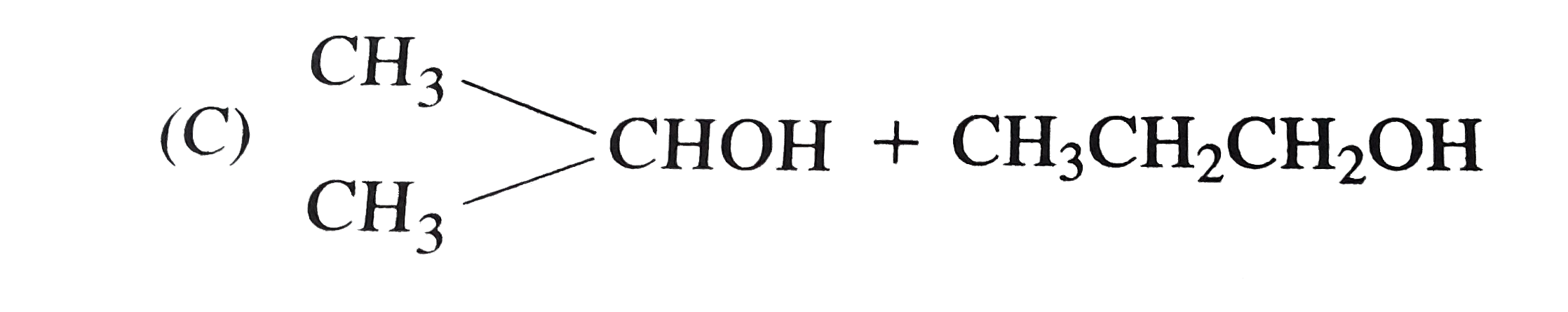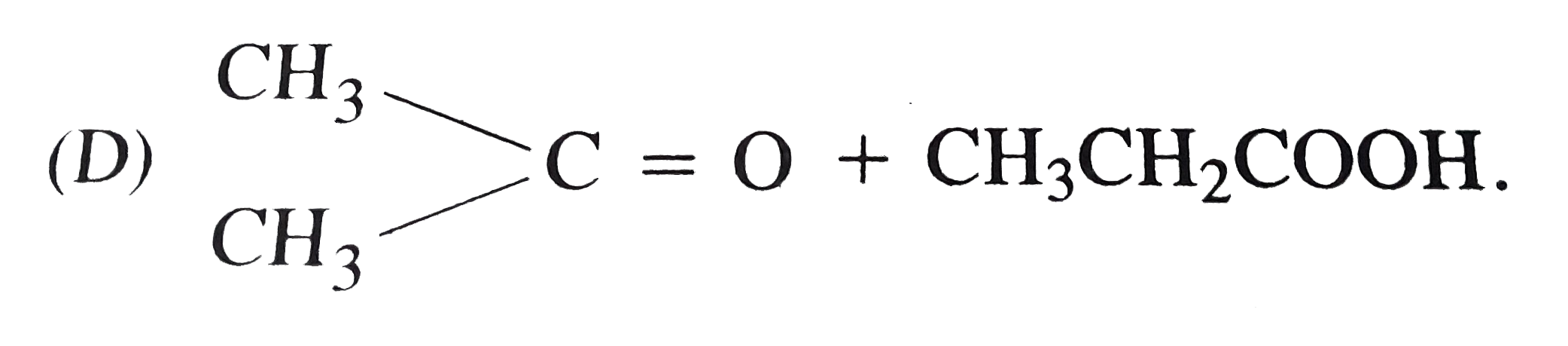Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
One among the following oxides cannot be formed |
|
Answer» `K_(2)O` |
|
| 2. |
One among the following pairs of compounds are not isomers? |
|
Answer» `(CH_(3))_(2)CH- O-C_(2)H_(5) and CH_(3) (CH_(2))_(2) - O-C_(2)H_(5)` |
|
| 3. |
One among the following is an example of ferroelectric compound. It is |
| Answer» Solution :Barium titanate. | |
| 4. |
Once mole of gas is subjected to a process causing a change in astate from(1.25 atm, 300 K) to a final state of (1 atm, 600 K). Calculate the enthalpy change from the following information [in atm-litre] Information 1: The process involvea 100 atm-litre of heat given to system of 20 atm-liter of work is done by the system. Information :2 Molar mass of the gas is 49.26. Information 3 : Density of gas as 1.25 atm and 300 K is 2 gm/liter. Information 4: Density of gas at 1 atm and 600 K is l gm/litre. [Given : R =0.0821 atm-litre/mol K=(18.47)/225 atm-litre/mole K] (Round off your answer to nearest integer). |
|
Answer» |
|
| 5. |
On which principle the solute moves on chromatography paper? |
| Answer» SOLUTION :The SOLUTE MOVES over on chromatography PAPER by the PRINCIPLE of capalary action. | |
| 6. |
On which of the following factors, inter- molecular force of attraction docs not depend ? |
|
Answer» Shape of molecules |
|
| 7. |
On which factors the enthalpy of dilution is depended? |
| Answer» Solution :Enthalpy of DILUTION depends UPON the original CONCENTRATION of solution and the amount of SOLVENT which is added in it. | |
| 8. |
On which factor does vapour pressure of liquid at fixed temperature depend upon ? |
|
Answer» Nature of LIQUID |
|
| 9. |
On which factor does dipole moment depend in case of polyatomic molecules. |
| Answer» SOLUTION :SEE TEXTBOOK | |
| 10. |
On which bases the non-viable particulates can be classified ? |
| Answer» SOLUTION :Non-viable particulates may be classified according to their nature and size : They are as FOLLOW. (i) SMOKE (ii) dust (iii) MISTS (iv) fumes (v) smog | |
| 11. |
On what parameter do the elements are classifed in the modern periodic table? |
|
Answer» |
|
| 12. |
On what basis is the position of an electron in a periodic table is decided ? |
| Answer» Solution :The POSITION of electron is DEPEND UPON its last orbital in which electron FILLED. | |
| 13. |
On what basis do you classify gases into permanent and temporary gases? Explain these types with example. |
|
Answer» Solution :Based on the values of the CRITICAL temperature, the gases are classified into permanent and temporary gases. (i) Gases having very low critical temperature belong to permanent type. EG: `H_(2),N_(2),He`, etc. (ii) Gases having critical temperature in the ordinary range of temperatures belong to the temporary type. Eg: `NH_(2),CO_(2),SO_(2),HCl`, etc. |
|
| 14. |
On what basis has periodic table made by Mendeleev? |
| Answer» Solution :(i) Initially order of atomic weight (II) SIMILARITIES of physical and chemical in PERIODIC table. (iii) EMPIRICAL formulas and properties of the compounds formed by the elements. | |
| 15. |
