Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Tin contains only 5 - 10 % of tin as SnO_(2), the rest being Siliceous matter, wolfram and pyrites of iron copper and arsenic. It is not attached by water. Only molten tin reacts with steam liberating hydrogen. In stannous oxide, tin is +2 oxidising state. So stannous oxide is also described as tin (II) oxide Tin (Ti) chloride is used |
|
Answer» As a modrant in DYEING |
|
| 3. |
The constant motion and high velocities of gas particles lead to some important practical consequences One such consequence is that is mixing rapidly when they come in contact. The mixing of different gases by random molecular motion and with frequent collisions is called diffusion.A similar process in which gas molecules escape through a tiny hole into vaccum is called effusion. 4 g of H_(2) effused through a pinhole in 10 sec at constant temperature and pressure The amount of oxygen effused in the same time interval and at the same conditions of temperature and pressure would be |
|
Answer» 4 g |
|
| 5. |
The constant motion and high velocities of gas particles lead to some important practical consequences One such consequence is that is mixing rapidly when they come in contact. The mixing of different gases by random molecular motion and with frequent collisions is called diffusion.A similar process in which gas molecules escape through a tiny hole into vaccum is called effusion. Helium gas at 1 atm and SO_(2) at 2 atm pressure temperature being the same are released seperately at the same moment into 1 m long evacuated tubes of equal diameters If helium reaches the other end of the tube in t sec what distance SO_(2) would traverse in the same time interval in the other tube? |
|
Answer» 25 cm |
|
| 6. |
The costancy of e/m ratio for electron shows that |
|
Answer» <P>`E, p,N,ALPHA` |
|
| 7. |
The constancy of e/m ratio for electron shows that |
|
Answer» Electron's mass is `1//1837^(th)` of the mass of proton |
|
| 8. |
The consequences of global warming may be______ |
|
Answer» increase in AVERAGE temperature of the EARTH |
|
| 9. |
The consequences of global warming are A) Increase in average temperature of the earth B) Melting of Himalayan glaciers C) Increased biochemical oxygen demand of natural water D)Eutrophication of water bodies The correct answer is |
| Answer» Answer :C | |
| 10. |
The conpound which on reducting with LiAiH_(4) gives two alcohols : |
|
Answer» `CH_(3)COOCH_(3)` |
|
| 11. |
The consequences of global warming may be ……………… . |
|
Answer» increase in AVERAGE temperature of the earth. |
|
| 12. |
The conjugate base of OH^(-) is |
|
Answer» `O^(2-)` `:.` Conjugate base of `OH^(-)=OH^(-)-H^(+)=O^(2-)` |
|
| 13. |
The conjugate base of hydrazoic acid is |
|
Answer» `N^(3-) ` |
|
| 14. |
The conjugate base of H_2PO_4^(-)is |
|
Answer» ` H_3 PO_4` |
|
| 15. |
The conjugate acid of OH^(-) ions is...........whereas its conjugate base is .......... . |
