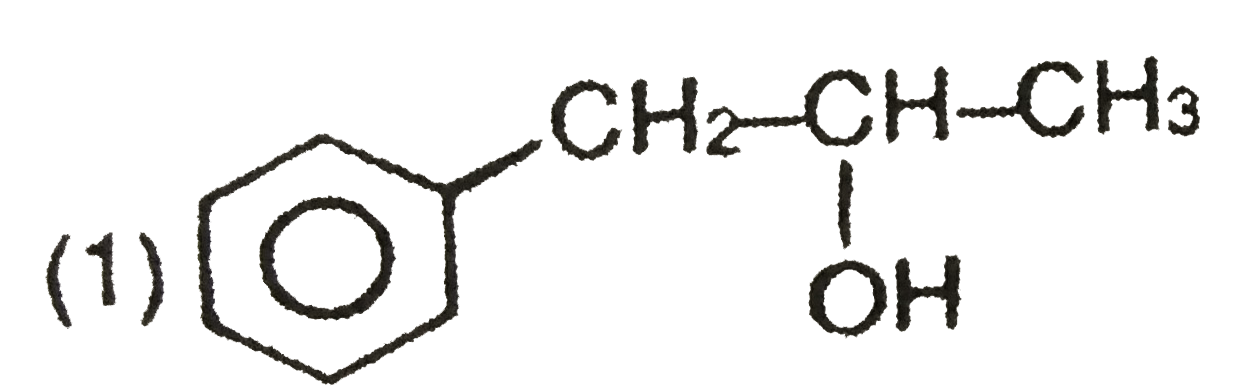
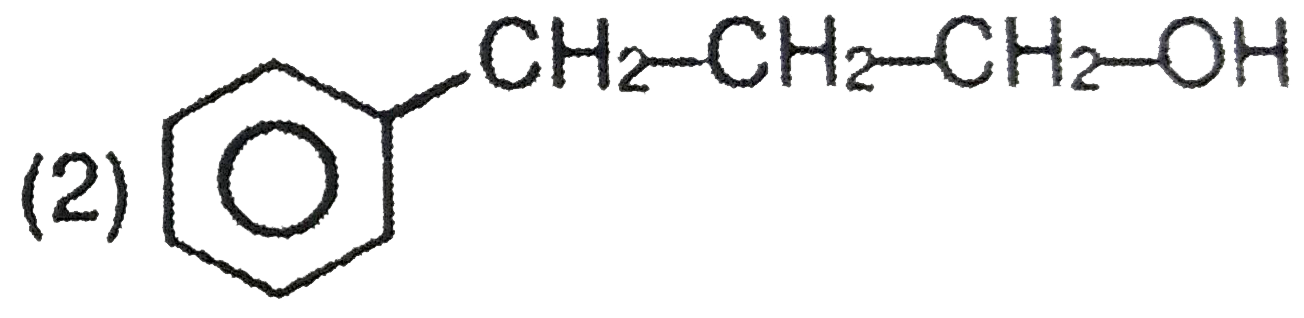
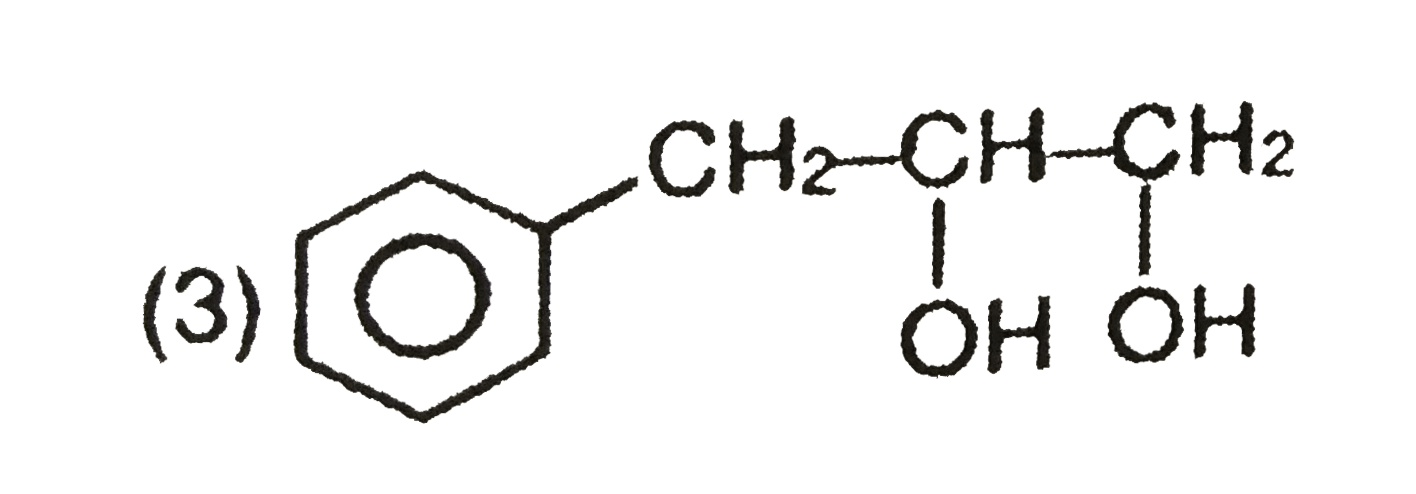
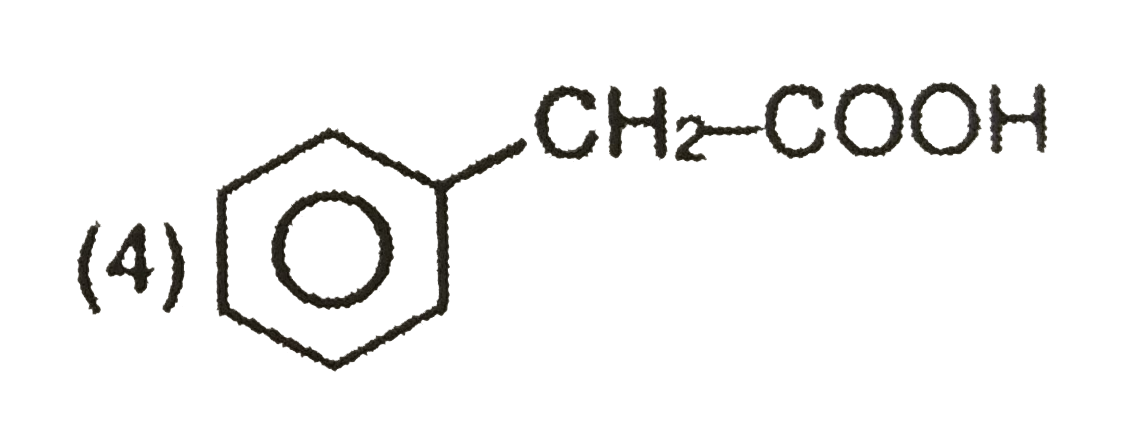
Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
On the basis of chemical reactions involved, explain how do CFC's cause depletion of ozone layer in stratosphere ? |
|
Answer» Solution :(i) The chloro-fluoro derivatives of methane and ethane are NAMED Freons (CFC.s). They slowly pass from troposphere to stratosphere. They stay for a very longer period of about 50-100 years. In the presence of UV radiations, CFC.s break up into chlorine free radicals, `CF_(2) overset(hv) rarr CF_(2)Cl+Cl^**` `CFCl_(3) overset(hv) CFCl_(2) + Cl^(**)` `Cl^(**)+ O_(3) rarr ClO^(**)+O_(2)` `ClO^(**) + O rarr Cl^(**) + O_(2)` (ii) Chlorine RADICAL is regenrated in the course of the REACTION. Due to this continuous ATTACK of Cl free radicals, thinning of ozone layer takes place which leads to the formation of ozone hole. (iv) It is estinated that for every reactive chlorine atom GENERATED in the stratosphere 1,00,000 molecules of ozone are depleted. |
|
| 2. |
On the basis of Bronsted concepts, which of the following are the correct representations? |
|
Answer» `CH_3CH_2OH + HCL HARR CH_3 CH_2 O^(+) H_2+Cl^(-) ` `CH_3 CH_2OH`stronger than ` NH_2^(-) ` `CH_3COOH ` stronger than ` CH_3 NH_2` |
|
| 3. |
On the basis bo VSEPR theory, prodict the shapes the shapes of the following :(i) CIF_(3) (ii) BrF_(5) (iii) NH_(2)^(-) (iv) H_(3) O^(+) |
|
Answer» Solution :(i) Shape of `CIF_(3)`<BR>No, of valence electrons of the central CL atom = 7 No. of atoms linked to it by single bonds = 3`therefore`Total no. of electron pairsaround`Cl=(7+3)/(2) = 5 ` No. of bond pairs = No. of atoms linked to Cl = 3 ` therefore ` No. of lone pairs = 5 - 3 = 2 Thus, the molecule is of the type `AB_(3) L_(2)` Hence, it is T-shaped. (ii) Shape of ` BrF_(5)` No. of valence electrons of centrons of central Br atom = 7 No. of atoms linked to it by single bonds = 5 ` therefore ` Total no. of electrons pairs around ` Br = (7+5)/(2) = 6 ` No. of bond pairs = No. of atoms linked to Br = 5 `therefore`Noof lone pairs = 6 - 5 = 1 Thus, the molecule is of the type ` AB_(5) L`. Hence,it has square