On what basis do you classify gases into permanent and temporary gases ? Expalin these types with example. |
|
Answer» SOLUTION :BASED on the values of the critical temperature, the gasesare calassified into permanent and temporary gases. (i) Gases having very low critical temperature belongs to permanent type. Eg: `H_(2), N_(2), He, ETC`. (ii) Gases having critical temperature is the ordinary range of temperatues belongs to the temporary type Eg: `NH_(3),CO_(2), SO_(2),HCl` etc. |
|
| 16. |
On vigorous oxidation by permanganate solution (CH_(3))_(2)C = CH - CH_(2) CH_(3) gives |
|
Answer» `CH_(3) - UNDERSET(CH_(3))underset(|)overset(OH)overset(|)(C) - overset(OH)overset(|)(CH) - CH_(2)CH_(3)` 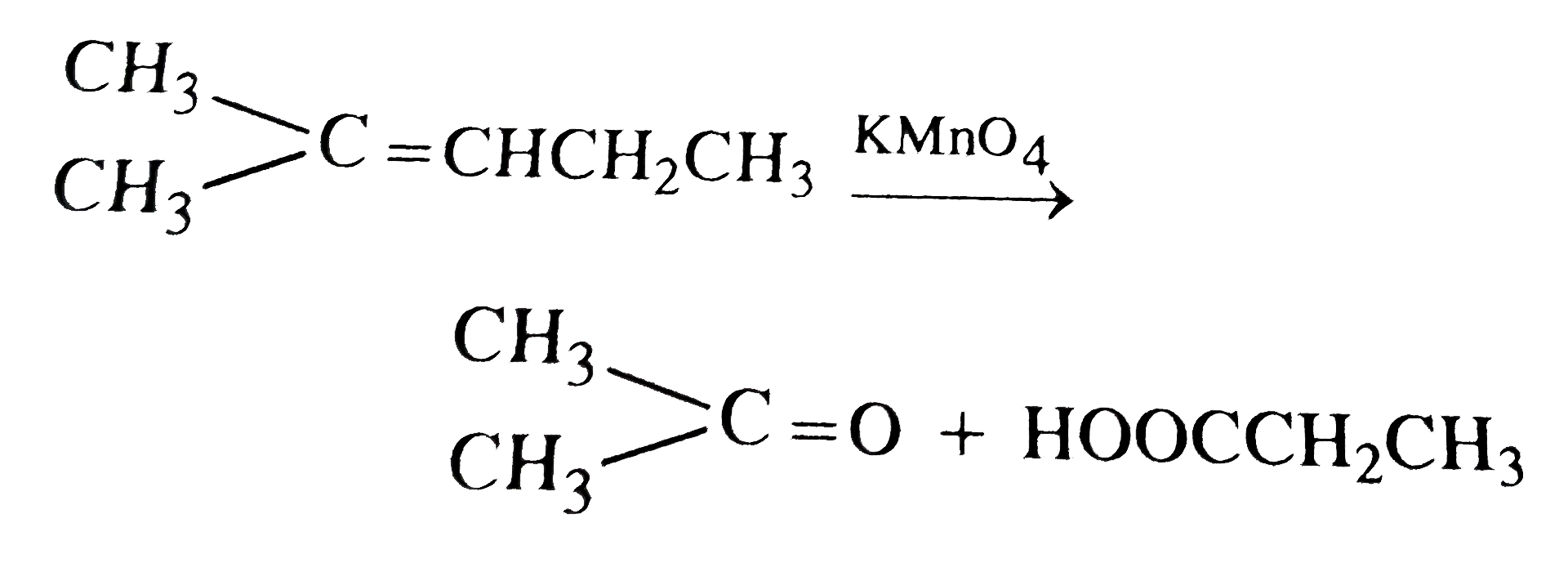
|
|
| 17. |
On very immediate reaction of sodium metal with water, it produces ...... gas. |
|
Answer» Oxygen |
|
| 18. |
On treatment with cold water, an element (A) reacted quietly liberating a colourless, odourless gas (B) and a basic solution (C ). Lithium reacted with (B) yielding a solid product (D) which effervesced with water to give a stongly basic solution (E) and gas (F). When CO_(2) was bubbled through solution (C ), initially a white ppt. (G) was formed, but this redissolved forming solution (H) when more CO_(2) was passed, precipitate (G) effervesced when moistened with conc HCl and gave a brick red colouration to the bunsen flame. When (G) was heated with carbon at 1000^(@)C, a caustic white compound (I) was formed, which when heated with N_(20 at 1000^(@)C gave a solid (J) of some commercial importance. Identify (A) to (J) and explain the reactions. |
|
Answer» Solution :`underset("Element")(Given, (A))+H_(2)Orarrunderset(gas)underset("odourless")underset("Colourless",)(B)+underset("Solution")underset("Basic")(C )` `Li+(B)rarrunderset("Solid product")(D)` `(D)+H_(2)Orarrunderset("Solution")underset("basic")underset("Strogly")(E)+(F)_(Gas)` `(C )+CO_(2(g))rarrunderset("White ppt".)((G))overset(CO_(2))(RARR)underset("Solution")underset("to give a")underset("redissolved")underset("White ppt".)((H))` `(G)+"conc" HClrarr{:("EFFEVESCENCE produced" and),("gave brick red colouration"),("to Bunshen flame".):}` `(G)+"Carbon"` `overset(1000^(@)C)(rarr)underset("Compound")underset("white")underset("Caustic")((I)) overset(N_(2))underset(1000^(@)C)(rarr) underset("importance")underset("commercial")underset("Solid having")((J))` Solution: Since `(G)`gives brick red colouration to Bunshen flame it SEEMS `(A)` is calcium. `underset((A))(Ca)+2H_(2)Orarrunderset("Basic solution")underset((C))(Ca(OH)_(2))+underset((B))(H_(2))` `2Li+underset((B))(H_(2))rarrunderset((D))(LiH_((s))` `LiH_((s))underset((D))+H_(2)Orarrunderset((G))(LiOH)+H_(2)` `underset((G))(CaCO_(3))+CO_(2)+H_(2)OrarrCaunderset(H)((HCO_(3))_(2))` `underset((G))(CaCO_(3))+underset(conc)(2HCl)rarrCaCl_(2)+H_(2)O+CO_(2)uarr` `underset((G))(CaCO_(3))+4Coverset(1000^(@)C)(rarr)underset((I))(CaC_(2))+3CO` `underset((G))(CaCO_(3))+4Coverset(1000^(@)C)(rarr)underset(I)(CaC_(2))+3CO` `underset((I))(CaC_(2))+N_(2)rarrunderset("Nitrolim")underset((J))(CaCN_((2))+C` |
|
| 19. |
On treatment of citric acid with conc. H_(2)SO_(4) which of the following is produced ? |
|
Answer» Acetone 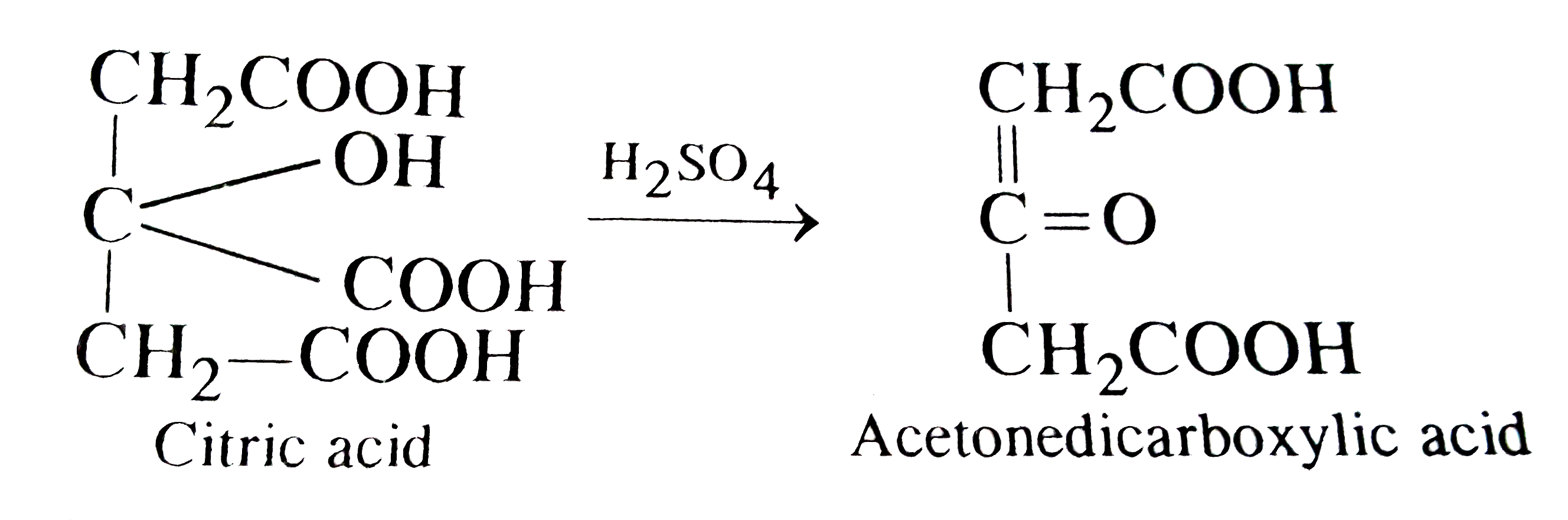
|
|
| 20. |