|
Answer» |
|
| 17. |
The configurrtions of the reactant and the product in the following reation ,respectively are: |
|
Answer» R,R 
|
|
| 18. |
The configuration fo the C - 2 epimer of D - glucose is |
|
Answer» 2R,3S,4R,5S 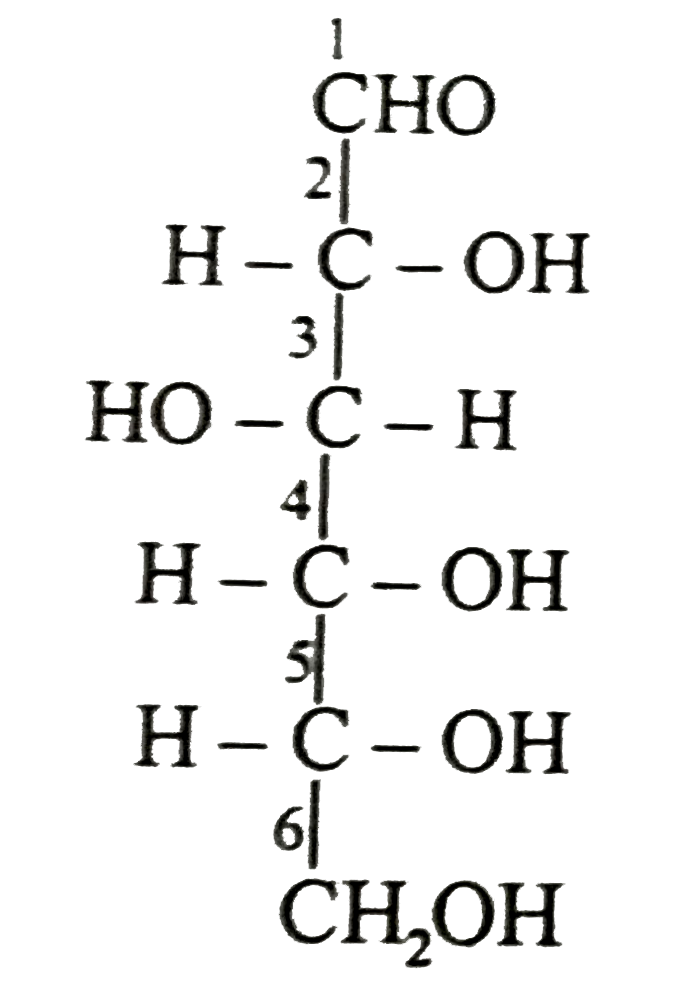
|
|
| 19. |
The configuration of Cr atom is 3d^(5)4s^(1) but not 3d^(4)4s^(2) due to reason R_(1) and the configuration of Cu atom is 3d^(10)4s^(1) but not 3d^(9)4s^(2) due to reason R_(2).R_(1) and R_(2) are: |
|
Answer» `R_(1):` The exchange energy of `3d^(5)4s^(1)`: is GREATER than that of `3d^(4)4s^(2)`  `3d^(5)4s^(1)` is correct because it has greater exchange possibilities of unpaired electron. `3d^(10)4s^(1)` is correct because `3d^(10)` orbitals are spherically symmetrical. |
|
| 20. |
The configuration of sugars is related to glyceraldehyde and that of amino acids is related to: |
|
Answer» SERINE |
|
| 21. |
The configuration of chromium atom in ground state is |
|
Answer» `[AR]3d^(4)4s^(2)` |
|
| 22. |
The configuration of 1s^(2)2s^(2)2p^(5)3s^(1) shows |
|
Answer» GROUND state of FLUORINE atom |
|
| 23. |
The configuration 1s^(2)2s^(2)2p^(6)3s^(2)3p^(3) corresponds to |
|
Answer» S |
|
| 24. |
The configuration [Ar]3d^(5)4s^(1) in the first excited states exists for |
|
Answer» `FE^(2+)` `""` EXCITED state `= 3d^5 4s^1` b) `Co^(3+)`: Ground state = `3d^6` `""` Excited state `=3d^5 4s^1` c) `Mn^(+) : ` Ground state =`3d^5` `""` Excited state `= 3d^5 4s^1` d) Cr :Ground state `=3d^5 4s^1` `""`Excited state :Excited state `=3d^5 4p^1` |
|
| 25. |
The configuration 1s^(2)2s^(2)2p^(5)3s^(1) shows |
|
Answer» GROUND state of FLUORINE |
|
| 26. |
The conditions for the combination of atomic orbitals to form molecular orbitals are stated below. Mark the incorrect condition mentioned here. |
|
Answer» The combining atomic orbitals must have nearly same energy. |
|
| 27. |
The condensed formula for CH_3CH_2CH_2CH(CH_3)NH_2 is _________ |
|
Answer» `CH_3(CH_2)_4NH_2` |
|
| 28. |
The concept of oxidation number (O.N) is very important in under standing redox reaction s it helps to identify the oxidant / reductant in a redox reaction it also helps to (i) find oiut thepossible molecular formula of any neutral compound and (ii) to balance redox reaction For the reaction M^(x+)+MnO_(4)^(-)rarrMO_(3)^(-)+Mn^(2+)+1//2 O_(2) if one mole of MnO_(4)^(-) oxidises 1.67moles of M^(x+) to MO_(3)^(-) then the value of x in the reaction is |
|
Answer» 5 since 1 MOLE of `MnO_(4)^(-)` accept 5 mole of electrons therefore 5 moles of electrons are LOST by 1.67moles of `M^(x+)` since `M^(x+)` change to `MO_(3)^(-)` by ACCEPTING 3 electrons `therefore` oxidation state of M x=+5-3=+2 |
|
| 29. |