pyramidal shape. (iii)Shape of ` NH_(2)^(-)` No . of valence electrons of central N atom = 5 + 1 (due to one unit -ve charge) = 6 No. of bond pairs = 2 ` therefore `Total no. of electron pairs around ` N = (6 +2)/(2) = 4 ` No, of bond pairs = 2 ` therefore ` No, of lone pairs = 4 - 2 = 2 Thus, the ion is of the type ` AB_(2) L_(2)`. Hence , it has a bent shape (V-shape). (iv) Shape of ` H_(3) O^(+)` ion No. of valenceelectrons of contral 'O' atom = 6 - 1(Due to one unit + ve charge ) = 5 No. of atoms linked 'O' atom = 3 |
|
| 4. |
On the addition of a solution containing CrO_(4)^(2-) ions to the solution of Ba^(2+), Sr^(2+) and Ca^(2+) ions, the precipitate obtained first will be of: |
|
Answer» `CaCrO_(4)` |
|
| 5. |
On taking 60 g CH_(3)COOH and 46g CH_(3)CH_(2)OH in a 5 lit. lask in tle presence of H_(3)O^(+) (catalyst), at 298K 44 g of CH_(3)COOC_(2)H_(5) is formed at equilibrium. If amount of CH_(3)COOH is doubled without affecting amount of CH_(3)CH_(2)OH then, CH_(3)"COOC"_(2)H_(5) formed is |
|
Answer» 20.33 g `CH_(3)COOH=(60)/(60)=1` MOL `CH_(3)CH_(2)OH=(46)/(46)=1` mol `CH_(3)COOC_(2)H_(5)=(44)/(88)=0.5` mol Case-II `CH_(3)COOH=2 mol, C_(2)H_(5)OH=1` mol `CH_(3)COOO_(2)H_(5)` =?  `K_(c)` is not affected at temperature is constant `(x^(2))/(1-x^(2))=(y^(2))/((2-y)(1-y)), (y^(2))/((2-y)(1-y))=1` `y^(2)=(2-4)(1-4)=y^(2)-3y` `y=(2)/(3)` mole of ester = `(2)/(3) xx 58 = 58.66` g |
|
| 6. |
On the account of osmotic pressure 'arive ' at the Van't Hoff equation. (##SUR_CHE_XI_V02_C09_E04_015_Q01.png" width="80%"> |
|
Answer» SOLUTION :Let us consider a simple apparatus as shown in the above figure . A semi-permeable membrane separtesa chamber into two compartments .WATER (pure solvent ) is added to the first compartment and the aqeuous NaCl ( solution ) is added to the second compartment such that the liquid LEVELS on the both sides are equal . Since there is a difference in concentration between the liquids present in the two compartments . the water molecules move from first compartment .to second compartment through the semi-permeable membrane allows only the water molecules to pass through ith in either direction but not allows the NaCl . The net flow of water is into the sodium chloride solution and