On the formation of SF_6 by the direct combination of Sand F_2, which is the limiting reagent? |
|
Answer» SOLUTION :`SF_6` is formed by burning Sulphur in an atmosphere of Fluorine. SUPPOSE 3 moles of S is allowed to react with 12 moles of Fluorine. `S_(I) + 3F_(2_(g)) to SF_(6_(g)` As per the stoichiometric reaction, ONE mole of REACTS with 3 moles of fluorine to complete the reaction. Similarly, 3 moles of Srequires only 9 moles of fluorine. `:.` It is understood that the limiting reagent is Sulphur and the excess reagent is Fluorine. |
|
| 21. |
On the reaction 2Ag + H_(2)SO_(4) to Ag_(2)SO_(4) + 2H_(2)O + SO_(2) . Sulphuric acid acts as |
|
Answer» REDUCING agent |
|
| 22. |
On the formation of SF_(6) by the direct combination of S and F_(2)which is the limiting reagent? Prove it. |
|
Answer» Solution :`SF_(6)` is formed by burning Sulphur in an atmosphere of Fluorine. Suppose 3 MOLES of S is ALLOWED to react with 12 moles of Fluorine. `S_((l))+3F_(2_((g))) rarr SF_(6_((g)))` As per the stoichiometric reaction, one mole of S reacts with 3 moles of fluorine to COMPLETE the reaction. Similarly, 3 moles of S requires only 9 moles of fluorine. `:.` It is understood that the limiting REAGENT is Sulphur and the excess reagent is Fluorine. |
|
| 23. |
On the basisof whichrulearrangementofelectrontakeplacein atom ? |
|
Answer» SOLUTION :the arrangementsof orbitals inatom arebasedon differentrulesand PRINCIPLE (i) Aufbauprinciple (II)Paulieexclusion principle (iii)Hund.sruleof maximum multiplicity (IV)Basedon orbital ENERGY |
|
| 24. |
On the basis of VSEPR theory, predict the shapes of the following molecules molecules /ions ? (i)SiF_(4) (ii) NH_(2)^(-) (iii) NH_(4)^(+) (iv) C_(2) H_(2) (v) H_(3)O^(+) (vi) F_(2)O (vii) PCl_(3) (viii) PF_(5). |
|
Answer» Solution : (i) Tetrhedral (ii) V-shape(iii) Tetrahedral (iv) CYLINDRICAL (v) TRIGONAL pyamidal(vi) V-shape (vii) Trigonal PYRAMIDAL (VIII) Trigonal bipyramidal. |
|
| 25. |
On the basis of VSEPR theory predict the shape of the Ozone . |
|
Answer» Solution :The RESONATING structure of ozone are : `:underset(..) overset(..)O - overset(..)O = underset(..) overset(..)O hArr underset(..) overset(..)O= overset(..)O - underset(..) overset(..)O:` The central o-atom is considered to have two BOND pairs and ONE lone pair of electrons (IE) it is of `AB_(2)E` type. Hence it is a bent molecule. |
|
| 26. |
On the basis of uncertainty principle show that an electron cannot exist with in atomic nucleus. (Given : Nuclear radius = 10^(-15) m) |
| Answer» SOLUTION :Taking `10^(-15)` m as `Deltax`, the `Delta`v comes much HIGHER than the velocity of LIGHT and hence is not possible | |
| 27. |
On the basis of thermochemical equations (i),(ii) and (iii), which of the algebric relationship is correct. C("graphite") + O_(2)(g) to CO_(2)(g), Delta_(r)H = x kJ"mol"^(-1) C("graphite") + 1/2 O_(2)(g) to CO(g), Delta_(r)H = y KJ"mol"^(-1) CO(g) + 1/2O_(2)(g) to CO_(2)(g), Delta_(r)H = z kJ"mol"^(-1) |
|
Answer» z = x + y |
|
| 28. |
On the basis of thermochemical equations (i), (ii) and(iii) , find out which of the algebric relationships given in options (a) to (d) is correct. (i) C(graphite) +O_(2)(g) rarr CO_(2)(g), Delta_(r)H = xkJ mol^(-1) (ii) C(graphite) + (1)/(2) O_(2)(g) rarr CO(g), Delta_(r)H= y kJ mol^(-1) (iii) CO(g)+(1)/(2)O_(2)(g) rarrCO_(2)(g), Delta_(r)H = z kJ mol^(-1) |