The concept of oxidation number (O.N) is very important in under standing redox reaction s it helps to identify the oxidant / reductant in a redox reaction it also helps to (i) find oiut thepossible molecular formula of any neutral compound and (ii) to balance redox reaction when copper is treated with a certain concentraton of nitric acid oxide and nitrogen dioxide are liberated in equal volume according to the equation XCu+YHNO_(3)rarrCu(NO_(3))_(2)+NO+NO_(2)+H_(2)O the coefficeents of x and y are |
|
Answer» 2 and 3 `3Cu +8HNO_(3) rarr 3Cu(NO_(3))_(2)+2NO+4H_(2)O` `Cu+4HNO_(3)rarrCu(NO_(3))_(2)+2NO_(2)+2H_(2)O` adding eqt (i) and (II) we have `4Cu+12 HNO_(3)rarr2Cu(NO_(3))_(2)+NO_(2)+NO+3H_(2)O` Thus coefficient x ANDY of Cu and `HNO_(3)` respecitively are 2and 6 |
|
| 30. |
The concept of oxidation number (O.N) is very important in under standing redox reaction s it helps to identify the oxidant / reductant in a redox reaction it also helps to (i) find oiut thepossible molecular formula of any neutral compound and (ii) to balance redox reaction A compound contains atoms of three elements A b and c if the oxidation umber of A is + 2 B is +5 and that of C is -2 the possible formula of the compund is |
|
Answer» `A_(3)(BC_(4))_(2)` `3xx(+2)+2xx(+5+4xx-2)=0` Thus option (a) is correct |
|
| 31. |
The concept of oxidation number (O.N) is very important in under standing redox reaction s it helps to identify the oxidant / reductant in a redox reaction it also helps to (i) find oiut thepossible molecular formula of any neutral compound and (ii) to balance redox reaction A mole of hydrazine (N_(2)H_(4)) loses ten moles of electrons to form a new compound X assuming that all the nitrogen appears in the new compound what is the oxidation state of nitrogen in x ? (there is no change in the oxidation numberof hydrogen in thereaction ) |
|
Answer» `-1` `therefore` O.N of each N in `X=6//2=3` |
|
| 32. |
The concept of hybridisation has been introduced to explain the shapes of molecules. It involves the intermixing of two or more atomic orbitals belonging to same atom but in or more atomic orbitals beloging to sasme atom but in different sub-shells so as to intermix and redistibute energies to from equivalent orbitals called hybrid orbitals.Depending upon toh enumber and nature of the orbitals involved, the hybridisation may be divided into sp (linear), sp^(2) (trigonal), sp^(3) (tetrahedral), sp^(3)d (trigonal bipyramidal), sp^(3)d^(3) (octahedral) and sp^(3)d^(3) (pentagonal bipyramidal) types. it may be noted that the orbitals of isolated atoms never hybridise and they do so at the time of bond formation. A hybrid orbital from s-and p-orbitals can contribute to |
|
Answer» `SIGMA`-BOND only |
|
| 34. |
The concentration of sulphide ion in 0.1 M HCl solution saturated with hydrogen sulphide is 1.0xx10^(-9)M. If 10 mL of this solution is added to 5 mL of 0.04 M solution of FeSO_(4), MnCl_(2), ZnCl_(2) and CdCl_(2), in which solutions precipitation will take place ? Given K_(sp) for Fes=6.3xx10^(-18), MnS = 2.5xx10^(-13), ZnS=1.6xx10^(-24) and CdS=8.0xx10^(-27). |
|