hence increase its VOLUME . This decreases its concentration and also creates a pressure difference between the compartment .This pressure difference push some of the water molecules back to the solvent side through the semipermeable membrane until an equilibrium is establised . At the equilibrium the rate of movement of solvent molecules on both directions are equal The pressure difference at the equilibrium is called osmotic pressure `(pi) ` .Thus osmotic preesure can be defined as " the pressure that must be applied to the solution to stop the influx of the solvent (to stop osmosis) through the semipermeable membrane " Van't Hoff found out that for dilute solution the osmotic pressure is directly proportional to the molar concentration of the solute and the tempeerature of the solution . He proposed the FOLLOWING equation to calculate osmotic pressure which is now called as Van't Hoff equation. ` "" pi =cRT ` Here , c= Concentration of the solution in molarity T=Temperature R= Gas constant |
|
| 7. |
On strong heating of H_(3)PO_(4) and H_(3)BO_(3) sum of oxidation number of P and B in the final producy obtained is: |
|
Answer» <P> `H_(3)PO_(3) overset(Delta)rarr B_(2)O_(3)(O.N. = +3)` |
|
| 8. |
On strong heating lead nitrate gives |
|
Answer» PbO, NO, `O_(2)` |
|
| 9. |
On solving Schrodinger wave equation for hydrogen atom, the values of the enery obtained are called......and the corresponding values of the wavefunction (Psi) are called....... |
| Answer» SOLUTION :EIGEN VALUES, eigen FUNCTIONS | |
| 10. |
On reactly discoveredallotrope, C_(60),is knownas "…………..". |
|
Answer» |
|
| 11. |
On reaction with Mg, very dilute nitric acid produces |
|
Answer» `NH_3` |
|
| 12. |
On reaction with Cl_(2), phosphorus forms two types of halides 'A' and 'B'. Halide A is yellowish-white powder but halide 'B' is colourless oily liquied. Identify A and B write the formulas of their hydrolysis products. |
|