|
Answer» `Z= x+y` |
|
| 29. |
On the basis of this equation pH = -log [H^+], the pH of 10^(-8) "mol dm"^(-3) solution of HCl should be 8. However, it is observed to be less than 7.0. Explain the reason. |
|
Answer» Solution :The CONCENTRATION `10^(-3)` mol `"dm"^(-3)` INDICATES that the solution is very dilute. So, we cannot neglect the contribution of `H_3O^+` ions PRODUCED from `H_2O` in the solution. Total `[H_3O^+]` = `10^(-8)` + `10^(-7)` M. From this we get the value of pH close to 7 but less than 7 because the solution is acidic. From CALCULATION, it is found that pH of `10^(-8) "mol dm"^(-3)` solution of HCl is equal to 6.96. |
|
| 30. |
On the basis of thermochemical equations (a),(b) and (c), find out which of the algebric relationship given in options(i) to (iv) is correct. (i)C("graphite ") + O_(2)(g) to CO_(2)(g), Delta_(r)H = x kJ"mol"^(-1) (ii)C("graphite") + 1/2 O_(2)(g) to CO(g), Delta_(r)H = y KJ"mol"^(-1) (iii)C(g) + 1/2O_(2)(g) to CO_(2)(g), Delta_(r)H = z kJ"mol"^(-1) |
|
Answer» z = x + y `C("graphite") + 1/2O_(2)(g) to CO(G), Delta_(r)H = y kJ"mol"^(-1)`...(b) Subtracting equations (a) and (b) we get `CO(g) + 1/2O_(2)(g) to CO_(2)(g), Delta_(r)H = z kJ"mol"^(-1)`...(c) Equation (c) is OBTAINED by subtracting (a) from (b) Therefore, z = x - y or x = y + z. |
|
| 31. |
On the basis of thermochemical equations (1), (2) and (3), find out which of the algebraic relationships given in options (A) to (D) is correct. (1) C_("(graphite)") + O_(2(g)) to CO_(2(g)) , Delta_(r) H= x "kJ mol"^(-1) (2) C_("(graphite)") + (1)/(2) O_(2(g)) to CO_((g)) , Delta_(r ) H= y "kJ mol"^(-1) (3) CO_((g)) + (1)/(2) O_(2(g)) to CO_((g)) , Delta_(r) H= z "kJ mol"^(-1) |
|
Answer» `z= x+y` (a) `C_("(graphite)") + O_(2(g)) , Delta_(R) H= x "kJ MOL"^(-1)` (b) `(C_("(graphite)") + (1)/(2) O_(2(g)) to CO_(2(g)) , Delta_(r) H = x "kJ mol"^(-1) )/(CO_((g)) + (1)/(2) O_(2(g)) to CO_(2(g)) , Delta_(r) H = z "kJ mol"^(-1) ) ` Hence, `x-y - z " or " x=y+z` |
|
| 32. |
On the basis of the postulates of kinetic theory of gases, it is possible to derive the mathematical expression, commonly known as kinetic gas equation. PV = 1/3 m n u^3? where, P= Pressure of the gas, V a volume of the gas, m=Mass of a molecule, n = Number of molecules present in the given amount of a gas and u = root mean square speed For one mole of gas, PV = RT and n=N_A 1/3 m N_a u^2 = RT or 2/3 .1/2m N_A u^2 = N_A [1/2mN_Au^2 = KE "per mole"] ,2/3K.E. = RT implies K.E. 3/2RT Average kinetic energy per mol does not depend on the nature of the gas but depends only on temperature. This, when two gases are mixed at the same temperature, there will be no rise or decrease in temperature unless both react chemically. Average kinetic energy per molecule = ("Average K.E. per mole")/N = 3/2(RT)/(N) implies 3/2kTwhere k is the Boltzmann constant Which of the following expressions correctly represents the relationship between the average molar kinetic energies of CO and N_2 molecules at the same temperature ? |
|
Answer» `K.E.(CO) = KE.(N_2)` |
|
| 33. |