Answer» SOLUTION :Precipitation will take PLACE in the solutionfor which ionic product is GREATER than solubility product. As 1o mL of solution containing `S^(2-)` ion is mixed with 5 mL of metal salt solution, after mixing `[S^(2-)]=1.0xx10^(-19)xx(10)/(15)=6.67 xx 10^(-20)`. `[Fe^(2+)]=[Mn^(2+)]=[ZN^(2+)]=[Cd^(2+)]=(5)/(15)xx0.04 = 1.33xx10^(-2)M` `:.` Ionic product for each of these will be `=[M^(2+)][S^(2-)]=(1.33xx10^(-2))(6.67xx10^(-20))=8.87xx10^(-22)` As this is greater than the solubility product of ZnS and CdS, therefore , `ZnCl_(2) and CdCl_(2)` solutions will be precipitated. |
|
| 35. |
The concentration of sulphide ion in 0.1 M HCl solution saturated with hydrogen sulphide is 1.0xx10^(-19) M. if 10 mL of this is added to 5 mL of 0.04 M solution of the following : FeSO_4 , MnCl_2 , ZnCl_2 and CdCl_2 |
|
Answer» Solution :`K_(SP)(FeS)=6.3xx10^(-18) , K_(sp) (MnS) = 2.5xx10^(-13)` `K_(sp)(ZnS)=1.6xx10^(-24) , K_(sp) (CdS)=8.0xx10^(-27)` CALCULATION `[S^(2-)]` in mixture : `[S^2]=1.0xx10^(-19) = M_1` its 10 mL solution + 5 mL SALT solution Total volume = (10+5)=15 mL `{:("Initial","In mixture"),(M_1V_1,M_2V_2),(1.0xx10^(-19)xx10,M_2xx15):}` `therefore M_2=(1.0xx10^(-19)xx10)/15=6.667xx10^(-20)` M Thus in mixture `[S^(2)]=6.667xx10^(-20)` M Concentration of metal ion in mixture : [metal salt ] =[metal ion] = 5 mol 0.004 M + volume of sulphide ion =10 mL `therefore` Volume of solution in mix two =15 mL So, concentrationof metal ion in mixture `{:("Initial","In mixture"),(M_1V_1,=M_2V_2),(0.04M xx 5"ml",M_2xx15 "ml"):}` `therefore M_2=(0.04M xx 5 mL)/"15 ml"=1.333xx10^(-2)` M Therefore in mixed solution `[Fe^(2+)]=[Mn^(2+)]` `=[Zn^(2+)]=[Cd^(2+)]` `=1.333xx10^(-2)` M Calculate value of `Q_(sp)` For salt M S `Q_(sp)=[M^(2+)][S^(2-)]=(1.333xx10^(-2))(6.667xx10^(-20))` `=8.8891xx10^(-22)` Ionic product Prediction of precipitation : For precipitation , `Q_(sp) gt K_(sp)` so, [`K_(sp)` of ZnS=`1.6xx10^(-24)` ] [`K_(sp)` of CdS=`8.0xx10^(-27)` ] Ionic product of both is more than their solubility product `Q_(sp)` is more. `therefore` Precipitation are formedfrom ZnS and CdS |
|
| 36. |
The concentration of saturated solution of Mg(OH)_2 is 8.2 xx 10^(-4) % w/V. Calculate its solubility product. Its molecular mass is 58.3 g "mol"^(-1) |
| Answer» SOLUTION :`1.12xx10^(-11)` | |
| 37. |
The concentration of saturated solution of Mg(OH)_2 is 8.2 xx 10^(-4) g L^(-1) at 298 K temp. Then calculate its solubility product. |
| Answer» SOLUTION :`1.121xx10^(-14) ("mol L"^(-1))^3` | |
| 38. |
The concentration of oxalic acid is 'x' mol "litre"^(-1). 40 mL of this solution reacts with 16 mL of 0.05 M acidified KMnO_(4). What is the pH of 'x' M oxalic acid solution ? (Assume that oxalic acid dissociates completely.) |
|
Answer» 1.3 `(M_(1)V_(1))/(n_(1))(KMnO_(4))=(M_(2)V_(2))/(n_(2)){:((COOH,),(|"",),(COOH,)):}` `(0.05xx16)/(2)=(X xx40)/(5)` x=0.05 M `[H^(+)]=2xx0.05=0.1 M` `pH= - " log" (H^(+))= - " log" (0.1)=1` |
|
| 39. |
The concentration of OH^- in water at 298 K |
|