Answer» Solution :Since `Cl_(2)` REACTS with phosphours to form two halids (A and B), therefore, thses halides must be `PCl_(3)andPCl_(3)`. Since halide 'A' is a yellowish-white solid but halide 'B' is a colourless oily liquid, therefore, due to HIGHER molecular MASS and HENCE stronger forces of ATTRACTION, halide 'A' must be a solid, i.e., `PCl_(5)` and `P_(4)+10Cl_(2)to4PCl_(3)` (yellowish-white solid) `P_(4)+6Cl_(2)tounderset((B))(4PCl_(3))` (colourless oily liquid) halide 'B' must be a liquid i.e., `PCl_(3)`. Their hydrolysis products are shown below : `underset("Phosphorus pentachloride")(PCl_(5))+4H_(2)Otounderset("Orthophosphoric acid")(H_(3)PO_(4))+5HCl` `underset("Phosphorus trichloride")(PCl_(3))+3H_(2)Otounderset("Phosphorus acid")(H_(3)PO_(3))+3HCl` |
|
| 13. |
On reaction of sodium amalgam with water produce ....... and ...... air. |
| Answer» SOLUTION :SODIUM HYDROXIDE, DIHYDROGEN | |
| 14. |
On reaction of Na/Hg with water, it produces NaOH and ......... gas. |
|
Answer» Dihydrogen |
|
| 15. |
On pauling scale, the electron negativity of fluorine. |
|
Answer» |
|
| 16. |
On passing CO_2through a concentrated aqueous solution of Na_2CO_3, …………… is formed. |
| Answer» SOLUTION :`NaHCO_3` | |
| 17. |
On passing CO_(2)gas for a certain period of time through lime water, it gives ........ color. |
|
Answer» Blue |
|
| 18. |
on passing a current of 1.0 ampre for 16 min and 5 sec through one litre solution of CuCl_(2) all the copper of the solution was solution was peposited at cathode. The strength of CuCl_(2) solution was (Molar mass of Cu=63.5 faraday constant =96500 C mol^(-1)) |
|
Answer» 0.07 M `=1Axx965s=965 C` `Cu^(2+)+UNDERSET(2xx96500 C)(2e^(-))to underset(1 "MOL")(Cu)` `2 xx 96500 C `deposit COPPER = 1 mol 965 C deposite copper `=5xx10^(-3)` mol `therefore` One litre of `CuCl_(2)` solution CONTAIN `=5xx10^(-3)` mol of solute `therefore` Molarity of solution =0.005 M |
|
| 19. |
On passing 10.0 L of a gaseous mixture of NO_(2) and N_(2) at STP, through an NaOH solution, a mixture of NaNO_(2) and NaNO_(3) is formed 6.32 g of KMnO_(4) is required to oxidise above NaNO_(2) in H_(2)SO_(4) medium. Determine the percentage by mass of gaseous mixture (N_2 does not react with NaOH) |
|
Answer» Solution :`2NO_(2)+2NaOHtoNaNO_(3)+NaNO_(3)+NaNO_(2)+H_(2)O` " EQ of "`KMnO_(4)=" Eq of "NO_(2)^(ɵ)` `{NO_(2)^(ɵ)toNO_(3)^(ɵ)(x=2)]` " Eq of "`KMnO_(4)=(6.32)/(31.5)=Eq. of NO_(2)^(ɵ)` Moles of `NO_(2)^(ɵ)=(6.32)/(31.5)xx(1)/(2)=0.1 MOL` Weight of `NO_(2)^(ɵ)=0.1xx69=6.9g` Weight of `NaNO_(2)=6.9g` From the above equation `0.1 " mol of "NaNO_(2)-=0.2 mol NO_(2)` `-=0.2xx22.4L at STP` `-=4.48 L NO_(2)` Volume of `N_(2)=(10-4.48)=5.52L` `=0.246L N_(2)` Mole of `NO_(2)=0.2=0.2xx46=9.2g` Mole of `N_(2)=0.246=0.246xx28=6.89g` `% of NO_(2)=57.18,% of N_(2)=42.82g` |