On the basis of the postulates of kinetic theory of gases, it is possible to derive the mathematical expression, commonly known as kinetic gas equation. PV = 1/3 m n u^3? where, P= Pressure of the gas, V a volume of the gas, m=Mass of a molecule, n = Number of molecules present in the given amount of a gas and u = root mean square speed For one mole of gas, PV = RT and n=N_A 1/3 m N_a u^2 = RT or 2/3 .1/2m N_A u^2 = N_A [1/2mN_Au^2 = KE "per mole"] ,2/3K.E. = RT implies K.E. 3/2RT Average kinetic energy per mol does not depend on the nature of the gas but depends only on temperature. This, when two gases are mixed at the same temperature, there will be no rise or decrease in temperature unless both react chemically. Average kinetic energy per molecule = ("Average K.E. per mole")/N = 3/2(RT)/(N) implies 3/2kTwhere k is the Boltzmann constant In deriving the kinetic gas equation, the use of the root mean square speed of the molecules is done, hecause it is |
|
Answer» AVERAGE SPEED of the molecules |
|
| 34. |
On the basis of the postulates of kinetic theory of gases, it is possible to derive the mathematical expression, commonly known as kinetic gas equation. PV = 1/3 m n u^3? where, P= Pressure of the gas, V a volume of the gas, m=Mass of a molecule, n = Number of molecules present in the given amount of a gas and u = root mean square speed For one mole of gas, PV = RT and n=N_A 1/3 m N_a u^2 = RT or 2/3 .1/2m N_A u^2 = N_A [1/2mN_Au^2 = KE "per mole"] ,2/3K.E. = RT implies K.E. 3/2RT Average kinetic energy per mol does not depend on the nature of the gas but depends only on temperature. This, when two gases are mixed at the same temperature, there will be no rise or decrease in temperature unless both react chemically. Average kinetic energy per molecule = ("Average K.E. per mole")/N = 3/2(RT)/(N) implies 3/2kTwhere k is the Boltzmann constant The average kinetic energy (in joule) of the molecules in 8g methane at 27&@C is. |
|
Answer» `62.14 XX 10^(-22)J` `implies KE = 3/2 xx KT = 3/2 xx (8.314)/(6 xx 10^23) xx 300 = 6 xx 10^(-21)`. |
|
| 35. |
On the basis of the following thermochemical data (Delta_(f)G^(@)H^(+)(aq) = 0) H_(2)O(l) to H^(+)(aq) + OH^(-)(aq), DeltaH = 57.32 kJ (ii) H_(2)(g) + 1/2 to H_(@)O(l), DeltaH = -286.2 kJ The value of enthalpy of formation of OH^(-) at 25^(@)C is |
|
Answer» `-22.88` kJ `H_(2)O(l) to H^(+)(aq) + OH^(-)(aq)DeltaH_(2) = 57.32 kJ` ..(i) `H_(2)(g) + 1/2O_(2)(g) to H_(2)O(l)DeltaH_(3) = -286.2` kJ ...(II) `DeltaH_(1)` will be OBTAINED by ADDING (i) and (ii) ` = 57.32 + (-286.2) = -228.88 kJ` |
|
| 36. |
On the basisof the following thermochemical data ( Delta_(f)G_(H^(+))(aq)= 0) H_(2)O(l) rarrH^(+)(aq)+ OH^(-), DeltaH= 57.32kJ H_(2)(g) +(1)/(2) O_(2)(g) rarr H_(2)O(l) , DeltaH = -282.20 kJthe value of enthalpy of formation ofOH^(-) ion at25^(@)C is |
|
Answer» `- 22.88 KJ ` `Delta_(R)H= [Delta_(f) H(H^(+))+Delta_(f)H^(@) ( OH^(-)) ]-Delta_(f)H^(@) (H_(2)O)` `:. 57.32 =0+ Delta_(f)H^(@) ( OH^(-)) - ( -286.2)` or `Delta_(f)H^(@) ( OH^(-)) =57.32 -286.2 = -228.88 kJ ` |
|
| 37. |
On the basis of the following E^(@) values the stongest oxidizing agent is [Fe(CN_(6)]^(4-)rarr[Fe(CN)_(6)]^(3-)+E^(-1),E^(@)=-0.35 v Fe^(2+)rarrFe^(3+)+E^(-1),E^(@)=-0.35 v |
|
