :`T = 127^(@)C = 127 + 273 = 400 K, P_(1) = 1` atm, `P_(2) = 5` atm`R = 8.314 J//K mol, n = 3 "mole"` `w = -2.303 nRT LOG ((P_(1))/(P_(2))) = -2.303 xx 3 xx 8.314 xx 400 xx log ((1)/(5))` `= -2.303 xx 3 xx 8.134 xx 400 xx BAR(1).3010` `= -2.303 xx 3 xx 8.314 xx 400 xx (-0.6990) = +16,060 "joule" = +16.06 kJ` |
|
| 45. |
Whichcatalystis usedin thebrominationofalkanes? |
|
Answer» `Cr_(2) O_(3)` |
|
| 46. |
The value of K_(p) for the reaction at 27^(@)C Br_(2)(l)+CI_(2)(g)hArr2BrCI(g) is 1atm. At equilibrium in a closed container partial pressure of BrCI gas 0.1atm and at this temperature the vapour pressure of Br_(2)(l) is also 0.1atm. Then what will be minimum moles of Br_(2)(l) to be added to 1 mole of CI_(2), initially, to get above equilibrium stiuation, |
|
Answer» `(10)/(6) MOL es` `t=0 1 0` `(1-x) 2x` `K_(p)=((P_(BrCl)^(2)))/(P_(Cl_(2)))=1 "so", P_(Cl_(2))=(P_(BrCl))^(2))=0.01atm` then at equilibrium, `(n_(BrCl))/(n_(Cl_(2))=(0.1)/(0.01)=10=(2x)/(1-x)` So, `10-10x=2x "or" x=(10)/(12)=(5)/(6)mol es` Mole of `Br_(2)(l)` required for maintaining VAPOUR PRESSURE of `01atm` `=2xx(5)/(6)mol es=(10)/(6)mol es="moles of" BrCl(g)`. Moles required for taking part in reaction`="moles of" Cl_(2)` used up`=(5)/(6)mol es`. Hence TOTAL moles required`=(5)/(6)+(10)/(6)=(15)/(6)mol es`. |
|
| 47. |
Which of the following has highest first ionisation potential? |
|
Answer» Al |
|
| 48. |
Which of the alkali metal is having least melting point? (a) Na (b) K (c ) Rb (d) Cs |
|
Answer» Solution : Atomic size increases as we move down the alkali group. As a result, the BINDING energies of their atoms in the crystal lattice decrease. Also, the strength of metallic bonds DECREASES on MOVING down a group in the periodic table. This CAUSES a decrease in the melting point. Among the given metals, Cs is the LARGEST and has the least melting point. |
|
| 49. |
Which of the following has maximum unpaired d-electrons? |
|
Answer» `Zn^+` |
|
| 50. |
Write the difference between Homogeneous equilibrium and Heterogeneous equilibrium. |
Answer» SOLUTION :
|
|