|
| 20. |
On passing 10.0 L of a gaseous mixture of nitrogen dioxide and N_2 at STP through a NaOH solution, a mixture of sodium nitrite and sodium nitrate in formed. A mass of 6.32 g of KMnO_4 is required to oxidise the above sodium nitrite in a H_2SO_4 medium. Determine the mass percentage of the gaseous mixture (N_2 does not react with NaOH.) |
|
Answer» Solution :`N_2` does not REACT with NaOH. So only `NO_2` reacts with NaOH. `2NO_2+2NaOHtoNaNO_3+NaNO_2+H_2O` `5NaNO_2+2KMnO_4+3H_2SO_4toK_2SO_4+2MnSO_4+5NaNO_3+3H_2O` `therefore2 MOL KMnO_4-=5 mol NaNO_2` ALSO: `2 mol NO_2-=1 mol NaNO_2` `implies0.1 mol NaNO_2-=0.2mol NO_2` `-=0.2xx22.4L at STP` `-=4.48 LIT. NO_2` or `(10-4.48)LN_2=5.52L of N_2-=0.246 " mol of "N_2` " mol of "`NO_2=0.2-=0.2xx46=9.2g` `% of NO_2=57.18%` `" mol of "N_2=0.246=0.246xx28=6.28g` `% of N_2=42.82%` |
|
| 21. |
On ozonolysis , one mole of a hydrocarbon produces two molecules of ethanal and one molecule of ethanedial. The hydrocarbon could be |
|
Answer» 1,3-hexadiene `underset"Ethanal"(CH_3CH=O)+ underset"Ethanedial"(O=CH-CH=O)+underset"Ethanal"(O=CHCH_3)` Remove the oxygen atoms, and combine the three fragments TOGETHER , the STRUCTURE of the HYDROCARBON is 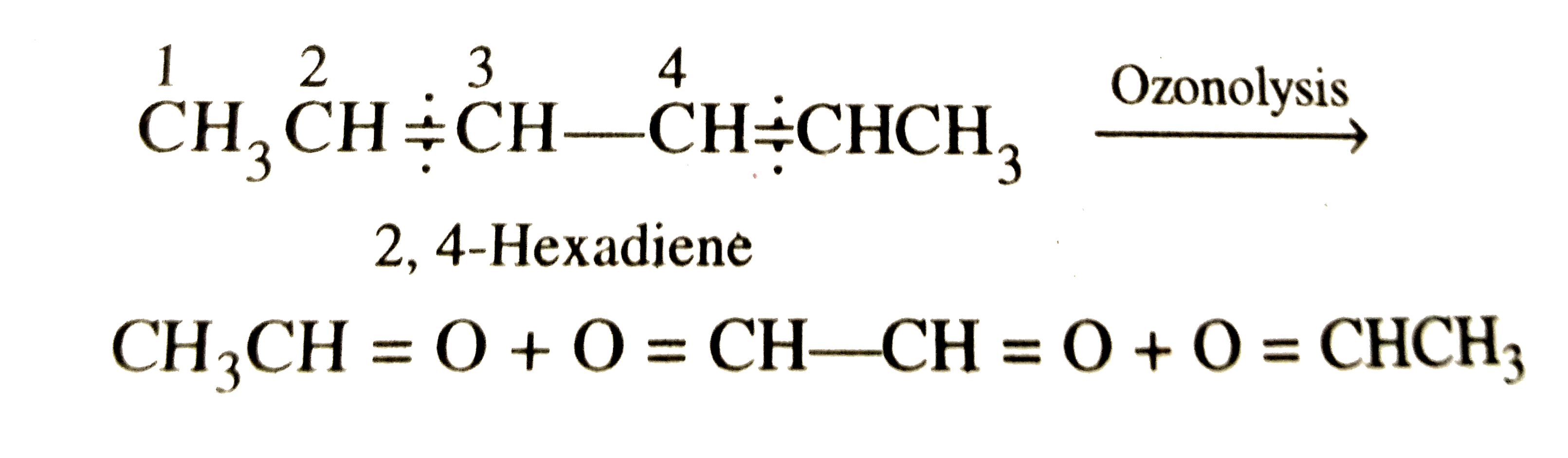
|
|
| 22. |
On oxidative ozonolysis of 3-Methylex-3-ene, two products A and B are formed . A gives CO_(2) gas with sodium bicarbonate , but B can not. The structure of A and B are respectively. |
|
Answer» `CH_(3)-CH_(2)-underset(O)underset(||)(C)-CH_(3) & CH_(3)-CH_(2)-CO OH` 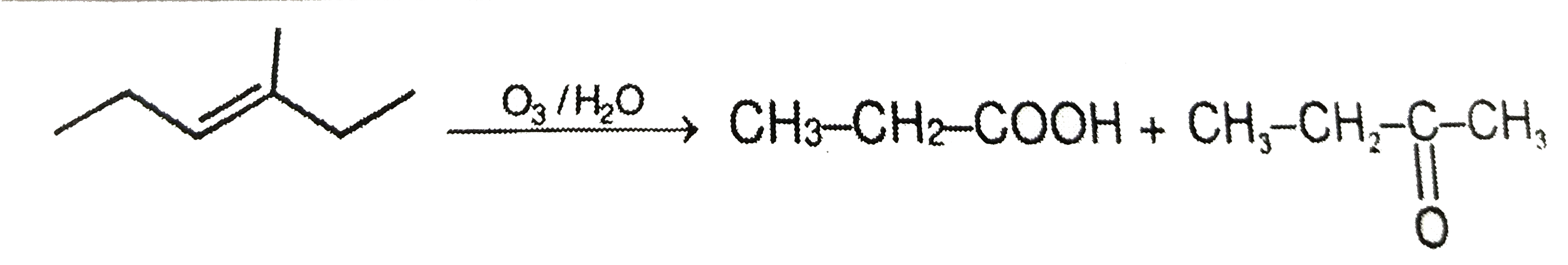
|
|
| 23. |
On moving from left to right across a period in the periodic table, the metallic character……….. |
|
Answer» INCREASES |
|
| 24. |
On monobromination of a hydrocarbon A(C_5H_(12)) the number of possible stereoisomers obtainedcan be ...... |
|
Answer» |
|
| 25. |
On mixing ethyl acetate with aqueous sodium chloride, the composition of the resultant solution is |
|
Answer» `CH_(3)COOC_(2)H_(5) + NaCl` |
|