Answer» `Fe^(3+)` `E^(2)+=0.35V` `Fe^(3+)+E^(-)rarrFe^(2+), E^(@)=+0.77` V higher +ve electrode POTENTIAL STRONGER is the oxidising agent thus OPTION (a) is CORRECT |
|
| 38. |
On the basis of the equation pH = - log [H^(+)], the pH of 10^(-8) mol dm^(-3) solution of HCl should be 8. However, it is observed to be less than 7.0 . Explain the reason. |
| Answer» Solution :pH of `10^(-8)` M HCl solution is not8 because this concentration is so low that `H^(+)` ion produced from `H_(2)O` in the solution (VIZ. `10^(-7)M`) connot be neglected. Total `[H^(+)] = 10^(-8) + 10^(-7)`M. SOLVING and calculating pH, we GET the value close to 7 but les than 7 as the solution is ACIDIC | |
| 39. |
On thebasisof symmetry (n-a) d^(4) ns^(2) and (n-1) d^(9) ns^(2) configurationsare lessstableand immediately changeover to thecorresponding more stable(n-1) d^(5) ns^(1)" and" (n-1) d^(10) ns^(1) configurations .Arethereany exceptions to thisgeneralization ? Comment |
|
Answer» Solution :The are many excetions to THISGENERALIZATION . Someof these are : `{:("Name of the element","ATOMIC NUMBER","Expected EC ","Actual EC"),("1. Tungsten (W) ",74,[Xe] 4f^(14) 5d^(5) 6s^(1),[Xe] 4f^(14) 5d^(4) 6s^(2)),("2.Platinum (Pt) ",78,[Xe] 4f^(14) 5d^(10) 6s^(0),[Xe] 4f^(14) 5d^(9) 6s^(1)),("3. Seaborgium (Sg) ",106,[Rn] 5f^(14) 6d^(5) 7S^(1),[Rn] 5f^(14) 6d^(4) 7s^(2)):}` |
|
| 40. |
On the basis of standard electrode potential values, suggest which of the following reactions would take place ? (Consult the book for E^(Ө) value) (a) Cu+Zn^(+2)toCu^(+2)+Zn (b) Mg+Fe^(+2)toMg^(+2)+Fe ( c) Br_(2)+2Cl^(-)toCl_(2)+2Br^(-) (d) Fe+Cd^(+2)toCd+Fe^(+2) |
|
Answer» Solution :`E_(Cu^(+2)//Cu)^(@)=0.34V,E_(Zn^(+2)//Zn)^(@)=-0.76V` `E_(Mg^(+2)//Mg)^(@)=-2.37V,E_(Fe^(+2)//Fe)^(@)=-0.74V` `E_(Br_(2)//Br^(-))^(@)=+1.08V,E_(Cl_(2)//Cl^(-))^(@)=+1.36V` `E_(CD^(+2)//Cd)^(@)=-0.44V` (a) `E_(Cu^(+2)//Cu)^(@)=-0.34V,E_(Zn^(+2)//Zn)^(@)=-0.76V` `Cu+Zn^(+2)TOCU^(+2)+Zn` `E_("cell")^(@)=E_("cathode")^(@)-E_("anode")^(@)` = `E_(Zn^(+2)//Zn)^(@)-E_(Cu^(+2)//Cu)^(@)` = `-0.76-(+0.34)=-1.10V` Value of `E_("cell")^(@)` is negative so reaction is not possible. (B) `Mg+Fe^(+2)toMg^(+2)+Fe` `E_("cell")^(@)=E_(Fe^(+2)//Fe)^(@)-E_(Mg^(+2)//Mg)^(@)` = `-0.74-(-2.37)=+1.63V` Value of `E_("cell")^(@)` is positive so reaction is possible. ( c) `Br_(2)+2Cl^(-)toCl_(2)+2Br^(-)` `E_("cell")^(@)=E_(Br^(-)//Br_(2))^(@)-E_(Cl^(-)//Cl_(2))^(@)` = `+1.08-(+1.36)=-0.28V` Value of `E_("cell")^(@)` is negative so reaction is not possible. (d) `Fe+Cd^(+2)toCd+Fe^(+2)` `E_("cell")^(@)=E_(Cd^(+2)//Cd)^(@)-E_(Fe^(+2)//Fe)^(@)` = `-0.44-(-0.74)=+0.30V` Value of `E_("cell")^(@)` is positive so reaction is not possible. |
|
| 41. |
On the basis of standard electrode potential values, suggest which of the following reactions would take place ? (Consult the book for E^@ value) (i) Cu+Zn^(2+)rarr Cu^(2+) +Zn (ii) Mg + Fe^(2+) rarr Mg ^(2+) +Fe (iii) Br_2+2Cl^(-) + 2Cl^(-) rarr Cl_2 +2Br^(-) (iv) Fe+Cd^(2+) rarr Cd +Fe^(2+) |
|