Answer» `10^(-14)` `H_2O_((l)) + H_2O_((l)) = H_3O_((aq))^(+) + OH_((aq))^(-)` |
|
| 40. |
The concentration of oxalic acid is 'X' mol lit^(-1) . 40 ml of this solution reacts with 16 ml of 0.05 M acidified KMnO_(4) . What is the pH of 'X' M oxalic acid solution ? (Assume that oxalic acid dissociates completely ) |
|
Answer» `1.3` |
|
| 41. |
The concentration of hydronium ions in a cup of black coffee is 1.3xx10^(-5) M. Find the pHof the coffee. Is this coffee acidic or alkaline ? |
|
Answer» |
|
| 42. |
The concentration of hydrogen ion in a sample of soft drink is 3.8xx10^(-3)M. What is its pH ? |
|
Answer» Solution :Here, we are given that `[H^(+)]=3.8xx10^(-3)M` `PH=-log [H^(+)]=-log (3.8xx10^(-3))=-(log 3.8 + log 10^(-3))=-0.58 + 3 = 2.42 = 2.42` As pH is LESS than 7, the SOFT drink is acidic. |
|
| 43. |
The concentration of hydrogen ion in a sample of soft drink is 3.8xx10^(-3) M. What is its pH ? |
| Answer» SOLUTION :`PH = - log [H^(+)]=- log (3.8xx10^(-3))= - log 3.8 + 3 = 3 - 0. 5798 = 2.4202 = 2.42` | |
| 44. |
The concentration of hydrogen ion in a sample of soft drink is 3.8 xx 10^(-3)M. what is its pH ? |
|
Answer» Solution :`[H^+]=[3.8xx10^(-2)]`M pH=-log `(3.8xx10^(-2))` `=-{log 3.8+ log 10^(3)}` =-(0.5798+(-3)) =-(-2.4202) =+2.42 pH `LT` 7.0 So, the solution is acidic. |
|
| 45. |
The concentration of H_2, I_2 and HI at 731K respectively 0.92 xx 10^(-2), 0.20 xx 10^(-2) and 2.96 xx 10^(-2) "mol L"^(-1), calculate equilibrium constant. H_(2(g)) + I_(2(g)) hArr 2HI_((g)) |
|
Answer» Solution :The given REACTION of SYNTHESIS of HI is as under. `H_(2(g)) + I_(2(g)) HARR 2HI_((g))` and its `K_c` can represent as under `K_c=([HI]^2)/([H_2][I_2])` where, [HI] =`0.92xx10^(-2) "mol L"^(-1)` `[I_2]=0.20xx10^(-2) "mol L"^(-1)` `[HI]=2.96xx10^(-2) "mol L"^(-1)` `=((2.96xx10^(-2))^2("mol L"^(-1))^2)/((0.92xx10^(-2))(0.20xx10^(-2))("mol L"^(-1))( mol L^(-1)))` =47.62 |
|
| 46. |
The concentration of H_(2)O_(2) in a solution containing 34g in 500ml is The correct match is |
|
Answer» `{:(A,B,C,D),(III,IV,I,II):}` |
|
| 47. |
The concentration of fluoride, lead, nitrate and iron in a water sample from an underground lake was found to the 1000 ppb, 40 ppb, 100 ppm and0.2 ppm respectively. This water is unsuitable for drinking due to high concentration of |
|
Answer» fluoride |
|
| 48. |
The concentration of fluoride, lead, nitrate and iron in a water sample from an underground lake was found to be 1000 ppb, 40 ppb, 100 ppm and 0.2 ppm, respectively. This water is unsuitable for drinking due to high concentration of: |
Answer» SOLUTION :
|
|
| 49. |
The concentration of electrolyte required to coagulate a given amount of As_(2)S_(3) sol.is minimum in the case of : |
|
Answer» Potassium sulphate |
|
| 50. |
The concentration of both Na_(2)CO_(3) and NaHCO_(3) is 5.2xx10^(-3) mol in their mixture. The amount of 0.1 (M) HCl required to nutralise this mixture completely is- |
|
Answer» 1.56 L |
|