| 26. |
On mixing certain alkane with chlorine and irradiating it with ultraviolet light, one forms only one monochloro alkane. To alkane could be |
|
Answer» neopentane |
|
| 27. |
On mixing a certain alkane with chlorine and irradiating it with ultraviolet light, it forms only one monochloroalkane. This alkane should be |
|
Answer» NEOPENTANE |
|
| 28. |
On mixing a certain alkane with chlorine and irradiating it with ultraviolet light, it forms only one monochloroalkane this alkane could be : |
| Answer» Answer :D | |
| 29. |
on mercuration-demercuration produces the major product . |
|
Answer»
|
|
| 30. |
On keeping Ag rod in CuSO_(4) |
|
Answer» No change in colour intensity of `CuSO_(4)` is observed. `Ag_((s))+Cu_((aq))^(+2)+SO_(4(aq))^(2-)to` No reaction Any reaction is not possible and not OCCURRING as a result of it - no .change in colour of `CuSO_(4)` is observed. |
|
| 31. |
On increasing the pressure, in which the direction will the gas phase reaction proceed to re-establish equilibrium, is predicted by applying the Le Chatelier's principle. Consider the reaction, N_(2) (g) + 3 H_(2) (g) hArr 2 NH_(3) (g) Which of the following is correct, if the total presurre at which the equilibrium is established, is increased without changing the temperature ? |
|
Answer» K will remain same |
|
| 32. |
On increasing the pressure, in which direction will the gas phase reaction proceed to reestablish equilibrium, is predicted by applying the Le-Chatelier's principle. Consider the reaction. N_(2(g)) + 3H_(2(g)) hArr 2NH_(3(g)) Which of the following is correct, if the total pressure at which the equilibrium is established, is increased without changing the temperature ? |
|
Answer» K will remain same If the total pressure at which the equilibrium is ESTABLISHED is increased WITHOUT changing the temperature. K will remain same. K changes only with change in temperature. |
|
| 33. |
On increasing temperature, which of the following statement is correct? |
|
Answer» AREA under `(dN)/(N)` vs V curve at different temperatures are different b) as `T uarr` fraction pocessing `u_(mp) darr`. c) as `T uarr` more molecules possess speeds nearer to `u_(mp)`. d) distribution of speeds depends on T also . |
|
| 34. |