Answer» Solution :(i) `Cu^(2+)+Zn^(2+) rarr Cu^(2+)+Zn` `E^@ (Cu^(2+)|Cu)=+0.34V,E^@(Zn^(2+)|Zn)=-0.76V` `E_("cell")^@=E^@("CATHODE")-E^@("ANODE")` `=-0.76-0.34=-1.10V` Since `E_("cell")^@` s - ve, the reaction does not OCCUR. (II) `Mg + Fe^(2+) rarr Mg ^(2+) +Fe ` `E^@(Mg^(2+)|Mg)=-2.37V,E^@(Fe^(2+)|Fe)=-0.74V` `E_("cell")^@=-0.74-(-2.37)=++1.63V` Since `E_("cell")^@` is +ve, the reaction will take place. (iii) `Br_2+2Cl^(-) + 2Cl^(-) rarr Cl_2 +2Br^(-)` `E_("cell")^@ = +1.08""1.36=-0.28V` Sine `E_("cell")^@ ` is ve - , this reaction does not occur . (iv) `Fe+Cd^(2+) rarr Cd +Fe^(2+)` `E_("cell")^@=-0.44-(-0.74)=+0.30` Since `E_("cell")^@` is +v, therefore , this redox reaction will occur. |
|
| 42. |
On the basis of standard electrode potential values suggest which of the following reactionwould take place ? (i) Cu+Zn^(2+)+Zn (ii) Mg+Fe^(2+)+Fe (iii) Br_(2)+2CI^(-)rarrCI_(2)+2Br (iv) Fe +Cd^(2+)rarrCd+Fe^(2+) |
|
Answer» Solution :(i) from the table `E_(Cu^(2+)//Cu)=+0.34 V "and" E_(Zn^(2+)//Zn))^(@)=-0.76V` The reaction `Cu+Zn^(2+) rarr Cu^(2+)+Zn` is FEASIBLE if `E_("cell")^(@)` is +v in this cell buy gets oxidised to `Cu^(2+)` therefore `Cu^(2+)//Cu` redox COUPLE acts as the anode further since `Zn^(2+)` getsreduced to Zn therefore `Zn^(2+)//Zn` acts as the cathode `therefore E_(cell)^(@)=E_("anode")^(@)=E_(Cu(Zn^(2+)//Zn)^(@)=-0.76-0.34=-1.10 v` since `E_("cell")^(@)` of the reaction `Cu+zn^(2+)rarrCu^(2+)rarrCu^(2+)Zn` is -ve therefore this reaction does not occur (ii) from `E_(Mg^(2+)//Mg)^(@)=-2.37 v` and `E_(Fe^(2+)//Fe)^(@)=-0.74 V` The reactoin `Mg+Fe^(2+)rarrMg^(2+)+Fe` is possible if `E_(cell)^(@)` is +ve here `Mg^(2+)//Mg`redox couple acts as the anode while `Fe^(2+)//Fe` acts the cathode (iii) From `E_(Br^(-)//Br_(2))^(@)=+1.08 V` and `E_(CI^(-)//CI_(2))^(@)=+1.36 v` The reaction `Br_(2)+21CI^(-)rarrCI_(2)+2Br^(-)` will occur if `E_(cell)^(@)`is +ve in this cel `Br_(2)//Br^(-)` redox couple wil actsas the cahode and `CI_(2)//CI^(-)` will act as the anode `E_(cell)^(@)=E_("cathode")^(@)-E_("anode")^(@)=E^(2)_(Br^(2+)//Fe)=-0.74 V and E_(Cd^(2+)//Cd)^(@)=-0.44V` The reaction `Fe+Cd^(2+)rarrCd+Fe^(2+)` wil occur if `E_(cell)^(@)` is +ve here in his reaction `Fe^(2+)//Fe` ELECTRODE will act as the anode while`Cd^(2+)//Cd` electrode will act as the cathode `E_(cell)^(@)=E_(cathode)^(@)-E_(anode)^(@)=E_(Cd^(2+)//Cd)^(@)-e_(Fe^(2+)//Fe)^(@)rarrCd+Fe^(2+)` will occur |
|
| 43. |
On the basis of quantum numbers justify that the sixth period table should have 32 elements. |
| Answer» SOLUTION :Each period starts with filling of a particular SHELL. The sixth period starts by filling the sixth shell (n = 6). (This corresponds to l = 0,1,2,3,4,5, (s,p,d,f,g,H subsshells) and HENCE there are 36 ORBITALS). As per Aufbau principle, the energy increases in the order 6s 4f 5d and 6p. | |
| 44. |
On the basis of quantum numbers, justify that the sixth period of the periodic table should have 32 elements. |
| Answer» Solution :The sixth period corresponds to sixth shell. The orbitals present in this shell are 6s, 4f, 5p and 6D. The maximum number of ELECTRONS which can be present in these sub-shell is 2 + 14 + 6 + 10 - 32. Since the number of ELEMENTS in a period corresponds to the number of electrons in the shells, the sixth period should have a maximum of 32 elements. | |
| 45. |
On thebasisof quantumnumber, justifythat thesixthperiodof theperiodictableshouldhave 32elements. |