On igniion, Rochelle salt NaKC_(4) H_(4) O_(6). 4H_(2)O (mol. Wt 282) is converted into NaKCO_(3) (mol wt. 122).0.9546g sample of the rochelle salt on ignition gives NaKCO_(3) which is titrated wih 41.72mL.H_(2)SO_(4). From the follwing data, find the percentage purtiy of the rochelle salt. The solution after neutralisation requires its 1.91 mL of 0.1297N NaOH. The H_(2)SO_(4) used for the neutralisationrequires its 10.27mL aganist 10.35mL of 0.1297N NaOH |
|
Answer» |
|
| 35. |
On heating with concentrated NaOH solution in an inert atmosphere of CO_(2), white phosphorus gives a gas. Which of the following statements is incorrect about the gas? |
|
Answer» It is highly poisonous and has smell like ROTTEN fish |
|
| 36. |
On heating with carbon, which of the following elements do not form carbides |
|
Answer» B |
|
| 37. |
On heating with carbon, which of the following elements donot from carbides? |
|
Answer» B |
|
| 38. |
On heating which of the following release CO_(2) most easily ? |
|
Answer» `Na_(2)CO_(3)` |
|
| 39. |
On heating which of the following releases CO_(2) most easily? |
|
Answer» `MgCO_(3)` |
|
| 40. |
On heating vapours of S_(8)(g) decomposes to S_(2)(g), Due to this, the van der Waal's constant 'b' for the resulting gas: |
|
Answer» increases |
|
| 41. |
On heating sodium metal in a current of dry ammonia'gas, the compound formed is |
|
Answer» Sodium nitrate |
|
| 42. |
On heating slowly mu of Cl-CH_(2)CH_(2)-Cl will: |
|
Answer» increase |
|
| 43. |
On heating of lithium nitrate it produce... |
|
Answer» `LiO_(2)` |
|
| 45. |
On heating, lead (II) nitrate gives a brown gas 'A'. The gas 'A' on cooling changes to colocurless solid 'B'. Solid 'B' on heating with NO changes to a blue solid 'C'. Identify 'A', 'B' and 'C' and also write reactions involved and draw the structures of 'B' and 'C'. |
|
Answer» Solution :(i) Since LEAD (II) nitrate on heating GIVES a brown gas 'A' therefore, gas 'A' MUST be nitrogen DIOXIDE `(NO_(2))`. `{:underset("Lead (II) nitrate")(2Pb(NO_(3))_(2))overset(Delta,673K)to2PbO+underset("Brown gas (A)")(4NO_(2))+O_(2):}` (ii) The brown gas 'A' on cooling dimerises to give a colourless SOLID 'B' therefore 'B' must be `N_(2)O_(4)` (dinitrogen tetroxide). `{:2NO_(2)underset("On