| Answer» Solution :In themodern periodic table , eachperiodstartswith thefillingsof a newprincipal energylevel. Thusthesixthperiodbeginswith thefillingof principalquantumnumber `n=6 ` . Whenn=6,L =0 , 1,2,3 . Butaccordingto aufbauprinciplethe electronsare addedto differentorbitalsin orderof theirincreasingenergies . Now theenergyof 6d- subshellis higherthan thatof 7s- subshellbutenergies of5d-and 4f- subshells arelower thanthat of6d-subshell . Thereforein 6thperiodelectronscan befilledin only 6s- 4f, 5d and6p- subshellswhoseenergiesincreasein THEORDER `: 6 s lt 4f lt5 d lt6 p`Now s- subshells hastwo p-subshell has threed-subshellhas fiveand f- subshellhas sevenorbitals. Hencein allthere are 16 (2+ 3 +5 + 7)orbitalsthat canbe filledin THISPERIOD. Sinceaccordingto pauli's exclusions PRINCIPLE eachorbitalat the maximumcan accommodate two elements therefore16 orbitals at the d maximum can have32electrons and hencesixthperiodhas 32elements. | |
| 46. |
On the basis of quantum numbers, justify that the 6th period of the periodic table should have 32 elements. |
|
Answer» |
|
| 47. |
On the basis of molecular orbital treatment of various aromatic compound, it has been observed that an aromatic compound must fulfil the following theoritical requirements It must have an uninterrupted cyclic cloud of pi-electrons above and below the plane of the molecule (often called as pi-cloud). Let us look what does this mean? (i). for the pi-cloud to be cyclic, the molecule must be cyclic. (ii). For the pi-cloud to be uninterrupted, every atom int he ring must have a p-orbital iii). for the pi-cloud to be fomred, each p-orbital must be able to overlap with the p-orbitals on either side of it. therefore, the molecule must be planar. Q. The stability order of the three compounds |
|
Answer» IgtIIgtIII |
|
| 48. |
On the basis of molecular orbital treatment of various aromatic compound, it has been observed that an aromatic compound must fulfil the following theoritical requirements It must have an uninterrupted cyclic cloud of pi-electrons above and below the plane of the molecule (often called as pi-cloud). Let us look what does this mean? (i). for the pi-cloud to be cyclic, the molecule must be cyclic. (ii). For the pi-cloud to be uninterrupted, every atom int he ring must have a p-orbital iii). for the pi-cloud to be fomred, each p-orbital must be able to overlap with the p-orbitals on either side of it. therefore, the molecule must be planar. Q. |
|
Answer» AROMATIC |
|
| 49. |
On the basis of molecular orbital treatment of various aromatic compound, it has been observed that an aromatic compound must fulfil the following theoritical requirements It must have an uninterrupted cyclic cloud of pi-electrons above and below the plane of the molecule (often called as pi-cloud). Let us look what does this mean? (i). for the pi-cloud to be cyclic, the molecule must be cyclic. (ii). For the pi-cloud to be uninterrupted, every atom int he ring must have a p-orbital iii). for the pi-cloud to be fomred, each p-orbital must be able to overlap with the p-orbitals on either side of it. therefore, the molecule must be planar. Q. Cyclo-octatetraene is: |
|
Answer» AROMATIC |
|
| 50. |
On the basis of molecular orbital theory, select the most appropriate option. |
|
Answer» The BOND ORDER of `O_(2)` is 2.5 and it is PARAMAGNETIC |
|