heating")overset("On cooling")hArrunderset("Colourless solid (B)")(N_(2)O_(4))` (iii) Since colourles solid 'B' on heating with NO, gives a blue solid 'C' therefore 'C' must be dinitrogen trioxide. `{:2NO+underset("Colourless solid (B)")(N_(2)O_(4))tounderset("Blue solid (C))(2N_(2)O_(3)):}` Thus, `A=NO_(2),B=N_(2)O_(4)andC=N_(2)O_(3)`. For structures, |
|
| 46. |
On heating glycerol with conc. H_2SO_4,a compound obtained which has an unpleasant odour. This compound is : |
|
Answer» ETHYLENE glycol |
|
| 47. |
On heating glycerol with conc. Sulphuric acid a compound with unpleasant odour is obtained. The compound is : |
|
Answer» METHYL alcohol |
|
| 48. |
On heating ethylene glycol at 775 K, the product obtained is |
|
Answer» OXIRANE 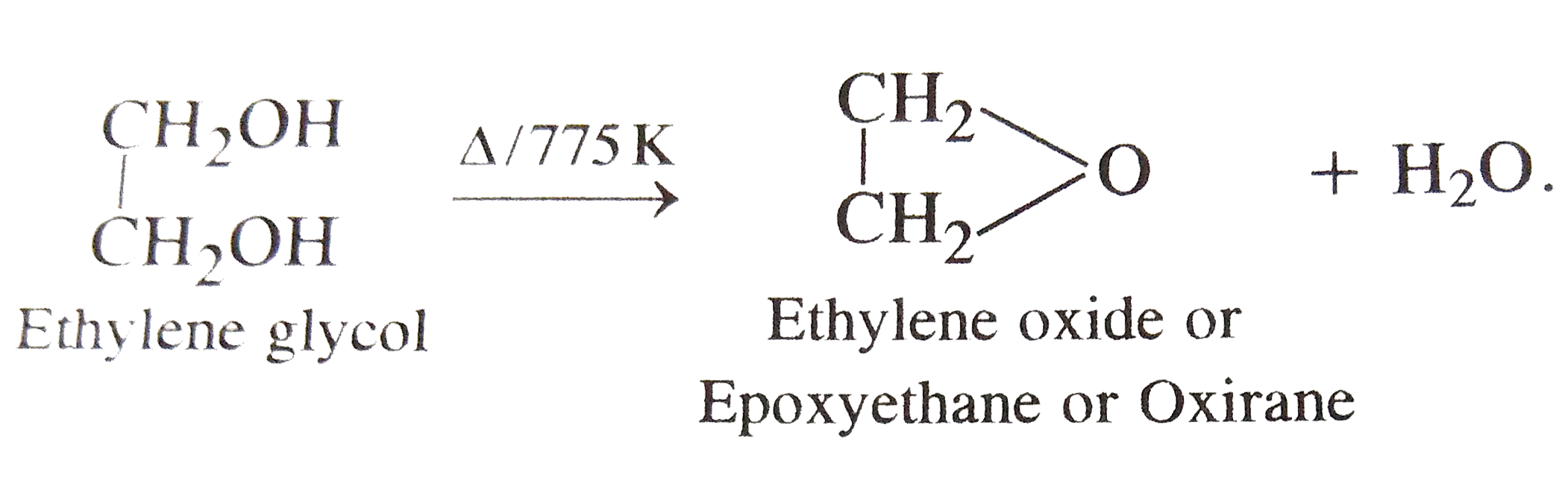
|
|
| 49. |
On heating compuond (A) gives a gas (B) which is a constituent of air. This gas when treated with 3 mol of hydrogen (H_(2)) in the presence of a catalyst gives anther gas (C) which is basic in nature. Gas C on further oxidation in moist condition gives a compound (D) which is a part of acid rain. Identify compounds (A)to (D) and also give necessary equations of all the steps involved. |
|
Answer» Solution :(i) Since gas `B` on heating with 3 moles of hydrogen in PRESENCE of a catalyst, gives a gas `C` which is BASIC in nature, THEREFORE, 'C' must be ammonia `(NH_(3))` and gas (B) must be `N_(2)` `underset((B))(N_(2)) + 3H_(2) overset(DELTA, "Catalyst")(to) underset("Ammonia(C)")(2NH_(3))` (ii) Since gas 'B' (i.e., `N_(2)`) is obtained by heating compound (A) , therefore , compound (A) must be either ammonium nitrite or ammonium dichromate `underset((A))(NH_(4)) NO_(2) overset(Delta)(to) underset((B))(N_(2)) + 2H_(2)O or (NH_(4))_(2) underset((A))(Cr_(2)) O_(7) overset(Delta)(to) underset((B))(N_(2)) + Cr_(2) O_(3) + 4H_(2)O` (iii) Since gas 'C', (i.e., `NH_(3)`) on further oxidation in moist conditions gives compound (D) which is a part of acid rain, therefore, compound (D) must be nitric acid `(HNO_(3)`). `underset((C))4NH_(3)+5O_(2)overset("Oxidation")to4NO+6H_(2)O` `2NO+O_(2)to2NO_(2),3NO_(3)+H_(2)Otounderset((D))2HNO_(3)+NO` Thus, `A=NH_(4)NO_(2)or(NH_(4))_(2)Cr_(2)O_(7),B=N_(2),C=NH_(3)andD=HNO_(3)`. |
|